Long-Acting Injectable Antipsychotics in High-Risk Pregnancy
Long-Acting Injectable Aripiprazole in High-Risk Pregnancy: A Case Report on Relapse Prevention, Obstetric Outcomes, and Infant Development
Dr. Parinda Parikh¹, Kanuja Sood², Himani J Suthar³, Arnesh Shukla⁴, Parthiv Pansuriya⁵, Shaurya Kumar Singh⁶, Zoe Gellert⁷, Sahia Manepalli⁸, Mahiya Buddhavarapu⁹, Arushi Kaushik Chandra¹⁰, Anika Shrivastava¹¹, Dr. Mina Oza¹²
- Department of Psychiatry, Weill Cornell Medical School, White Plains, USA
- SGT Medical College, Hospital and Research Institute, Gurugram, India
- GMERS Medical College and Civil Hospital, Gandhinagar, India
- St. Martinus University, Willemstad, Curaçao
- Government Medical College Surat, India
- Pravara Institute of Medical Sciences, Loni, Maharashtra, India
- Tulane University, New Orleans, USA
- University of South Florida, Tampa, USA
- University of Pittsburgh, Pennsylvania, USA
- NYU Steinhardt School, New York, USA
- Cassady School, Oklahoma City, USA
- 2nd Arc Associates, White Plains, New York, USA
OPEN ACCESS
PUBLISHED: 31 August 2025
CITATION: Parikh, P., 2025. Long-Acting Injectable Aripiprazole in High-Risk Pregnancy: A Case Report on Relapse Prevention, Obstetric Outcomes, and Infant Development. Medical Research Archives, [online] 13(8).
https://doi.org/10.18103/mra.v13i8.6905
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6905
ISSN 2375-1924
ABSTRACT
Pregnancy in women with bipolar disorder (BD) is considered high-risk due to various clinical and pharmacotherapeutic factors. The administration of pharmacological treatment during pregnancy requires a thorough evaluation of the exposure to psychotropic drugs and the risk of BD relapses. According to a recent systematic review published in the American Journal of Psychiatry, 37% of patients with a history of bipolar disorder relapsed during pregnancy. Failure to address bipolar disorder during pregnancy can lead to adverse outcomes for both the mother and the infant. This case report aims to explore the utilization of long-acting injectable aripiprazole (LAI) in a pregnant woman, to maintain stability and prevent relapse, while also considering the safety of the fetus. The patient experienced significant psychiatric symptoms, including mania and depression. Subsequently, throughout the 6 months of her pregnancy, the medication was adjusted to ensure optimal management of her bipolar disorder. Ultimately, the patient delivered a healthy baby boy via cesarean section at 39 weeks, weighing 9 lbs and 7 oz, and exhibiting an APGAR score of 9. The findings suggest that LAIs can be a viable option for managing bipolar disorder in pregnant women.
Keywords
Bipolar disorder, pregnancy, long-acting injectable aripiprazole, relapse prevention, obstetric outcomes
Introduction
Pregnancy in women with bipolar disorder (BD) is considered high-risk due to various clinical and pharmacotherapeutic factors. The administration of pharmacological treatment during pregnancy requires a thorough evaluation of the exposure to psychotropic drugs and the risk of BD relapses. According to a recent systematic review published in the American Journal of Psychiatry, 37% of patients with a history of bipolar disorder relapsed during pregnancy. Failure to address bipolar disorder during pregnancy can lead to adverse outcomes for both the mother and the infant.
This case report aims to explore the utilization of long-acting injectable aripiprazole (LAI) in a pregnant woman, to maintain stability and prevent relapse, while also considering the safety of the fetus.
Case Report
A 28-year-old female with a history of manic, psychotic, or depressive episodes. Her monthly Young Mania rating scale (YMRS) ranged from the range of 4-8. In addition to her primary treatment, the patient reported experiencing anxiety regarding her pharmacological needs. Regular monitoring by a multidisciplinary team, comprising her psychiatrist and obstetrician, revealed ongoing manic symptoms and no teratogenic effects on the growing fetus. Notably, the patient experienced a weight gain of 45 lbs within the initial trimester and subsequently developed gestational diabetes in the 8th month, necessitating insulin therapy. In this period, she reported an overall sense of well-being and good mood. The patient delivered a healthy baby boy via cesarean section at 39 weeks, weighing 9 lbs and 7 oz, and exhibiting an APGAR score of 9.
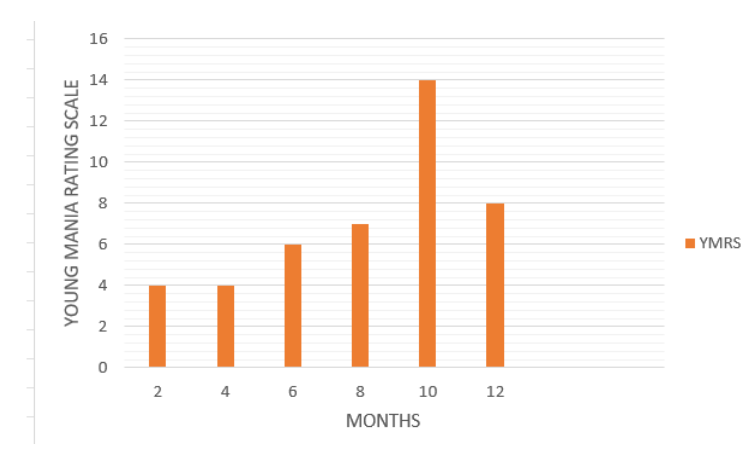
Discussion
Long-acting injectable (LAI) antipsychotics play a crucial role in enhancing adherence among clinicians regarding medication adherence, reduced risk of overdose, and improved, standardized interaction between clinicians and patients. In contrast to oral antipsychotics, LAIs have demonstrated greater efficacy in preventing psychiatric hospitalization. Additional advantages of LAIs include heightened awareness among clinicians regarding medication adherence, reduced risk of overdose, and improved, standardized interaction between clinicians and patients. The majority of available data suggest that the adverse effects of LAIs during pregnancy are comparable to those of oral counterparts, except for long-acting injectable, which necessitates a three-hour observation period following injection.
Conclusion
In the case of long-acting injectable antipsychotics (LAIs), there is a greater tendency for clinicians not to begin or discontinue LAI prescriptions during pregnancy. Given the prevalence of significant psychiatric conditions and their associated risks of decompensation during the perinatal period, healthcare providers must be at ease with the use of antipsychotic medications during pregnancy. While acknowledging the limited safety data on long-acting injectable (LAI) formulations during pregnancy, we recommend transcending apprehensions related to prescribing LAIs to pregnant women when substantial risks of psychiatric decompensation outweigh the absence of data.
References
2. Wesseloo R, Kamperman AM, Munk-Olsen T, Pop VJ, Kushner SA, Bergink V. Risk of Postpartum Relapse in Bipolar Disorder and Postpartum Psychosis: A Systematic Review and Meta-Analysis. Am J Psychiatry. 2016;173(2):117-127. doi:10.1176/appi.a jp.2015.15010124
3. Rusner M, Berg M, Begley C. Bipolar disorder in pregnancy and childbirth: a systematic review of outcomes. BMC Pregnancy Childbirth. 2016;16(1): 331. Published 2016 Oct 28. doi:10.1186/s12884-016-1127-1
4. Nguyen T, Frayne J, Watson S, Lebedevs T, Teoh S, Galbally M. Long-acting injectable antipsychotic treatment during pregnancy: Outcomes for women at a tertiary maternity hospital. Psychiatry Res. 2022; 313:114614. doi:10.1016/j.psychres.202 2.114614
5. Kishimoto T, Hagi K, Nitta M, et al. Effectiveness of Long-Acting Injectable vs Oral Antipsychotics in Patients With Schizophrenia: A Meta-analysis of Prospective and Retrospective Cohort Studies. Schizophr Bull. 2018;44(3):603-619. doi:10.1093/sc hbul/sbx090
6. Okoli CTC, Abufarsakh B, Wang T, Makowski A, Cooley A. Assessing the impact of long-acting injectable compared to oral antipsychotic medications on readmission to a state psychiatric hospital. J Psychiatr Ment Health Nurs. 2024;31(6):1155-1163. doi:10.1111/jpm.13075
7. Terrana N, Koren G, Pivovarov J, Etwel F, Nulman I. Pregnancy Outcomes Following In Utero Exposure to Second-Generation Antipsychotics: A Systematic Review and Meta-Analysis. J Clin Psychopharmacol. 2015;35(5):559-565. doi:10.109 7/JCP.0000000000000391
8. Greene M, Yan T, Chang E, Hartry A, Touya M, Broder MS. Medication adherence and discontinuation of long-acting injectable versus oral antipsychotics in patients with schizophrenia or bipolar disorder. J Med Econ. 2018;21(2):127-134. doi:10.1080/13696998.2017.1379412
9. Vieta E, Tohen M, McIntosh D, Kessing LV, Sajatovic M, McIntyre RS. Early use of long-acting injectable antipsychotics in bipolar disorder type I: An expert consensus. Bipolar Disord. 2025;27(1):7-16. doi:10.1111/bdi.13498
10. Bartoli F, Cavaleri D, Nasti C, et al. Long-acting injectable antipsychotics for the treatment of bipolar disorder: evidence from mirror-image studies. Ther Adv Psychopharmacol. 2023;13:20451253231163682. Published 2023 Mar 25. doi:10.1177/20451253231 163682
11. Patel R, Liman C, Oyesanya M, et al. Retrospective cohort study of long-acting injectable (LAI) antipsychotic initiation in the inpatient setting: impact of LAI characteristics on transition and continuation of care among patients with schizophrenia in the USA. BMJ Open. 2025;15(3): e092216. Published 2025 Mar 24. doi:10.1136/bmj open-2024-092216
12. Tiihonen J, Mittendorfer-Rutz E, Majak M, et al. Real-World Effectiveness of Antipsychotic Treatments in a Nationwide Cohort of 29 823 Patients With Schizophrenia. JAMA Psychiatry. 2017;74(7):686-693. doi:10.1001/jamapsychiatry.2017.1322
13. McDonnell DP, Detke HC, Bergstrom RF, et al. Post-injection delirium/sedation syndrome in patients with schizophrenia treated with olanzapine long-acting injection, II: investigations of mechanism. BMC Psychiatry. 2010;10:45. Published 2010 Jun 10. doi:10.1186/1471-244X-10-45
14. Bruno C, Cesta CE, Hjellvik V, et al. Antipsychotic use during pregnancy and risk of specific neurodevelopmental disorders and learning difficulties in children: a multinational cohort study [published correction appears in EClinicalMedicine. 2025 Mar 01;81:103139. doi: 10.1016/j.eclinm.202 5.103139.]. EClinicalMedicine. 2024;70:102531. Published 2024 Mar 17. doi:10.1016/j.eclinm.202 4.102531
15. Taylor CL, Broadbent M, Khondoker M, Stewart RJ, Howard LM. Predictors of severe relapse in pregnant women with psychotic or bipolar disorders. J Psychiatr Res. 2018;104:100-107. doi:10.1016/j.j psychires.2018.06.019
16. Reinstein SA, Cosgrove J, Malekshahi T, Deligiannidis KM. Long-Acting Injectable Antipsychotic Use During Pregnancy: A Brief Review and Concise Guide for Clinicians. J Clin Psychiatry. 2020;81(6):20ac13597. Published 2020 Nov 24. doi:10.4088/JCP.20ac13597
17. Caregiver discussion guide on long-acting injectables (LAIs): a treatment option for individuals living with bipolar I disorder and schizophrenia [PDF]. Otsuka Patient Education. Accessed June 16, 2025. https://www.otsukapatiented.com/00US23EUC0013_LAI_Caregiver_Discussion_Guide.pdf
18. Pejčić AV, Stefanović SM, Milosavljević MN, et al. Outcomes of long-acting injectable antipsychotics use in pregnancy: A literature review. World J Psychiatry. 2024;14(4):582-599. Published 2024 Apr 19. doi:10.5498/wjp.v14.i4.582
19. Voit FAC, Kajantie E, Lemola S, Räikkönen K, Wolke D, Schnitzlein DD. Maternal mental health and adverse birth outcomes. PLoS One. 2022;17 (8):e0272210. Published 2022 Aug 31. doi:10.137 1/journal.pone.0272210
20. Cattane N, Räikkönen K, Anniverno R, et al. Depression, obesity and their comorbidity during pregnancy: effects on the offspring’s mental and physical health [published correction appears in Mol Psychiatry. 2021 Feb;26(2):482. doi: 10.1038/s41380-020-01014-9.]. Mol Psychiatry. 2021;26(2):462-481. doi:10.1038/s41380-020-0813-6
21. Eleftheriou G, Butera R, Sangiovanni A, Palumbo C, Bondi E. Long-Acting Injectable Antipsychotic Treatment during Pregnancy: A Case Series. Int J Environ Res Public Health. 2023;20(4): 3080. Published 2023 Feb 9. doi:10.3390/ijerph20 043080
22. A Practical Review of Long‑Acting Injectable Antipsychotics. U.S. Pharm. Published [date not provided]. Accessed June 24, 2025.
https://www.uspharmacist.com/article/a-practical-review-of-longacting-injectable-antipsychotics
23. Deligiannidis KM, Meltzer-Brody S, Maximos B, et al. Zuranolone for the Treatment of Postpartum Depression [published correction appears in Am J Psychiatry. 2025 Mar 1;182(3):311. doi: 10.1176/appi.a jp.20220785correction.]. Am J Psychiatry. 2023; 180(9):668-675. doi:10.1176/appi.ajp.20220785
24. Marks JM, Spatz DL. Medications and lactation: what PNPs need to know. J Pediatr Health Care. 2003;17(6):311-319.

