Isoniazid Mono-resistant TB: Treatment Outcomes in SA
Time to Sputum Culture Conversion and the Intensive Phase Treatment Outcomes in South African Patients with Isoniazid Mono-resistant Tuberculosis
Osaretin Christabel Okonji 1, Pierre Mugabo 1, Osaro Mgbere 2,3,4,5
- School of Pharmacy, Faculty of Natural Sciences, University of the Western Cape, Private Bag X17, Bellville, 7535, South Africa.
- Department of Pharmaceutical Health Outcomes and Policy, College of Pharmacy, University of Houston, Houston, TX, USA
- Department of Health Systems and Population Health Sciences, Tilman J. Fertitta Family College of Medicine, University of Houston, Houston, TX, USA
- Institute of Community Health, University of Houston, Houston, TX, USA
- Public Health Science and Surveillance Division, Houston Health Department, Houston, TX, USA
ORCID: Osaretin Christabel Okonji – https://orcid.org/0000-0002-1511-2094 Pierre Mugabo – https://orcid.org/0000-0001-5653-0551 Osaro Mgbere – https://orcid.org/0000-0002-2863-6284
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Okonji, OC., Mugabo, P., Mgbere, O., 2026. Time to Sputum Culture Conversion and the Intensive Phase Treatment Outcomes in South African Patients with Isoniazid Mono-resistant Tuberculosis. Medical Research Archives, [online] 14(1).
DOI: https://doi.org/10.18103/mra.v14i1.7138
ISSN 2375-1924
ABSTRACT
Background: Isoniazid mono-resistant tuberculosis defined by resistance to isoniazid while remaining susceptible to other first-line drugs, presents significant treatment challenges. This study aimed to determine the time to sputum culture conversion and assess the intensive phase treatment outcomes among Isoniazid mono-resistant tuberculosis patients in South Africa.
Methods: This retrospective cohort study analysed 145 Isoniazid mono-resistant tuberculosis patients aged 18–65 years treated at Brewelskoof Hospital, Western Cape, South Africa, from 2009 to 2015. Demographic and clinical characteristics were obtained from the hospital’s tuberculosis registry. Data analyses used descriptive statistics, chi-square, analysis of variance, Kaplan–Meier survival, and Cox proportional hazards models to assess the time to sputum culture conversion and the intensive phase treatment outcomes, adjusting for relevant covariates. All statistical analyses were conducted using SAS 9.4 (SAS Institute, Cary, NC, USA).
Results: The median age of Isoniazid mono-resistant tuberculosis patients was 37 years, with an interquartile range (IQR) of 30–44, with 76 (52.41%) being males. After six months of treatment, 71 patients (48.97%) achieved sputum culture conversion, while 74 (51.03%) did not. The overall median time to sputum culture conversion was 55 days (IQR: 25–91), with significant variation across age groups (p=0.0074). The shortest time to sputum culture conversion was observed among patients aged 50–59 years (33.71±7.89 days). Overall, most Isoniazid mono-resistant tuberculosis patients (72.41%, 95%CI: 64.63–79.04) experienced unfavourable intensive phase treatment outcomes. Patients with unfavourable outcomes were 79% more likely to have delayed sputum culture conversion compared to those with favourable outcomes (HRR: 1.79, 95% CI: 1.07–2.98, p=0.0264). Conversely, patients who were 50-59 years were 4 times at risk of early conversion compared to individuals <20 years of age (HRR: 4.19, 95% CI: 1.13–15.61, p=0.0327).
Conclusion: Isoniazid mono-resistant tuberculosis patients in this cohort experienced prolonged time to sputum culture conversion and a high proportion of unfavourable intensive phase treatment outcomes. Delayed sputum culture conversion was strongly associated with poor treatment outcomes, highlighting the critical role isoniazid plays in drug-resistant tuberculosis therapy. Consequently, there is need for intensified monitoring and targeted interventions to improve early culture conversion and treatment success during the intensive phase treatment.
Keywords
Isoniazid mono-resistant tuberculosis, time to sputum culture conversion, intensive phase treatment outcome, South Africa
INTRODUCTION
Isoniazid (INH) is a bactericidal first-line drug essential for the treatment of tuberculosis (TB). Isoniazid mono-resistant tuberculosis (IMR-TB) is a condition in which Mycobacterium. tuberculosis is resistant to isoniazid and sensitive to rifampicin regardless of other first-line resistance. The prevalence of IMR-TB varies between 4–12%, with a global prevalence of 8.1%. In 2012–2014, the overall prevalence of IMR-TB in the Province of Western Cape, South Africa was 7.3%. Although earlier studies reported that INH resistance was not linked to poorer outcomes, more recent evidence indicates that patients with IMR-TB experience worse outcomes compared with those who have drug-susceptible tuberculosis (DS-TB). This highlights the critical importance of early detection and proper management of IMR-TB to improve patient prognosis.
Despite the progress made in TB diagnostics, particularly with automated molecular tests, the evaluation of treatment response in both drug-susceptible TB (DS-TB) and DR-TB continues to rely on microbiological methods such as sputum smear examination and sputum culture conversion. Sputum culture conversion (SCC), defined as “two successive negative culture results obtained at least one month apart” plays a crucial role in monitoring treatment response, forecasting regimen effectiveness, identifying therapeutic limitations, and determining the appropriate treatment duration and outcomes for patients with DR-TB. Nevertheless, the success of DR-TB treatment is influenced by a multitude of factors, including co-infection with HIV, the presence of multiple drug resistance, and the type of treatment regimen.
Isoniazid mono-resistant tuberculosis serves as a precursor to further resistance and is linked with the development of MDR-TB. Isoniazid mono-resistant tuberculosis, in instances of non-identification or improper treatment, may pose the risk for progression to MDR-TB and poor treatment outcomes. Early diagnosis, timely intervention, and efficacious strategies to curtail transmission becomes paramount in tackling the challenges presented by IMR-TB.
There is limited literature on the intensive phase treatment (IPT) outcomes for individuals with IMR-TB, with reported outcomes varying across different geographical regions and settings. Retrospective studies conducted in Southern Mexico and South Africa have reported an elevated risk of treatment failure associated with IMR-TB in comparison to DS-TB. Two recent systematic reviews that have evaluated treatment options for IMR-TB concluded that treatment relying on first-line drugs resulted in suboptimal outcomes and extending the duration of rifampicin use as well as increasing the number of effective drugs reduced the likelihood of unfavourable outcomes.
There are limited studies that have explored the time to sputum culture conversion (TSCC) in IMR-TB patients in South Africa and the African regions. Therefore, this study aimed to generate empirical evidence regarding the TSCC and IPT outcomes among adult patients diagnosed with IMR-TB in South Africa between 2009-2015.
METHODS
STUDY SETTING, DESIGN AND PATIENTS
The data for this study were collected at Brewelskloof Hospital, situated in the Worcester/Robertson health district of the Cape Winelands, South Africa. Brewelskloof Hospital serves as a specialized healthcare facility, providing comprehensive diagnostic and therapeutic services to patients diagnosed with DR-TB. The dataset was sourced from the hospital’s DR-TB outpatient clinic database, covering patient records spanning from January 1, 2009, to January 31, 2015. The hospital caters for a diverse demographic, including individuals from both urban and rural communities. This was a retrospective study of adult male and female patients who were admitted for DR-TB treatment during the study period, including some who were co-infected with HIV.
DATA SOURCE AND NEED FOR BASELINE EVIDENCE
This dataset was selected because, despite the ten-year gap since the last observation year, there remains limited information on intensive-phase treatment outcomes among IMR-TB patients in South Africa. There is inadequate research evaluating patient outcomes under the previous treatment protocols. Therefore, this dataset provides essential baseline evidence for understanding trends prior to the updated guidelines and helps address critical gaps in the literature regarding earlier treatment strategies, while also informing future programmatic and clinical approaches.
This study therefore, provides an important historical benchmark by re-examining clinical outcomes from a decade ago, at a time when treatment for IMR-TB was less standardized. Since then, both global and national policies have shifted: for example, the South African Standard Treatment Guidelines (2024) now recommend a six-month RHZE (Rifampicin, Isoniazid, Pyrazinamide, and Ethambutol) regimen plus daily Levofloxacin for IMR-TB. Meanwhile, the World Health Organization’s 2018 guidelines on the treatment of IMR-TB recommends a six-month RHZE regimen plus daily Levofloxacin for IMR-TB. By comparing earlier treatment regimens’ safety, efficacy, and tolerability to current best practices, this study fills a gap in knowledge and helps assess whether past practices continue to inform, validate, or challenge today’s policy-driven approaches. Ultimately, the findings can help guide future treatment strategies and contribute to policy-making in resource-limited settings.
STUDY POPULATION
The study population includes male and female patients, both HIV-positive and HIV-negative, 18–65 years old, with confirmed IMR-TB who commenced and completed treatment between January 1, 2009, and January 31, 2015. The study population is marked by a significant burden of coexisting conditions, most notably the high prevalence of HIV co-infection in the region.
INCLUSION AND EXCLUSION CRITERIA
The inclusion criteria for the study were as follows: 18–65 years, with a laboratory-confirmed diagnosis of IMR-TB, who have documented IPT treatment outcomes for at least six months and had baseline drug sensitivity test (DST) results as well as monthly culture results for a duration of six months.
Patients were excluded from the study if they were younger than 18 years or older than 65 years, diagnosed with pre-extensively drug-resistant tuberculosis (XDR-TB), other forms of mono-resistant TB, MDR-TB, or XDR-TB, lacked a definitive interim treatment outcome at six months, had no confirmed evidence of IMR-TB, or lacked baseline drug sensitivity test results.
SAMPLE SIZE CALCULATION
From a total sampling frame of 236 patients, 145 met the predefined inclusion criteria for IMR-TB and were included in the analyses. Given this sample size, and accounting for the finite population correction, the study had approximately 80% statistical power (two-sided, α = 0.05) to detect a minimum detectable absolute difference of about 7.3 percentage points from a null proportion of p0=0.50. The flow diagram of IMR-TB Patients selection process is displayed in Figure 1.
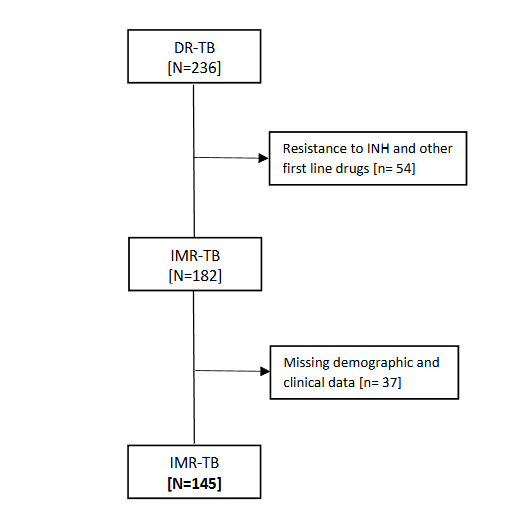
DATA COLLECTION
Data were extracted from patient records and folders on the following variables: date of hospital admission, age, sex, DR-TB treatment initiation date, HIV status, antiretroviral therapy (ART) history, type of DR-TB, diagnostic category, TB history, confirmatory tests for DR-TB, DST results, site of DR-TB localization, treatment regimen, six-month treatment outcomes (e.g., died, alive, default, failed, transferred out), smear and sputum microscopy, and monthly culture and sensitivity (MCS) test results. Data collection was carried out using a structured form, and all extracted information was entered into a Microsoft Excel database (Microsoft Office 2007).
ANTI-TUBERCULOSIS AND ANTIRETROVIRAL DRUGS USED AT BREWELSKOOF HOSPITAL FROM 2009–2015
The six-month IPT for DR-TB patients consisted of a combination of first and second-line anti-TB drugs: ethambutol (ETM), isoniazid (INH), pyrazinamide (PZA), rifampicin (RIF), kanamycin (KM) or amikacin (AM) or streptomycin (SM), terizidone (TRZ)/cycloserine [CS]), ofloxacin (OFX)/ moxifloxacin (MFX), and ethionamide (ETO). The injectable agents, aminoglycosides (KM or AM or SM) were discontinued at the end of the intensive phase once the TB cultures were confirmed negative. All drugs were administered daily, except for the injectable aminoglycosides, which were administered only five days a week. The antiretroviral drugs used included combinations of Nucleoside reverse transcriptase inhibitors (NRTIs): (abacavir [ABC], lamivudine [3TC], stavudine [D4T], emtricitabine [FTC] and tenofovir [TFV]) and Non-nucleoside reverse transcriptase inhibitors (NNRTIs): nevirapine [NVP], efavirenz [EFV]; and Protease inhibitors [PIs]: lopinavir/ritonavir [LPV/RTV] in accordance with the South African Department of Health (DOH) treatment guidelines.
INTENSIVE PHASE TREATMENT OUTCOME AND DEFINITIONS
A patient with a favourable treatment outcome was one whose sputum culture converted from positive to negative. The World Health Organization DR-TB treatment outcome definitions were used together with the South African Department of health (DOH) DR-TB Policy Guidelines of 2013. Unfavourable treatment outcome: A patient was classified as having an unfavourable treatment outcome if he/she met any of the following criteria: remained culture-positive at the end of six months of DR-TB treatment; died from any cause during the six-month treatment period; interrupted treatment for two or more consecutive months without a medical reason by the end of the six months; or lacked an assigned treatment outcome by the end of the six-month period, including those transferred to another treatment unit or with unknown treatment outcomes.
Efficacy endpoints: The primary efficacy endpoint was the incidence of an unfavourable outcome, defined as treatment failure, disease relapse, or death. Patients were considered to have a favourable outcome if their clinical TB disease had resolved, they had a negative sputum culture status at six months after the end of the therapy, and they had not already been classified as having had an unfavourable outcome. Sputum culture conversion: Defined as two consecutive negative cultures collected at least 30 days apart after initiation of treatment. Time to stable sputum culture conversion (TSCC): TSCC was defined as duration in days from the initiation of standardized anti-tuberculosis treatment to the date of the first of two consecutive negative sputum cultures, collected at least 30 days apart, with no subsequent positive cultures. Patients who did not achieve stable conversion during the follow-up period were censored at their last available culture result.
DEPENDENT VARIABLES
Study endpoints: The primary outcome was the proportion of IMR-TB patients whose sputum culture converted from positive to negative during the six-month IPT. The dependent variable of this study was TSCC, classified as early or delayed SCC. The intensive phase treatment outcomes were categorised as favourable and unfavourable according to predetermined criteria.
INDEPENDENT VARIABLES
The independent variables that were included are socio-demographic characteristics (age, and sex) and clinical variables (HIV status, ART history, type of DR-TB, smear baseline result, sputum culture results, baseline DST, HIV status, baseline CD4 count, patient’s diagnostic category of TB (new, return after lost to follow-up, treatment failure, relapse, failure and other), TB history, DR-TB localisation site and treatment regimen.
DATA MANAGEMENT
Data were initially extracted from the hospital registry records and entered into a Microsoft Access database. A thorough review and quality assurance process was then conducted to prepare the dataset for analysis. This process included systematic data cleaning procedures, beginning with the exclusion of patients who did not meet the predefined eligibility criteria for the study. Variables that were irrelevant or not pertinent to the research objectives were also removed. To ensure data integrity, missing values were assessed across all key demographic and clinical variables. Where feasible, inconsistencies and incomplete entries were cross-checked against original records to minimize information loss. The final analytic dataset included only patients and variables that met the study’s inclusion criteria and were essential for the statistical analyses.
STATISTICAL ANALYSIS
Descriptive statistics were first generated to summarize demographic and clinical characteristics of the study population. Continuous variables were expressed as means with standard deviations (SD) or standard error mean (SEM) in cases of normal distribution, while medians with interquartile ranges (IQR) were used for skewed data, depending on the distribution. Categorical variables were summarized using frequencies and percentages. Associations between intensive-phase treatment outcomes and demographic or clinical factors were assessed using Pearson’s chi-square test or Fisher’s exact test, as appropriate. In a separate analysis, IPT outcomes were dichotomized as favourable or unfavourable. Time to sputum culture conversion was compared across demographic and clinical factors using either Student’s t-test or analysis of variance (ANOVA). Where significant differences existed, post-hoc comparisons were performed using Tukey’s Honestly Significant Difference (HSD) test to determine which specific group means differed significantly. Kaplan–Meier survival functions were used to estimate the probability of SCC over time, and the log-rank test was applied to compare survival distributions across subgroups. Cox proportional hazard risk ratio models’ analyses were employed to identify independent predictors of TSCC and IPT outcomes, adjusting for relevant covariates. Hazard rate ratios (HRRs) with corresponding 95% CIs were calculated. Model fit was evaluated using likelihood ratio tests and Akaike’s information criterion (AIC). All statistical tests conducted were 2-tailed, and a probability of ≤0.05 was used as the threshold for declaring statistical significance. All statistical analyses were conducted using SAS 9.4 (SAS Institute, Cary, NC, USA).
ETHICAL CONSIDERATIONS
This study was registered and approved by the University of the Western Cape Ethics Committee under the ethics clearance registration number 07/6/12. Permission to access patients’ information was granted by the provincial DOH, as well as the medical superintendent of Brewelskoof Hospital in the Western Cape Province, South Africa. Patient information was captured anonymously, and all the data obtained were treated as confidential. The study was conducted according to the Helsinki and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines. The Protection of Personal Information Act (POPIA) policy of the University of the Western Cape ethics committee was applied in conducting the study.
RESULTS
BASELINE DEMOGRAPHIC AND CLINICAL CHARACTERISTICS OF IMR-TB PATIENTS
Table 1 summarizes the demographic and clinical characteristics of IMR-TB patients on treatment regimens. Of the 145 patients, 69 (47.6%) were male and 76 (52.4%) were female, with a median age of 37 years and an interquartile range (IQR) of 30 to 44. About 44.9% (n=65) of them were HIV-positive patients. Nearly two-thirds (65.5%) had a history of previous TB treatment, comprising 18.6% after default, 34.5% after relapse, and 12.4% after treatment failure. Most patients (96.4%) had pulmonary TB, 62.3% were smear-negative at baseline, and 86.7% were culture-positive. Drug susceptibility testing showed 98.6% were resistant to isoniazid alone, and 1.4% to both isoniazid and ethionamide. HIV co-infection was present in 44.9% of patients, of whom 92.2% were on ART, typically a combination therapy consisting of two nucleoside reverse transcriptase inhibitors (stavudine and lamivudine) along with one non-nucleoside reverse transcriptase inhibitor (efavirenz) at TB treatment initiation. Among patients with available data for the history of ART, 62.50% were on ART before TB diagnosis, and 33.33% were on ART after TB diagnosis with regards to the duration of ART, 50% of patients were on ART for less than 365 days and the other 50% were on ART 365-1096+days. Most HIV-positive patients (65.9%) had a CD4 count <200 cells/mm³, indicating advanced immunosuppression.
| Characteristics | IMR-TB patients | n | % |
|---|---|---|---|
| Sex | Male | 69 | 47.59 |
| Female | 76 | 52.41 | |
| Age group (Years) | < 20 | 5 | 3.45 |
| 20-29 | 31 | 21.38 | |
| 30-39 | 59 | 40.69 | |
| 40-49 | 33 | 22.76 | |
| 50-59 | 16 | 11.03 | |
| 60-69 | 1 | 0.69 | |
| Previous TB Treatment | New | 50 | 34.48 |
| Previous treatment | 95 | 65.52 | |
| DR-TB localisation sites | EPTB | 4 | 3.47 |
| PTB | 139 | 96.43 | |
| Patient diagnostic category | After Default | 27 | 18.62 |
| After Failure | 18 | 12.41 | |
| After Relapse | 50 | 34.48 | |
| New | 50 | 34.48 | |
| Sputum smear test results at baseline | Negative | 43 | 62.32 |
| Positive | 26 | 37.68 | |
| Sputum culture results at baseline | Negative | 13 | 13.27 |
| Positive | 85 | 86.73 | |
| Baseline drug sensitivity test | INH | 143 | 98.62 |
| INH+ETO | 2 | 1.38 | |
| HIV status | Negative | 80 | 55.17 |
| Positive | 65 | 44.85 | |
| Currently on ART | No | 5 | 7.81 |
| Yes | 59 | 92.19 | |
| On ART before TB diagnosis | No | 9 | 37.50 |
| Yes | 15 | 62.50 | |
| On ART after TB diagnosis | No | 16 | 66.67 |
| Yes | 8 | 33.33 |
Abbreviations: DR-TB: Drug-resistant tuberculosis; IMR-TB: Isoniazid mono-resistant tuberculosis; ART: Antiretroviral therapy; TB: Tuberculosis; INH: Isoniazid; ETO: Ethionamide; EPTB: Extrapulmonary tuberculosis, PTB: Pulmonary tuberculosis. Note: Due to missing responses, the frequencies reported for individual characteristics may not add up to the overall sample total.
TIME TO SPUTUM CULTURE CONVERSION
Out of the 145 IMR-TB patients, 71 (48.97%) achieved SCC within six months of treatment, while 74 (51.03%) did not attain SCC. The overall median time to sputum culture conversion for IMR-TB patients was 55 days (IQR 25-91). The median TSCC was 61 days (IQR 22.5–103) for HIV-negative individuals and 53 days (IQR 34–66) for those who were HIV-positive. Among the 71 patients who achieved SCC within six months of treatment, 44 (61.97%) were HIV-negative, and 27 (38.03%) were HIV-positive.
There were no statistically significant differences (p=0.5994) in the meantime to SCC based on HIV status among IMR-TB patients.
FACTORS INFLUENCING THE TIME TO SPUTUM CULTURE CONVERSION AMONG IMR-TB PATIENTS
Among all the factors assessed in the study, only age group showed a statistically significant (p=0.0074) influence on the time to SCC. Patients under 20 years and those aged 40–49 years had the longest mean SCC times, at 106.00±3.00 days and 103.27±13.96 days, respectively. The shortest mean SCC time was observed in the 50–59 years age group, at 33.71±7.89 days. Meanwhile, intermediate mean SCC times were recorded for patients aged 20–29 years (55.21±8.02 days) and 30–39 years (63.20±11.41 days). The median time to SCC was longer for IMR-TB patients aged 40–49 years at 87 (IQR: 65-146) and those below 20 years at 106 (IQR: 103-109).
| Characteristic | Sputum culture conversion time (days) | Test statistics | |
|---|---|---|---|
| Sex | Female | 73.00±9.990 | |
| Male | 62.65±8.090 | ||
| Age group (years) | <20 | 106.00±3.000ab | |
| 20-29 | 55.21±8.020bc | ||
| 30-39 | 63.20±11.410bc | ||
| 40-49 | 103.27±13.960ab | ||
| 50-59 | 33.71±7.890c | ||
| Previous TB treatment | New | 57.36±10.658 | |
| PT | 66.27±6.023 | ||
| Diagnostic Category | After default | 53.39±9.547 | |
| After failure | 59.18±10.137 | ||
| After relapse | 76.08±9.546 | ||
| New | 57.36±10.658 | ||
| DR-TB localisation site | EPTB | 23.50±5.315 | |
| PTB | 65.89±5.461 | ||
| Initial sputum smear result at baseline | Negative | 67.32±10.870 | |
| Positive | 47.64±9.473 | ||
| Characteristics | IMR-TB patients | n | % |
| Duration of ART before TB treatment (Days) | <365 | 50.00 | |
| 365-730 | 6.25 | ||
| 731-1095 | 12.50 | ||
| 1096+ | 31.25 | ||
| CD4+ category (Cells/mm3) | <200 | 65.91 | |
| 200-499 | 25.00 | ||
| >500 | 9.09 |
Abbreviations: DR-TB: Drug-resistant tuberculosis; IMR-TB: Isoniazid mono-resistant tuberculosis; ART: Antiretroviral therapy; PT: Previous TB treatment; INH: Isoniazid; ETO: Ethionamide; EPTB: Extrapulmonary tuberculosis, PTB: Pulmonary tuberculosis; βT=T-ratio; €F= F-ratio. Note: Due to missing responses, the frequencies reported for individual characteristics may not add up to the overall sample total.
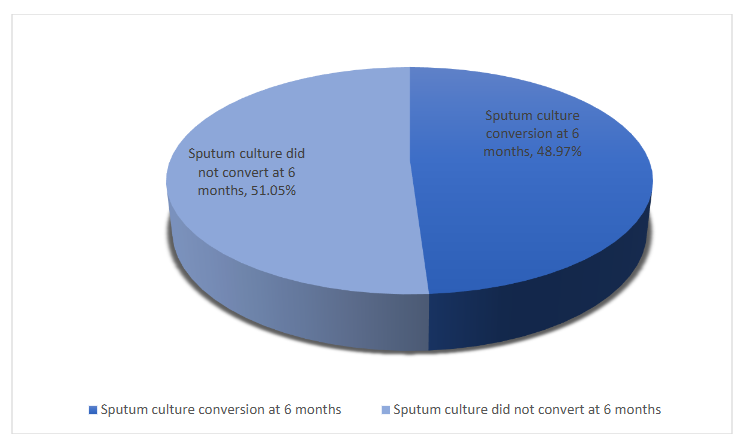
Figure 2: Sputum culture conversion at six months of treatment
INTENSIVE PHASE TREATMENT OUTCOMES OF IMR-TB PATIENTS ON TREATMENT REGIMEN
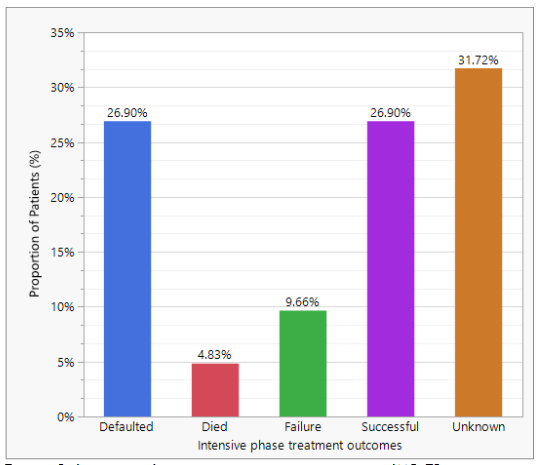
Among the 145 IMR-TB patients included in the study, only 26.90% achieved favourable outcome, 26.90% defaulted treatment, 4.83% died, 9.66% experienced treatment failure, and 31.72% had unknown outcomes. Majority of IMR-TB patients had unfavourable IPT outcome (72.41% 95% CI: 64.63-79.04) as shown in Figure 3.
FACTORS INFLUENCING THE TREATMENT OUTCOMES OF IMR-TB PATIENTS ON TREATMENT REGIMENS
Among the 145 IMR-TB patients included in the analysis, 40 (27.59%) achieved a favourable outcome during the intensive phase of treatment, while 105 (72.41%) experienced unfavourable outcomes. Differences in the treatment outcomes during the intensive phase were noted between patients with negative and positive baseline sputum culture results (p=0.0197). Patients with a positive baseline sputum culture exhibited a higher percentage (51.02%) of unfavourable treatment outcomes compared to those with a negative baseline sputum culture (12.24%). Significant variations (p=0.0132) in outcomes were observed among HIV-positive patients depending on the duration of ART. Specifically, all HIV-positive patients receiving ART for less than 356 days (equivalent to almost one year) prior to TB diagnosis experienced unfavourable outcomes (50.00%), while those on ART for 1096 days or more (equivalent to three years or more) before TB diagnosis also displayed higher rates of unfavourable outcomes (25.00%). Differences in treatment outcomes were identified based on the use of aminoglycosides (p=0.0141). Patients who were administered streptomycin (24.03%) showed elevated rates of unfavourable outcomes. Conversely, patients (42.64%) who did not receive any aminoglycosides demonstrated the highest rates of unfavourable outcomes.
| Characteristic | Total n (%) | Favourable † IMR-TB outcome n (%) | Unfavourable ‡ IMR-TB outcome n (%) | X2 value | p-Value | |
|---|---|---|---|---|---|---|
| Overall | 145 (100.00) | 40 (27.59) | 105 (72.41) | 29.138 | <.0001**** | |
| Sex | Female | 69 (47.59) | 16 (11.03) | 53 (36.55) | 1.275 | 0.2589ns |
| Male | 76 (52.41) | 24 (16.55) | 52 (35.86) | |||
| Age group (years) | <20 | 5 (3.45) | 3 (2.07) | 2 (1.38) | 4.492 | 0.4809ns |
| 20-29 | 31 (21.38) | 7 (4.83) | 24 (16.55) | |||
| 30-39 | 59 (40.69) | 14 (9.66) | 45 (31.03) | |||
| 40-49 | 33 (22.76) | 11 (7.59) | 22 (15.17) | |||
| 50-59 | 16 (11.03) | 0 (0.00) | 16 (11.03) | |||
| Previous TB treatment | New | 50 (34.48) | 14 (9.66) | 36 (24.83) | 0.007 | 0.9355ns |
| PT | 95 (65.52) | 26 (17.93) | 69 (47.59) | |||
| Diagnostic category | After default | 27 (18.62) | 5 (3.45) | 22 (15.17) | 2.271 | 0.5181ns |
| After failure | 18 (12.41) | 7 (4.83) | 11 (7.59) | |||
| After relapse | 50 (34.48) | 14 (9.66) | 36 (24.83) | |||
| New | 50 (34.48) | 14 (9.66) | 36 (24.83) | |||
| DR-TB localisation site | EPTB | 4 (3.47) | 2 (2.08) | 2 (2.08) | 0.386 | 0.5346ns |
| PTB | 139 (96.43) | 38 (26.39) | 101 (70.14) | |||
| Initial sputum smear result at baseline | Negative | 43 (62.32) | 9 (13.04) | 34 (49.28) | 0.326 | 0.5681ns |
| Positive | 26 (37.68) | 4 (5.80) | 22 (31.88) | |||
| Initial sputum culture at baseline | Negative | 13 (13.27) | 1 (1.02) | 12 (12.24) | 5.440 | 0.0197** |
| Positive | 85 (86.73) | 35 (35.71) | 50 (51.02) | |||
| Baseline drug sensitivity test | INH | 143 (98.62) | 39 (26.90) | 104 (71.72) | 0.510 | 0.4751ns |
| INH+ETO | 2 (1.38) | 1 (0.69) | 1 (0.69) | |||
| HIV status | Negative | 80 (55.17) | 27 (18.62) | 53 (36.55) | 3.394 | 0.0654ns |
| Positive | 65 (44.85) | 13 (8.97) | 52 (35.86) | |||
| Currently on ART | No | 5 (7.81) | 0 (0.00) | 5 (7.81) | 1.383 | 0.2397ns |
| Yes | 59 (92.19) | 13 (20.31) | 46 (71.88) | |||
| On ART before TB diagnosis | No | 9 (37.50) | 2 (8.33) | 7 (29.17) | 0.017 | 0.8967ns |
| Yes | 15 (62.50) | 3 (12.50) | 12 (50.0) | |||
| Duration of ART before TB treatment (days category) | < 365 | 8 (50.00) | 0 (0.00) | 8 (50.00) | 10.749 | 0.0132** |
| 365-730 | 1 (6.25) | 0 (0.00) | 1 (6.25) | |||
| 731-1095 | 2 (12.50) | 0 (0.00) | 2 (12.50) | |||
| 1096+ | 5 (31.25) | 0 (0.00) | 5 (31.25) | |||
| CD4+ category (Cells/mm³) | <200 | 29 (65.91) | 5 (11.36) | 24 (54.55) | 0.142 | 0.9313ns |
| 200-499 | 11 (25.00) | 2 (4.55) | 9 (20.45) | |||
| >500 | 4 (9.09) | 0 (0.00) | 4 (9.09) | |||
| Aminoglycosides | Kanamycin | 14 (10.85) | 6 (4.65) | 8 (6.20) | 8.526 | 0.0141** |
| Streptomycin | 34 (26.36) | 3 (2.33) | 31 (24.03) | |||
| Not receiving any aminoglycosides | 81 (62.79) | 6 (4.65) | 55 (42.64) |
Abbreviations: DR-TB: Drug-resistant tuberculosis; IMR-TB: Isoniazid mono-resistant tuberculosis; ART: Antiretroviral therapy; PT: Previous TB treatment; INH: Isoniazid; ETO: Ethionamide; EPTB: Extrapulmonary tuberculosis, PTB: Pulmonary tuberculosis; C: no data (not considered in the applicable analysis). Note: Due to missing responses, the frequencies reported for individual characteristics may not add up to the overall sample total. Significance level: *=p<0.05; **=p<0.01; ****=p<.0001; ns=Not significant (p>0.05).
RELATIONSHIPS BETWEEN TIME TO SPUTUM CULTURE CONVERSION, AGE GROUP, AMINOGLYCOSIDES AND INTENSIVE PHASE TREATMENT OUTCOMES
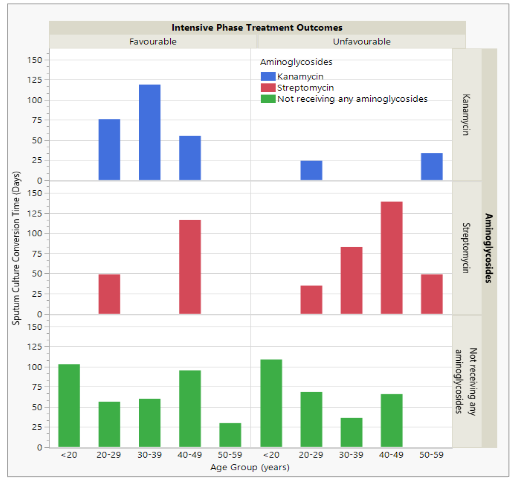
Figure 4 depicts the relationships between TSCC, age, Aminoglycosides and intensive phase treatment outcomes. Significant variations in the treatment outcomes (favourable vs. unfavourable) existed with patients who received streptomycin and had unfavourable outcomes, they experienced longer TSCC in comparison to those treated with kanamycin as well as patients who did not receive any aminoglycosides. The TSCC tended to increase with age and decreased subsequently for patients who were age 45 years and above taking streptomycin. Kanamycin demonstrated superior efficacy over streptomycin. However, patients receiving kanamycin experienced rapid TSCC and exhibited significantly more favourable outcomes, with SCC remaining relatively stable between the ages of 35 and 45 years.
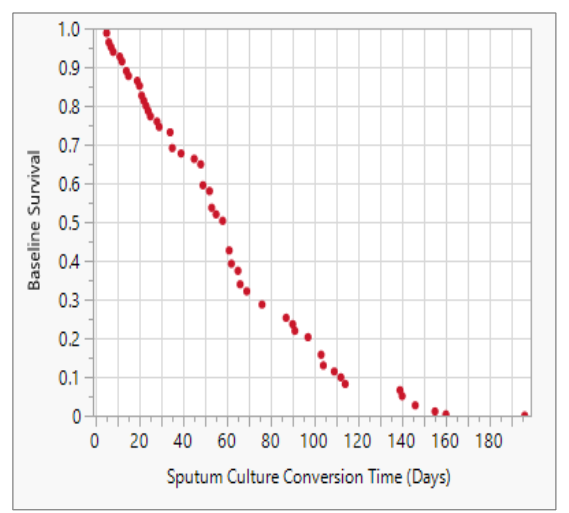
Figure 5 displays the baseline survival rate at the mean time to SCC in relation to the impact of the covariates. At 60 days (2 months) after start of therapy, approximately 44% of the patients attained SCC, indicating a favourable treatment response and a lower risk of treatment failure. By 90 days (3 months), the proportion of patients achieving SCC declined to 25%, suggesting the possible influence of factors such as drug resistance, suboptimal adherence or advanced disease at baseline. By 180 days (6 months), the survival rate had dropped to 0% indicating that all patients who were going to convert had done so by this point. The baseline survival rates at mean sputum culture conversion time (days) is presented in Appendix Table 1.
PROPORTIONAL HAZARD RISK RATIO FOR SPUTUM CULTURE CONVERSION TIME
| Level 1 /Level 2 (Ref) | Risk Ratio | p-value | Lower 95% | Upper 95% | |
|---|---|---|---|---|---|
| Age Group (years) | 20-29 <20 | 2.44 | 0.1626 | 0.70 | 8.53 |
| 30-39 <20 | 1.85 | 0.3212 | 0.55 | 6.20 | |
| 40-49 <20 | 1.04 | 0.9502 | 0.29 | 3.71 | |
| 50-59 <20 | 4.19 | 0.0327* | 1.13 | 15.61 | |
| <20 50-59 | 0.24 | 0.0327* | 0.06 | 0.89 | |
| 20-29 50-59 | 0.58 | 0.1744 | 0.27 | 1.27 | |
| 30-39 50-59 | 0.44 | 0.0321* | 0.21 | 0.93 | |
| 40-49 50-59 | 0.25 | 0.0012* | 0.11 | 0.58 | |
| Intensive Phase Treatment Outcomes | Unfavourable Favourable | 1.79 | 0.0264* | 1.07 | 2.98 |
| Favourable Unfavourable | 0.56 | 0.0264* | 0.34 | 0.93 |
TIME TO STABLE SPUTUM CULTURE CONVERSION IN IMR-TB PATIENTS
| Characteristic | Stable time to sputum culture conversion (months) | Test statistics |
|---|---|---|
| Sex | Female | 1.78±0.350 |
| Male | 1.89±0.330 | |
| Age group (years) | <20 | 0.50±0.500 |
| 20-29 | 1.52±0.420 | |
| 30-39 | 1.66±0.380 | |
| 40-49 | 3.20±0.650 | |
| 50-59 | 1.52±0.430 | |
| Previous TB treatment | New | 4.23±0.411 |
| PT | 3.78±0.265 | |
| Diagnostic category | After default | 3.00±0.548 |
| After failure | 3.56±0.338 | |
| After relapse | 4.11±0.403 | |
| New | 4.23±0.411 | |
| DR-TB localisation site | EPTB | 2.00±0.000 |
| PTB | 4.00±0.223 | |
| Initial sputum smear result at baseline | Negative | 4.63±0.532 |
| Positive | 3.67±0.615 | |
| Baseline drug sensitivity test | INH | 3.86±0.227 |
| INH+ETO | 5.00±1.000 | |
| HIV status | Negative | 3.87±0.283 |
| Positive | 4.00±0.365 | |
| On ART before TB diagnosis | No | 4.50±1.500 |
| Yes | 2.50±0.289 | |
| CD4+ category (Cells/mm³) | <200 | 3.40±0.748 |
| 200-499 | 3.00±0.000 | |
| >500 | 5.00±0.000 | |
| Aminoglycosides | Kanamycin | 4.50±0.619 |
| Streptomycin | 3.52±0.314 | |
| Not receiving any aminoglycosides | 5.00±0.447 |
Abbreviations: DR-TB: Drug-resistant tuberculosis; IMR-TB: Isoniazid mono-resistant tuberculosis; ART: Antiretroviral therapy; PT: Previous TB treatment; INH: Isoniazid; ETO: Ethionamide; EPTB: Extrapulmonary tuberculosis, PTB: Pulmonary tuberculosis; βT=€T-ratio; F= F-ratio; c =missing data/ was not considered/used in the statistical test in the analysis. Note: Due to missing responses, the frequencies reported for individual characteristics may not add up to the overall sample total. Significance Level: *=p<0.05; ns = not significant (p>0.05).
INTENSIVE PHASE TREATMENT OUTCOMES BY DEMOGRAPHICS AND CLINICAL FACTORS AMONG IMR-TB PATIENTS WHO ACHIEVED STABLE SPUTUM CULTURE CONVERSION ON TREATMENT REGIMENS
| Characteristic | Intensive phase treatment outcome at six months | Test statistics | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Overall | 85 (100.00) | 30.118 | <.0001**** | |||||||
| Sex | Female | 40 (47.06) | 6 (7.06) | 1 (1.18) | 7 (8.24) | 14 (16.47) | 12 (14.12) | 6 (7.06) | 5.450 | 0.2441 |
| Male | 45 (52.94) | 12 (14.12) | 2 (2.35) | 5 (5.88) | 20 (23.53) | 6 (7.06) | 3 (3.53) | 0.6254 | 0.6254 | |
| Age group (years) | <20 | 4 (4.71) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| 20-29 | 21 (24.71) | 5 (5.88) | 11 (12.94) | 1 (1.18) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| 30-39 | 38 (44.71) | 11 (12.94) | 24 (16.47) | 1 (1.18) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| 40-49 | 15 (17.65) | 1 (1.18) | 1 (1.18) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| 50-59 | 7 (8.24) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| Previous TB treatment | New | 25 (29.41) | 5 (5.88) | 1 (1.18) | 4 (4.71) | 11 (12.94) | 0.724 | 0.9483 | ||
| PT | 60 (70.59) | 1 (1.18) | 1 (1.18) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| Diagnostic category | After default | 20 (23.53) | 7 (8.24) | 1 (1.18) | 3 (3.53) | 4 (4.71) | 0.724 | 0.9483 | ||
| After failure | 10 (11.76) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| After relapse | 30 (35.29) | 1 (1.18) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| New | 25 (29.41) | 1 (1.18) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0 (0.00) | 0.6254 | 0.6254 |
| DR-TB localisation site | EPTB | 2 (2.38) | 0 ( |
*Significance level: *=p<0.05; ***=p<0.0001.
DISTRIBUTION OF AT-RISK POPULATION, SURVIVAL
AND FAILURE RATES BY TSCC AMONG IMR-TB PATIENTS
DURING INTENSIVE PHASE TREATMENT
Table 8 shows the survivability probabilities with respect to the IPT outcomes among IMR-TB patients who were at-risk (n=40). Among the 40 IMR-TB patients with SSCC, the likelihood of surviving after two months of therapy was recorded at 75.00%, which decreased to 57.50% after three months, further dropping to 35.00% after four months, and ultimately reaching 0.00% after six months of treatment. The likelihood of failure among the IMR-TB after two months of therapy was 25.00% (n=10). It increased to 42.50% and 65.00% at three and four months, respectively, and ultimately reached 100% after six months of treatment.
Table 8: Distribution of at-risk population, survival and failure rates for TSCC during intensive phase treatment among IMR-TB patients.
| Type of drug-resistant/TSCC (months) | Survival | Failure | Survival std error | Number failed | Number censored | At-risk population |
|---|---|---|---|---|---|---|
| 0.0 | 1.000 | 0.000 | 0.000 | 0 | 0 | 40 |
| 2.0 | 0.750 | 0.250 | 0.068 | 10 | 0 | 40 |
| 3.0 | 0.575 | 0.425 | 0.078 | 7 | 0 | 30 |
| 4.0 | 0.350 | 0.650 | 0.075 | 9 | 0 | 23 |
| 5.0 | 0.225 | 0.775 | 0.066 | 5 | 0 | 14 |
| 6.0 | 0.000 | 1.000 | 0.000 | 9 | 0 | 9 |
Discussion
This study explored the sputum culture conversion and the intensive phase treatment outcomes among IMR-TB patients treated at Brewelskloof Hospital in the Province of Western Cape in South Africa. The median TSCC for IMR-TB patients was 55 days (IQR 25–91). Our findings were unfavourable, demonstrating that only 48.97% of IMR-TB patients achieved SCC within the first six months of treatment, irrespective of HIV status. This outcome is considerably lower than that reported in the United States study conducted over 16 years (1993–2008) in which 82.9% of 3434 IMR-TB patients achieved sputum culture conversion during IPT.²⁶
In this study, age group emerged as the main factor influencing delayed SCC. Patients aged 40–49 years and those under 20 years experienced a prolonged time to SCC. These findings suggest potential age-related differences in response to IMR-TB treatment, highlighting the need for closer monitoring and targeted interventions in younger and middle-aged patients. Previous study has shown that patients above 45 years old face delays in achieving SCC.²⁷ In our study, TSCC tended to increase with age and decreased subsequently for patients who were age 45 years and above who were receiving streptomycin. The shortest TSCC was attained in the age group 50–59 years.
This study revealed an overall poor IPT outcome for IMR-TB patients, with only 27.59% having favourable outcomes and 72.41% experiencing unfavourable outcomes. The elevated incidence of unfavourable outcomes may be associated with the significant number of “unknown” or “default cases within the DR-TB cohort that inflate the rates of unfavourable outcomes, masking true treatment effectiveness. Additionally, in our investigation, a significant proportion (78.6%) of patients did not receive RIF, potentially attributable to concurrent health conditions such as HIV infection. Additionally, majority of the HIV-positive patients (83.1%) were not taking ART while they were taking. The exclusion of rifampicin in the treatment of IMR-TB during the intensive phase treatment of IMR-TB was linked with unfavourable outcomes in a study conducted in the United States reported only a 2% unfavourable outcome rates among patients with IMR-TB treated with RIF during the intensive phase treatment.⁵ Conversely, another study conducted in the United States reported that only 4.9% of patients were HIV-positive and 8.2% of patients had previously undergone TB treatment; thus, the current study observed a notably higher percentage of patients who had received prior TB treatment (66%), this could be another explanation for the poor treatment outcomes.
The limited use of RIF can have important implications for TB treatment outcomes. RIF is among the most potent bactericidal agents in first-line therapy, and its absence may lead to delayed sputum culture conversion, longer treatment duration, and a higher likelihood of suboptimal clinical outcomes.⁹¹ These findings highlight the ongoing complexity of treating TB/HIV co-infection in real-world settings and emphasize the need for integrated care approaches and broader access to ARV regimens compatible with rifampicin-containing therapy as recommended by international guidelines.²⁸
The alarming rate of unfavourable outcomes observed at the end of the IPT surpasses that reported by previous studies conducted in South Africa. Jacobson et al.¹¹ found a 16% rate of unsuccessful outcomes, with 15% classified as failed and 1% as deceased. In the investigation conducted in Durban South Africa from 2000–2012 among 557 IMR-TB patients, a 30.3% rate of unfavourable outcomes was reported after the long-term treatment, including those who failed, died, or defaulted.¹⁸ Differences in the findings on the unfavourable outcomes could stem from variations in study duration, study population, co-medication such as HIV-TB treatment and DR-TB regimen. The high number of unknown and default cases contributes significantly to thisstudy’s high rate of unfavourable outcomes, warranting further investigation.
The issue of patients lost to follow-up is pervasive and challenges across TB programmes in South Africa, including IMR-TB patients, leading to treatment discontinuation. The favourable outcomes observed in this study, at 27.59%, are notably lower than those reported in prior studies in South Africa. For example, a study conducted in the Western Cape Province covering the period from 2000 to 2009, documented a success rate of 65%.¹¹ Similarly, another study conducted in Durban, South Africa between 2000 and 2012 reported a 42% success rate in the long-term treatment of IMR-TB patients.¹⁸ Discrepancies in findings on treatment success could stem from variations in study duration, study population, co-medication such as HIV-TB treatment and DR-TB regimen.
Moreover, in this study, all deaths among IMR-TB patients occurred in those who were HIV-positive. Similarly, a study conducted on IMR-TB in Europe, identified HIV infection as a significant risk factor for unfavourable treatment outcomes.²⁹ Likewise, research conducted among IMR-TB patients in South Africa and Brazil found HIV infection to be associated with poor treatment outcomes,¹⁸,¹⁹ HIV remains the most significant risk factor for tuberculosis, as it increases the likelihood of progression to active TB disease, facilitates reactivation of latent TB infection, worsens treatment outcomes, and contributes to higher mortality rates.³⁰ The high rate of treatment failure underscores the critical need for early detection and thorough monitoring of IMR-TB patients. It also highlights the importance of developing tailored treatment regimens to improve outcomes.
The notable unfavourable outcomes observed in IMR-TB patients are a cause for concern and may be linked to the rise in MDR-TB cases. Jacobson et al.¹¹ pointed out that 61% of IMR-TB patients who failed treatment progressed to MDR-TB in a study conducted in the Western Cape Province in South Africa from 2000–2009.
We observed a significant association between TSCC and IPT outcomes in IMR-TB patients. Specifically, those who experienced unfavourable outcomes were found to have a 79% increased likelihood of encountering delayed or absent SCC compared to their counterparts with favourable outcomes. These results suggest that monitoring and addressing factors that contribute to unfavourable IPT outcomes may be critical in improving patient care and reducing the incidence of prolonged SCC. Further research is warranted to explore the underlying mechanisms and to develop targeted interventions that could improve IPT treatment outcomes. Previous studies that have explored TSCC and outcomes among DR-TB patients have reported similar findings.¹²,¹⁴
We observed the enhanced efficacy of kanamycin compared to streptomycin in the treatment of patients, particularly highlighting the rapid TSCC and improved patient outcomes associated with kanamycin administration. Our findings stand in stark contrast to a study conducted in the United States, which indicated that kanamycin was associated with unfavourable treatment outcomes in patients with DR-TB when juxtaposed with the use of streptomycin.³¹ The TSCC tended to increase with age and decreased subsequently for patients who were age 45 years and above who were taking streptomycin. This contradicts Jacobson’s study that found no link between the use of streptomycin and treatment outcomes.¹¹
According to the Kaplan–Meier curve, there was minimal change in survival time to stable sputum culture conversion beyond six months. Although further well-designed studies are needed, a possible explanation for the lower SSCC rate at later stages is that the patients may be immunocompromised or present with more severe illness making them less likely to achieve SSCC on any drug regimen. Previous studies have found that patients identified with indicators of severe disease experienced delayed time to SCC even on longer anti-TB drug regimens.¹³,²² This study has demonstrated that the regimen indicated for IMR-TB in South Africa results in unfavourable outcomes. A prospective study is recommended to confirm these findings.
Study Limitations
There are certain limitations associated with our current study that needs to be taken into consideration. Given the high loss to follow-up, the true death and failure rate is difficult to assess. It is important to acknowledge the possibility that there may have been more patients who failed treatment or died among this group of patients. This was a single-cohort study, with no control group, which limits the ability to draw direct comparisons or establish causal relationships. The lack of a control group means that observed outcomes cannot be directly attributed to the specific intervention administered, as there was no baseline for comparison. Future research should consider incorporating control groups or comparative designs to strengthen the validity and generalisability of findings.
Due to the retrospective nature of this study, certain data were missing, such as information on co-morbidities, CD4 counts, viral load, height and weight, history of alcohol use, smoking history, prior use of second-line drugs, BMI <18.5 kg/m², lung cavity and sputum smear grading at baseline. Hence the influence of these variables on the TSCC was not explored. Patient deaths during treatment were recorded without further investigation into the specific cause of death, whether TB-related or otherwise. A substantial proportion of patients had missing data on sociodemographic factors, behaviours, adverse drug reactions, adverse events, key laboratory parameters, and treatment adherence status. As a result, these variables were excluded from the analysis, limiting our ability to comprehensively investigate treatment failure and mortality predictors. Consequently, factors influencing mortality may extend beyond those examined in this study. Future prospective research is needed to explore these unexamined variables and identify definitive treatment failure and mortality predictors. However, since data came from only one centre, the findings may not be generalisable to broader population. We categorised default, lost to follow-up,and unknown into unfavourable treatment in our analysis, which may have influenced the study’s results.
This study provides a critical perspective on IMR-TB treatment outcomes, offering a benchmark against which contemporary WHO and national guideline-based regimens can be evaluated. By highlighting how management strategies have evolved, these insights not only contextualize current clinical practices but also inform ongoing improvements in patient care and future policy development. Ultimately, these findings underscore the enduring value of understanding past outcomes to guide more effective and evidence-based approaches to managing isoniazid-mono-resistant TB moving forward.
Recommendations
Efforts should be made to trace patients who default or are reported as unknown outcomes in order to prevent the spread of DR-TB within communities. Comprehensive monitoring, confirmation through DST, and effective treatment during the intensive phase are essential for IMR-TB patients to prevent inappropriate management. Periodic drug susceptibility testing during the intensive phase is crucial to monitor the acquisition of additional drug resistance among patients. Male patients, as well as patients of both sexes aged 40–49 years, require heightened clinical oversight due to their increased risk of adverse treatment outcomes. Furthermore, IMR-TB patients with a history of previous TB diagnosis or poor treatment outcomes should be prioritized for intensive monitoring throughout the treatment course to improve adherence, ensure timely interventions.
The early fatalities, defaults, and unknown outcomes underscore the necessity for vigilant monitoring of patients during the IPT, intensification of adverse event monitoring, and active reporting by both patients and healthcare providers. This underscores the importance of a short-course treatment regimen, excluding injectable therapies. Our study findings support the need for a shift towards a short course treatment regimen that exclude injectable agents, aiming to improve treatment adherence and outcomes. However, the success of such regimens depends on addressing key factors contributing to early unfavourable outcomes, including patient defaulting and mortality.
Conclusion
This study identified delayed sputum culture conversion and poor intensive phase treatment outcomes during the intensive phase treatment period of six months. Default and unknown outcomes occurred at notably high rates, warranting urgent programmatic attention. Male gender and older age (40–49 years) were associated with poorer IPT outcomes, highlighting the need for targeted interventions in these groups. The significant proportion of patients classified as defaulted or with unknown outcomes emphasizes the need for TB programmes to enhance patient tracking systems and ensure effective continuity of care and treatment. Furthermore, the occurrence of early deaths, defaults, and unknown outcomes reinforces the importance of intensive patient monitoring and adherence support throughout the IPT course to improve treatment success rates.
Acknowledgements
The authors acknowledged the South African Medical Research Council (SAMRC) for funding the study and the University of the Western Cape, South Africa, for their support.
Ethic approval and Consent to participate
This study was registered and approved by the University of the Western Cape Ethics Committee under the ethics clearance registration number 07/6/12. Permission to access patients’ information was granted by the Provincial DOH, as well as the medical superintendent of Brewelskloof Hospital in the Western Cape Province, South Africa. Patient information was captured anonymously, and all the data obtained were treated as confidential.
Authors contributions
All authors contributed equally to the development of this manuscript. Each author read and approved the final version of the manuscript for publication.
Funding
This study was conducted under the umbrella of a bigger research project funded by the South African Medical Research Council (SAMRC) through the “Self-Initiated Research Grant” allocated to Prof. P. Mugabo.
Competing interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data used for this study can be obtained upon reasonable request from Prof. P. Mugabo, the Principal Investigator.
References
1. WHO. WHO, 2014. In: Companion Handbook to the WHO Guidelines for the Programmatic Management of Drug-Resistant Tuberculosis. World Health Organization, https://www.ncbi.nlm.nih.gov/books/NBK247413/ (2014, accessed 22 March 2024).
2. Hoopes AJ, Kammerer JS, Harrington TA, et al. Isoniazid-Monoresistant Tuberculosis in the United States, 1993 to 2003. Archives of Internal Medicine 2008; 168: 1984–1992.
3. WHO W. WHO, 2008, https://www.who.int/publications/i/item/9789241563611 (2008, accessed 14 September 2024).
4. National Institute for communicable diseases, South African Tuberculosis Drug Resistance Survey 2012–14’ (2018). Available at: https://www.nicd.ac.za/assets/files/K-12750%20NICD%20National%20Survey%20Report_Dev_V11-LR.pdf (Accessed: 4 January 2025).
5. Cattamanchi A, Dantes RB, Metcalfe JZ, et al. Clinical Characteristics and Treatment Outcomes of Patients with Isoniazid-Monoresistant Tuberculosis. Clinical Infectious Diseases 2009; 48: 179–185.
6. Bang D, Andersen PH, Andersen ÅB, et al. Isoniazid-resistant tuberculosis in Denmark: Mutations, transmission and treatment outcome. Journal of Infection 2010; 60: 452–457.
7. Chien J-Y, Chen Y-T, Wu S-G, et al. Treatment outcome of patients with isoniazid mono-resistant tuberculosis. Clin Microbiol Infect 2015; 21: 59–68.
8. Menzies D, Benedetti A, Paydar A, et al. Standardized Treatment of Active Tuberculosis in Patients with Previous Treatment and/or with Mono-resistance to Isoniazid: A Systematic Review and Meta-analysis. PLoS Med 2009; 6: e1000150.
9. Menzies D, Benedetti A, Paydar A, et al. Effect of Duration and Intermittency of Rifampin on Tuberculosis Treatment Outcomes: A Systematic Review and Meta-Analysis. PLOS Medicine 2009; 6: e1000146.
10. Gegia M, Winters N, Benedetti A, et al. Treatment of isoniazid-resistant tuberculosis with first-line drugs: a systematic review and meta-analysis. The Lancet Infectious Diseases 2017; 17: 223–234.
11. Jacobson KR, Theron D, Victor TC, et al. Treatment Outcomes of Isoniazid-Resistant Tuberculosis Patients, Western Cape Province, South Africa. Clin Infect Dis 2011; 53: 369–372.
12. Kurbatova EV, Gammino VM, Bayona J, et al. Predictors of sputum culture conversion among patients treated for multidrug-resistant tuberculosis. The International Journal of Tuberculosis and Lung Disease 2012; 16: 1335–1343.
13. Kurbatova EV, Cegielski JP, Lienhardt C, et al. Sputum culture conversion as a prognostic marker for end-of-treatment outcome in patients with multidrug-resistant tuberculosis: a secondary analysis of data from two observational cohort studies. The Lancet Respiratory Medicine 2015; 3: 201–209.
14. Holtz TH, Sternberg M, Kammerer S, et al. Time to Sputum Culture Conversion in Multidrug-Resistant Tuberculosis: Predictors and Relationship to Treatment Outcome. Ann Intern Med 2006; 144: 650–659.
15. Javaid A, Ahmad N, Afridi AK, et al. Validity of Time to Sputum Culture Conversion to Predict Cure in Patients with Multidrug-Resistant Tuberculosis: A Retrospective Single-Center Study. Am J Trop Med Hyg 2018; 98: 1629–1636.
16. Brust JCM, Gandhi NR, Carrara H, et al. High treatment failure and default rates for patients with multidrug-resistant tuberculosis in KwaZulu-Natal, South Africa, 2000–2003. The International Journal of Tuberculosis and Lung Disease 2010; 14: 413–419.
17. Gegia M, Cohen T, Kalandadze I, et al. Outcomes among tuberculosis patients with isoniazid resistance in Georgia, 2007–2009. The international journal of tuberculosis and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease 2012; 16: 812.
18. van der Heijden YF, Karim F, Mufamadi G, et al. Isoniazid mono-resistant tuberculosis is associated with poor treatment outcomes in Durban, South Africa. Int J Tuberc Lung Dis 2017; 21: 670–676.
19. Báez-Saldaña R, Delgado-Sánchez G, García-García L, et al. Isoniazid Mono-Resistant Tuberculosis: Impact on Treatment Outcome and Survival of Pulmonary Tuberculosis Patients in Southern Mexico 1995-2010. PLoS One 2016; 11: e0168955.
20. Stagg Hr, Mc L, Td M, et al. Isoniazid resistant tuberculosis- a cause for concern? The international journal of tuberculosis and lung disease : the official journal of the International Union against Tuberculosis and Lung Disease 2017; 21: 129.
21. The National Department of Health; Essential Drugs Programme. Primary Healthcare Standard Treatment Guideline and Essential Medicine List. 8 th ed. South Africa: National Department of Health; 2024. Available from: https://www.health.gov.za/nhi-edp-stgs-eml/.
22. WHO. WHO treatment guidelines for isoniazid-resistant tuberculosis: supplement to the WHO treatment guidelines for drug-resistant tuberculosis, https://www.who.int/publications/i/item/9789241550079 (2018, accessed 25 October 2024).
23. Department of Health (DOH) South African tuberculosis treatment guidelines NDOH, 2014.https://www.tbonline.info/media/uploads/documents/national_tuberculosis management_guidelines_%282014%29.
24. World Health Organization. Definitions and reporting framework for tuberculosis – 2013 revision: updated December 2014 and January 2020. Geneva: World Health Organization, https://apps.who.int/iris/handle/10665/79199.
25. UWC-POPIA-Policy, 2021, https://www.uwc.ac.za/files/files/UWC-POPIA-Policy-approved-C202103.pdf (accessed 3 June 2024).
26. Prach LM, Pascopella L, Barry PM, et al. Rifampin monoresistant tuberculosis and HIV comorbidity in California, 1993-2008: a retrospective cohort study. AIDS 2013; 27: 2615–2622.
27. Iqbal Z, Khan MA, Aziz A, et al. Time for culture conversion and its associated factors in multidrug-resistant tuberculosis patients at a tertiary level hospital in Peshawar, Pakistan. Pak J Med Sci 2022; 38: 1009–1015.
28. Sculier D, Getahun H. World Health Organization. WHO policy on collaborative TB/HIV activities: guidelines for national programmes and other stakeholders. Geneva: World Health Organization; 2012. https://iris.who.int/server/api/core/bitstreams/d38d86fb-a877-471b-88a6-9f62fba35790/content. Geneva: World Health Organization, 2012.
29. Elliott E, Draper HR, Baitsiwe P, et al. Factors affecting treatment outcomes in drug-resistant tuberculosis cases in the Northern Cape, South Africa. Public Health Action 2014; 4: 201–203.
30. Narasimhan P, Wood J, MacIntyre CR, et al. Risk Factors for Tuberculosis. Pulmonary Medicine 2013; 2013: 828939.
31. Cegielski JP, Chan P-C, Lan Z, et al. Aminoglycosides and Capreomycin in the Treatment of Multidrug-resistant Tuberculosis: Individual Patient Data Meta-analysis of 12 030 Patients From 25 Countries, 2009–2016. Clinical Infectious Diseases 2021; 73: e3929–e3936.
32. Tierney DB, Franke MF, Becerra MC, et al. Time to Culture Conversion and Regimen Composition in Multidrug-Resistant Tuberculosis Treatment. PLOS ONE 2014; 9: e108035.

