International Use of BIG-IV for Infant Botulism Treatment
International Experience with Human Botulism Immune Globulin for the Treatment of Infant Botulism
Ashley P. Scarborough, MPH1; Jessica M. Khouri, MD1; Connie H. Chung, MPH1; Haydee A. Dabritz, PhD1 Jennifer S. Read, MD1
- Program, Infectious Diseases Laboratories Division, Center for Laboratory Sciences, California Department of Public Health, Richmond, California, USA
OPEN ACCESS
PUBLISHED: 31 October 2025
CITATION: Scarborough, AP, Khouri, JM, et al. 2025. International Experience with Human Botulism Immune Globulin for the Treatment of Infant Botulism. Medical Research Archives, [online] 13(10).
https://doi.org/10.18103/mra.v13i10.7026
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i10.7026
ISSN 2375-1924
Abstract
Background: Human Botulism Immune Globulin Intravenous (BIG-IV) was licensed in the United States in October 2003 for the treatment of the rare, life-threatening infectious disease, infant botulism (IB) due to botulinum toxin types A or B. International use of BIG-IV began in July 2005 on a case-by-case basis following approval by the respective countries’ medicines regulatory agencies authorizing its importation. Since 2005, BIG-IV has been distributed to 16 countries on five continents.
Aims: To review the international utilization of BIG-IV for the treatment of IB over a 20-year period, describe the mechanisms by which BIG-IV is strategically stored and deployed domestically and internationally, and compare international and domestic patients according to toxin type, demographic and clinical characteristics, transit time, time to diagnosis, time to BIG-IV infusion, and length of hospital stay (LOS).
Methods: Clinical, demographic, operational, and epidemiological data maintained in the California Department of Public Health’s Infant Botulism Treatment and Prevention Program comprehensive database were compiled and reviewed for BIG-IV-treated patients with laboratory confirmation of IB from July 1, 2005, through June 30, 2025.
Results: Of the 2,792 treated IB patients between July 1, 2005, and June 30, 2025, 113 (4%) were outside of the U.S. and 2,679 (96%) were in the U.S. Toxin types differed for international vs U.S.-treated patients by region (p<0.001), in accordance with known geographic distribution of Clostridium botulinum. Infant feeding regimens were similar, but honey exposure was significantly lower in U.S. patients compared to international patients (p<0.001). Median BIG-IV transit time was 43.2 hours for international patients compared to 14.4 hours for U.S. patients (p<0.001). International patients were also diagnosed and treated later in hospitalization than U.S. patients (p-values <0.001) and had longer hospital LOS (p<0.001).
Conclusion: Treatment of international patients was more likely to be delayed until laboratory confirmation was obtained. Longer time to BIG-IV infusion likely contributed to longer hospital LOS in international patients. Time to infusion differences could also be attributed to process delays in obtaining required approvals to import given BIG-IV is not licensed outside the U.S., transport challenges related to distance, and customs clearance delays.
Keywords
botulinum neurotoxin, botulism, human botulism immune globulin, infant botulism, orphan drug, rare diseases
Abbreviations and Acronyms:
- BAT – Botulism Antitoxin Heptavalent (A, B, C, D, E, F, G) – (Equine)
- BIG-IV – Human Botulism Immune Globulin Intravenous
- BoNT – botulinum neurotoxin
- CDPH – California Department of Public Health
- DC – distribution center
- EqBA – equine botulinum antitoxin
- Hrs – hours
- IB – infant botulism
- IBTPP – Infant Botulism Treatment and Prevention Program
- IVIG – intravenous immunoglobulins
- LOS – length of stay
- TT – transit time
- U.S. – United States
Introduction
Infant botulism (IB) is a rare, life-threatening disease that occurs when swallowed spores of the bacterium Clostridium botulinum or related species temporarily colonize an infant’s large intestine and produce botulinum neurotoxin (BoNT). The neurotoxin enters the infant’s systemic circulation and binds to cholinergic presynaptic neurons, cleaving key intracellular proteins necessary for the release of the neurotransmitter acetylcholine. Common clinical signs at presentation include constipation, difficulty feeding, ptosis, decreased facial expression, head lag, and a symmetrical descending paralysis that can affect respiratory muscles and necessitate ventilatory support. Botulism is a medical emergency; antitoxin treatment should be initiated based on clinical diagnosis without waiting for confirmatory laboratory testing.
Human Botulism Immune Globulin Intravenous (BIG-IV; BabyBIG®) was licensed to the California Department of Public Health (CDPH), a State of California Department, by the United States (U.S.) Food and Drug Administration (FDA) on October 23, 2003, for the treatment of IB caused by toxin types A or B in patients under a year of age. Prompt treatment with antitoxin results in shorter hospital stays and concomitant decreased hospital costs. The CDPH is the sponsor, sole producer and distributor of BIG-IV domestically as an orphan biologic under the U.S. federal Orphan Drug Act and California state law (California Health and Safety Code Sections 123700-123709). Production and distribution of BIG-IV is a public service (not-for-profit) activity of CDPH. Market authorization for BIG-IV has not been sought in other countries. The Infant Botulism Treatment and Prevention Program (IBTPP) under CDPH has distributed BIG-IV on a case-by-case basis internationally when an unsolicited request is made by clinicians caring for a patient with confirmed or suspected infant botulism once written approval to import has been obtained from the respective country’s medicines regulatory authority.
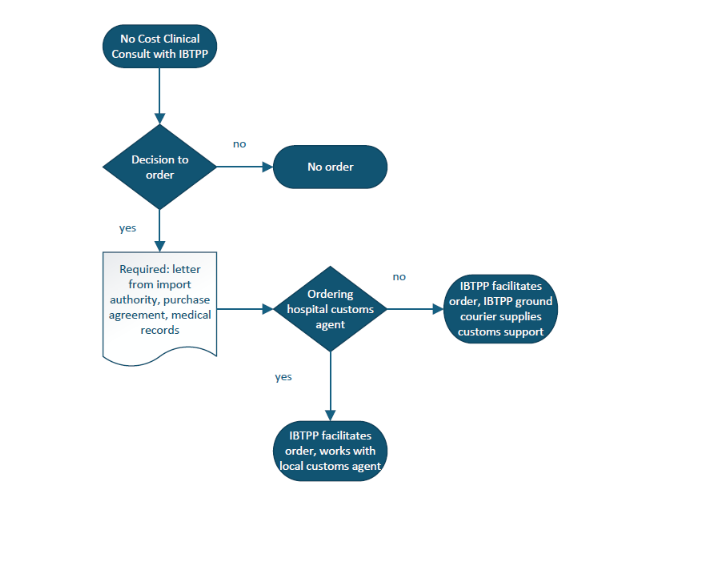
Outreach to IBTPP for clinical consultation and the decision to treat is at the determination of the physician caring for the patient.
Since U.S. licensure, BIG-IV has been distributed to treat patients in all 50 U.S. states, the District of Columbia and in 16 countries on five continents. The first international distribution occurred in July 2005. A limited-supply resource, BIG-IV, is staged at two distribution centers (DCs) in the United States and distributed from states on the East and West Coasts (North Carolina and California, respectively). Pharmacies do not store BIG-IV due to the U.S. FDA-requirements including expiry dating. Centralized inventory control avoids potential risk to product integrity. Each of the two DCs are located within 300 miles of the regions with the highest domestic distribution of BIG-IV to facilitate ease of delivery via ground transportation. Additionally, the two locations allow selection of the most expedited air transport from either DC when outside of the driving radius. Both DCs have proximate international airports. The split allows for mitigating risk to BIG-IV should a catastrophic event occur at one site. Only one lot of BIG-IV is in distribution at a time and a new lot is manufactured approximately once every five years. Therefore, supply is tightly controlled and monitored to minimize wasting medication and BIG-IV is not available for stockpiling outside of the two approved, qualified DCs.
Our objectives were to review the international utilization of BIG-IV for the treatment of IB over a 20-year period and describe the mechanisms by which BIG-IV is strategically stored and deployed internationally. In addition, we compared international and domestic patients according to toxin type, demographic and clinical characteristics, transit time, time to diagnosis, time to infusion, and length of hospital stay (LOS).
Methods
STUDY POPULATION
The study population for this analysis was all patients treated with BIG-IV from July 1, 2005, through June 30, 2025, with laboratory confirmation of IB. The study population was stratified into two groups depending on the location of the hospital where they were treated with BIG-IV (within or outside the United States). Data on U.S. and international IB cases were obtained from the IBTPP database. The IBTPP 24/7/365 no-cost clinical consultation on IB serves as a surveillance mechanism to recognize U.S. and international cases. The IBTPP maintains a detailed database of patients referred for medical consultation with the program. Data for all treated, laboratory-confirmed cases were obtained from the referring clinician and abstracted from medical records. The U.S. Code of Federal Regulations (CFR) allows for post-marketing surveillance for both effectiveness and safety. Specifically, the Health Insurance Portability and Accountability Act (HIPAA) has exemptions for both the investigation of public health activities and for the purposes of evaluation of quality, safety, or effectiveness of FDA-regulated products. The IBTPP is not a covered entity as defined by HIPAA. U.S. hospitals governed by the above generally provide more complete data than international hospitals.
STUDY DEFINITIONS
A case of IB was defined as a clinically compatible case with laboratory confirmation occurring in a child 0-17 months of age without associated risk or exposure for other types of human botulism (i.e., foodborne, wound, or iatrogenic). Laboratory confirmation was defined as detection of BoNT in a diagnostic stool sample. Text search of medical records was used to determine whether laboratory confirmation of IB was or was not completed prior to consultation with IBTPP. The date of disease onset (DOO) was defined as the date when the patient’s family first contacted a medical provider regarding clinical signs of IB or the date of initial hospital admission if the former was not available. The hospital length of stay (LOS) was the difference in days between the date of hospital discharge and the date of hospital admission for all hospitalizations associated with IB illness, excluding emergency room visits. The LOS at each hospital, if more than one, was summed. Hospital admission and discharge dates were obtained through review of medical records or verbal reports from the patient’s clinicians. Number of emergency room visits and non-consecutive hospitalizations due to IB were based on review of medical records, excluding emergency room visits resulting in hospitalization at the same facility. Clinical signs at the time of onset of illness, hospital procedures, and serious events were assessed at the time of consultation with IBTPP. The time-to-diagnosis was defined as the duration of time between the DOO and the date of referral to (medical consultation with) the IBTPP. The time to infusion was calculated as the duration of time in days between the DOO and the date of BIG-IV infusion. Additionally, transit time (TT) was approximated and defined as the time in hours between hospital order date/time and date/time of infusion. All order date/times are tracked according to the Pacific Time zone in the U.S. and infusion date/times are recorded according to the infusing hospital’s local time zone. Hospitals were grouped by their respective time zones to convert the local infusion time to Pacific Time. Daylight and standard time were not accounted for in this analysis. Therefore, the TT in hours is estimated within +/- two hours.
STATISTICAL ANALYSES
Comparison of characteristics of the study population were stratified according to whether patients were hospitalized within or outside the U.S. was performed using Microsoft Excel V2505. The following statistical tests were performed in SAS Viya v.03.05: a) the Mann-Whitney U test to compare medians of continuous variables with non-normal distributions, and b) Chi-squared or Fisher exact tests to evaluate differences in categorical variables according to whether patients were hospitalized within or outside the U.S. A p-value of less than 0.05 was considered statistically significant. Unknown values were excluded from all statistical tests.
Results
Of the 2,792 laboratory-confirmed IB patients who were treated with BIG-IV from July 1, 2005, through June 30, 2025, 113 (4%) were hospitalized outside of the United States and 2679 (96%) in the United States. The majority (77%) of international IB patients were hospitalized in Canada, Australia, the United Kingdom, and Spain. The remaining patients were hospitalized in 12 other countries.
| Toxin Type | Country | A* | B | Bivalent | E | F | Pos† | Total n (%) |
|---|---|---|---|---|---|---|---|---|
| Argentina | 1 | 1 (1) | ||||||
| Australia | 8 | 6 | 1 | 15 (13) | ||||
| Austria | 2 | 1 | 1 | 4 (4) | ||||
| Canada | 35 | 15 | 1 | 1 | 52 (46) | |||
| Denmark | 2 | 1 | 3 (3) | |||||
| France | 1 | 1 (1) | ||||||
| Germany | 1 | 1 | 2 (2) | |||||
| Greece | 1 | 1 (1) | ||||||
| Ireland | 1 | 1 | 2 | 4 (4) | ||||
| Netherlands | 1 | 1 (1) | ||||||
| Norway | 1 | 1 (1) | ||||||
| Portugal | 1 | 2 | 1 | 4 (4) | ||||
| Singapore | 2 | 2 (2) | ||||||
| Spain | 6 | 2 | 8 (7) | |||||
| United Arab Emirates | 1 | 1 | 2 (2) | |||||
| United Kingdom | 5 | 3 | 3 | 1 | 12 (11) | |||
| International Total | 61 (54) | 36 (32) | 8 (7) | 2 (2) | 1 (<1) | 5 (4) | 113 | |
| United States n (%) | 1058 (40) | 1564 (58) | 37 (1) | 2 (<1) | 16 (<1) | 2 (<1) | 2679 |
| International (n = 113, 4%) | U.S. (n = 2679, 96%) | P value†‡ |
|---|---|---|
| Toxin | <0.001** | |
| A | 61 (54) | 1058 (39) |
| B | 36 (32) | 1564 (58) |
| Bivalent | 8 (7) | 37 (1) |
| E | 2 (2) | 2 (<0.1) |
| F | 1 (1) | 16 (1) |
| Not identified | 5 (4) | 2 (<0.1) |
| Sex | 0.09 | |
| Male | 47 (42) | 1331 (50) |
| Female | 66 (58) | 1348 (50) |
| Feeding modality | 0.57 | |
| Exclusively breastfed | 59 (56) | 1616 (61) |
| Mixed feeding (breastmilk & formula) | 33 (31) | 724 (27) |
| Exclusively formula fed | 14 (13) | 321 (12) |
| Unknown | 7 | 18 |
| Honey exposure | <0.001** | |
| Yes | 17 (16) | 95 (4) |
| No | 92 (84) | 2576 (96) |
| Unknown | 4 | 8 |
| Number of emergency room visits and non-consecutive hospitalizations | <0.001** | |
| 1 | 88 (78) | 1481 (55) |
| 2 | 24 (21) | 1010 (38) |
| 3 | 1 (1) | 170 (6) |
| 4 | 0 (0) | 14 (<1) |
| 5 | 0 (0) | 4 (<1) |
| International (n = 113, 4%) | U.S. (n = 2679, 96%) | P value†‡ | |||||
|---|---|---|---|---|---|---|---|
| Clinical signs | Yes | No | Unknown | ||||
| Generalized weakness | 112 (100) | 0 (0) | 1 | 2645 (99) | 29 (1) | 5 | 0.629** |
| Decreased Head Control | 111 (100) | 0 (0) | 2 | 2624 (99) | 18 (1) | 37 | 1.000** |
| Poor Suck | 105 (99) | 1 (1) | 7 | 2586 (98) | 45 (2) | 48 | 1.000** |
| Weak cry | 104 (99) | 1 (1) | 8 | 2569 (99) | 36 (1) | 74 | 1.000** |
| Hypotonia | 110 (98) | 2 (2) | 0 | 2277 (86) | 29 (1) | 5 | <0.001 |
| Ptosis | 82 (80) | 20 (20) | 11 | 1770 (67) | 878 (33) | 31 | 0.004 |
| Decreased deep tendon reflexes | 71 (69) | 32 (31) | 10 | 1438 (61) | 904 (39) | 337 | <0.001 |
| Respiratory Difficulty | 70 (63) | 42 (38) | 1 | 980 (37) | 1686 (63) | 13 | <0.001 |
| Intubated | 58 (54) | 49 (46) | 6 | 602 (23) | 2035 (77) | 42 | <0.001 |
| Cardiorespiratory arrest | 18 (17) | 88 (83) | 7 | 203 (8) | 2392 (92) | 84 | 0.001 |
| International | United States | P-value†† | |
|---|---|---|---|
| Age (months) | 4 (0-10) | 4 (0-17) | 0.86‡‡ |
| Time to diagnosis (days) | 4 (0-32) | 2 (0-110) | <0.001 |
| Time to infusion (days) | 7 (2-36) | 3 (0-111) | <0.001 |
| Transit time (hours) | 43.2 (12.9-116.5) | 14.4 (1.4-93.1) | <0.001‡‡ |
| Time to treatment (days) | <0.001† | ||
| 0-3 | 11 (10) | 1558 (58) | |
| 4-7 | 53 (47) | 782 (29) | |
| 8-14 | 25 (22) | 273 (10) | |
| > 14 | 24 (21) | 66 (3) | |
| Length of hospital stay (days) | 22 (4-298) | 12 (1-163) | <0.001 |
The proportion of patients with preliminary positive laboratory testing at the time of clinical consultation with IBTPP differed by location. Less than one percent (26/2679) of U.S. patients had received preliminary positive laboratory results prior to IBTPP consultation vs 15% (17/113) of international patients (p < 0.001). During this time period, five patients in the U.S. were treated with both BIG-IV and Botulism Antitoxin Heptavalent (Equine) (BAT). Five type F IB patients in the United States were treated with BAT after laboratory diagnosis confirmed disease due to BoNT/F. One toddler in the U.S. was treated first with BAT and subsequently with BIG-IV after laboratory confirmation for type A infant botulism along with clinical deterioration. Two international patients were treated with BAT and subsequently treated with BIG-IV after clinical regression and/or plateau; one of these patients also received non-specific IG therapy.
There was one U.S. BIG-IV treated IB patient who experienced an out-of-hospital cardiopulmonary arrest prior to admission to the hospital who was subsequently compassionately treated with BIG-IV though the patient had experienced significant hypoxic ischemic injury and the family elected to subsequently withdraw support. There were no other deaths among U.S. or international patients included in this analysis. For those patients for whom IBTPP obtained follow up information, the vast majority of patients recovered without any known long-term sequelae.
Discussion
The vast majority of recognized IB cases occur in the U.S. The reason for this higher occurrence may be due to soil ecology or availability of certain health and public health infrastructure elements. Rapid recognition and treatment of IB depends on physician awareness and training while diagnostic testing is resource intensive and involves highly sensitive and specialized techniques.
Significantly longer time to diagnosis, BIG-IV TT, time to infusion, and longer hospital LOS were observed in internationally treated patients compared to patients treated in the U.S. In a global summary of IB cases from 2007 to 2021, the median hospital LOS for patients outside the United States was 27 days vs 12 days for U.S. patients, but included many IB patients who did not receive antitoxin. In the same study, only 25% of BIG-IV-treated international patients received BIG-IV in 0 to 3 days of hospitalization, compared to 82% of U.S. patients. More rapid administration of BIG-IV is associated with improved clinical outcomes.
Overall, shortened time to BIG-IV infusion requires prompt clinical recognition of IB and contact with IBTPP to obtain BIG-IV (time to diagnosis) and completion of the necessary steps to import BIG-IV, i.e., time to order (unmeasured) and TT. International hospitals may face barriers at any step contributing to overall longer time to infusion. The rate-limiting steps are clinical decision to treat, coordination of import authorization, availability of outbound international flights, and customs clearance.
While most international and U.S. IB patients had illness caused by either BoNT/A or /B, a smaller proportion of international patients (86%) had these toxin types versus 98% of U.S. patents. International IB patients were more likely to have illness caused by BoNT/E or /F or unidentified BoNT. International patients had fewer emergency room visits and non-consecutive hospitalizations due to IB but were more likely than U.S. patients to have severe disease (to be intubated and/or suffer cardiorespiratory arrest) by the time of IBTPP medical consultation. Since international patients had more severe illness at the time of IBTPP referral, it is possible they were admitted to the hospital immediately and may have had fewer outpatient visits for early symptoms preceding hospitalization or may have presented later in their course of illness.
Alternative therapies (i.e., equine botulinum antitoxins [EqBA], pooled human intravenous immunoglobulins [IVIGs]) are available in multiple countries. A longitudinal retrospective study in Argentina showed that prompt treatment with EqBA to be safe and effective when compared to IB patients who did not receive any treatment. A retrospective multicenter study of IB patients in Spain showed shorter duration of hospital LOS for patients treated with pooled human IVIG if treated within the first 72 hours of illness. Prospective, randomized controlled studies would be needed to determine the efficacy of pooled IVIG for the treatment of IB. Twenty-two IB patients in Italy were treated with trivalent or heptavalent antitoxin, of whom 10 were intubated and 12 were not at the time of treatment. None of the latter subsequently required intubation. Only three of the 22 (14%) had hypotension or transient rash following antitoxin administration. Six IB patients in China were treated with repeated doses of BoNT/A and /B antitoxin one to nine days after detection of BoNT in stool and still experienced relatively long hospital LOS ranging from 30 to 52 days.
The mean half-life of equine antitoxin serotypes is significantly shorter than human IVIG. Given that IB patients are colonized by C. botulinum bacteria for weeks to months, there is the potential for clinical deterioration after treatment with EqBA as has been described in adult intestinal toxemia botulism. There are also concerns for adverse events such as anaphylaxis, serum sickness, and potential long-term sensitization to equine proteins. Pooled human IVIGs will not contain anti-botulinum antibodies unless some portion of donors/population has been immunized against BoNT, or the donor pool includes individuals who have recently had botulism. Of note, immunogens against BoNT remain investigational and are not available for use in the general population.
This is the first study of its kind comparing BIG-IV-treated international patients with U.S. patients. Strengths include clinician review of medical records at the time of consultation with IBTPP using a standardized intake form, review of all records to confirm hospital LOS and number of emergency room and/or hospital visits due to IB, and follow-up with providers to determine outcome and final disposition of patients treated with BIG-IV. Limitations of the analysis include TT calculations that were limited to later IBTPP records of BIG-IV order date and time, excluding the initial 95 IB cases. Additionally, the infusion time was not available for some patients. Transit time (TT) is not the same as time to delivery. In some cases, flights to international destinations occur as infrequently as once per 24 hours. BIG-IV-qualified international shippers have validated windows of 96 hours for maintaining temperature. To account for the time between order and available flights, BIG-IV is not packed until the latest possible point to mitigate the risk of going beyond the validated window, rendering the product unusable. The proportion of patients with laboratory confirmation prior to infusion was determined by text search of the patient medical records. It is possible that some patients with laboratory confirmation prior to infusion were missed using this methodology of data ascertainment.
Conclusion
In summary, BIG-IV has been successfully deployed internationally to 16 countries on five continents. International patients were more likely to have laboratory confirmation of IB prior to the hospital’s outreach for BIG-IV. Prompt recognition and treatment is imperative to minimize complications and shorten hospital stay for IB patients. Botulinum antitoxin should be administered once the clinical diagnosis is established to maximize effectiveness. Preparation by establishing pathways to engage import authorities and customs for importing BIG-IV could assist with minimizing TT and ultimately time to infusion. For requests to import BIG-IV, possible solutions to minimize delays to delivery include utilizing the clinical consultation service at IBTPP/CDPH (available 24 hours per day, 7 days per week, 365 days per year) and expediting the customs clearance process by submitting and pre-clearing customs entry with the country’s import authority while the flight is in transit. Additionally, working with a local customs broker can help facilitate medicine import. Careful coordination among the requesting hospital, the distributor and international couriers is essential to facilitate timely administration of BIG-IV.
Conflict of Interest Statement:
The authors have no conflicts of interest to disclose.
Funding Statement:
This study was supported by the California Department of Public Health Infant Botulism Treatment and Prevention Fund.
Acknowledgements:
We thank all the patients and their families who help advance our understanding of infant botulism and its treatment. We also thank the Infant Botulism Treatment and Prevention Program clinical and regulatory team and our distribution partners who work tirelessly to ensure that the antitoxin reaches the patients who need it.
Disclaimer:
The findings and conclusions in this article are those of the authors and do not necessarily represent the views or opinions of the California Department of Public Health or the California Health and Human Services Agency.
References
- Abe Y, Negasawa T, Monma C, Oka A. Infantile botulism caused by Clostridium butyricum type E toxin. Case Report. Pediatr Neurol. 2008 2008;38(1):55-57. In File.
- Shelley EB, O’Rourke D, Grant K, et al. Infant botulism due to C. butyricum type E toxin: a novel environmental association with pet terrapins. Case Report. Epidemiol Infect. 2/2015 2014;143(3):461-469. In File. doi:10.1017/S0950268814002672
- Halpin AL, Khouri JM, Payne JR, et al. Type F infant botulism: investigation of recent clusters and overview of this exceedingly rare disease. Review. Clin Infect Dis. Dec 27 2018;66(1)(Suppl):S92-S94. doi:10.1093/cid/cix818
- Aureli P, Fenicia L, Pasolini B, Gianfranceschi M, McCroskey LM, Hatheway CL. Two cases of type E infant botulism caused by neurotoxigenic Clostridium butyricum in Italy. Case Report. J Infect Dis. 1986 1986;154(2):207-211. Not in File.
- Arnon SS. Infant botulism. Annu Rev Med. 1980 1980;31:541-560. In File.
- Long SS. Infant botulism and treatment with BIG-IV (BabyBIG). Pediatr Infect Dis J. 2007 2007;26(3):261-262. doi:10.1097/01.inf.0000256442.06231.17
- American Academy of Pediatrics. Botulism and infant botulism (Clostridium botulinum). In: Kimberlin DW, Barnett ED, Lynfield R, Sawyer MH, eds. Red Book 2021-2024 Report of the Committee on Infectious Diseases. 32nd ed. American Academy of Pediatrics; 2021:266-269:chap Clostridial infections.
- Rummel A. The long journey of botulinum neurotoxins into the synapse. Toxicon. 9/9/2015 Epub ahead of print 2015;107(Pt A):9-24. In File.
- Arnon SS, Chin J. The clinical spectrum of infant botulism. Rev Infect Dis. 1979 1979;1(4):614-621. In File.
- Arnon SS. Creation and development of the public service orphan drug Human Botulism Immune Globulin. Pediatrics. 2007 2007;119(4):785-789. In File.
- Payne JR, Khouri JM, Jewell NP, Arnon SS. Efficacy of Human Botulism Immune Globulin for the treatment of infant botulism: the first 12 years post-licensure. J Pediatr. 2018;193:172-177. doi:10.1016/j.jpeds.2017.10.035
- Arnon SS, Schechter R, Maslanka SE, Jewell NP, Hatheway CL. Human Botulism Immune Globulin for the treatment of infant botulism. N Engl J Med. 2006 2006;354(5):462-471. In File.
- Dabritz HA, Chung CH, Khouri JM, Read JS. Global occurrence of infant botulism: 2007-2021. Review. Pediatrics. 2025;155(4):e2024068791. doi:10.1542/peds.2024-068791
- Primdahl D, Derrer MA, Allen S, et al. A case of infant botulism caused by Clostridium baratii type F: evaluation and experience with administration of equine-derived heptavalent botulism antitoxin. Case Report. Arch Pediatr. 2023;8:262. doi:10.29011/2575-825X
- Crowley CH. Race for a remedy. Smithsonian. 2000 2000;31(12):38-43. In File.
- Koepke R, Sobel J, Arnon SS. Global occurrence of infant botulism, 1976-2006. Pediatrics. 2008 2008;122(1):e373-e82. In File.
- Griese SE, Kisselburgh HM, Bartenfeld MT, et al. Pediatric botulism and use of equine botulinum antitoxin in children: a systematic review. Review. Clin Infect Dis. 2017;66(Suppl 1):S17-S29. doi:10.1093/cid/cix812
- Vanella de Cuetos EE, Fernández RA, Bianco MI, et al. Equine botulinum antitoxin for the treatment of infant botulism. Clin Vaccine Immunol. 2011 2011;18(11):1845-1849. doi:10.1128/CVI.05261-11
- Palomino-Fernandez L, Villarejo-Perez A, Fernandez-Fuentes C, et al. Could intravenous immunoglobulin be an alternative therapy, for treating infant botulism in area where Human Botulism Immunoglobulin is not easily available? Our experience in Andalusia, Spain. Pediatr Infect Dis J. 2025;44(6):e193-e198. doi:10.1097/INF.0000000000004727
- Lonati D, Schicchi A, Petrolini VM, et al. Infant botulism: Pavia Poison Centre clinical experience (2009-2022). 2024:104-105.
- Wang LJ, Li KC, Qian SY, et al. Clinical characteristics and prognosis of 8 cases of severe infant botulism. [Chinese]. Zhonghua Er Ke Za Zhi. 2024;62(3):218-222. doi:10.3760/cma.j.cn112140-20230908-00169
- Yu PA, Lin NH, Mahon BE, et al. Safety and improved clinical outcomes in patients treated with new equine-derived heptavalent botulinum antitoxin. Clin Infect Dis. 2018;66(Suppl 1):S57-S64. doi:10.1093/cid/cix816
- Dabritz HA, Payne JR, Khouri JM. Duration of fecal excretion of Clostridium Botulinum and botulinum neurotoxin in patients recovering from infant botulism. Brief Report. The Journal of Pediatrics. 2023;258(113396):1-5. doi:10.1016/j.jpeds.2023
- Fagan RP, Neil KP, Sasich R, et al. Initial recovery and rebound of type F intestinal colonization botulism after administration of investigational heptavalent botulinum antitoxin. Clin Infect Dis. 2011 2011;53(9):e125-e128. In File.
- Parrera GS, Astacio H, Tunga P, Anderson DM, Hall CL, Richardson JS. Use of botulism antitoxin heptavalent (A, B, C, D, E, F, G)-(equine)(BAT) in clinical study subjects and patients: a 15-year systematic safety review. Review. Toxins (Basel). 2021;14(1):19. doi:10.3390/toxins14010019
- Richardson JS, Parrera GS, Astacio H, et al. Safety and clinical outcomes of an equine-derived heptavalent botulinum antitoxin treatment for confirmed or suspected botulism in the United States. Clin Infect Dis. 2020;70(9):1950-1957. doi:10.1093/cid/ciz515
- Rasetti-Escargueil C, Popoff MR. Antibodies and vaccines against botulinum toxins: Available measures and novel approaches. Toxins (Basel). Sep 12 2019;11(9):1-23. doi:10.3390/toxins11090528
- Rao AK, Sobel J, Chatham-Stephens K, Lúquez C. Clinical guidelines for the diagnosis and treatment of botulism, 2021. MMWR Recomm Rep. 2021;70(2):1-30. doi:10.15585/mmwr.rr7002a1

