Infection Triggered Encephalopathy in Children: Insights
Beyond The Usual – Infection Triggered Encephalopathy Syndromes (ITES) In Children
Dr. Prerna Choudhury, MD, MRCPCH¹
- Consultant, Pediatric Neurology, Bharati Vidyapeeth Medical College and Hospital, Pune
OPEN ACCESS
PUBLISHED: 31 July 2025
CITATION: Choudhury, P., 2025. Beyond The Usual – Infection Triggered Encephalopathy Syndromes (ITES) In Children. Medical Research Archives, [online] 13(7).
https://doi.org/10.18103/mra.v13i7.6828
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i7.6828
ISSN 2375-1924
Abstract
Infections of the central nervous system have significant long-term morbidity and mortality in the paediatric population in both developing and developed countries. One of the dreaded presentations seen in children is that of acute febrile encephalopathy that is triggered by infectious or non-infectious agents. The COVID 19/SARS-CoV pandemic gave us newer insights into the role of immune dysregulation that is triggered by an antecedent infection. Since then, multiple new terminologies have been defined based on imaging characteristics in children presenting with acute encephalopathy. This is now collectively termed as infection triggered encephalopathy syndrome (ITES). This review looks at the expanding spectrum of infective conditions of the CNS in children and how early differentiation of the various conditions can impact the management and overall clinical outcome.
Keywords:
- Central nervous system
- infections
- artificial intelligence
- MRI patterns
- ITES
Abbreviations:
- CNS – Central nervous system
- ITES – Infection triggered encephalopathy syndrome
- BBB – Blood brain barrier
- ADEM – Acute demyelinating encephalomyelitis
- ADS – Acute demyelinating syndrome
- ON – Optic Neuritis
- TM – Transverse myelitis
- MOG – Myelin oligodendrocyte
- NMO – Neuromyelitis optica
- ALERD – Acute leukoencephalopathy with restricted diffusion
- AESD – Acute encephalopathy with biphasic seizures and diffusion restriction
- FIRES – Fever induced refractory epilepsy syndrome
- ANEC – Acute necrotising encephalopathy of childhood
- AFCE – Acute fulminant cerebral edema
- AERRPS – Acute encephalopathy with refractory repetitive partial seizures
- MERS – Mild encephalopathy with reversible splenial lesions
- HHV – Human herpes virus
- DAI – Diffuse axonal injury
- PRES – Posterior reversible encephalopathy syndrome
- DEE – Developmental epileptic encephalopathy
Introduction
Central nervous system (CNS) infections in the paediatric population represent a significant cause of morbidity and mortality. These conditions, encompassing a spectrum from meningitis to encephalitis, present unique diagnostic, and therapeutic challenges due to the ongoing development of the paediatric brain and the diverse range of potential aetiologies.
The CNS, comprising of the brain and spinal cord, is protected by anatomical and physiological barriers, most notably the blood-brain barrier (BBB). This highly selective membrane restricts the passage of substances from the systemic circulation into the CNS and is crucial for maintaining homeostasis. However, the BBB can also impede the entry of immune cells and antimicrobial agents, complicating infection management. The relative immaturity of these protective mechanisms in children increases their susceptibility to a wide array of infectious agents.
The terminologies classically used for CNS infections include:
- a) Meningitis – inflammation/infection of the meninges
- b) Encephalitis – inflammation of the brain parenchyma
- c) Meningoencephalitis – diffuse inflammation/infection of both the meninges as well as the brain parenchyma
- d) Encephalomyelitis – involvement of the brain parenchyma and spinal cord
Encephalopathy is a state of altered sensorium resulting from disturbances in brain function. This can present as deterioration in consciousness, behavioural disturbances, difficulties in speech or higher mental functions. It can be divided into 4 categories:
- a) Hyperacute (seconds to minutes)
- b) Acute (hours to days)
- c) Subacute (weeks- months)
- d) Chronic (months – years)
Table 1: Causes of encephalopathy in children
| Hyperacute | Acute | Subacute | Chronic/Recurrent |
|---|---|---|---|
| Primary Neurological | 1. Infections – bacterial/Viral 2. Acute Demyelinating syndromes (ADEM, MOG antibody disease) 3. Infection triggered encephalopathy syndromes (ITES – ANE, HSES/AFCE) 4. Metabolic – Mitochondrial encephalopathy 5. New onset refractory status epilepticus (NORSE) 6. Vascular – subdural haemorrhage |
1. Infection triggered encephalopathy syndromes (AESD/ALERD, FIRES) 2. Infectious – Fungal, tubercular, parasitic, HIV 3. Neoplastic |
1. Genetic – RANBP2 mutation 2. Glycine encephalopathy 3. Developmental epileptic encephalopathies (DEE) |
| 1. Moderate to severe head trauma, Diffuse axonal injury (DAI) 2. Intracranial haemorrhage (Arterio-venous malformation/aneurysm) 3. Migraine |
1. Systemic infection/Sepsis 2. Hypovolemic shock 3. Adrenal crisis 4. Toxins – carbon monoxide, organophosphorous, opioids |
1. Thyroid encephalopathy (SREAT) 2. Toxins – Heavy metal (Arsenic, Lead, Mercury) |
Table 2: Key differences between auto-inflammatory versus autoimmune encephalopathy
| Auto-inflammation | Auto-immune |
|---|---|
| Neurological syndromes Virus associated encephalitis, neurovasculitis syndromes, neuro-sarcoidosis | Acute demyelinating syndromes-ADS (ADEM, TM, ON), autoimmune encephalitis (Anti-NMDAR, Anti-GAD65) |
| Relation to infection Para-infectious | Post infectious |
| Immune mechanism Innate immunity | Adaptive immunity |
| Genetic syndromes Aicardi-Goutiere syndrome, RANBP2 associated recurrent ANE | Not well known |
| Imaging May be normal in early stages, then show characteristic patterns | May be normal in autoimmune encephalitis or associated with changes in the temporal lobe- hippocampus complex. In ADS, classical patterns are noted. |
| CSF Normal or shows pleocytosis, elevated proteins | May be normal |
| Additional investigations Elevated pro-inflammatory cytokines such as IL-6, IL-1β, CSF neopterin, quinolic acid | Positive antibody testing in serum (MOG/NMO), CSF (NMDAR, GAD -65), oligoclonal bands in CSF. Antibody testing may also be normal throughout or become seropositive at later stages. |
Unlike in infective encephalitis where there is a direct invasion of the parenchyma by pathogen, in ITES there is stimulation of the cytokine cascade in response to an infection outside the CNS. In autoimmune encephalitis, there is commonly antibody production in response to a subacute infection.
Epidemiology
In children, the most common causes of encephalopathy are infectious – bacterial or viral. A systematic analysis of the burden of meningitis performed by the WHO global disease burden study 2019 noted that there were 112,000 reported deaths from meningitis in children < 5 years of age in 2019. The most common organisms across all ages were Streptococcus pneumoniae and Neisseria meningitidis.
A prospective observational study in 176 Indian children found viral aetiology in 46.59% of the cohort followed by pyogenic and tubercular meningitis. Highest incidence was seen in the age group of 3-6 years with increased mortality in younger age groups (1 month-3 years). Tubercular meningitis was noted to have significant sequelae.
Rickettsial infections, specifically with Orientia tsutusgamushi, is an important cause of acute encephalitis in areas with dense bush vegetation, classically known as the ‘tsutsugamushi triangle’ (Asia, Northern Australia and islands in the Indian and Pacific Ocean). A study from Southern India found scrub typhus positive in nearly half of their study population by rapid testing although real time PCR showed positivity in only 18% of these. Both Rickettsial and arthropod borne diseases show seasonal variation with higher incidence during the monsoon seasons in Southeast Asia. Epidemics of arthropod-borne diseases with CNS manifestations include dengue encephalitis, Japanese encephalitis, chikungunya encephalitis, and cerebral malaria.
We reported a case of Listeria meningoencephalitis that was community acquired with rare spinal involvement implicating a dual mechanism of direct invasion as well as immune mediated injury that responded well to a combination of appropriately chosen anti-microbials and steroids.
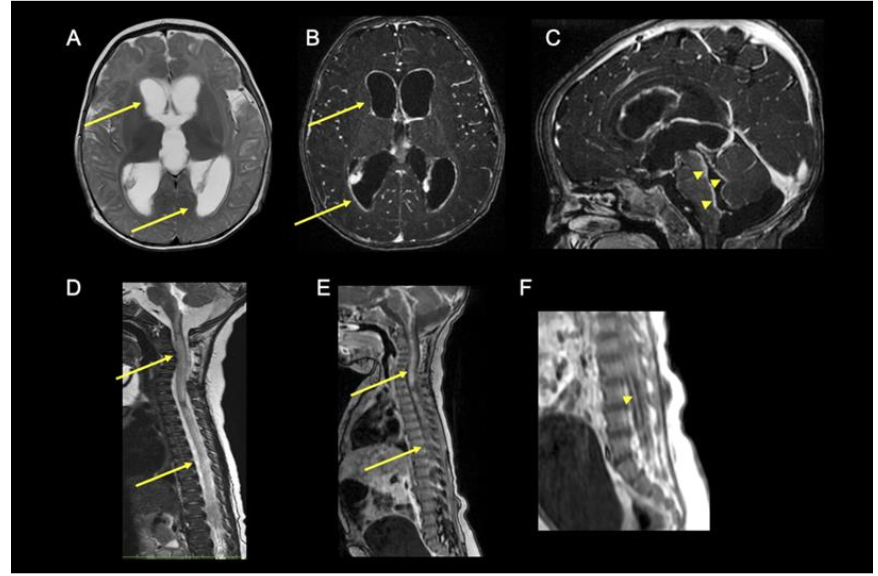
Herpes simplex encephalitis is the most common cause of viral encephalitis. There are several case reports and observational studies that show HSV association with autoimmune encephalitis, particularly anti-NMDAR encephalitis, although exact figures are missing. The most plausible theory behind this association suggests that direct invasion of the CNS by Herpesviridae triggers release of neuronal proteins that can induce auto-antibody production.
The differentiation between the various pathophysiological mechanisms between infectious, para-infectious and autoimmune encephalitis can be difficult with significant overlap in clinical presentations and initial management strategies.
Infection triggered encephalopathy syndromes (ITES) is a recent term that denotes a group of conditions that present with rapid decline in sensorium following a febrile illness and are associated with changes on neuroimaging. These may be associated with other symptoms such as seizures or movement disorders. ITES as an entity is different from other autoimmune or post infectious conditions such as autoimmune encephalitis and acute demyelinating encephalomyelitis (ADEM).
The International Consensus definitions for ITES by Sakuma et al published in August 2024 highlighted the various conditions that encompass this term and the pathophysiological mechanisms that differentiate the ITES group from other conditions. ITES comprises five conditions that are based on the MRI findings:
- Acute encephalopathy with biphasic seizures and restricted diffusion (AESRD)
- Acute necrotising encephalopathy (ANEC)
- Acute fulminant cerebral edema (AFCE)
- Acute shock with encephalopathy and multi-organ failure (ASEM)
- Mild encephalopathy with reversible splenial lesion (MERS)
Acute encephalopathy with biphasic seizures and restricted diffusion (AESRD/AESD) is the most common para-infectious encephalopathy in children. It commonly follows infection with pathogens such as influenza A, rhinovirus, mycoplasma and HHV 6. The average age group of children is around 9 months -2 years which is younger than those with FIRES. It usually presents with prolonged febrile seizures lasting >30 mins followed by a period of normal consciousness and then a decline in sensorium. This is followed by complex partial seizures on day 4-6 of illness. The initial MRI may be normal but later images can show characteristic pattern of restricted diffusion or atrophy. The diagnostic criteria is as below:
Diagnosis of AESRD
A diagnosis of AESRD is made if essential plus any of the additional criteria are present:
A) Essential
- Occurs during the course of a febrile illness
- Onset is characterised by prolonged febrile seizures lasting >30 mins in the first 2 days of illness
B) Additional
- Secondary seizures (usually cluster of complex partial seizures) or alteration in level of consciousness on Day 3-7 of illness
- DWI shows high signal in the subcortical white matter (bright tree appearance) and/or in the cortex at day 3 to 14. Sparing of pre- and postcentral gyrus (central sparing) is usually observed.
- After 2 weeks, residual lesions or atrophy on CT/MRI, or decreased blood flow on SPECT are observed in the frontal or fronto-parietal region, often with central sparing.
Acute necrotising encephalopathy of childhood (ANE/ANEC) is a rare but devastating condition following viral infection. Although, the most implicated organisms are influenza A and HHV 6, dengue is a known cause in the Indian subcontinent. Dengue associated necrotising encephalitis (DANE) affects similar areas as those in classical ANE and is now considered a variant of sporadic ANE. In contrast to AESD, there is a rapid deterioration of sensorium at onset (within a few hours). Seizures and encephalopathy are the most common presentation; movement disorders occur as sequelae of the disease. The diagnostic criteria for ANEC given is as follows:
- Acute encephalopathy related to a febrile viral infection: rapid reduction in consciousness and seizures
- CSF examination shows normal counts and increased proteins
- Symmetrical and multiple brain lesions on CT/MRI. These include bilateral thalamic lesions (most common) and lesions in periventricular areas, putamen, upper brainstem tegmentum, and cerebellum
- Elevated transaminase level with no elevation in serum ammonia levels
- Exclusion of other diseases such as severe viral/bacterial infections, metabolic conditions – mitochondrial disorders (Leigh’s), Reye’s syndrome, Wernicke encephalopathy, carbon monoxide poisoning, methylmalonic acidemia; hypoxic brain injury, vasculitis.
Genetic predisposition is seen in children with missense RANBP2 mutations (ANE 1). RAN binding protein 2 (RANBP2) is a key component in the nuclear pore complex (NPC) and has various functions that include intracellular signalling and mitochondrial trafficking. Mutations in this, cause dysregulation of mitochondrial function and disruption of the blood brain barrier allowing cytokines to cause injury. The presentation is with single or multiple episodes of ANEC with or without an infective trigger and may be confused with ADEM or Leigh’s disease for the uninitiated physician. Sequelae are noted in the form of dystonia, seizures and cognitive issues that tend to persist or worsen with repeated episodes. Unlike sporadic ANE, transaminitis and coagulation disturbances are usually not associated with ANE.
Mild encephalopathy with reversible splenial lesion (MERS) is the least severe form of ITES as the name suggests and is characterised by alteration in sensorium during a febrile illness for 1-2 days followed by improvement to baseline GCS without any long-term sequelae. During the acute phase, the imaging shows reversible lesion in the brain with restricted diffusion. Based on imaging findings, there are 2 types of MERS – Type 1 has only splenial involvement and Type 2 and splenial plus symmetric deep white matter lesions.
Familial or recurrent MERS is seen with Type 2 lesions and are associated with functional defects in MYRF gene. MYRF regulates oligodendrocyte differentiation and maintenance of myelin integrity. Therefore, defects result in encephalopathy with vacuolisation. The transient diffusion restriction seen in MERS is postulated to result from intramyelinic oedema.
Essential
- Onset of neurological symptoms after a febrile illness
- Neurological manifestations – altered sensorium, reduced level of consciousness, personality change >24 hours
- MRI lesion in splenium of corpus callosum with restricted diffusion
- Exclusion of other diseases – acquired demyelinating syndromes, RESLES – reversible splenial lesion syndrome seen with anti-seizure medication, hyponatremia, hypoglycemia, high altitude induced cerebral oedema, etc.
Common
- Other clinical symptoms such as seizures/hallucination
- Spontaneous recovery with a self-limited illness course
- MRI involvement of corpus callosum, periventricular white matter and other areas of the cortex
Acute shock with encephalopathy and multi-organ failure (ASEM)/Acute fulminant cerebral edema- Both these conditions represent a continuum of hyperacute conditions under the term ITES. ASES was previously known as HSES (haemorrhagic shock with encephalopathy syndrome). AESM presents with high grade fever, haemorrhage, disseminated intravascular coagulation followed by a rapid onset of encephalopathy/coma and decline in renal functions. AFCE shows similar radiological findings as ASEM but without features of haemorrhage or coagulopathy.
There are two other related conditions that are currently classified under epilepsy syndromes but possibly share similar pathophysiological mechanisms – Fever induced refractory epilepsy syndrome (FIRES) and hemiconvulsion-hemiplegia epilepsy syndrome (HHE). These usually show normal imaging in the early stages and atrophy in later stages.
Diagnosis
The diagnosis of CNS infections requires timely and appropriate samples. Classically, cerebro-spinal fluid (CSF) is collected from L1-L2 space in children and tested for biochemical, cellular and microbiological parameters using various methods such as gram staining and culture. If blockage is suspected due to lack of CSF flow during lumbar puncture and the child is less than 2 years of age with an open anterior fontanelle, samples can be collected by an expert by placing the needle directly into the lateral ventricles. Other samples that can be collected include pus for intracranial abscess or tissue from suspected infectious lesion (for eg-fungal). This requires the expertise of a neurosurgeon.
Although, CSF sample remains the 1st line diagnostic test especially in resource poor settings, the results depend on the timing and quality of the sample and the expertise of the laboratory specialist. The results are also modified by the concurrent use of anti-microbial agents making detection of pathogens difficult.
Current methods for pathogen detection have improved the yield with shorter turnaround times (TAT). These include pathogen specific PCR (eg HSV 1 and 2 PCR) as well as multi-panel PCR which can detect pathogens even when the CSF cultures are negative.
One such example was our case report on community acquired Listeria meningitis in a 4-month-old female child. Routine CSF study showed neutrophilic leucocytosis with high protein, but culture was sterile. Listeria monocytogenes was detected in the multiplex PCR panel (BIOFIRE®) within 6 hours of sample collection and the child improved on appropriate antibiotics (High dose Ampicillin and single dose of Gentamycin).
The Paediatric Infections point of care (PI-POC) trial compared the utility of several diagnostic tools that would increase the yield of pathogen detection in low resource settings (Uganda). These included novel DNA based diagnostic assay, multiplex PCR based meningoencephalitis panel, proteomics profiling from blood samples of cases and myxovirus resistance protein A (MxA) as a biomarker for viral CNS infections. This group found commercially available multiplex PCR kits to be useful in detecting bacterial pathogens at quicker turnaround time, thus helping in appropriate management and shortening duration of hospital stay.
O’Brien et al reported that the multiplex PCR panels had 93.8% sensitivity and 90% specificity as compared to microbiological culture in detecting organisms causing paediatric CNS infections with a reduction in detection time from 6 days to 2 days and duration of hospital stay from 5 days to 3 days.
Metagenomic next generation testing (mNGS) is the latest in the diagnostic armamentarium of childhood CNS infections with high throughput, sensitivity and low risk of bias. It can be done either on a whole cell DNA or a cell free DNA specimen. Studies on paediatric patients have shown the mNGS significantly increases the detection rate of pathogens that are difficult to be picked up by conventional methods such as Listeria monocytogenes, Mycobacterium tuberculosis, Nocardia etc. Cell free DNA mNGS appeared to have higher detection rates for viral pathogens and tuberculosis as compared to whole cell DNA if done 1-6 days after starting anti-microbial therapy and correlated with the clinical picture in >67.5% of the cases.
As mentioned earlier the differentiation between infective, para-infectious and autoimmune aetiologies, especially in the early stages is nearly impossible due to similarities in the clinical presentation. Opportunity to target the exact aetiology may be missed unless there are reliable clinical and diagnostic parameters to predict the evolution and manage them appropriately.
Uematsu et al looked at predictors for AES in children presenting with prolonged febrile status. They developed a simple scoring system based on laboratory parameters which included an elevated creatinine, hyperglycemia (RBS>200mg/dl) and elevated AST. Tada et al devised a similar predictive scoring system that included the GCS level at admission, duration of seizures, age of patient, mechanical ventilation and laboratory parameters (Creatinine, AST and blood glucose). A score of more than 4 was suggestive of AES.
The elevation in creatinine levels which was common in both the above studies was attributed to the increased energy consumption due to prolonged seizures with leakage of creatinine through damaged blood brain barrier. Both the studies did not report abnormal renal function in any of the patients.
Serum hyponatremia is often seen in MERS. Serum sodium levels were significantly lower in MERS patients as compared to age matched controls in a study by Takanashi et al in 2009. Whether the systemic hyponatremia directly causes or partly contributes to the intra-myelin oedema seen in MERS is still not well understood. The authors also found it difficult to differentiate MERS from hyponatremic encephalopathy.
Monitoring the level of cytokines in CSF is helpful for administering targeted treatment. Tomioka et al noted that cytokine levels peaked within 12-24 hours of onset of encephalitis symptoms and pro-inflammatory cytokines (IL-1β, IL-6) reached baseline levels by 48 hours. The levels were higher in those with Haemorrhagic shock encephalopathy syndrome (HSES) and AESRD as compared to those with febrile seizures.
Dale et al compared CSF neurotransmitters in children with ITES vs those with acute encephalitis. They measured CSF neopterin, quinolic acid, kynurenine and kynurenine/tryptophan ratio using a combination of liquid chromatography and tandem mass spectrometry. The results were available in 4 hours and an elevated CSF neopterin level significantly favoured ITES over acute encephalitis. This is particularly helpful as imaging may be normal in the early stages, especially in FIRES and initiating early treatment has shown better outcomes across several studies.
Imaging
The role of neuroimaging in paediatric CNS infections is pivotal and crucial. It helps to detect and differentiate various infections, identify complications and helps in guiding treatment. MRI brain is the preferred modality. Contrast enhanced and diffusion weighted images are used to help differentiate infective conditions from other causes such as metabolic or demyelinating conditions. MR spectroscopy is used to differentiate infective masses from tumours.
In the subcontinent, tubercular meningitis continues to be the most common CNS infection after viral encephalitis. The radiological features like the clinical findings are varied and often mimic other conditions. MRI brain with contrast is the preferred technique as non-contrast scans may miss the diagnosis in paucibacillary infection. Classically, exudates in the basal cisterns and near the optic chiasma are noted. Complications such as hydrocephalus, infarcts, vasculitis and cranial nerve involvement may be noted. Single tuberculomas are often difficult to differentiate from neuro-cysticercosis. Diffusion weighted imaging shows diffusion restriction in tuberculomas and spectroscopy may help to differentiate in the absence of scolex or other classical features – tuberculomas show lipid-lactate peaks due to areas of necrosis while neuro-cysticercosis shows amino acid peaks.
The diagnosis of ITES is based on the imaging findings in addition to the clinical features. MRI is preferred over CT brain. Diffusion weighted imaging and FLAIR sequences are key modalities in the imaging protocol.
In AESD, there is a biphasic pattern of illness which correlates with radiological evolution. The initial scans are usually normal followed by diffuse restriction of the frontal or fronto-parietal subcortical white matter with central sparing. Later scans show cerebral atrophy. Based on the imaging findings, Kamate et al suggested a change of the terminology to ALERD (Acute Leukoencephalopathy with restricted diffusion) as it depicts the condition better.
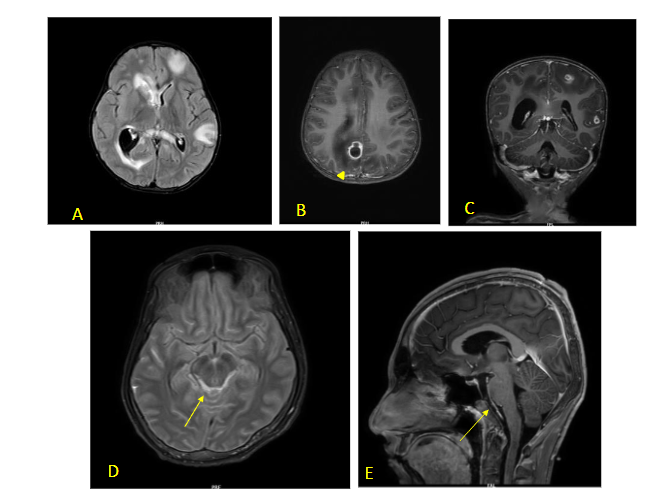
In ANEC/ANE1, there is involvement of bilateral thalami in the form of areas of diffusion restriction with haemorrhagic necrosis (‘trilaminar sign’). Involvement of midbrain, brainstem and cortex are also noted and have been associated with poorer outcomes. In milder forms of sporadic ANE, spontaneous resolution of lesions with residual hemosiderin deposits are noted with minimal sequelae.
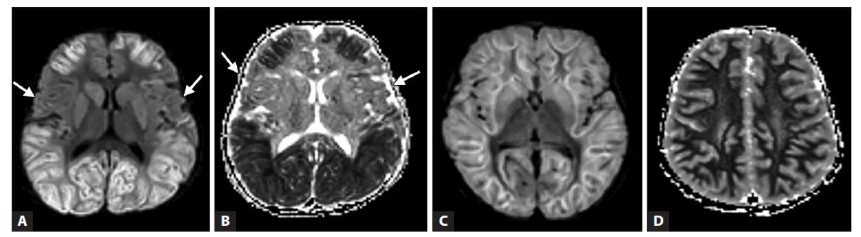
FIRES is usually associated with normal imaging at the outset. Some cases show swelling in the mesial temporal or hippocampal regions presumably from prolonged seizure activity. Late scans show diffuse cortical atrophy due to a combination of brain injury and use of prolonged steroids.
MERS is characterised by reversible splenial lesions predominantly and a milder course with minimal sequelae as compared to the other ITES. DWI sequences on MRI help to identify lesions with homogenous reduction in diffusion in the corpus callosum or at least the splenium without any contrast enhancement. Those with MERS secondary to rotavirus encephalitis may progress to develop Cerebellitis and cerebellar atrophy in later stages. Takanashi et al also reported that MERS type 2 lesions tend to resolve to MERS 1 in the initial 1-2 days before complete resolution by day 7 of illness.
AFCE and AEM/HSES represent severe end of the spectrum of ITES, characterised by life threatening hyperacute onset of cerebral oedema or multi-organ dysfunction respectively. They carry a high mortality rate of about 80%. Both the entities have similar clinical presentations and radiological findings and probably are a part of the same severe neuro-inflammatory process.
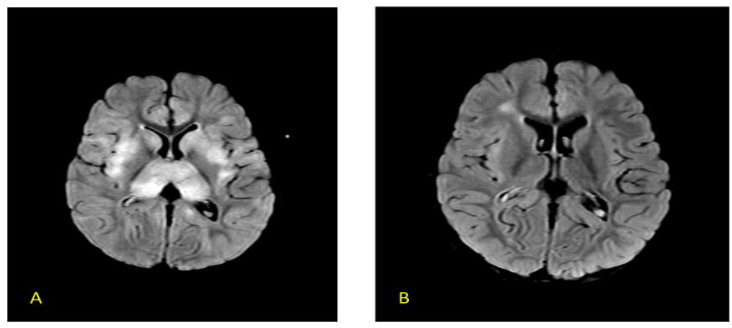
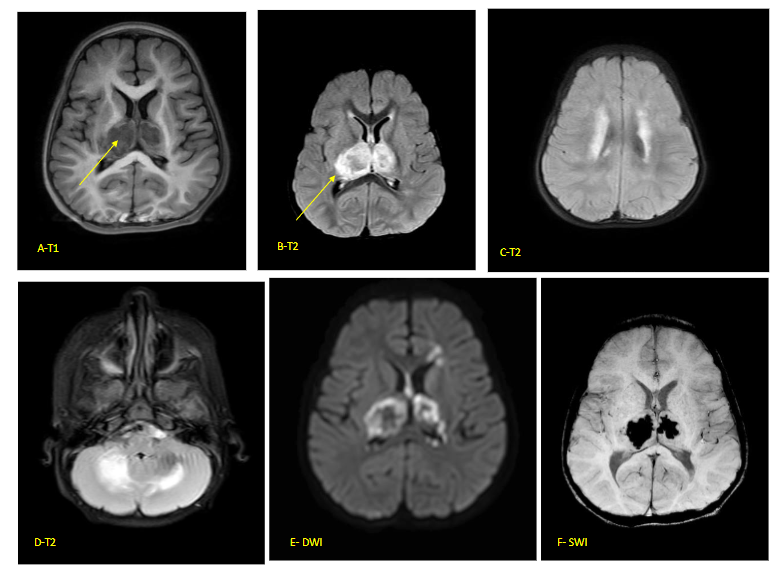
Fig 5 (A–F) – Acute necrotising encephalopathy of childhood (ANEC) in a 12-month-old child with viral gastroenteritis 2 days prior to rapid decline in sensorium. Classical ‘Trilaminar’ sign (arrow) noted in the T1 (A) and T2 (B–D) axial images in bilateral thalamic, hyperintensities also present in cerebellum and periventricular regions with restricted diffusion on DWI (E) and blooming on SWI (F) (s/o micro-hemorrhages). MR spectroscopy did not show inverted lactate doublet. FIRES is usually associated with normal imaging at the outset. Some cases show swelling in the mesial temporal or hippocampal regions presumably from prolonged seizure activity. Late scans show diffuse cortical atrophy due to a combination of brain injury and use of prolonged steroids.
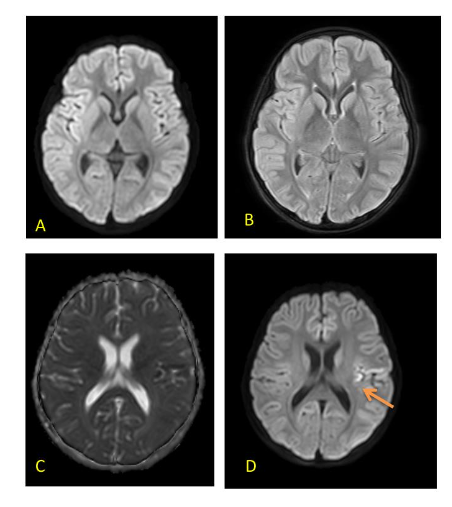
FIG 6 A–D: Fever induced refractory epilepsy syndrome (FIRES) – 8-year-old with onset of refractory seizures on Day 7 of febrile illness. Initial MRI (T2/FLAIR) images are normal (A,B). Subsequent scan done 10 days later showed diffusion restriction in left peri-Rolandic area – arrow (C,D). EEG showed persistent electrical seizures in left > right occipital regions which responded well to Midazolam and Ketamine infusion along with ketogenic diet.
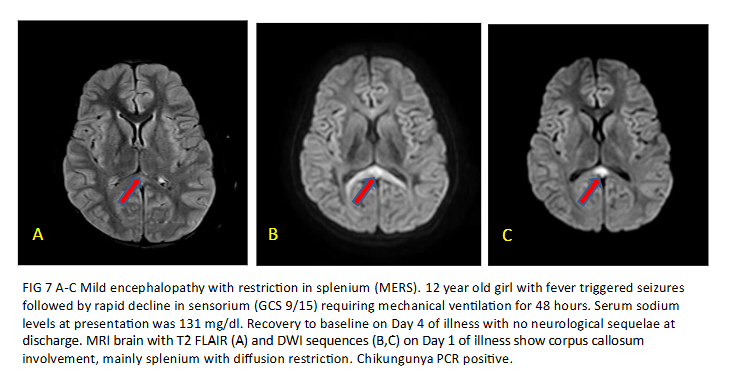
MERS is characterized by reversible splenial lesions predominantly and a milder course with minimal sequelae as compared to the other ITES. DWI sequences on MRI help to identify lesions with homogeneous reduction in diffusion in the corpus callosum or at least the splenium without any contrast enhancement. Those with MERS secondary to rotavirus encephalitis may progress to develop cerebellitis and cerebellar atrophy in later stages. Takanashi et al also reported that MERS type 2 lesions tend to resolve to MERS 1 in the initial 1–2 days before complete resolution by day 7 of illness²⁹.
AFCE and AEM/HSES represent severe end of the spectrum of ITES, characterised by life-threatening hyperacute onset of cerebral oedema or multi-organ dysfunction respectively. They carry a high mortality rate of about 80%. Both the entities have similar clinical presentations and radiological findings and probably are a part of the same severe neuro-inflammatory process.
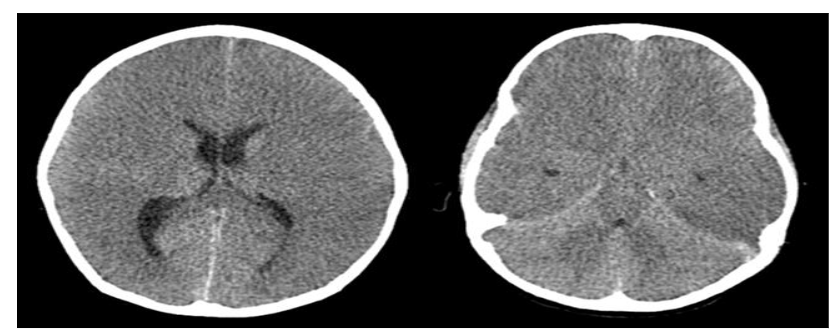 FIG 8 (A–B) Axial CT scan in a 7-year-old with AFCE showing diffuse cerebral edema (A) with ‘white’ cerebellum sign (reversal) (B) – prognosis is very poor in this case (Image courtesy – Dr Nihal Reddy, Consultant Neuro-radiologist, Hyderabad)
FIG 8 (A–B) Axial CT scan in a 7-year-old with AFCE showing diffuse cerebral edema (A) with ‘white’ cerebellum sign (reversal) (B) – prognosis is very poor in this case (Image courtesy – Dr Nihal Reddy, Consultant Neuro-radiologist, Hyderabad)
Treatment
The initial management of a child with suspected CNS infection is stabilisation. This includes taking care of airway, breathing and circulation primarily and to rule out features of raised intracranial pressure/ impending brainstem herniation as children often decompensate rapidly.
General neuroprotective measures such as maintaining normotension, normoglycemia and normothermia is essential. 20% Mannitol/3% saline is commonly used to reduce intracranial hypertension. Both have been shown to be equally efficacious in adult studies. In children, however, hypertonic saline (HTS) is preferred due to ease of administration and better safety profile.
A prospective study comparing hypertonic saline with 20% mannitol in 57 children with acute CNS infection reported better fall in intracranial pressure (ICP) and improvement in cerebral perfusion pressure (CPP) with hypertonic saline. Shein et al looked at real time data using invasive intracranial monitoring device to look at the effectiveness of various pharmacological agents to reduce raised intracranial pressure and found HTS to be most effective in acute reduction in raised ICP when compared to phenobarbitone, fentanyl and mannitol.
The use of antimicrobial treatment in paediatric CNS infections is guided by the epidemiology of pathogens in that geographical region and seasonal variation. The protocol may differ from unit to unit. In acute encephalopathy, a combination of antibiotic (usually ceftriaxone) and anti-viral (acicyclovir/oseltamivir) is used. Azithromycin or doxycycline is used in cases of atypical organisms such as rickettsia. Anti-malarial agents are used if the child is positive for P. Vivax or P. falciparum on the blood smear or antigen test.
Treatment in ITES is based on the understanding of the pattern of cytokine activation and targeted therapy is recommended.
IMMUNOMODULATORY AGENTS:
Steroids form the first line of immunomodulation in ITES due to ease in availability, administration and pricing. High dose of methylprednisolone 30mg/kg/dose or intravenous dexamethasone is given over 3-5 days. In the subcontinent, care must be taken to rule out subclinical tuberculosis prior to initiating steroids. Several studies reported better outcomes when steroids are given in the first 24 hours of presentation.
Horino et al studied the outcomes of intrathecal dexamethasone (IT-DEX) in 6 children with FIRES after the first week of illness onset. This is based on the theory that FIRES is a neuro-inflammatory condition with increased levels of pro-inflammatory cytokines in CSF than in serum. All patients could be weaned off sedation and ventilation by approximately day 5 of IT – DEX with reduced duration of ICU stay and improved seizure control.
Intravenous immunoglobulin (IvIg) is the 2nd most common agent used for immunomodulation in ITES. It suppresses cell activation and cytokine production. In ANE, a combination of steroids and IvIg has been shown to reduce mortality. The outcomes in the group treated with IvIg were better as compared to placebo. However, the efficacy of IvIg in acute encephalitis is still debatable.
Tocilizumab is an anti-interleukin 6 (IL-6) receptor monoclonal antibody that has shown promising results in children with ANE. It has also been shown to help in repairing the blood brain barrier and thereby limiting the entry of pro-inflammatory cytokines. It can be given as a single dose of 8-12 mg/kg or in 2 doses 12 hours apart. It is believed that early dosing has better outcome but there is a lack of consensus regarding the timing and dosing of Tocilizumab in ANE. The outcomes are better when Tocilizumab is used along with IvIG and intravenous steroids.
Lee et al reported the beneficial role of Tocilizumab in Rituximab-refractory autoimmune encephalitis one month after treatment with 1st line immunomodulation (steroids and IvIg) and standard Rituximab regimen (375mg/m2). The Tocilizumab group had better modified Rankin scores (mRS) compared to those treated with repeated doses of Rituximab. The delayed role of tocilizumab may be related to blocking the action of IL-6 in B and T cell differentiation and proliferation.
Anakinra is an interleukin 1 antagonist that has been tried mainly in FIRES/AERPS. In a case series of 25 patients with FIRES across an international population, early use of Anakinra (Median = 20 days) showed reduction in seizure burden, shorter duration of ventilation and ICU stay but didn’t necessarily improve the final outcome. Anakinra is started at a dose of 3-6mg/kg/day (max 10mg/kg/day) and continued for a duration of 12-24 months. Some case studies have also reported the beneficial role of Anakinra and Tocilizumab in the chronic phase of FIRES.
OTHER THERAPIES
Targeted therapeutic hypothermia (TTM) differs from head cooling that is done in neonates for hypoxic ischaemic encephalopathy (HIE) in that the whole body is moderately cooled to maintain temperature of 34.5-37-degree centigrade as compared to brain hypothermia therapy where the temperatures range from 32-34-degree centigrade.
Hypothermia has been a known therapeutic option for many years in the adult cardiac and trauma ICUs. In children, except neonates, therapeutic hypothermia is not routinely administered and there is a lack of protocol for using TTM in Paediatric ICUs.
TTM acts by lowering the body temperature, lowering the brain basal metabolic rate and therefore reducing the cytokine mediated injury. It is used as an adjunctive treatment in children with AESD or FIRES in addition to other anti-seizure medications. A combination of head cooling (BHT) in the acute phase (72 hours) followed by TTM for 96 hours showed reduction in seizure recurrence on rewarming. No neurological sequelae was detected for the child at discharge on day 28.
Other therapies – drugs such as Dextromethorphan (an anti-NMDAR antagonist) have been tried in a few cases with refractory seizures and was associated with better seizure control, mitochondrial cocktail drugs such a thiamine, biotin, CoQ10, pyridoxine, vitamin E etc may show reduced seizure occurrence in acute encephalopathy. Pyridoxine is beneficial in refractory seizures as it acts as a coenzyme to GAD (glutamic acid decarboxylase) receptors.
Early ketogenic diet and steroids have been shown to have benefit in FIRES in a review by Kessi et al. All other therapies did not show any significant values in the study.
Scope for future research
ITES and other immune mediated conditions in the paediatric age is still in its incipient stages but has tremendous potential in terms of early prevention of devastating sequelae if picked up early. Most of the breakthrough research in these conditions have come from the dedicated group of paediatric neurology teams in pockets across the globe (Japan, Australia, United Kingdom). More data is required from other high burden countries such as India to understand the effect of geographical and ethnic factors. It is also important to look into economical therapeutic options especially in developing nations where financial constraints, logistics (drug availability, healthcare access) and delays in referral play an important role in deciding treatment.
Machine learning/Artificial intelligence in medicine is picking up a lot of steam in recent years. Its use in paediatric neurology has expanded to aid in tumour diagnosis, neonatal seizure detection and in autism spectrum disorder. It could prove beneficial in collating all the clinical and imaging data in ITES to create algorithms for quicker diagnosis and appropriate management for doctors practicing in peripheral areas.
Conclusion
This short article highlights the expanding spectrum of Paediatric CNS infections and the immunological mechanisms that are at play in the causation of ITES group of disorders. Early recognition with clinical, basic laboratory and radiological parameters can help in diagnosis and appropriate management.
References
- Vries L, Volpe J. Intracranial Infections. Chapter 34-35. Volpe’s Neurology of the Newborn (Sixth Edition). Elsevier; 2018. p. 973-1090.
- Erkkinen MG, Berkowitz AL. A Clinical Approach to Diagnosing Encephalopathy. Am J Med. 2019 Oct;132(10):1142-1147. doi: 10.1016/j.amjmed.2019.07.001. Epub 2019 Jul 19. PMID: 31330129.
- Imataka G, Kuwashima S, Yoshihara S. A Comprehensive Review of Pediatric Acute Encephalopathy. J Clin Med. 2022 Oct 7;11(19):5921. doi: 10.3390/jcm11195921. PMID: 36233788; PMCID: PMC9570744.
- Rossor T, Lim M. Immune-mediated encephalitis. Dev Med Child Neurol. 2024 Mar;66(3):307-316. doi: 10.1111/dmcn.15694. Epub 2023 Jul 12. PMID: 37438863.
- Wunrow, Han Yong et al. Global, regional, and national burden of meningitis and its aetiologies, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019, The Lancet Neurology, Volume 22, Issue 8, 685 – 711.
- Bokade C, Gulhane R, Bagul A, Thakre S. Acute febrile encephalopathy in children and predictors of mortality. J Clin Diagn Res. 2014 Aug;8(8):PC09-11. doi: 10.7860/JCDR/2014/9115.4686. Epub 2014 Aug 20. PMID: 25302241; PMCID: PMC4190766.
- Damodar T, Singh B, Prabhu N, Marate S, et al. Association of Scrub Typhus in Children with Acute Encephalitis Syndrome and Meningoencephalitis, Southern India. Emerg Infect Dis. 2023 Apr;29(4):711-722. doi: 10.3201/eid2904.221157. PMID: 36957990; PMCID: PMC10045701.
- Choudhury P, Mohanty S, Reddy Y, Reddy N. A Rare Case of Listeria Meningitis with Spinal Involvement. Pediatr Infect Dis J. 2023 Dec 1;42(12):e498-e499. doi: 10.1097/INF.0000000000004100. Epub 2023 Oct 11. PMID: 37851965.
- Susanna E, Giovanni A, Alberto A, et al. Autoimmune encephalitis after herpes simplex encephalitis: A still undefined condition, Autoimmunity Reviews, Volume 21, Issue 12, 2022, 103187, ISSN 1568-9972, https://doi.org/10.1016/j.autrev.2022.103187.
- Sakuma H, Thomas T, Debinski C, et al. International consensus definitions for infection-triggered encephalopathy syndromes. Dev Med Child Neurol. 2025 Feb;67(2):195-207. doi: 10.1111/dmcn.16067. Epub 2024 Aug 14. PMID: 39143740; PMCID: PMC11695768.
- Masashi M, Takashi I, George I, et al. Guidelines for the diagnosis and treatment of acute encephalopathy in childhood, Brain and Development, Volume 43, Issue 1,2021,Pages 2-31, ISSN 0387-7604, https://doi.org/10.1016/j.braindev.2020.08.001.
- Barron S, Han V, Gupta J, et al. Dengue-Associated Acute Necrotizing Encephalopathy Is an Acute Necrotizing Encephalopathy Variant Rather than a Mimic: Evidence From a Systematic Review, Pediatric Neurology, Volume 161, 2024, Pages 208-215, ISSN 0887-994. https://doi.org/10.1016/j.pediatrneurol.2024.09.021.
- Levine JM, Ahsan N, Ho E, Santoro JD. Genetic Acute Necrotizing Encephalopathy Associated with RANBP2: Clinical and Therapeutic Implications in Pediatrics. Mult Scler Relat Disord. 2020 Aug;43:102194. doi: 10.1016/j.msard.2020.102194. Epub 2020 May 15. PMID: 32426208; PMCID: PMC7228726.
- Kurahashi, H., Azuma, Y., Masuda, A., Okuno, T., Nakahara, E., Imamura, T., et al. (2018). MYRF is associated with encephalopathy with reversible myelin vacuolization. Ann. Neurol. 83, 98–106. doi: 10.1002/ana.25125.
- Gaudenzi G, Kumbakumba E, Rasti R, et al. Point-of-Care Approaches for Meningitis Diagnosis in a Low-Resource Setting (Southwestern Uganda): Observational Cohort Study Protocol of the “PI-POC” Trial. JMIR Res Protoc. 2020 Nov 4;9(11):e21430. doi: 10.2196/21430. PMID: 33146628; PMCID: PMC7690656.
- Rasti, R., Kumbakumba, E., Nanjebe, D. et al. Clinical utility of the FilmArray® meningitis/encephalitis panel in children with suspected central nervous system infection in a low-resource setting – a prospective study in Southwestern Uganda. BMC Infect Dis 25, 396 (2025). https://doi.org/10.1186/s12879-025-10732-w.
- O’Brien, Matthew P, et al. Impact of Cerebrospinal Fluid Multiplex Assay on Diagnosis and Outcomes of Central Nervous System Infections in Children: A Before and After Cohort Study. The Pediatric Infectious Disease Journal 37(9):p 868-871, September 2018. | Doi: 10.1097/INF.0000000000001936.
- He, S., Xiong, Y., Tu, T. et al. Diagnostic performance of metagenomic next-generation sequencing for the detection of pathogens in cerebrospinal fluid in pediatric patients with central nervous system infection: a systematic review and meta-analysis. BMC Infect Dis 24, 103 (2024). https://doi.org/10.1186/s12879-024-09010-y.
- Li X, Yang L, Li D et al. Diagnosis of Neurological Infections in Pediatric Patients from Cell-Free DNA Specimens by Using Metagenomic Next-Generation Sequencing. Microbiol Spectr 11 2023: e02530-22. https://doi.org/10.1128/spectrum.02530-22.
- Yuan, L., Zhu, X.Y., Lai, L.M. et al. Clinical application and evaluation of metagenomic next-generation sequencing in pathogen detection for suspected central nervous system infections. Sci Rep 14, 16961 (2024). https://doi.org/10.1038/s41598-024-68034-1.
- Uematsu K, Matsumoto H, Zaha K, et al. Prediction and assessment of acute encephalopathy syndromes immediately after febrile status epilepticus. Brain Dev. 2023 Feb;45(2):93-101. doi: 10.1016/j.braindev.2022.10.004. Epub 2022 Oct 31. PMID: 36328834.
- Tada H, Takanashi J, Okuno H, et al. Predictive score for early diagnosis of acute encephalopathy with biphasic seizures and late reduced diffusion (AESD), Journal of the Neurological Sciences, Volume 358, Issues 1–2, 2015, Pages 62-65, ISSN 0022-510X, https://doi.org/10.1016/j.jns.2015.08.016.
- Takanashi J, Tada H, Maeda M, et al. Encephalopathy with a reversible splenial lesion is associated with hyponatremia. Brain Dev 2009;31:217–20.
- Tomioka K, Nishiyama M, Tokumoto S, et al. Time course of serum cytokine level changes within 72 h after onset in children with acute encephalopathy and febrile seizures. BMC Neurol. 2023 Jan 7;23(1):7. doi: 10.1186/s12883-022-03048-8. PMID: 36609211; PMCID: PMC9824967.
- Dale RC, Thomas T, Patel S, et al. CSF neopterin and quinolinic acid are biomarkers of neuroinflammation and neurotoxicity in FIRES and other infection-triggered encephalopathy syndromes. Ann Clin Transl Neurol. 2023 Aug;10(8):1417-1432. doi: 10.1002/acn3.51832. Epub 2023 Jun 20. PMID: 37340737; PMCID: PMC10424664.
- Triulzi, F., Doneda, C., & Parazzini, C. Neuroimaging of pediatric brain infections. Expert Review of Anti-infective Therapy. 2011; 9. https://doi.org/10.1586/eri.11.38.
- Maheswarappa RP, Agarwal C, Bansal J. Tuberculoma versus neurocysticercosis: can Magnetic resonance Spectroscopy and Diffusion Weighted Imaging solve the diagnostic conundrum? Original article. Journal of Clinical and Diagnostic Research. 2019 Jun, Vol-13(6): TC01-TC06 doi: 10.7860/JCDR/2019/41334.12957.
- Kamate M. Acute Leukoencephalopathy with Restricted Diffusion. Indian J Crit Care Med. 2018 Jul;22(7):519-523. doi: 10.4103/ijccm.IJCCM_139_18. PMID: 30111927; PMCID: PMC6069315.
- Takanashi J, Imamura A, Hayakawa F, Terada H. Differences in the time course of splenial and white matter lesions in clinically mild encephalitis/encephalopathy with a reversible splenial lesion (MERS). J Neurol Sci. 2010 May 15;292(1-2):24-7. doi: 10.1016/j.jns.2010.02.013. Epub 2010 Mar 16. PMID: 20236662.
- Rameshkumar R, Bansal A, Singhi S, et al. Randomized Clinical Trial of 20% Mannitol Versus 3% Hypertonic Saline in Children With Raised Intracranial Pressure Due to Acute CNS Infections. Pediatr Crit Care Med. 2020 Dec;21(12):1071-1080. doi: 10.1097/PCC.0000000000002557. PMID: 33003179.
- Shein SL, Ferguson NM, et al. Effectiveness of Pharmacological Therapies for Intracranial Hypertension in Children with Severe Traumatic Brain Injury–Results From an Automated Data Collection System Time-Synched to Drug Administration. Pediatr Crit Care Med. 2016 Mar;17(3):236-45. doi: 10.1097/PCC.0000000000000610. PMID: 26673840; PMCID: PMC4779724.
- Horino A, Kuki I, et al. Intrathecal dexamethasone therapy for febrile infection-related epilepsy syndrome. Ann Clin Transl Neurol. 2021 Mar;8(3):645-655. doi: 10.1002/acn3.51308. Epub 2021 Feb 5. PMID: 33547757; PMCID: PMC7951105.
- Wagner, J.N., Leibetseder, A., Troescher, A. et al. Efficacy and safety of intravenous immunoglobulins for the treatment of viral encephalitis: a systematic literature review. J Neurol 269, 712–724 (2022). https://doi.org/10.1007/s00415-021-10494-w.
- Lee WJ, Lee ST, Moon J, et al. Tocilizumab in Autoimmune Encephalitis Refractory to Rituximab: An Institutional Cohort Study. Neurotherapeutics. 2016 Oct;13(4):824-832. doi: 10.1007/s13311-016-0442-6. PMID: 27215218; PMCID: PMC5081109.
- Lai YC, Muscal E, Wells E, et al. Anakinra usage in febrile infection related epilepsy syndrome: an international cohort. Ann Clin Transl Neurol. 2020 Dec;7(12):2467-2474. doi: 10.1002/acn3.51229. Epub 2020 Dec 4. PMID: 33506622; PMCID: PMC7732241.
- Angel A S, Roshan H, Alicia G M, et al. Anakinra and tocilizumab in the chronic phase of febrile infection-related epilepsy syndrome (FIRES): Effectiveness and safety from a case-series, Seizure: European Journal of Epilepsy, Volume 100, 2022, Pages 51-55, ISSN 1059-1311. https://doi.org/10.1016/j.seizure.2022.06.012.
- Imataka G, Fujita Y, Kikuchi J, et al. Brain Hypothermia Therapy and Targeted Temperature Management for Acute Encephalopathy in Children: Status and Prospects. Journal of Clinical Medicine. 2023; 12(6):2095. https://doi.org/10.3390/jcm12062095.
- Fujita, Y.; Imataka, G.; Kikuchi, J.; Yoshihara, S. Successful mild brain hypothermia therapy followed by targeted temperature management for pediatric hemorrhagic shock and encephalopathy syndrome. Eur. Rev. Med. Pharm. Sci. 2021, 25, 3002–3006.
- Kessi M, Liu F, Zhan Y et al (2020) Efficacy of different treatment modalities for acute and chronic phases of the febrile infection-related epilepsy syndrome: a systematic review. Seizure 79:61–68.
- Gombolay GY, Gopalan N, Bernasconi A, et al. Review of Machine Learning and Artificial Intelligence (ML/AI) for the Pediatric Neurologist. Pediatr Neurol. 2023 Apr;141:42-51. doi: 10.1016/j.pediatrneurol.2023.01.004. Epub 2023 Jan 13. PMID: 36773406; PMCID: PMC10040433.
- Peng J, Kim DD, Patel JB, et al. Deep learning-based automatic tumor burden assessment of pediatric high-grade gliomas, medulloblastomas, and other leptomeningeal seeding tumors. Neuro Oncol. 2022;24(2):289–299.
- Yildiz I, Garner R, Lai M, Duncan D. Unsupervised seizure identification on EEG. Comput Methods Programs Biomed. 2022;215:106604.
- Megerian JT, Dey S, Melmed RD, et al. Evaluation of an artificial intelligence-based medical device for diagnosis of autism spectrum disorder. NPJ Digit Med. 2022;5(1):57.




