Intramuscular Immunoglobulins for ASD and PANS Treatment
Intramuscular Immunoglobulins as a Therapeutic Modality for Neural Inflammation in patients with ASD and PANS: A Combined Case Report
OPEN ACCESS
PUBLISHED:30 September 2024
CITATION: PR, Fourie., and JC, Armstrong., 2024. Intramuscular Immunoglobulins as a Therapeutic Modality for Neural Inflammation in patients with ASD and PANS. Medical Research Archives, [online] 12(9). https://doi.org/10.18103/mra.v12i9.5984
DOI https://doi.org/10.18103/mra.v12i9.5984
ISSN 2375-1924
ABSTRACT
This report explores the increasing link between autoimmune disorders and neurodiverse conditions, particularly Autism Spectrum Disorder (ASD) and Pediatric Acute-onset Neuropsychiatric Syndrome (PANS). A small study included aims to evaluate the effectiveness of intramuscular immunoglobulins (IMIG) in reducing neuroinflammation in these conditions. Seven children with Autism Spectrum Disorder and five with Pediatric Acute-onset Neuropsychiatric Syndrome were treated with monthly intramuscular immunoglobulins injections. Parents rated improvements in concentration, communication, and behaviour for Autism Spectrum Disorder, and reduction of tics for Pediatric Acute-onset Neuropsychiatric Syndrome. Results showed notable improvements, particularly in Pediatric Acute-onset Neuropsychiatric Syndrome patients. Intramuscular immunoglobulins offer a promising, cost-effective alternative to intravenous immunoglobulins (IVIG) in managing autoimmune neuroinflammation, warranting further investigation.
Keywords
Intramuscular immunoglobulins, Autism Spectrum Disorder, Pediatric Acute-onset Neuropsychiatric Syndrome, neuroinflammation, autoimmune disorders
1. Introduction
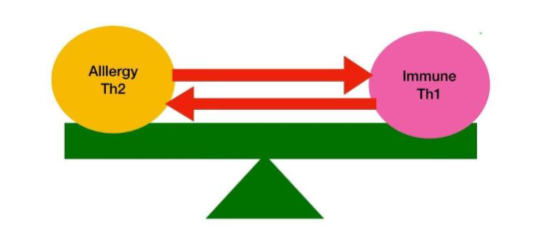
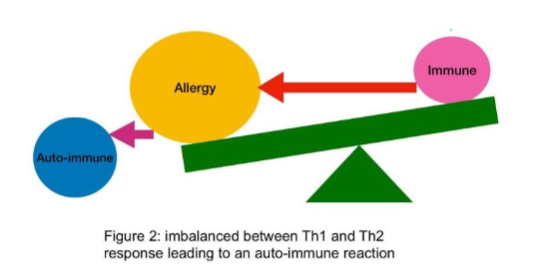
The balance between allergy related symptoms, the immune system (See figure 1.) and auto-immune diseases has been discussed extensively in numerous research publications. The hygiene theory, supported by an equal amount of information, indicates that there has been a shift away from a Th1 to a Th2 response potentially caused by the industrialisation, overuse of antibiotics and our under exposure to unrefined nutrients and soil bacteria. This is backed up by changes in the gut commensal of infants born via caesarean route. The balance between the immune system and the sensitivity to allergies depends on the exposure to specific allergens. The more allergic, the more the balance moves towards an “allergic overweight”. Finally the system moves so far towards an allergic overweight that the immune system becomes deranged and starts “attacking” the body, so called auto-immune reaction. See figure 2.
The recent increase in auto-immune diseases has been characterised by a parallel increase in the neurodiverse population with autism leading the pack and paediatric acute onset neuropsychiatric syndrome following suite. Personal observations of both these neuropathologies have been met with an upsurge that is both disturbing and challenging. Although a direct causal relationship is difficult to establish, research has demonstrated the presence of neuro-inflammation as a result of an increase in the Th2 response and a subsequent auto-immune derangement.
In order to restore the imbalance towards a Th1 response, behavioural changes should be instituted as early as possible including more natural and home births, playing outside often thereby increasing exposure to soil bacteria and increase in uncooked nutrients such as vegetables and fruit. Optimisation of the iron levels, the vitamin D status and adequate intake of essential B vitamins such as folic acid and cyanocobalamin should be included in the realignment of our immune system. In the case where a change in the allergy status (IgE increase) and/or decrease in the immune system response (decrease in IgA and IgG) can be demonstrated, the challenge will be to decrease exposure to the offending allergens (dietary and environmental avoidance) and an improvement in the immune response through natural interventions such as echinacea. If this proves to be unsuccessful, then pharmacological agents such as antihistamines, leukotriene receptor blockers and, in serious cases, pooled immunoglobulins should be considered and administered.
It becomes a challenge once the neuro-inflammation has already led to structural neural changes and apoptosis. Fortunately, the brain demonstrates neuroplasticity up to the third decade aiding in combating the neural onslaught. Therapeutic modalities such as neurofeedback have demonstrated some benefit whereas pharmacological agents such as sodium valproate, selective serotonin reuptake inhibitors and prefrontal cortex stimulants have clear benefits in improving neural conductivity.
The future star wars will be aimed at our own immune system and hopefully we will see a return to a more natural and healthy way of living. In the meantime, we will have to make use of all the means we have at our disposal including the drug armamentarium mentioned above.
A large amount of research has gone into understanding the neurobiology of both ASD and Pediatric Acute-onset Neuropsychiatric Syndrome. An underlying autoimmune derangement is clearly at play here. It has been demonstrated that in some children with autism, an abnormal gut microbiome played a role in triggering an immune response. There again, in PANS, the basic underlying inflammatory processes causing the derangement of the basal ganglia are the result of antibodies directed against the latter.
2. Material and Methods
Management for both pathologies are very similar with the aim of addressing the target symptom domains i.e. concentration, irritability, OCD, tics, mood disorders and anxiety. Psychological interventions seem to be limited and there is a move towards more pharmacological support. IVIG has recently been demonstrated to have a dramatic effect in PANS where in ASD it has not been so effective. One reason may have been the timing of the intervention, as with ASD the neural apoptosis had already taken place by the time IVIG is instituted. The rationale behind the administration of IVIG is based on the effect it has on the neuroinflammation that exists in children with either entities.
In the developing world, the exorbitant cost of IVIG excludes most patients from the benefit. Recently a program administering intramuscular immunoglobulins (IMIG) for PANS and ASD has demonstrated a surprising advantage in curtailing the severity of the ticks. Although anecdotal, it opens up a cost-effective opportunity to administer pooled immunoglobulins to a wider population. At the same time, it might also lead to the administration of immunoglobulins as an early intervention for children with profound Autism Spectrum Disorder.
This informal study was designed for the use of IMIG as an alternative to IVIG in the management of neuropsychiatric symptoms in young children with specific focus on distressing verbal and motor tics. A protocol involving the intramuscular immunoglobulins (IMIG) was developed and this was delivered on a 4 to 6 weekly basis at 0.2ml/kg body weight Intragram. (Intragam, National Bioproducts Institute, South Africa, 16%) The Intragram was injected intramuscularly to the upper lateral buttock area in an antiseptic manner. The thick viscosity of the product resulted in the need to deliver the total dose in two divided injections if the total volume exceeded 7-10ml, based on patient tolerance. Feedback was delivered by patient guardians on the day of treatment as well as regularly in between doses. General health checks were performed before and after treatment.
3. Selection criteria
In order to evaluate the response of IMIG, 7 children (ages 3 to 12, all males) in the ASD group and 5 (ages 4 to 14, three females and 2 males) were included in the study. The ASD group were specifically selected being diagnosed with level 2 ASD (non verbal) whereas in the PANS group, the criteria met the international guidelines as set out by Pfeiffer et al.
4. Results
As not all treatment started at the same time, at the time of the assessment for this report, the total doses delivered ranged from 1 to 4 given at monthly intervals. Parents were requested to gauge the response on a scale -5 to +5 in terms of the following improvements; concentration, verbal, communication and general behaviour. For children with PANS the response included the improvement of the tics.
| Group | Average Score |
|---|---|
| ASD | 2.9 |
| PANS | 4.4 |
Although a relative informal assessment, these results are significant in showing parents’ perceived improvement in their children’s symptoms after a short period of treatment. Some of the patient’s themselves noticed the improvements as well.
Side effects were minimal. As expected, due to the viscosity of the treatment, pain on injection with residual local discomfort was a common complaint. This was resolved in all patients after 12 hours. One patient experienced acute nausea and vomiting shortly after treatment, but this did not occur in any other patients. The patient also recovered rapidly and no further episodes occurred with successive doses.
5. Discussion and analysis
Except for the local discomfort, patients experience no adverse effects. Some parents actually commented on an improvement of appetite and general health which is in keeping with the expected response for intramuscular immunoglobulins.
Although this is a very limited study without statistical analysis performed, it does demonstrate an improvement of the symptomatology of both clinical groups. The results of this case report, although limited in scope, are encouraging and highlight the positive response to the IMIG, which is to be expected, given the rationale of antibodies being directed towards the basal ganglia, producing inflammation. The IMIG suppresses this derangement with subsequent improvement of the behaviour and tics. The improvement observed in both groups aligns with the underlying hypothesis that autoimmune and neuroinflammatory mechanisms play a significant role in the pathophysiology of these disorders.
For PANS, with a high average score of 4.4, the notable improvements in tic reduction and overall symptom management corroborate the theory that autoantibodies targeting the basal ganglia contribute to the neuropsychiatric symptoms, and that IMIG effectively modulates the immune response. Compared to the PANS group, the response to the IMIG in the ASD group was less dramatic, with an average score of 2.9, although most parents reported an improvement of behaviour, concentration and verbal communication. This could be due to the difference in the pathophysiology where in the ASD group, the neuro-inflammation commences at a very early age and by the time the IMIG was instituted, a significant amount of neural damage and apoptosis could have already occurred. Nevertheless, the fact that a response was detected potential points towards an ongoing auto-immune inflammation. The logical deduction implies that by administering IMIG on an ongoing basis, the neuro-inflammation is limited whilst supporting the neuroplasticity of the brain to improve cognition, verbal communication and concentration. The more modest response in ASD could be attributed to the timing of intervention, as neuroinflammatory processes in ASD likely begin much earlier in life, making early intervention crucial. Future studies might investigate whether starting IMIG therapy earlier in the course of ASD could yield more significant improvements.
When compared to the traditional IVIG treatment avenue, this inexpensive and manageable protocol demonstrated to be a strong potential and viable alternative route to IVIG, especially in cases where cost is the main barrier to treatment. Cost aside, the ease of monthly injections at a clinic or practice compared to hospitalisation and IV administration also provides significant benefits to these patients, specifically towards symptoms of anxiety. The environment is much more pleasant, manageable and acceptable to them.
While the results are promising, the limitations of this study must be acknowledged. The small sample size, lack of a control group, and reliance on subjective parental reports limit the generalizability of the findings. Further research with larger, randomised controlled trials is necessary to substantiate these preliminary results and to better understand the long-term effects and optimal dosing of IMIG in these populations.
6. Conclusion
Intramuscular immunoglobulins present a promising, cost-effective alternative to intravenous immunoglobulins for the management of neuroinflammation in patients with Autism Spectrum Disorder and Pediatric Acute-onset Neuropsychiatric Syndrome. This case report demonstrates that IMIG can lead to meaningful improvements in symptoms, particularly in PANS, where the autoimmune response is more directly targeted. While the improvements in ASD were less pronounced, the observed benefits still support the potential for IMIG to be part of a broader therapeutic strategy, especially in resource-limited settings where IVIG is not feasible. Future research should focus on expanding the sample size, incorporating objective measures of neuroinflammation, and exploring the timing and duration of IMIG therapy to maximise its efficacy. If these findings are validated in larger trials, IMIG could become a valuable tool in the treatment of autoimmune-related neuroinflammation, providing a more accessible option for patients with Autism Spectrum Disorder and Pediatric Acute-onset Neuropsychiatric Syndrome.
7. References
- Krishna MT, Subramanian A, Adderley NJ, Zemedikun DT, Gkoutos GV, Nirantharakumar K. Allergic diseases and long-term risk of autoimmune disorders: longitudinal cohort study and cluster analysis. Eur Respir J. 2019;54(5):1900476. Published 2019 Nov 14. doi:10.1183/13993003.00476-2019
- D’Auria E, Minutoli M, Colombo A, et al. Allergy and autoimmunity in children: non-mutually exclusive diseases. A narrative review. Front Pediatr. 2023;11:1239365. Published 2023 Nov 2. doi:10.3389/fped.2023.1239365
- Okada H, Kuhn C, Feillet H, Bach JF. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: an update. Clin Exp Immunol. 2010;160(1):1-9. doi:10.1111/j.1365-2249.2010.04139.x
- Zhou D, Zhang H, Bai Z, et al. Exposure to soil, house dust and decaying plants increases gut microbial diversity and decreases serum immunoglobulin E levels in BALB/c mice. Environ Microbiol. 2016;18(5):1326-1337. doi:10.1111/1462-2920.12895
- Taniya MA, Chung HJ, Al Mamun A, et al. Role of Gut Microbiome in Autism Spectrum Disorder and Its Therapeutic Regulation. Front Cell Infect Microbiol. 2022;12:915701. Published 2022 Jul 22. doi:10.3389/fcimb.2022.915701
- Dietert RR, Coleman ME, North DW, Stephenson MM. Nourishing the Human Holobiont to Reduce the Risk of Non-Communicable Diseases: A Cow’s Milk Evidence Map Example. Applied Microbiology. 2022; 2(1):25-52. https://doi.org/10.3390/applmicrobiol2010003
- Arenella M, Fanelli G, Kiemeney LA, McAlonan G, Murphy DG, Bralten J. Genetic relationship between the immune system and autism. Brain Behav Immun Health. 2023;34:100698. Published 2023 Nov 3. doi:10.1016/j.bbih.2023.100698
- Iinuma T, Okamoto Y, Morimoto Y, et al. Pathogenicity of memory Th2 cells is linked to stage of allergic rhinitis. Allergy. 2018;73(2):479-489. doi:10.1111/all.13295
- Lampiasi N, Bonaventura R, Deidda I, Zito F, Russo R. Inflammation and the Potential Implication of Macrophage-Microglia Polarisation in Human ASD: An Overview. Int J Mol Sci. 2023;24(3):2703. Published 2023 Jan 31. doi:10.3390/ijms24032703
- Kolios A, Immune balance and loss of immune tolerance, Lecture presented at: Inflammatory Brain Disorders Conference on 23 June 2024, San Diego, USA.
- Rice CE, Rosanoff M, Dawson G, et al. Evaluating Changes in the Prevalence of the Autism Spectrum Disorders (ASDs). Public Health Rev. 2012;34(2):1-22. doi:10.1007/BF03391685
- Pallanti S, Di Ponzio M. PANDAS/PANS in the COVID-19 Age: Autoimmunity and Epstein-Barr Virus Reactivation as Trigger Agents?. Children (Basel). 2023;10(4):648. Published 2023 Mar 30. doi:10.3390/children10040648
- Toscano CVA, Barros L, Lima AB, Nunes T, Carvalho HM, Gaspar JM. Neuroinflammation in autism spectrum disorders: Exercise as a “pharmacological” tool. Neurosci Biobehav Rev. 2021;129:63-74. doi:10.1016/j.neubiorev.2021.07.023
- Chiarotti F, Venerosi A. Epidemiology of Autism Spectrum Disorders: A Review of Worldwide Prevalence Estimates Since 2014. Brain Sci. 2020;10(5):274. Published 2020 May 1. doi:10.3390/brainsci10050274
- Stone R. The inflamed brain. Science. 2024;384(6697):728-733. doi:10.1126/science.adq4311
- D’Auria E, Minutoli M, Colombo A, et al. Allergy and autoimmunity in children: non-mutually exclusive diseases. A narrative review. Front Pediatr. 2023;11:1239365. Published 2023 Nov 2. doi:10.3389/fped.2023.1239365
- Wang L, Wang B, Wu C, Wang J, Sun M. Autism Spectrum Disorder: Neurodevelopmental Risk Factors, Biological Mechanism, and Precision Therapy. Int J Mol Sci. 2023;24(3):1819. Published 2023 Jan 17. doi:10.3390/ijms24031819
- Usui N, Kobayashi H, Shimada S. Neuroinflammation and Oxidative Stress in the Pathogenesis of Autism Spectrum Disorder. Int J Mol Sci. 2023;24(6):5487. Published 2023 Mar 13. doi:10.3390/ijms24065487
- Mateos-Aparicio P, Rodríguez-Moreno A. The Impact of Studying Brain Plasticity. Front Cell Neurosci. 2019;13:66. Published 2019 Feb 27. doi:10.3389/fncel.2019.00066
- Chen Z, Wang X, Zhang S, Han F. Neuroplasticity of children in autism spectrum disorder. Front Psychiatry. 2024;15:1362288. Published 2024 Apr 25. doi:10.3389/fpsyt.2024.1362288
- de Souza DLS, Costa HMGES, Idalina Neta F, et al. Brain Neuroplasticity after Treatment with Antiseizure: A Review. Clin Psychopharmacol Neurosci. 2023;21(4):665-675. doi:10.9758/cpn.23.1058
- Connery K, Tippett M, Delhey LM, et al. Intravenous immunoglobulin for the treatment of autoimmune encephalopathy in children with autism. Transl Psychiatry. 2018;8(1):148. Published 2018 Aug 10. doi:10.1038/s41398-018-0214-7
- Citrigno L, Muglia M, Qualtieri A, et al. The Mitochondrial Dysfunction Hypothesis in Autism Spectrum Disorders: Current Status and Future Perspectives. Int J Mol Sci. 2020;21(16):5785. Published 2020 Aug 12. doi:10.3390/ijms21165785
- Björkstén B, Sepp E, Julge K, Voor T, Mikelsaar M. Allergy development and the intestinal microflora during the first year of life. J Allergy Clin Immunol. 2001;108(4):516-520. doi:10.1067/mai.2001.118130
- Hardin H, Shao W, Bernstein JA. An updated review of paediatric autoimmune neuropsychiatric disorders associated with Streptococcus/paediatric acute-onset neuropsychiatric syndrome, also known as idiopathic autoimmune encephalitis: What the allergist should know. Ann Allergy Asthma Immunol. 2023;131(5):567-575. doi:10.1016/j.anai.2023.08.022
- Rossignol DA, Frye RE. A Systematic Review and Meta-Analysis of Immunoglobulin G Abnormalities and the Therapeutic Use of Intravenous Immunoglobulins (IVIG) in Autism Spectrum Disorder. J Pers Med. 2021;11(6):488. Published 2021 May 30. doi:10.3390/jpm11060488