Innovative SCD Therapy for Acute Kidney Injury Treatment
Autologous Cell Processing with the Selective Cytopheretic Device for the Treatment of Acute Kidney Injury
Sadie J Jones¹, Christopher J Pino¹˒², Kimberly A Johnston¹, David Humes¹˒²*
- Innovative BioTherapies
- Division of Nephrology, Department of Internal Medicine, University of Michigan School of Medicine
OPEN ACCESS
PUBLISHED 31 August 2025
CITATION: Jones, SJ., Pino, CJ., etal.,2025. Autologous Cell Processing with the Selective Cytopheretic Device for the Treatment of Acute Kidney Injury. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6874
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6874
ISSN 2375-1924
ABSTRACT
Acute kidney injury continues to be a common and deadly condition despite medical advancements, occurring in upwards of half of all critically ill patients. Whether resulting from sepsis, the most common etiology, or from ischemic or nephrotoxic insults, acute kidney injury is associated with a systemic hyperinflammatory state that not only propagates additional kidney injury but often leads to injury of other organs as well. The Selective Cytopheretic Device is an autologous cell processing device that selectively targets and modifies the most inflammatory effector cells it is a novel approach to treating acute kidney injury by using cell-based rather than drug-based therapy with the aim of reducing inflammation, the causative factor of numerous disease states. Across multiple clinical trials, the selective cytopheretic device has demonstrated safety and efficacy as a treatment for acute kidney injury with reduced long term chronic dialysis sequelae, increased survival rates, and no device-related adverse events to date in both adult and pediatric populations. This innovative device has been granted FDA clearance under a Humanitarian Device Exemption to treat pediatric patients with sepsis associated acute kidney injury and its currently under evaluation in a pivotal trial in adults with renal injury as a step towards Pre-Market Approval. As the mechanism of action of the selective cytopheretic device lends it to be agnostic of the pathological cause, there is potential for it to be beneficial in treating other hyperinflammatory conditions as well.
Keywords
Acute Kidney Injury, Selective Cytopheretic Device, Inflammation, Autologous Cell Processing, Clinical Trials
Introduction
Acute Kidney Injury (AKI) is a common condition characterized by reduced renal function and structural abnormalities, occurring in up to 15% of all hospitalized patients and increasing to 50-60% in critically ill patients. It is a frequently-deadly complication among patients in the intensive care unit (ICU) with mortality rates of 50% or greater. The heterogeneous condition, AKI, has diverse causes including sepsis, ischemia, and nephrotoxins. Often, multiple etiologies actively contribute to the condition simultaneously. Despite the cause, there is a strong association between a dysregulated inflammatory immune state and AKI. In the case of sepsis, one of the most common causes of AKI, the response to infection activates a complex cascade of inflammatory mediators which are meant to eradicate infectious pathogens. However, this response can become dysregulated and promote tissue injury. The kidneys are especially vulnerable to this damage and are often among the first organs to be affected during sepsis. The damage typically begins with acute tubular injury, because the renal tubular epithelial cells are especially susceptible to oxidative stress and inflammation, which can progress into full-blown AKI. In contrast, with aseptic causes of AKI (ischemia, nephrotoxins, etc.) initial tubular cell injury is primarily caused through reduced perfusion or direct toxicity, rather than cytokine-mediated damage due to a dysregulated immune state. Regardless of the initial insult, injury triggers the tubule cells to release inflammatory mediators and set off an inflammatory cascade which exacerbates the initial damage. This not only amplifies the local injury but can induce a hyperinflammatory state where there previously was not one. It is thought to significantly contribute to the propagation of the systemic inflammatory response and potential progression to multi-organ dysfunction with increased morbidity, though the exact mechanism is not known. Thus, a hyperinflammatory state can both cause AKI, such as in sepsis, and AKI itself can initiate and perpetuate hyperinflammation.
The hyperinflammatory response associated with AKI plays a critical role in the severity and the progression of the condition to other organs. During a hyperinflammatory response, capillary microvasculature and leukocytes, particularly neutrophils and monocytes, enter a state of activation. This highly activated state leads to changes in expression of cytokines where proinflammatory variants become more prevalent, impairing capillary blood flow via interactions between microvasculature and leukocytes. Additionally, while in this state, degradative enzymes will be released by leukocytes infiltrating the tissues. Both processes of impaired capillary blood flow and degradative enzyme release will result in ischemic and toxic damage to the kidneys, and if this state of hyperinflammation is not resolved, the condition will progress to other organs with likely fatal outcomes. Accordingly, efforts to understand and create therapeutics that modulate this immune response are among the most promising approaches to improving clinical outcomes of AKI.
The Selective Cytopheretic Device (SCD) is an immunomodulatory device developed from a serendipitous discovery during a previous cell therapy clinical trial in ICU patients with severe AKI. The SCD houses fibers of synthetic, biocompatible, biomimetic membranes in a polycarbonate cylinder, with a surface area determined therapeutic dose ranging from 1.0-2.5 m². Treatment with the SCD is administered via incorporation of the device into an extracorporeal blood circuit (Figure 1) such as that used for hemodialysis, where the design of the cartridge utilizes low-velocity, low-shear force blood flow around the bundled fibers in conjunction with a low ionized-calcium (iCa) environment within the SCD to facilitate autologous immune cell processing. The combination of the low iCa produced via regional citrate anticoagulation (RCA) and the low-shear environment mimicking capillary beds, result in activated leukocytes binding temporarily, for up to several hours, before releasing and returning to the patient. The low iCa environment is crucial for SCD treatment as it enables the immunomodulation of the bound cells as well as the maintenance of anticoagulation within the circuit. The effectiveness and safety of SCD has been examined in multiple preclinical animal and clinical studies in various hyperinflammatory disease states, including AKI, with promising results. To date, there have been no reports of leukopenia or clinically significant hypocalcemia because of SCD treatment with RCA, suggesting that the immunomodulatory effect of SCD with RCA is not immunosuppressive and is safe when managed appropriately.
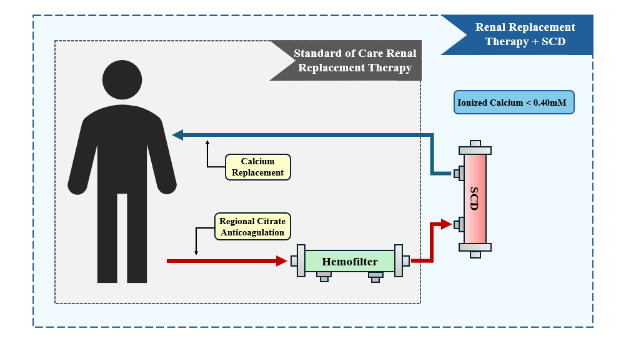
Table 1. Selective Cytopheretic Device Mortality Rates vs. Contemporaneous or Historical Controls Across Studies.
| Study | Patient Population | SCD Tx (n) | Control Tx (n) | SCD Mortality Rate | Control Mortality Rate | p value |
|---|---|---|---|---|---|---|
| Pilot (China) | Adult AKI | 9 | HC | 22.2% | 77.8%² | p = 0.027 |
| ARF-002 | Adult AKI | 35 | HC | 31.4% | 50%³ | NR |
| SCD-003 | Adult AKI | 19¹ | 27¹ | 15.8% | 40.7% | p = 0.11 |
| SCD-PED | Pediatric AKI | 22 | HC | 22.7% | 45.2%⁴ | p = 0.04 |
| SCD-005 | Adult COVID-19 AKI/ARDS | 16¹ | HC | 31.3% | 81%⁵ | p = 0.012 |
Table 1 reports a summary of mortality rates in patients receiving SCD therapy compared to contemporaneous or historical control groups from each completed clinical trial. The trend for higher survival rates in patients that received SCD treatment suggests a favorable risk-benefit relationship.
HC = historical control, NR = not reported, Tx = treatment, SCD-PED = combined data from SCD-PED 01 & 02 trials
¹ – only includes analysis of subjects treated per protocol.
² – case-matched controls based on Sequential Organ Failure Assessment scores and age.
³ – historical data²⁸,³³
⁴ – contemporaneous controls from the prospective pediatric CRRT registry⁵⁹
⁵ – contemporaneous controls from the CRRTnet registry⁸
A key area of investigation for treatment with the SCD has been AKI, with multiple clinical trials in both pediatric and adult patients with severe AKI. Initial pilot studies and clinical trials were conducted in patients with AKI before expanding to other patient populations, including those suffering from sepsis, multi-organ failure, and COVID-19.³⁶ Currently, the SCD has been approved by the Food and Drug Administration (FDA) for use in pediatric AKI patients weighing more than 10 kg under a Humanitarian Device Exemption. Additionally, a phase 3 clinical trial is currently underway to gain Pre-Market Approval for the treatment of AKI in adults (ClinicalTrials.gov Identifier: NCT05758077).⁸
The SCD is a unique approach to treat AKI via autologous modulation of the immune cells without becoming immunosuppressive (corticosteroids) or narrowly targeting a single inflammatory mediator.³⁵,³⁶
Autologous Cell Processing with the SCD for the Treatment of AKI
An advantage in such a multifaceted disease condition. The SCD works to modulate the extreme immune response following acute injury by selectively sequestering the most activated neutrophils and monocytes, while leaving the reparative population intact. Once sequestered, the activated cells are processed into a less inflammatory state before being released back into the bloodstream (Figure 2). This is believed to have a biofeedback effect which quells the negative effects of the dysregulated immune state and allows the body to achieve a naturally reparative process and support recovery³⁴,³⁷,³⁸. This report will detail how the SCD works to treat AKI, explore why an autologous cell processing device offers a better option than other narrower therapeutic targets, and provide an overview of the work done so far to bring SCD from bench to bedside to treat critically ill patients with AKI.
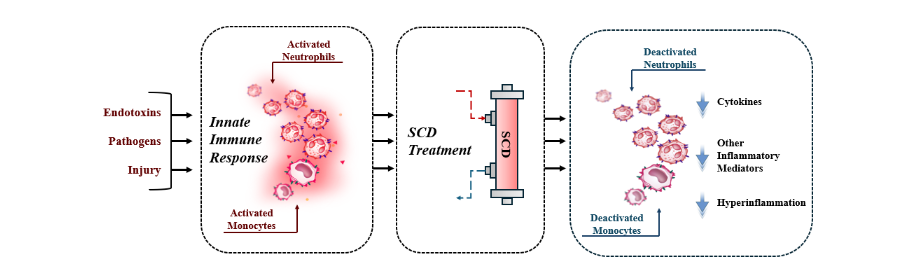
Figure 2. The SCD Mechanism of Action diagram illustrates the impact of SCD therapy on activated neutrophils and monocytes. Activated neutrophils adhere to the SCD, and there, undergo autologous processing towards a pro-apoptotic phenotype before being released back into circulation where they will be cleared via phagocytosis. Activated monocytes that adhere to the SCD shift the circulating population to favor the reparative monocyte/macrophage phenotype.
Mechanism of Action of the Selective Cytopheretic Device
The mechanism of action of SCD therapy selectively targets the most activated and inflammatory neutrophils and monocytes, but not lymphocytes, to undergo autologous processing. This selectivity is thought to occur because of the low iCa levels within the SCD and the calcium dependency of leukocyte cell surface integrin binding reactions. Thus, highly activated cells, which express more adhesion molecules and therefore have a greater capacity for adhesion, continue to bind in low iCa environments while less inflammatory cells do not bind. This ensures the immunomodulatory processes do not become immunosuppressive. This process has been shown in both preclinical and clinical applications in multiple disease states.
Previous work has demonstrated that neutrophils degranulate upon binding to the SCD membranes, supported by a release of exocytotic vesicles and reflected by an increase in carcinoembryonic antigen (CD66b), which is a component of exocytotic vesicles that move to the cell surface upon degranulation. During states of inflammation the neutrophil lifespan is elongated, due to a delay in apoptosis. Through cell processing in the SCD neutrophils that are bound, degranulated, and then released from the membranes of the SCD become apoptotic. This is reflected by a decrease in a cell surface marker (CD16), which has been shown to be associated with neutrophil apoptosis. Also, an increase in the receptor that is critical for the clearance of apoptotic neutrophils from the blood, CD184, is observed. The increase in apoptotic neutrophils released from the SCD is due to the low iCa environment within the cartridge, since a decline in calcium entry in neutrophils has been shown to promote apoptosis. After being released from the membranes back into circulation, the apoptotic neutrophils are then phagocytosed and digested by macrophages of the bone marrow, spleen and liver. This clearance of senescent neutrophils promotes the return to a balanced immune state. Elevated white blood cell count (WBC) is a hallmark of hyperinflammatory disease states. Treatment with SCD has been shown to significantly decrease the WBC in patients with multi-organ failure due to AKI, with subsequent reductions in inflammatory cytokines. A recent analysis investigated the effects of SCD treatment on neutrophil-to-lymphocyte ratios (NLR), which has been shown to correlate with mortality and disease progression across several disease states including AKI. In AKI, specifically, work has demonstrated that a high NLR is associated with worsening disease severity, progression, and clinical outcomes. The results of the analysis of 76 adult SCD-treated patients and 32 control patients demonstrated that, even though both groups had similar NLRs on day 0 of treatment (SCD=23.6 vs. control=21.7, p=0.636), a distinct divergence emerged around day 3. The SCD group had a sustained decline in NLR by around day 2 until day 6 of treatment, while the control group experienced a reversal in trend shortly after day 3 and an increase in NLR by day 6 (SCD=13.3 vs. control=25.7, p=0.002). The association between the SCD and a reduction in NLR is driven by both a reduction in absolute neutrophil count and an increase in absolute lymphocyte count. While the control group also experienced an initial decline in NLR, this is likely due to the clearance of low molecular weight inflammatory mediators by Continuous Renal Replacement Therapy (CRRT) and was short-lived as the trend reversed with an ultimate upward trend in the NLR. The reduction in absolute neutrophil count of the SCD-treated cohort supports the conclusion drawn from in-vitro studies that the SCD modifies bound and subsequently released neutrophils towards apoptosis.
Additionally, within hours of initiating SCD therapy, patients experience a reduction in vasopressor requirements, improved respiratory function, and can tolerate a greater net volume of fluid removed with CRRT. This is believed to be a result of the selective modulation and then clearance of activated neutrophils. When activated neutrophils release inflammatory mediators and other harmful constituents, these molecules interact with and damage the microvascular endothelium. This damage increases the vascular permeability and results in transudation and accumulation of fluid and inflammatory mediators within the interstitial tissues, contributing to the need for vasopressors, impaired respiratory function, and edema. Thus, the reduction of activated neutrophils in circulation via autologous modulation towards apoptosis results in less capillary leak and an improvement in the clinical effects of hyperinflammation.
As with neutrophils, SCD also selectively binds the most inflammatory subset of monocytes and transforms them into a less inflammatory state, while leaving the reparative subset less altered. This has been shown in previous work, where the bound and subsequently released monocytes have significantly higher secretion rates of proinflammatory cytokines compared to the circulating monocytes. Also, the monocytes released from the membranes were found to have a less inflammatory phenotype and a lower secretion rate of inflammatory cytokines than those bound to the SCD. The change in monocyte phenotype has also been demonstrated through a change in the cell surface markers (CD14 and CD16), which denote a circulating monocyte as either classical and intermediate (proinflammatory) or non-classical (anti-inflammatory). During AKI, circulating inflammatory monocytes will infiltrate the kidneys to become inflammatory macrophages (M1), which have a significant role in the progression of the renal injury. When treated with SCD, the proportion of circulating inflammatory monocytes is reduced and prevents them from exacerbating the renal injury. Instead, bound inflammatory monocytes are modified towards the more anti-inflammatory phenotype. This is believed to support the natural transition of the initial pro-inflammatory (M1) phenotype to the reparative phenotype (M2) and promote improved tissue repair after AKI. This transition from an inflammatory to a reparative population is often hindered by the dysregulated immune response after AKI and may not occur on its own before long-term damage has been done to the tissues. The net effect of initiating and supporting the transformation of the monocyte population to a more anti-inflammatory phenotype has been observed with SCD therapy in various hyperinflammatory disease states, including the treatment of chronically inflamed end stage renal disease patients. The shift in monocyte phenotype could explain the phenomena that in both pediatric and adult clinical trials of AKI requiring CRRT have demonstrated an improvement in renal recovery and dialysis independence of the SCD-treated cohort compared to control patients receiving CRRT alone. Changes in monocyte phenotype and activity are highly plastic and complex. As such, more evaluation is required to fully elucidate the mechanism of action of the changes occurring in monocytes during SCD therapy.
A project is currently underway to provide further insight into how the SCD modifies monocytes during treatment by utilizing single cell ribonucleic acid sequencing (RNAseq) methodology. Preliminary data has demonstrated that SCD treated monocytes have a lower expression of proinflammatory gene products as well as a downregulation of calcium dependent gene pathways (data not shown, a full manuscript is in preparation). Other groups using single cell RNAseq have also shown that citrate anticoagulation itself alters multiple genes involved in the cell cycle regulation, metabolism and cytokine signaling of monocytes. This affirms that RCA plays a crucial role in SCD therapy by not only acting as the anticoagulation within the circuit and selectively targeting the activated cells to bind, but now it seems likely to also affect the modulation of monocyte phenotype. So far, SCD therapy has mainly been targeted at treating acute conditions, like AKI, which are dominated by neutrophil driven pathologic processes. As it has been shown that the SCD modifies the monocyte population as well, with a deeper understanding of the mechanism, SCD therapy could conceivably be utilized for the treatment of both acute and chronic disease states. The SCD has the potential to transform the treatment of a diverse range of both acute and chronic conditions by utilizing a cell-directed therapy to target inflammation, the primary causative factor in so many clinical disorders.
Clinical Use of SCD for the Treatment of AKI
Promising clinical results of SCD therapy have been seen in both adult and pediatric populations. The initial pilot study to evaluate the safety and efficacy of SCD treatment for adults with AKI requiring kidney replacement therapy in the ICU was conducted in a single-center in China. This prospective study enrolled a total of 9 patients and demonstrated a promising safety profile with no device related adverse events, no reports of neutropenia, nor subsequent infections. The resulting significant decrease in mortality rates on the SCD treatment group compared to the control group (22.2% vs. 77.8%, p=0.027) was also encouraging. Following the success of the pilot study, the Acute Renal Failure-002 (ARF-002) trial was a single-arm, multicenter study in the United States designed to test the safety and efficacy of SCD in a larger population of adult patients (n=35) with AKI requiring CRRT in the ICU. Again, there were no device related adverse events, and a reduced mortality rate (31.4%) was seen compared to the upwards of 50% mortality expected from standard treatment of patients with AKI in the ICU. The success of these two pilot studies led to the development of a larger-scale, two-arm study, the SCD-003 trial.
The SCD-003 trial is among the most significant recent clinical trials, designed to compare survival and kidney outcomes in patients receiving CRRT with and without SCD therapy. Unfortunately, the trial was terminated early after enrolling 134 patients due to national shortages of calcium which created difficulties maintaining safe systemic calcium levels and appropriate iCa levels within the circuit during the RCA required for optimal SCD therapy. Consequently, iCa levels within the circuit were frequently outside of the recommended therapeutic range and as a result no difference in mortality rate was found between the SCD and control cohorts. However, post hoc analysis revealed that patients treated with the proper levels of iCa trended towards a lower mortality rate (16% vs. 41%, p=0.11) and a reduction in dialysis dependence (0% vs. 25%, p=0.10) by day 60 than the control group. These findings highlighted the importance of maintaining the proper levels of iCa within the circuit for effective SCD therapy. As a result, subsequent adult and pediatric trials have implemented strict RCA protocols, ensuring the safe and effective use of the SCD.
Following the early termination of the SCD-003 trial due to the national calcium shortage, several pediatric trials were conducted. Critically ill children in the ICU are a particularly vulnerable population, with recent data indicating that 1 in 3 will develop AKI. In children, the mortality rate of AKI remains around 50% with CRRT alone as the treatment. The SCD has been shown across multiple clinical trials to have the potential to improve outcomes in this context. The safety and feasibility in pediatric patients with AKI was SCD-PED-01, a single-arm trial. The study consisted of 16 patients who required CRRT, weighing 15 kg or more with AKI and at least one other organ dysfunction. The results were promising with 94% of the subjects surviving through the end of SCD therapy, and 75% surviving through day 60. Of those who survived, all were dialysis-independent on day 60 and 91% had completely normal kidney function. In addition to the promising survival and renal recovery rates, there were no device related adverse events. The limited sample size of the study prevented any claims of efficacy on patient outcomes, but the results did successfully establish the safety of SCD for a Humanitarian Device Exemption application. However, it is worth noting that the 75% survival rate in SCD-PED-01 compares favorably to the historical survival rate near 50% for critically ill children requiring CRRT. A subsequent single-arm trial that focused on patients weighing between 10 and 20 kg, SCD-PED-02, produced similar results to the previous trial. Of a total of 6 patients enrolled, 5 subjects survived through the end of SCD therapy and through day 60. All 5 survivors were also dialysis-independent with completely normal kidney function by day 60. Again, there were no device related adverse events.
The combined data from the two SCD-PED trials resulted in an overall 77% survival rate after receiving CRRT+SCD treatment. This was considerably higher when compared to the ICU survival rate of 51.7% seen in a comparative control cohort receiving CRRT alone found in the Prospective Pediatric CRRT registry. Additionally, there was a substantially higher proportion of patients successfully becoming dialysis-independent and recovering complete kidney function in these studies (100%) than those in the cohort found in the ppCRRT registry. Interestingly, extracorporeal membrane oxygenation (ECMO) provision was an exclusion criterion in the comparator studies in the ppCRRT, so a potentially more relevant statistic is a 94.1% survival rate (excluding five patients receiving ECMO in the CRRT-SCD cohort) compared to the 51.7% in the ppCRRT cohort. The promising results from these SCD-PED trials were sufficient for the FDA to approve the Humanitarian Device Exemption for the use of SCD to treat AKI due to sepsis or a sepsis-like condition in pediatric patients weighing more than 10 kg who require CRRT.
More recently, the results of these studies have been compared to a more contemporary registry, the Worldwide Exploration of Renal Replacement Outcomes Collaborative in Kidney Disease (WE-ROCK), as the original comparator cohort data found in ppCRRT was 20 years old. Preliminary analysis has resulted in a similar improvement of SCD patient survival rate compared to the contemporaneous control. While this more recent study is still limited by the small sample size of the SCD cohort, it is strengthened by the fact that the two cohorts received treatment in the same era, reducing the potential for temporal differences in ICU care having an impact on outcomes. Therefore, it is a reasonable conclusion that SCD therapy may be beneficial in the treatment of critically ill children with AKI and multi-organ dysfunction receiving CRRT. Regardless, larger scale pediatric studies are needed to validate these results.
One of the following trials, SCD-005, was a study designed to evaluate the use of SCD in adult patients with COVID-19, AKI, and acute respiratory distress syndrome by comparing those treated with the SCD to a contemporaneous control group found in the CRRTnet database. Results indicated patients treated with SCD for at least 4 days (per protocol) had a reduction in mortality compared to the control group (31% vs. 80%, p=0.012). Moreover, SCD treatment was associated with a significant reduction in white blood cells and a substantial decrease in levels of key cytokines, including interleukin(IL)-6, IL-2, IL-15, and IL-10, all of which are predictive of mortality in COVID-19 patients.
To validate the findings of the previous clinical trials, a current phase 3 NEUTRALIZE-AKI trial (ClinicalTrials.gov Identifier: NCT05758077) is an ongoing study designed to find Pre-Market Approval from the FDA for the use of SCD to treat adult AKI. The NEUTRALIZE-AKI trial will be the largest pivotal trial to date with a planned enrollment of 200 patients and the aims of reducing mortality and dialysis dependence of adult patients with AKI and multiple organ dysfunction requiring CRRT. Based on several lessons learned in previous clinical trials, it has been designed such that the clinical sites must strictly adhere to the protocol of keeping the circuit iCa levels within the optimal range (less than 0.4mM). Also, from previous work on the SCD-005 trial, which indicated that in severely hyperinflammatory disease states at least 4 days of SCD treatment may be necessary to see significant clinical improvement, the minimum duration of treatment for 96 hours with SCD was made a key inclusion criterion in the NEUTRALIZE-AKI trial. Additionally, there have been several indications that SCD therapy could potentially have long term effects on organ recovery, well beyond the ICU. For this reason, the primary endpoint for NEUTRALIZE-AKI is 90 days, rather than 60 days, with a composite of both mortality and dialysis dependence. The results of this study have the potential to demonstrate the effectiveness of SCD therapy to reduce mortality and dialysis dependence in critically ill adult patients with AKI and multiple organ dysfunction.
As an autologous cell processing device that acts on effector cells to be in a less inflammatory state, it shifts the treatment paradigm from drug-based therapeutics to cell-based therapy, making it broadly applicable to a diverse range of diseases by focusing on inflammation as the fundamental origin of the disorders.
Medical Devices used in the Treatment of Acute Kidney Injury
The SCD is not the only device that is placed into an extracorporeal blood circuit with the aim of treating AKI, particularly in the case of sepsis. However, it is the only device utilizing cell-based therapy which allows SCD treatment to have broad, sustained impact that is agnostic to the root cause of the condition. Others, such as the Polymyxin B (PMX) cartridge and the Seraph cartridge, focus on binding and removing specific pathogens or components of pathogens to alleviate the effects of sepsis and hopefully improve clinical outcomes. The PMX cartridge utilizes polystyrene fibers on which polymyxin B is bound to selectively remove endotoxin (lipopolysaccharide), a major component of the outer membrane of gram-negative bacteria, from the bloodstream. As blood passes through the column of the cartridge, polymyxin B binds to the lipid A portion of endotoxin, which neutralizes its toxic effects on the body. The PMX cartridge has demonstrated its potential to improve the clinical outcomes of patients afflicted with sepsis caused by gram-negative bacteria. It has been in clinical practice for over three decades outside of the United States and has been used in more than 200,000 patients, indicating its efficacy and safety. In the most recently completed multicenter, randomized control trial, EUPHRATES, it was found that when the endotoxin activity assay (EAA) score did not exceed the absorption capacity of the device the PMX treated group had a significantly reduced 28-day mortality rate than the control group (26% vs. 37%, p=0.04). However, when the EAA score did exceed the absorption capacity of the device no difference was seen in the mortality rates between the control and PMX treated subjects. This has narrowed the therapeutic target of the PMX cartridge further by suggesting that it may be specifically beneficial for patients with moderate endotoxin levels. While the PMX cartridge has had a long history of safety and efficacy, the narrow therapeutic target greatly limits how many critically ill patients suffering from sepsis can benefit from PMX therapy. Historical data indicates that only approximately 45% of sepsis infections were caused by gram-negative bacteria, highlighting how the specific pathway targeting gram-negative bacteria used by the PMX cartridge would lead it to be ineffective in many cases of sepsis.
The Seraph cartridge is composed of 300 µm micro polyethylene beads chemically bonded with heparin sulfate to bind a wide range of pathogens including viruses, bacteria, and fungi. The heparin sulfate coated beads work by mimicking the protective brush border of endothelial cells, and binding pathogens via electrostatic interactions between the negatively charged beads and the positively charged amino groups on the surface proteins of pathogens. So far, results have been promising with the filter able to achieve more than 99% reduction in most blood pathogens within one 4-hour session. The first randomized control trial, PURIFY-RCT, to evaluate the Seraph cartridge in patients with septic shock is currently underway. While no conclusions can be drawn about efficacy, previous retrospective observational studies have had promising results. One study on critically ill COVID-19 patients treated with the Seraph demonstrated a greatly reduced mortality rate between Seraph treated subjects and the matched control cohort (32% vs. 64%, p=0.001). Although the Seraph targets a broader range of pathogens than the PMX, it still is focused on targeting and removing a pathogen to improve clinical outcomes which is still limited. While the Seraph may prove to be effective at improving clinical outcomes of AKI due to sepsis or another similar pathogenic condition, it likely would not be effective in treating a substantial number of cases of AKI where pathogens may not actively be in circulation at the time of treatment or are not even the root cause. The direct comparison of SCD and other comparable devices in development highlights how innovative the SCD is as a therapeutic device. By allowing effector cells to be in a less inflammatory state, it shifts the therapeutic target from neutralizing pathogens or specific inflammatory mediators to modulating the overactivated immune response. In doing so, it also broadens the scope of conditions where SCD therapy may be effective at improving clinical outcomes.
Potential of the Selective Cytopheretic Device Beyond Acute Kidney Injury
Even though AKI has been the focus for a significant amount of the clinical evaluation of SCD therapy, multiple pilot feasibility studies of other acute dysregulated immunologic conditions have demonstrated promising safety and efficacy. These other conditions so far include acute respiratory distress syndrome (ARDS) from COVID-19, hemolytic uremic syndrome from Shiga toxin, toxic shock syndrome, hemophagocytic lymphohistiocytosis, acute-on-chronic liver failure (ACLF), and acute-on-chronic cardiac failure.
A two center, prospective, single-arm clinical trial enrolling 22 COVID-19 patients with ARDS requiring mechanical ventilation demonstrated substantial improvements with SCD treatment compared to the selected control cohort. Specifically, improvements in leukocytosis and Po2/Fio2 ratios were seen during SCD therapy that were not observed in the control group. Additionally, the mortality rate of the SCD-treated patients was reduced compared to the control group (50% vs. 81%) and was even further reduced when only comparing those that received greater than 96 hours of SCD treatment (31% vs 81%, p=0.012). While further investigation is needed, the results demonstrated safety without any device related serious adverse events and a potential to improve clinical outcomes of COVID-19 ICU patients progressing to multiple organ failure.
The SCD has also been utilized to treat two cases of severe ACLF with multiple organ failure. The first case had severe alcohol associated hepatitis and was rapidly deteriorating with more than 4 organ failures. The second case had non-alcoholic steatohepatitis and 3 organ failures. Both cases, prior to initiating SCD treatment, had systemic hyperinflammation related to their clinical decline. Treatment with the SCD in both patients was associated with rapidly reduced systemic levels of cytokines, decreased neutrophil activation, and shifted circulating monocytes to a less inflammatory state. In both cases, the patients survived either through 90 days or to successful liver transplantation. The success in these two cases demonstrated the potential of the SCD to act as a bridge between ACLF and successful liver transplantation, although further investigation is needed.
The proof of concept, first use of SCD to treat chronic systolic heart failure in humans was performed under the ongoing NEUTRALIZE-CRS (clinicaltrials.gov, NCT03836482), a multicenter single-arm trial to evaluate the safety and feasibility of the SCD as a bridge to left ventricular assist device (LVAD) implantation. The subject was a longstanding slowly progressive idiopathic cardiomyopathy, deemed ineligible for heart transplant due to age and co-morbidities. The subject was also ineligible for LVAD implantation due to age, decreased renal function, and moderate right ventricular dysfunction. Despite aggressive treatment with diuretics and inotropic agents, the lack of improvement in right atrial pressure or renal function led to his enrollment in the trial for SCD treatment as a potential bridge to LVAD implantation. During the 6 days of SCD treatment significant improvements were observed in the cardiac output and cardiac index (p=0.023), the left ventricular stroke volume index (p=0.0009), and the right ventricular stroke volume index (p=0.008) compared to values during the six days prior to SCD treatment initiation. During SCD therapy, diuretic treatment continued resulting in net fluid loss without worsening renal function. Three days after discontinuing SCD therapy, the improved cardiac parameters and stable renal function allowed him to successfully undergo LVAD implantation. The preliminary results are encouraging that the SCD may be successful as a bridge to a lifesaving procedure for refractory patients that are ineligible for heart transplantation or LVAD implantation who would otherwise have a life expectancy of days to weeks.
Currently, SCD therapy is only administered in the ICU in patients as it requires CRRT pump systems, RCA, and a central dialysis catheter. It would not be possible to administer SCD therapy in its current form in the general hospital nor in ambulatory, outpatient settings. In this regard, the need for development of a form of SCD therapy that utilizes a simple outpatient blood pump system with a peripheral inserted central catheter and short-term RCA is indicated. If successfully developed, the number of patients SCD treatment has the potential to benefit would expand substantially. In sepsis, of which clinical results to date provide a strong signal for efficacy in the ICU, earlier intervention with the SCD may prevent organ failure and transfer to the ICU altogether. Preclinical large animal data suggests that SCD could be beneficial in the treatment of type 2 diabetes mellitus as the condition develops from insulin resistance due to adipose tissue inflammation. The use of SCD therapy would be a novel intervention to reduce inflammation-induced insulin resistance and may provide a more effective and sustainable approach rather than GLP-1 agonists or glucose management with insulin. It has also been proposed that SCD therapy may be able to support and improve the treatment of certain metastatic cancers. In pancreatic ductal adenocarcinoma, specifically, surgical and chemotherapy fail to significantly impact patients with advanced stages due to the immunosuppression of T-cells by tumor associated neutrophils (TANs). Since the SCD shifts neutrophils to apoptosis, SCD therapy may reduce the number of circulating TANs towards apoptosis and allow T-cells to promote their cytotoxic effect on the tumor cells. However, much development and investigation are needed before the therapeutic application of the SCD can be broadened to such disease conditions.
Conclusions
Acute kidney injury is a common condition that continues to have elevated mortality rates despite advancements in CRRT. SCD is a novel cell-based therapy and works to modify the effector cells via autologous processing to ameliorate the dysregulated immune state without becoming immunosuppressive. So far, results of both adult and pediatric clinical trials of AKI have demonstrated the safety and efficacy of SCD therapy as a treatment for AKI. Since the SCD targets effector cells directly, the treatment is agnostic to the cause of the dysregulated immune state and has the potential to be beneficial in numerous disease states beyond AKI. However, further exploration is still needed.
Disclosure
CJP, KAJ and HDH are shareholders of SeaStar Medical, which is commercializing the SCD as Quellimmune™. SJJ, CJP, KAJ and HDH are employees of Innovative BioTherapies, which HDH owns.
Conflict of Interest Statement: None.
Funding Statement: None.
Acknowledgements: None.
References
- Hoste EA, Bagshaw SM, Bellomo R, et al. Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med. 2015;41(8):1411-1423.
- Kister TS, Remmler J, Schmidt M, et al. Acute kidney injury and its progression in hospitalized patients-Results from a retrospective multicentre cohort study with a digital decision support system. PLoS One. 2021;16(7):e0254608.
- Liano F, Junco E, Pascual J, Madero R, Verde E. The spectrum of acute renal failure in the intensive care unit compared with that seen in other settings. The Madrid Acute Renal Failure Study Group. Kidney Int Suppl. 1998;66:S16-24.
- McCarthy JT. Prognosis of patients with acute renal failure in the intensive-care unit: a tale of two eras. Mayo Clin Proc. 1996;71(2):117-126.
- Metnitz PG, Krenn CG, Steltzer H, et al. Effect of acute renal failure requiring renal replacement therapy on outcome in critically ill patients. Crit Care Med. 2002;30(9):2051-2058.
- Uchino S, Kellum JA, Bellomo R, et al. Acute renal failure in critically ill patients: a multinational, multicenter study. JAMA. 2005;294(7):813-818.
- Makris K, Spanou L. Acute Kidney Injury: Definition, Pathophysiology and Clinical Phenotypes. Clin Biochem Rev. 2016;37(2):85-98.
- Yessayan L, Humes HD, Scribe EC, Iyer SPN, Chung KK. Rationale and Design of NEUTRALIZE-AKI: A Multicenter, Randomized, Controlled, Pivotal Study to Assess the Safety and Efficacy of a Selective Cytopheretic Device in Patients with Acute Kidney Injury Requiring Continuous Kidney Replacement Therapy. Nephron. 2024;148(1):43-53.
- Deng J, Wu Z, He Y, Lin L, Tan W, Yang J. Interaction Between Intrinsic Renal Cells and Immune Cells in the Progression of Acute Kidney Injury. Front Med (Lausanne). 2022;9:954574.
- Bagshaw SM, Uchino S, Bellomo R, et al. Septic acute kidney injury in critically ill patients: clinical characteristics and outcomes. Clin J Am Soc Nephrol. 2007;2(3):431-439.
- Mehta RL, Bouchard J, Soroko SB, et al. Sepsis as a cause and consequence of acute kidney injury: Program to Improve Care in Acute Renal Disease. Intensive Care Med. 2011;37(2):241-248.
- Pais T, Jorge S, Lopes JA. Acute Kidney Injury in Sepsis. Int J Mol Sci. 2024;25(11).
- Manrique-Caballero CL, Del Rio-Pertuz G, Gomez H. Sepsis-Associated Acute Kidney Injury. Crit Care Clin. 2021;37(2):279-301.
- Kadatane SP, Satariano M, Massey M, Mongan K, Raina R. The Role of Inflammation in CKD. Cells. 2023;12(12).
- Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012;2(2):1303-1353.
- Rabb H, Griffin MD, McKay DB, et al. Inflammation in AKI: Current Understanding, Key Questions, and Knowledge Gaps. J Am Soc Nephrol. 2016;27(2):371-379.
- Hanif M, Bali A, Ramphul K. Acute Renal Tubular Necrosis. Treasure Island (FL): StatPearls; 2023.
- Akcay A, Nguyen Q, Edelstein CL. Mediators of inflammation in acute kidney injury. Mediators Inflamm. 2009;2009:137072.
- Baker ML, Cantley LG. Adding insult to injury: the spectrum of tubulointerstitial responses in acute kidney injury. J Clin Invest. 2025;135(6).
- Cantaluppi V, Quercia AD, Dellepiane S, Ferrario S, Camussi G, Biancone L. Interaction between systemic inflammation and renal tubular epithelial cells. Nephrol Dial Transplant. 2014;29(11):2004-2011.
- Armutcu F. Organ crosstalk: the potent roles of inflammation and fibrotic changes in the course of organ interactions. Inflamm Res. 2019;68(10):825-839.
- Doyle JF, Forni LG. Acute kidney injury: short-term and long-term effects. Crit Care. 2016;20(1):188.
- Fajgenbaum DC, June CH. Cytokine Storm. N Engl J Med. 2020;383(23):2255-2273.
- Himmelfarb J, McMonagle E, Freedman S, et al. Oxidative stress is increased in critically ill patients with acute renal failure. J Am Soc Nephrol. 2004;15(9):2449-2456.
- Mutunga M, Fulton B, Bullock R, et al. Circulating endothelial cells in patients with septic shock. Am J Respir Crit Care Med. 2001;163(1):195-200.
- Simmons EM, Himmelfarb J, Sezer MT, et al. Plasma cytokine levels predict mortality in patients with acute renal failure. Kidney Int. 2004;65(4):1357-1365.
- Pino CJ, Yevzlin AS, Lee K, et al. Cell-based approaches for the treatment of systemic inflammation. Nephrol Dial Transplant. 2013;28(2):296-302.
- Humes HD, Sobota JT, Ding F, Song JH, Group RADI. A selective cytopheretic inhibitory device to treat the immunological dysregulation of acute and chronic renal failure. Blood Purif. 2010;29(2):183-190.
- Pino CJ, Westover AJ, Johnston KA, Buffington DA, Humes HD. Regenerative Medicine and Immunomodulatory Therapy: Insights From the Kidney, Heart, Brain, and Lung. Kidney Int Rep. 2018;3(4):771-783.
- Yessayan L, Sohaney R, Puri V, et al. Regional citrate anticoagulation “non-shock” protocol with pre-calculated flow settings for patients with at least 6 L/hour liver citrate clearance. BMC Nephrol. 2021;22(1):244.
- Brophy PD, Somers MJ, Baum MA, et al. Multi-centre evaluation of anticoagulation in patients receiving continuous renal replacement therapy (CRRT). Nephrol Dial Transplant. 2005;20(7):1416-1421.
- Finn A, Rebuck N. Measurement of adhesion molecule expression on neutrophils and fixation. J Immunol Methods. 1994;171(2):267-270.
- Hamblin A, Taylor M, Bernhagen J, et al. A method of preparing blood leucocytes for flow cytometry which prevents upregulation of leucocyte integrins. J Immunol Methods. 1992;146(2):219-228.
- Westover AJ, Humes HD, Pino CJ. Immunomodulatory effects of a cell processing device to ameliorate dysregulated hyperinflammatory disease states. Sci Rep. 2024;14(1):12747.
- Yessayan L, Pino CJ, Humes HD. Extracorporeal therapies in sepsis: a comprehensive review of the Selective Cytopheretic Device, Polymyxin B and Seraph cartridges. Ren Fail. 2025;47(1):2459349.
- Humes HD, Goldstein SL, Yessayan LT, et al. Safety Summary of the Selective Cytopheretic Device: A Review of Safety Data Across Multiple Clinical Trials in ICU Patients With Acute Kidney Injury and Multiple Organ Failure. Crit Care Explor. 2023;5(10):e0995.
- Ding F, Yevzlin AS, Xu ZY, et al. The effects of a novel therapeutic device on acute kidney injury outcomes in the intensive care unit: a pilot study. ASAIO J. 2011;57(5):426-432.
- Yessayan LT, Neyra JA, Westover AJ, Szamosfalvi B, Humes HD. Extracorporeal Immunomodulation Treatment and Clinical Outcomes in ICU COVID-19 Patients. Crit Care Explor. 2022;4(5):e0694.
- Goldstein SL, Humes HD. Current Experience Using the Selective Cytopheretic Device for Continuous Immunomodulation in Acute Kidney Injury and Multiorgan Failure. Blood Purif. 2025.
- Humes HD, Aaronson KD, Buffington DA, et al. Translation of immunomodulatory therapy to treat chronic heart failure: Preclinical studies to first in human. PLoS One. 2023;18(4):e0273138.
- Yessayan L, Szamosfalvi B, Napolitano L, et al. Treatment of Cytokine Storm in COVID-19 Patients With Immunomodulatory Therapy. ASAIO J. 2020;66(10):1079-1083.
- Yessayan LT, Sharma P, Westover AJ, Szamosfalvi B, Humes HD. Extracorporeal Immunomodulation Therapy in Acute Chronic Liver Failure With Multiorgan Failure: First in Human Use. ASAIO J. 2024;70(3):e53-e56.
- Ribon M, Mussard J, Semerano L, Singer BB, Decker P. Extracellular Chromatin Triggers Release of Soluble CEACAM8 Upon Activation of Neutrophils. Front Immunol. 2019;10:1346.
- Zhao L, Furebring M, Xu S, Venge P. Subcellular localization and mobilization of carcinoembryonic antigen-related cell adhesion molecule 8 in human neutrophils. Br J Haematol. 2004;125(5):666-673.
- Silvestre-Roig C, Hidalgo A, Soehnlein O. Neutrophil heterogeneity: implications for homeostasis and pathogenesis. Blood. 2016;127(18):2173-2181.
- Moulding DA, Hart CA, Edwards SW. Regulation of neutrophil FcgammaRIIIb (CD16) surface expression following delayed apoptosis in response to GM-CSF and sodium butyrate. J Leukoc Biol. 1999;65(6):875-882.
- Martin C, Burdon PC, Bridger G, Gutierrez-Ramos JC, Williams TJ, Rankin SM. Chemokines acting via CXCR2 and CXCR4 control the release of neutrophils from the bone marrow and their return following senescence. Immunity. 2003;19(4):583-593.
- Ayub K, Hallett MB. Ca2+ influx shutdown during neutrophil apoptosis: importance and possible mechanism. Immunology. 2004;111(1):8-12.
- Whyte MK, Hardwick SJ, Meagher LC, Savill JS, Haslett C. Transient elevations of cytosolic free calcium retard subsequent apoptosis in neutrophils in vitro. J Clin Invest. 1993;92(1):446-455.
- Iyer SPN, Ollberding NJ, Koyner JL, Yessayan LT, Chung KK, Humes HD. The Impact of the Selective Cytopheretic Device on Neutrophil-to-Lymphocyte Ratios and Hematological Parameters in Acute Kidney Injury: A Pooled Analysis. Nephron. 2025:1-13.
- DiStasi MR, Ley K. Opening the flood-gates: how neutrophil-endothelial interactions regulate permeability. Trends Immunol. 2009;30(11):547-556.
- Brown KA, Brain SD, Pearson JD, Edgeworth JD, Lewis SM, Treacher DF. Neutrophils in development of multiple organ failure in sepsis. Lancet. 2006;368(9530):157-169.
- Kapellos TS, Bonaguro L, Gemund I, et al. Human Monocyte Subsets and Phenotypes in Major Chronic Inflammatory Diseases. Front Immunol. 2019;10:2035.
- Ozanska A, Szymczak D, Rybka J. Pattern of human monocyte subpopulations in health and disease. Scand J Immunol. 2020;92(1):e12883.
- Meng X, Jin J, Lan HY. Driving role of macrophages in transition from acute kidney injury to chronic kidney disease. Chin Med J (Engl). 2022;135(7):757-766.
- Han HI, Skvarca LB, Espiritu EB, Davidson AJ, Hukriede NA. The role of macrophages during acute kidney injury: destruction and repair. Pediatr Nephrol. 2019;34(4):561-569.
- Szamosfalvi B, Westover A, Buffington D, Yevzlin A, Humes HD. Immunomodulatory Device Promotes a Shift of Circulating Monocytes to a Less Inflammatory Phenotype in Chronic Hemodialysis Patients. ASAIO J. 2016;62(5):623-630.
- Tumlin JA, Galphin CM, Tolwani AJ, et al. A Multi-Center, Randomized, Controlled, Pivotal Study to Assess the Safety and Efficacy of a Selective Cytopheretic Device in Patients with Acute Kidney Injury. PLoS One. 2015;10(8):e0132482.
- Goldstein SL, Ollberding NJ, Askenazi DJ, et al. Selective Cytopheretic Device Use in Continuous Kidney Replacement Therapy in Children: A Cohort Study With a Historical Comparator. Kidney Medicine. 2024;6(4).
- Di Marco GS, Chasan AI, Boeckel GR, et al. Monocytes as Targets for Immunomodulation by Regional Citrate Anticoagulation. Int J Mol Sci. 2024;25(5).
- Tumlin JA, Chawla L, Tolwani AJ, et al. The effect of the selective cytopheretic device on acute kidney injury outcomes in the intensive care unit: a multicenter pilot study. Semin Dial. 2013;26(5):616-623.
- Kaddourah A, Basu RK, Bagshaw SM, Goldstein SL, Investigators A. Epidemiology of Acute Kidney Injury in Critically Ill Children and Young Adults. N Engl J Med. 2017;376(1):11-20.
- Menon S, Starr MC, Zang H, et al. Characteristics and outcomes of children ≤ 10 kg receiving continuous kidney replacement therapy: a WE-ROCK study. Pediatr Nephrol. 2025;40(1):253-264.
- Stanski NL, Gist KM, Hasson D, et al. Characteristics and Outcomes of Children and Young Adults With Sepsis Requiring Continuous Renal Replacement Therapy: A Comparative Analysis From the Worldwide Exploration of Renal Replacement Outcomes Collaborative in Kidney Disease (WE-ROCK). Crit Care Med. 2024;52(11):1686-1699.
- Goldstein SL, Askenazi DJ, Basu RK, et al. Use of the Selective Cytopheretic Device in Critically Ill Children. Kidney Int Rep. 2021;6(3):775-784.
- Goldstein SL, Somers MJ, Baum MA, et al. Pediatric patients with multi-organ dysfunction syndrome receiving continuous renal replacement therapy. Kidney Int. 2005;67(2):653-658.
- Shimizu T, Miyake T, Tani M. History and current status of polymyxin B-immobilized fiber column for treatment of severe sepsis and septic shock. Ann Gastroenterol Surg. 2017;1(2):105-113.
- Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. N Engl J Med. 2003;348(2):138-150.
- Fujimori K, Tarasawa K, Fushimi K. Effects of Polymyxin B Hemoperfusion on Septic Shock Patients Requiring Noradrenaline: Analysis of a Nationwide Administrative Database in Japan. Blood Purif. 2021;50(4-5):560-565.
- Klein DJ, Foster D, Walker PM, Bagshaw SM, Mekonnen H, Antonelli M. Polymyxin B hemoperfusion in endotoxemic septic shock patients without extreme endotoxemia: a post hoc analysis of the EUPHRATES trial. Intensive Care Med. 2018;44(12):2205-2212.
- Moreno RP, Metnitz B, Adler L, et al. Sepsis mortality prediction based on predisposition, infection and response. Intensive Care Med. 2008;34(3):496-504.
- Seffer MT, Cottam D, Forni LG, Kielstein JT. Heparin 2.0: A New Approach to the Infection Crisis. Blood Purif. 2021;50(1):28-34.
- Mattsby-Baltzer I, Bergstrom T, McCrea K, Ward R, Adolfsson L, Larm O. Affinity apheresis for treatment of bacteremia caused by Staphylococcus aureus and/or methicillin-resistant S. aureus (MRSA). J Microbiol Biotechnol. 2011;21(6):659-664.
- Chitty SA, Mobbs S, Rifkin BS, et al. A Multicenter Evaluation of the Seraph 100 Microbind Affinity Blood Filter for the Treatment of Severe COVID-19. Crit Care Explor. 2022;4(4):e0662.
- Hambrick HR, Short K, Askenazi D, et al. Hemolytic Uremic Syndrome-Induced Acute Kidney Injury Treated via Immunomodulation with the Selective Cytopheretic Device. Blood Purif. 2023;52(9-10):812-820.
- Goldstein SL, Yessayan LT, Krallman KA, et al. Use of extracorporeal immunomodulation in a toddler with hemophagocytic lymphohistiocytosis and multisystem organ failure. Pediatr Nephrol. 2023;38(3):927-931.
- Rodriguez PJ, Zhang V, Gratzl S, et al. Discontinuation and Reinitiation of Dual-Labeled GLP-1 Receptor Agonists Among US Adults With Overweight or Obesity. JAMA Netw Open. 2025;8(1):e2457349.
- Osborn O, Olefsky JM. The cellular and signaling networks linking the immune system and metabolism in disease. Nat Med. 2012;18(3):363-374.
- Jiang W, Li X, Xiang C, Zhou W. Neutrophils in pancreatic cancer: Potential therapeutic targets. Front Oncol. 2022;12:1025805.
- Pratt HG, Steinberger KJ, Mihalik NE, et al. Macrophage and Neutrophil Interactions in the Pancreatic Tumor Microenvironment Drive the Pathogenesis of Pancreatic Cancer. Cancers (Basel). 2021;14(1).
- Raskov H, Orhan A, Christensen JP, Gogenur I. Cytotoxic CD8(+) T cells in cancer and cancer immunotherapy. Br J Cancer. 2021;124(2):359-367.
- Mehta RL, Pascual MT, Soroko S, et al. Spectrum of acute renal failure in the intensive care unit: the PICARD experience. Kidney Int. 2004;66(4):1613-1621.
- Waikar SS, Curhan GC, Wald R, McCarthy EP, Chertow GM. Declining mortality in patients with acute renal failure, 1988 to 2002. J Am Soc Nephrol. 2006;17(4):1143-1150.




