Long-lasting Drying of Diabetic Macular Edema Insights
Long-lasting, Pathogeneses-related Drying of Diabetic Macular Edema
Avinoam Ophir, MD1
- 1 81 Sokolov St. Ramat-Hasharon, Israel
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Ophir, A., 2024. Long-lasting, Pathogeneses-related Drying of Diabetic Macular Edema. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.6035
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.6035
ISSN 2375-1924
ABSTRACT
Center-involved diabetic macular edema (DME) is the major cause of vision loss in the working-age population. Enduring DME might progressively injure the foveal layers with subsequent visual acuity loss. Therefore, the primary aim of DME therapy is to achieve an early, long-lasting dry macula to improve or sustain visual acuity. Current drug treatment has not achieved this aim. Diffuse DME (DDME), the most challenging DME type, is characterized by a compromised diffuse vasculature. Its pathogeneses include vitreofoveal traction and tractional epimacular membrane, as well as two newly-recognized pathogeneses: a) extrafoveal traction, the most common one, which is primarily detectable by 3D optical coherence tomography, and b) transitional-phase type, which represents the early tractional process and is detectable only ultra-structurally. Hence, all DDME eyes are apparently tractional. Consequently, treatment of naïve-treated DDME eyes by early pars plana vitrectomy has achieved long-lasting dry maculae in 92%–100% of eyes, typically in one step, and habitually associated with improved visual acuity. The surgery also naturally included the elimination of leaking microaneurysms (the “focal DME” component) when they were present. The transitional-phase type presents circumstances for attaining efficacious outcome by grid laser photocoagulation as well. Hence, DME seems to approach a curative situation. Accordingly, a revised pathogenesis-related DME classification is presented.
Keywords: Dry diabetic macular edema; Extrafoveal traction; 3D-OCT; Curative DME; Diffuse DME; Focal DME; Pars plana vitrectomy; DME pathogenesis; DME classifications; Grid laser photocoagulation; Tractional DME; Transitional-phase DME; Vitreopapillary traction; Anti-VEGF.
INTRODUCTION
Diabetic retinopathy is a leading cause of blindness worldwide. Center-involving diabetic macular edema (DME) is the most common cause of vision loss in diabetic patients. Thus, the primary aim of DME therapy is to achieve early, long-lasting macular dryness to improve or maintain visual acuity (VA). However, since the pathogenesis of diffuse DME (DDME) in the treated eyes during key randomized controlled trials (RCTs) involving anti-vascular endothelial growth factor (-VEGF) agents, with or without modified grid laser photocoagulation (GLP) was obscure, treatment has largely been approached as trial-and-error (T&Er). Consequently, repeated treatments often fall short of the therapeutic goal.
This article aims to introduce two previously overlooked DDME pathogeneses and their clinical significance in reaching the goal of DME therapy. It is based on peer-reviewed therapeutic, meta-analytic, and review studies published (in PubMed Central and Google Scholar) until September 2024.
Leaking microaneurysms (MAs) are considered the major source of focal DME, associated with capillary nonperfusion. In diffuse DME, VEGF and inflammatory cytokine upregulation and advanced glycation end products lead to compromised retinal vasculature. While RCTs show that intravitreal treatments temporarily improve VA and central sub-field thickness (CST), durable macular dryness is not consistently achieved, signaling a treatment failure.
The Protocol-T RCT of DME by Diabetic Retinopathy Clinical Research Network (DRCR.net) included 660 eyes with undetected traction, comparing three anti-VEGF agents – bevacizumab (BVZ; Avastin; Genentech/Roche), ranibizumab (RBZ; Lucentis, Genentech/Roche), and aflibercept (Eylea, Regeneron) – for DME treatment. The unknown DDME pathogenesis necessitated T&Er administration. Focal and diffuse DME were combined for analysis, and recommendations were made for both DME types as a single group. First two years of the three anti-VEGF medications, aided by laser rescue therapy in ~45% (mean) of eyes, achieved temporary VA gains and CST improvements (mean). However, VA decline was found in the subsequent three years, continuing as a real-world study. This led to a call by the authors that “a change in therapeutic strategy from anti-VEGF to a long-lasting efficacious treatment is required” in order to save sight. The DRCR.net request has been met. This key RCT has reasserted the critical importance of DME therapy to attain early and long-lasting macular drying.
A more recent DRCR.Retina.net study on BVZ monotherapy showed that 70% of eyes required an agent switch, highlighting medication failure. Corticosteroids like dexamethasone implant (Ozurdex, Allergan, Irvine, CA, USA) are acceptable DME treatments. However, cataract formation and intraocular pressure concerns limit their use as a primary option. A recent study summarized that diabetic blindness remains a substantial challenge despite all the recent advancements in diagnostics and treatments.
Laser studies using OCT also report poor outcomes for GLP in DDME which were worse pre-OCT era due to undetected vitreoretinal interface (VRI) abnormalities. Another challenge in reviewing GLP literature is that GLP improvement is gradual and necessitates studies that have at least 12 months of follow-up. Furthermore, most RCTs have focused on VA as the key measure, with less available data of CST except for the statistical mean. As well, the RCTs typically combined outcomes of focal laser and GLP as one group. Arevalo et al. reported on GLP monotherapy (n=120). Mean CST gradually decreased from base-line of 379µm by 20% and 28% (271µm) after 12 and 24 months, respectively. The percentage of patients who achieved durable dry macula was not reported. The VA improved by ≥2 lines in 30% of eyes at month 12, but decreased to 21% of eyes by month 24. In DME protocol-I of DRCR.net, 39% of 211 eyes achieved CST <250µm post-laser at two years, but the specific contribution of focal laser and GLP to these results is indeterminate.
Novel Pathogeneses of Diffuse Diabetic Macular Edema
A) EXTRAFOVEAL TRACTION: THE MOST COMMON PATHOGENESIS
Most existing OCT scans utilize 2D image slices, thereby missing the complex 3D structure of the VRI. When OCT B-scans cross the fovea, they may detect two of the DDME pathogeneses: vitreofoveal traction (VFT) and epimacular membrane (ERM). OCT scans often employ raster or radial lines, which leave the significant areas between scanned lines unexamined, as previously discussed. By comparison, continuous scanning systems, such as the 3D spectral-domain OCT (SD-OCT) 1000 (Topcon, Japan), provide more precise imaging of the VRI by scanning every point in the examined area. These continuous scans also provide the ability to view the examined field in 3D images and video clips (which differ from a 3D block). These 3D images thus exposed the overlooked vitreoretinal extrafoveal traction (ExFT) membranes and their traction sites, as well as their association with the centrally-involved DDME (Figure 1). ExFT site may appear in any spot in the area centralis, as explained.
The 3D diagnostic information is essential since the meridian of the posterior vitreous cortex (PVC; posterior hyaloid =PH) at the ExFT site is regularly different from that of the macula. The rest of the PH is anteriorly detached and appears in the different B-scan meridians as several free-floating membranes, as previously explained. Vitreopapillary traction (VPT) is a relatively common type of ExFT, detectable by B-scans (Figure 1). ExFT is often linked to splitting of the PVC into anterior and posterior lamellae, i.e., vitreoschisis.
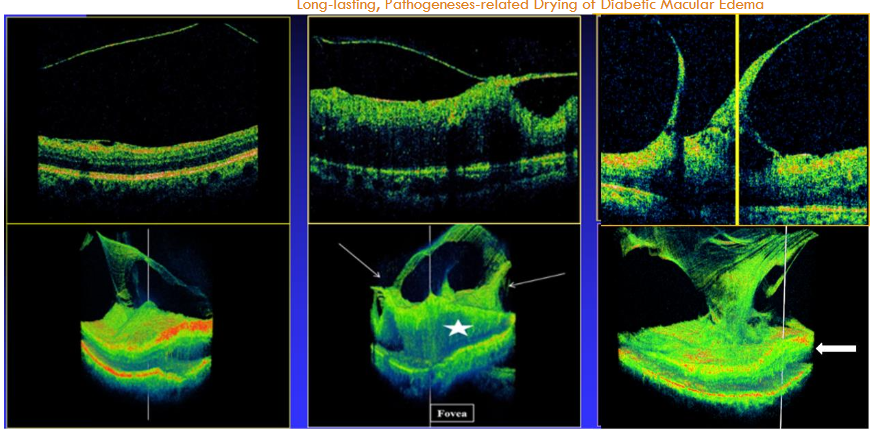
A study (n= 58 eyes/ patients) using the 3D SD-OCT 1000 identified the following prevalence of DDME pathogeneses: VFT in 19%, ERM in 22.4%, ExFT (including VPT) in 34.5% (making ExFT the most common pathogenesis), and transitional-phase (previously termed ‘vasogenic’) DDME, after excluding ExFT, accounted for 24.1%. Other studies have verified the association between ExFT and DDME. (Adie’s study termed ExFT “adhesions-pegs,” and reported a prevalence of 41%. Therefore, based on these data, in eyes treated with medications or laser following the exclusion of VFT and ERM, the mean expected ratio of ExFT to transitional-phase DDME is >34.5/24.1= ≥60/40%. Researchers have emphasized the importance of 3D-OCT imaging in evaluating the VRI in pathologies other than DME.
Recently, widefield OCT B-scans were shown to enable detection of ExFT membranes associated with center-involved pathologies, mainly of tractional retinoschisis in high myopia.
B) TRANSITIONAL-PHASE: THE EARLY TRACTIONAL PROCESS
Clinicopathologic studies have examined the VRI in DME using electron microscopy and immunocytochemistry following PPV and internal limiting membrane (ILM) peeling. The investigations found tractional membranes, irrespective of OCT classification of DME as tractional or non-tractional. The membranes were multilayered, with cell clusters embedded in collagen masses, representing an early stage of extracellular membrane (ECM) formation triggered by oxidative stress and inflammatory cytokines. Hyalocytes, cells residing in the preretinal PVC, are key players in this process, as they transdifferentiate into myofibroblasts, secrete profibrotic cytokines, and produce VEGF. These cellular components can thus promote collagen production and tangential ECM contraction, thereby leading to diffuse compromised vasculature, subsequent capillary leakage and diffuse macular edema. Consequently, the authors claim for early PPV with ILM peeling, regardless of the presence of traction formation on OCT. Johns et al. reviewed the issue and also called for early surgery, which can potentially prevent proliferative vitreoretinal diseases by addressing the role of hyalocytes.
Understanding treatments in the DDME pathogeneses era
A) INTRAVITREAL MEDICATIONS
It seems evident that T&Er DDME studied using medications have regularly targeted the ExFT (including VPT) membranes and/or the early-tractional (transitional-phase) process. These medications included anti-VEGFs, their biosimilars, steroids, second-generation intravitreal medications and their combinations. However, these therapies have not achieved the key aim of the therapy, durable dry maculae. In DDME, the medications may temporarily reduce VEGF, capillary leakage and inflammation in both the present tractional membranes (ExFT) and the early tractional process, improving VA and edema temporarily. However, they would not address the tractional component in each. In fact, intravitreal injections may sometimes exacerbate traction and worsen edema and VA, possibly by displacing the anterior vitreous anteriorly on removal of the needle, or increasing intravitreal inflammation. Similar complications have been observed in high myopia and retinoschisis.
B) GRID LASER PHOTOCOAGULATION
A study on GLP in transitional-phase (earlier termed ‘vasogenic’) DDME (n=18), after excluding ExFT, achieved durable macular drying in 13 (72%) eyes over an average of 15.9 months. Recurrent DDME occurred in four eyes between months 5 and 12, secondary to emergence of incomplete posterior vitreous detachment (iPVD) and ExFT (n=3), and ERM formation (n=1), all of which were operable. Overt macular ischemia following a second GLP during the second year resulted in recurrent edema in the fifth eye. GLP has possibly reduced macular oxygen consumption sufficiently enough to halt and reverse the early tractional and leaking process, leading to a durable dry macula in most (13) eyes. Noteworthy, new ExFT membranes were detected in two more eyes, each at month-15. Each has mildly increased the CST, but did not affect outcome, behaving actually as extrafoveal adherent membrane. This GLP study outcome may explain the circumstances of positive outcome in T&Er GLP studies. Further studies are required to improve GLP outcome in the transitional-phase process. Other studies may investigate the effect of prevention of hyalocytes transdifferentiation into myofibroblasts.
C) PARS PLANA VITRECTOMY
Studies on early PPV with ILM peeling in treatment-naive DDME eyes in which traction was not detected, have achieved complete and long-lasting macular drying in 92-100% of eyes, along with improved VA. The largest, multi-national study, included 120 patients with the longest follow-up (two years). Surgery resulted in 100% dry maculae, with a reduction of CST from a mean of 593μm to 260μm, in just one month post-surgery. After two years, all maculae remained dry. The authors noted, “for each day PPV is delayed, the chance of gaining more than 5 letters at month 24 decreases by 1.8%.” Notably, no distinction was made between fully attached posterior hyaloid and complete PVD prior to surgery. The surgery outcomes may be explained by the elimination of existing overlooked ExFT membranes, releasing ILM tangential traction and addressing the early tractional (transitional-phase) process located on the PVC. The latter include elimination of hyalocytes and myofibroblasts, removing as well as avoiding proinflammatory and profibrotic cytokines and VEGF production, and improving macular oxygenation. Other studies have reported on PPV’s effectiveness for VPT and retinal ExFT, as previously discussed. An exclusive T&Er study (n=870) found that PPV with ILM peeling was significantly superior to anti-VEGF, intravitreal triamcinolone acetonide (TCA) or GLP monotherapies after 6-24 months of follow-up. Today, a comparative study between early-PPV in naïve-treated eyes versus the already-approved failed current drug treatment will raise an ethical conflict.
When PPV was performed in refractory DME eyes following anti-VEGF treatments, statistically significant improvements in CST and VA were commonly observed. However, many eyes – 64% and 34% in some cases – continued with persistent macular edema. Some reports also describe combining surgery with panretinal photocoagulation, steroid implants, macular laser and/or intravitreal TCA. By contrast, when PPV with ILM peeling followed failed focal/GLP, including eyes with taut PH (which essentially are ExFT membranes), it appears that durable macular drying was attained more frequently, in 82-100% of cases (n = 6-58 eyes). VA improved by ≥2 lines in only 40% to ≥92% of these eyes, likely due to advanced preoperative foveal injury, as accurate diagnosis was limited before the OCT era. Notably, Kim’s study also suggested potentially worse outcome when PPV followed failed anti-VEGF therapy compared to failed laser treatments. These potential differences in PPV outcomes warrant further investigation. When PPV was performed as a last resort after prolonged treatment failures, it was often too late to restore VA probably due to irreversible foveal layers injuries. Noteworthy, the reason for poorer CST outcomes in intractable DME following PPV and ILM peeling compared to treatment-naive eyes remains to be elucidated.
In cases of residual macular edema following PPV, treatment typically involves anti-VEGF agents and/or intraocular steroids. However, after ruling out overt macular ischemia postoperatively, focal/GLP treatment may be added to the armamentarium, for achieving durable outcome. PPV alters the DDME status, and thus the emergence of iPVD and ExFT following GLP, a potential cause of recurrent edema, will not occur.
Focal DME treatment
After decades of focal laser treatment to leaking MAs, the short-acting anti-VEGFs have improved focal DME but have failed to achieve durable macular drying. New approaches, such as advanced laser instruments or surgical removal of MAs, are currently under investigation. Faricimab, a bispecific agent, has shown promise in drying focal DME for ≥8 weeks after three monthly intravitreal injections, due to its effects on MAs. Given the common presence of leaking MAs in DDME eyes as well, their differentiation into focal and diffuse DME is often based on calculating the relative percentage of leaking MAs in the DME area. Based on the outstanding efficacy of early PPV in treatment-naïve DDME eyes, it is anticipated that PPV may also eradicate the leaking MAs and halt new MAs production, probably by improving macular oxygenation and removing oxidants and VEGFs from the PVC. Thus, if further proven, PPV might also be an option for durably drying MA-related DME in selected cases.
Managing DME with Second-Generation Therapies
Second-generation treatments for DME, such as intravitreal faricimab and anti-VEGF biosimilars, have been studied using a T&Er approach. The faricimab RCT included patients with DDME and focal DME (n=1,891) in one combined group for calculations followed by recommendations, similar to earlier pivotal RCTs. Central macular anatomic drying, as observed on OCT, was achieved in 40% of eyes in this study and 39% in another study (n=51) for periods of eight weeks or more. (Notably, study outcomes based on an alternative DME definition, which have classified CST as ≥325 microns, were disregarded. This is because extending injection intervals requires evidence of an anatomically dry macula as seen on OCT). Takamura et al. identified that the macular drying by faricimab (n=27) was related to its effect on focal DME, with most MAs collapsing associated with a significant reduction in the formation of new MAs. This efficacy has been further supported by a clinical observation (n=2). The faricimab efficacy on focal DME was anticipated, since it won’t dry maculae for 8 weeks in DDME, which is associated with tractional processes.
In this RCT faricimab study, the 2 mg bi-monthly dose of aflibercept was chosen as the control group. However, this control group could be expected to attain poorer outcomes compared to the 2 mg aflibercept administered monthly, which had already been deemed unsuccessful. In selecting a control group for studying a new medication, the best available treatment should be chosen. Given that all non-surgical treatments have failed to meet the therapeutic goal, early PPV in treatment-naïve DDME eyes ought to be considered the best alternative.
The T&Er approach with faricimab is expected to reduce the frequency of injections by half compared to anti-VEGF treatments (8 weeks vs. 4-5 weeks, respectively). However, this improvement was observed in these studies in only 40% of eyes, probably the MA-related DME. It means that the remaining 60% with DDME experienced persistent edema throughout the studies. Therefore, this T&Er treatment preference may not be fully justified. If future DME trials separate focal and diffuse DME into distinct groups, it could lead to more clinically useful outcomes for each DME subtype. The use of anti-VEGF biosimilars, despite their comparable outcomes to the original agents, does not seem to address the underlying failure of anti-VEGFs in treating DDME, as was previously claimed.
Proposal to revise DME classifications
There is little evidence that the characteristics of DME described by the terms focal and diffuse explain variations in VA, CST, or response to treatment. A revised classification that is also based on objective diagnosis of the novel pathogeneses is proposed.
- MAs-related DME (for microaneurysm-dominant cases)
- Tractional DME (including VFT, ERM, VPT and ExFT)
- Transitional-phase DME (the early-tractional stage, undetectable clinically)
Its algorithm would personally define each DME eye (instead of DME and DDME), and consequently direct its optimal therapeutic approach.
In practice
To achieve long-lasting macular drying following exclusion of MA-related DME and overt macular ischemia, early PPV and ILM peeling in naïve-treatment eyes may be considered the primary treatment, irrespective whether traction is detected or not. Systemic factors influencing DME should also be best controlled. However, in cases in which GLP is preferred, advanced OCT imaging is essential to exclude ExFT before proceeding. When appropriate 3D OCT is unavailable, or when widefield OCT images are indefinite, detecting free-floating PH segments by B-scans should guide a search for ExFT membrane(s). This, by rescanning the PH route in various areas, looking for its contact site in the area centralis. In case of detecting ExFT, rescanning between the edema underlying the traction site (Figure 1, central) and the foveal edema is essential to verify or exclude continuity of the ExFT-related edema and the central DDME. Other options for detecting ExFT sites were earlier described. When ExFT associated with the DDME is unequivocally excluded, GLP may be considered.
Conclusions
Based on the newly-recognized pathogeneses of DDME, current scientific evidence provides the clinical support, which has achieved the aim of therapy. This, by early PPV in naïve-treated eyes, or by GLP in the transitional-phase type as an alternative. These pathogeneses may also explain the failure of achieving the goal of DME monotherapy by the current medications or by T&Er GLP. PPV treatment is accessible in numerous locations worldwide, offering a cost-effective alternative to the frequent and costly intraocular injections previously needed to manage the disease. Accordingly, a revised pathogenesis-related DME classification (instead of Diffuse and Focal DME) is proposed, which may direct a personalized optimal therapeutic approach. Further investigations are needed to fully elucidate and improve the potential effects in achieving durable DME drying by PPV when it is encountered in recalcitrant DME eyes, and of GLP in the transitional-phase DME.
Conflict
The author declares no conflict of interest.
References
- Bourne RRA, Jonas JB, Bron AM, Cicinelli MV, Das A, Flaxman SR, et al. Prevalence and causes of vision loss in high income countries and in Eastern and Central Europe in 2015: magnitude, temporal trends and projections. Br J Ophthalmol. 2018;102(5):575–585. doi: org/ 10.1136/ bjoph thalmol- 2017- 311258.
- Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema. Early treatment diabetic retinopathy study report number 1. Arch Ophthalmol. 1985;103(12):1796–1806. PMID: 2866759.
- Bhagat N, Grigorian RA, Tutela A, Zarbin MA. Diabetic macular edema: pathogenesis and treatment. Surv Ophthalmol. 2009;54(1):1-32. doi: 10.1016/j.survophthal.2008.10.001.
- Sorour OA, Levine ES, Baumal CR, Elnahry AG, Braun P, Girgis J, et al. Persistent diabetic macular edema: Definition, incidence, biomarkers and treatment methods. Surv Ophthalmol. 2023;68(2):147-174. doi: 10.1016/j.survophthal.2022.11.008.
- Sadda SR, Campbell J, Dugel PU, Holekamp NM, Kiss S, Loewenstein A, et al. Relationship between duration and extent of edema and visual acuity outcome with ranibizumab in diabetic macular edema: A post hoc analysis of Protocol I data. Eye (Lond) 2020;34(3):480-490. doi: 10.1038/s41433-019-0522-z.
- Sakamoto A, Nishijima K, Kita M, Oh H, Tsujikawa A, Yoshimura N. Association between foveal photoreceptor status and visual acuity after resolution of diabetic macular edema by pars plana vitrectomy. Graefes Arch Clin Exp Ophthalmol. 2009;247(10):(1325-1330. doi: 10.1007/s00417-009-1107-5.
- Cennamo G, Montorio D, Fossataro F, Fossataro C, Tranfa F. Evaluation of vessel density in disorganization of retinal inner layers after resolved diabetic macular edema using optical coherence tomography. PLoS One. 2021;16(1):e0244789. doi: 10.1371/journal.pone.0244789.
- Schmidt-Erfurth U, Garcia-Arumi J, Bandello F, Berg K, Chakravarthy U, Gerenda BS, et al. Guidelines for the Management of Diabetic Macular Edema by the European Society of Retina Specialists (EURETINA). Ophthalmologica. 2017;237(4):185-222. doi: 10.1159/000458539.
- The DRCR.net, Elman MJ, Aiello LP, Beck RW, Bressler NM, Bressler SB, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2010;117:1064-1077. doi:10.1016/j.ophtha.2010.02.031
- Elman MJ,Bressler NM,Qin H,beck RW, Ferris 3rd FL, Fridman SM, et al. Expanded 2-year follow-up of ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2011;118(4):609-614. doi: 10.1016/j.ophtha.2010.12.033.
- MitchellP,BandelloF,Schmidt-ErfurthU, Lang GE, Massin P, Schlingemann RO, et al. The RESTORE Study: Ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology. 2011;118(4): 615-625. doi: 10.1016/j.ophtha.2011.01.031.
- Diabetic Retinopathy Clinical Research Network; Wells JA, Glassman AR, Ayala AR, Jampol LM, Aiello LP, Antoszyk AN, et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. New Eng J Med. 2015;372(13):1193-1203. doi: 10.1056/NEJMoa1414264.
- Wells JA, Glassman AR, Ayala AR, Jampol LM, Bressler NM, Bressler SB, et al. Diabetic Retinopathy Clinical Research Network. Aflibercept, Bevacizumab, or Ranibizumab for diabetic macular edema: Two-year results from a comparative effectiveness randomized clinical trial. Ophthalmology. 2016;123(6):1351-1359. doi: 10.1016/j.ophtha.2016.02.022.
- Glassman AR, Wells III JA, Josic K, maguire MG, Antoszyk AN, Baker C, et al. Five-year outcomes after initial Aflibercept, Bevacizumab, or Ranibizumab treatment for diabetic macular edema (Protocol T Extension Study). Ophthalmology. 2020;127(9):1201-1210. doi: 10.1016/j.ophtha.2020.03.021.
- La Haij EC, Hendrikse F, Kessels AGH, Derhaag PJ. Vitrectomy results in diabetic macular edema without evident vitreomacular traction. Graefe’s Arch Clin Exp Ophthalmol. 2001;239(4):264-270. doi: 10.1007/s004170000251.
- Yanyali A, Nohutcu AF, Horozoglu F, Celik E. Modified grid laser photocoagulation versus pars plana vitrectomy with internal limiting membrane removal in diabetic macular edema. Am J Ophthalmol.2005;139(5):795-801. doi: 10.1016/j.ajo.2004.12.017.
- Iglicki M, Lavaque A, Ozimek M, Negri HP, Okada M, Chhablani J, et al. Biomarkers and predictors for functional and anatomic outcomes for small gauge pars plana vitrectomy and peeling of the internal limiting membrane in naïve diabetic macular edema: The VITAL Study. PloS One. 2018:13(7):e0200365. doi: 10.1371/journal.pone.0200365.
- Michalewska Z, Stewart MW, Landers III MB, Bednarski M, Adelman RA, Nawrocki J. Vitrectomy in the management of diabetic macular edema in treatment-naïve patients. Can J Ophthalmol. 2018;53(4): 402-407. doi: 10.1016/j.jcjo.2017.10.011.
- Jhaveri CD, Glassman AR, Ferris FL 3rd, Liu D, Maguire MG, Allen JB, et al. Aflibercept monotherapy or bevacizumab first for diabetic macular edema. New Engl J Med. 2022; 387(8):692-703. doi: 10.1056/NEJMoa2204225.
- Zhang J, Wang M, Chen L, Radke N. Diabetic blindness remains a big challenge despite all recent advancements in diagnostics and treatments. Asia Pac J Ophthalmol. 2024;13(5):100105. doi: 10.1016/j.apjo/2024/100105.
- Jorge EC, Jorge EN, Botelho M, Farat JG, Virgili G, Dib REI. Monotherapy laser photocoagulation for diabetic macular oedema. Cochrane Database of Systematic Reviews. 2018;10(10):CD010859. doi: 10.1002/14651858.CD010859.pub2
- LadasID, TheodossiadisGP. Long-term effectiveness of modified grid laser photocoagulation for diffuse diabetic macular edema. Acta Ophthalmologica. 1993;71(3):393-397. doi: 10.1111/j.1755-3768.1993.tb07154.x.
- Arevalo JF, Lasave AF, Wu L, Diaz-Llopis M, Gallego-Pinazo R, Alezzandrini AA, et al. Pan-American Collaborative Retina Study Group (PACORES). Intravitreal bevacizumab plus grid laser photocoagulation or intravitreal bevacizumab or grid laser photocoagulation for diffuse diabetic macular edema. Retina. 2013;33(2):403-413. doi:10.1097/IAE.0b013e3182695b83.
- Ophir A, Martinez MR, Mosqueda P, Trevino A. Vitreous traction and epiretinal membranes in diabetic macular edema using spectral-domain optical coherence tomography. Eye (London). 2010;24(10):1545-1553 . doi:10.1038/eye.2010.80.
- Ophir A. Full-field 3-D optical coherence tomography imaging and treatment decision in diffuse diabetic macular edema. Letter. Invest Ophthalmol Vis Sci. 2014;55(5):3052-3053. doi: 10.1167/iovs.14-14414.
- Thi A, Freund KB, Engelbert M. A pixel-based machine-learning model for three-dimensional reconstruction of vitreous anatomy. Trans Vis Sci Technol. 2022;11(7):1-10. doi:10.1167/tvst.11.7.3
- Spaide RF, Valmaggia P, Maloca P, Scholl HPN, Otto TP, Caujolle S. Imaging the vitreous with a novel boosted optical coherence tomography technique: vitreous degeneration and cisterns. Retina. 2022;42(8):1433–1441. doi:10.1097/IAE.0000000000003474.
- Ohno-MatsuiK,TakahashiH,MaoZ, Nakao N. Determining posterior vitreous structure by analysis of images obtained by AI-based 3D segmentation and ultra widefield optical coherence tomography. Br J Ophthalmol.2023;107(5):732-737.doi:10.1136/bjophthalmol-2021-320131.
- Nguyen TX, Jiang M, Yang D, Ran A, Ziqi T, Zhang S, et al. Advancing diabetic macular edema detection from 3D optical coherence tomography scans: Integrating privacy-preserving AI and generalizability techniques – a prospective validation in Vietnam. New Eng J Med. AI 2024;1(9). doi: 10/1056/Aloa2400091.
- Ophir A, Trevino A, Fatum S. Extrafoveal vitreous traction associated with diabetic diffuse macular oedema. Eye (Lond). 2010;24(2):347-353. doi:10.1038/eye.2009.106.
- Ophir A. Achieving durable dry macula in diffuse diabetic macular edema: a meta-analysis. Acta Sci Ophthalmol. 2021;4.3:91-104. https://actascientific.com > ASOP-04-0225.
- Ophir A. Re: Mylonas G, et al. The impact of the vitreomacular interface on functional and anatomical outcomes in diabetic macular edema treated with three different anti-VEGF agents: Post hoc analysis of the protocol T study. Correspondence. Retina. 2023;43(7):e39-41. doi:10.1097/IAE.0000000000003803.
- Ophir A. Diabetic macular edema: Approaching a curative situation. Editorial. Acta Sci Ophthalmol. 2024;7.6:01-05. https://actascientific.com › ASOP › ASOP-07-0767.
- Karatas M, Ramirez JA, Ophir A. Diabetic vitreopapillary traction and macular edema. Eye (London). 2005;19(6):676-682. doi:10.1038/sj.eye.6701622.
- Kakehashi A, Schepens CL, de Sousa-Neto A, Jalkh AE, Trempe CL. Biomicroscopic findings of posterior vitreoschisis. Ophthalmic Surg. 1993;24(12):846-850. PMID: 8115102.
- Sebag J. “Vitreoschisis”. Graefes Arch Clin Exp Ophthalmol. 2008;246(3):329-332. https://doi.org/10.1007/s00417-007-0743-x.
- Ophir A, Martinez MR. Epiretinal membranes and incomplete posterior vitreous detachment in diabetic macular edema, detected by spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2011;52(9):6414-6420. doi:https://doi.org/10.1167/iovs.10-6781
- Adhi M, Badaro E, Liu JJ, Kraus MF, Baumal CR, Witkin AJ, et al. Three-dimensional enhanced imaging of vitreoretinal interface in diabetic retinopathy using swept-source optical coherence tomography. Am J Ophthalmol. 2016;162:140-149.e1. doi:10.1016/j.ajo.2015.10.025
- Fatima N, Islam QU, Shafique M. Frequency of vitreomacular traction in diabetic macular edema on optical coherence tomography. Pak Armed Forces Med J. 2017;67(1):47-50. ISSN 0030-9648.
- Martinez MR, Ophir A. Extrafoveal traction in retinal vein occlusion using spectral domain optical coherence tomography. Graefes Arch Clin Exp Ophthalmol. 2011;249(6):811-820. doi: 10.1007/s00417-011-1666-0.
- Krebs I, Glittenberg C, Zeiler F, Binder S. Spectral domain optical coherence tomography for higher precision in the evaluation of vitreoretinal adhesions in exudative age-related macular degeneration. Br J of Ophthalmol. 2011;95(10):1415-1418. https://doi.org/10.1136/bjo.2010.192385.
- Ota-Itadani M, Takahashi H, Mao Z, Igarashi-Yokoi T, Yoshida T, Ohno-Matsui K. Deep learning-based 3D OCT imaging for detection of lamina cribrosa defects in eyes with high myopia. Sci Rep. 2022;12(1):22195. doi: 10.1038/s41598-022-26520-4.
- Stanga PE, Pastor-Idoate S, Reinstein U, Vatas P, Patel U, Dubovy S, et al. Navigated single-capture 3D and cross-sectional wide-field OCT of the mid and peripheral retina and vitreoretinal interface. Eur J Ophthalmol. 2022;32(3):1642-1651. doi: 10.1177/11206721211026100.
- Gandorfer A, Messmer EM, Ulbig MW, Kampik A. Resolution of diabetic macular edema after surgical removal of the posterior hyaloid and the inner limiting membrane. Retina. 2000;20(2):126-133.
- Matsunaga N, Ozeki H, Hirabayashi Y, Shimada S, Ogura Y. Histopathologic evaluation of the internal limiting membrane surgically excised from eyes with diabetic maculopathy. Retina. 2005;25(3):311–316. https://doi.org/10.1097/00006982-200504000-00010.
- Hagenau F, Vogt D,Ziada J,Guenther S, Haritoglou, Wolf A, et al. Vitrectomy for diabetic macular edema: Optical coherence tomography criteria and pathology of the vitreomacular interface. Am J Ophthalmol. 2019 Apr;200:34-46. doi: 10.1016/j.ajo.2018.12.004.
- SchumannRG,Hagenau F,Guenther SR,WOLF A, Priglinger SG, Vogt D. Premacular cell proliferation profiles in tangential traction vitreo-maculopathies suggest a key role for hyalocytes. Ophthalmologica 2019;242(2):106-112. doi:10.1159/000495853.
- Jones CH, Gui W, Schumann RG, Boneva S, Lange CA, van Overdam K, et al. Hyalocytes in proliferative vitreoretinal diseases. Expert Rev Ophthalmol. 2022;17(4):263-280. doi: 10.1080/17469899.2022.2100764.
- Sensoy E, Citirik M. Current biosimilar anti-VEGF drugs in retinal diseases. Eur Eye Res. 2024;4:00-00. Ahead of Print: EER-29291. doi: 10.14744/eer.2024.29291.
- Bressler SB, Barve A, Ganapathi PC, Beckmann K, Apte RS, Marcus DM, et al. Aflibercept biosimilar MYL-1701P vs. reference aflibercept in diabetic macular edema: The INSIGHT randomized clinical trial. Jama Ophthalmol. 2024:142(10):952-960. doi: 10.1001/jamaophthalmol.2024.3458.
- Wykoff CC, Abreu F, Adamis AP, Basu K, Eichenbaum DA, Haskova Z, et al. Efficacy, durability, and safety of intravitreal faricimab with extended dosing up to every 16 weeks in patients with diabetic macular oedema (YOSEMITE and RHINE): two randomized, double-masked, phase 3 trials. Lancet. 2022;399(10326);741-755. doi: 10.1016/S0140-6736(22)00018-6.
- Yoon D, Rusu I, Barbazetto I. Reduced effect of anti-vascular endothelial growth factor agents on diabetics with vitreomacular interface abnormalities. Int Ophthalmol. 2014;34(4):817-823. doi: 10.1007/s10792-013-9884-6.
- Russo A, Longo A, Avitabile V, Bonfiglio V, Fallico M, Boscia F, et al. Incidence and risk factors for traction macular detachment after anti-vascular endothelial growth factor agent pretreatment before vitrectomy for complicated proliferative diabetic retinopathy. J Clin Med. 2019;8(11):1960. doi:10.3390/jcm8111960.
- Patil NS, Dhoot AS, Popovic MM, Kertes PJ, Muni RH. Risk of intraocular inflammation after injection of anti-vascular endothelial growth factor agents. Retina. 2022;42(11): 2134-2142. doi: 10.1097/IAE.0000000000003582.
- Gopalakrishnan N, Joshi A, Yadav NK, Prabhu V, Bevaskar S, Ccablani J, et al. Progression of macular retinoschisis following intravitreal aflibercept injection for myopic macular neovascularization- a case report and review of literature. BMC Ophthalmol. 2024;24(1):224. doi: 10.1186/s12886-024-03497-4.
- Ophir A, Hanna R, Martinez MR. Importance of 3-D image reconstruction of spectral-domain OCT on outcome of grid laser photocoagulation for diffuse diabetic macular edema. Int J Ophthalmol. 2013;6(6):836-843. doi: 10.3980/j.issn.2222-3959.2013.06.17.
- Stefansson E,Landers, MB 3rd,Wolbarst ML. Increased retinal oxygen supply following pan-retinal photocoagulation and vitrectomy and lensectomy. Trans Am Ophthalmol Soc. 1981;79:307-334. PMID: 7200671; PMCID: PMC1312190.
- Nguyen QD, Shah SM, Van Anden E, Sung JU, Vitale S, Campochiaro PA. Supplemental oxygen improves diabetic macular edema: a pilot study. Invest Ophthalmol Vis Sci. 2004;45(2):617–624. doi:10.1167/iovs.03-0557.
- Kroll P, Wiegand W, Schmidt J. Vitreopapillary traction in proliferative diabetic vitreoretinopathy. Br J Ophthalmol. 1999;83(3):261-264. doi:10.1136/bjo.83.3.261.
- Kashani AH, Zhang Y, Capone A Jr, Drenser KA, Puliafito C, Moshfeghi AA, et al. Impaired retinal perfusion resulting from vitreoretinal traction: A mechanism of retinal vascular insufficiency. Ophthalmol Surg Lasers Imaging Retina. 2016;47(3):1-11. doi: 10.3928/23258160-20160229-03.
- Adelman R,Parnes AMichalewska Z, Parolini B, Boscher C, Ducournau D. Strategy for the Management of Diabetic Macular Edema: The European Vitreo-Retinal Society Macular Edema Study. BioMed Res Internat. 2015;Article ID352487, http://doi.org/10.1155/2015/352487
- Browning DJ, Lee C, Stewart MW, Landers III MB. Vitrectomy for center-involved diabetic macular edema. Clin Ophthalmol. 2016 Apr 26;10:735-742. doi: 10.2147/OPTH.S104906
- Miyamoto N, Ishida K, Ghassemi YK. Restoration of photoreceptor outer segments up to 24 months after pars plana vitrectomy in patients with diabetic macular edema. Ophthalmol Retina. 2017;1(5):389-394.doi.org/10.1016/j.oret.2017.01.017.
- Kim JH, Kang SW, Ha HS, Kim JR. Vitrectomy combined with intravitreal triamcinolone acetonide injection and macular laser photocoagulation for nontractional diabetic macular edema. Korean J Ophthalmol. 2013;27(3):186-193. http://dx.doi.org/10.3341/kjo.2013.27.3.18661.
- Hwang S, Kang SW, Kim KT, Noh H, Kim SJ. Three‑year outcomes of vitrectomy combined with intraoperative dexamethasone implantation for non‑tractional refractory diabetic macular edema. Sci Rep. 2021;11(1):1292. https://doi.org/10.1038/s41598-020-80350-w.
- Lewis H, Abrams GW, Blumenkranz MS, Campo RV. Vitrectomy for diabetic macular traction and edema associated with posterior hyaloidal traction. Ophthalmology. 1992;99(5):753-759. doi: 10.1016/s0161-6420(92)31901-3.
- Tachi N, Ogino N. Vitrectomy for diffuse macular edema in cases of diabetic retinopathy. Am J Ophthalmol. 1996;122(2):258-260. Doi: 10.1016/s0002-9394(14)72018-5.
- Pendergast SD, Hassan TS, Williams GA, Cox MS, Margherio RR, Ferrone PJ, et al. Vitrectomy for diffuse diabetic macular edema associated with a taut premacular posterior hyaloid. Am J Ophthalmol. 2000;130(2):178-186. doi: 10.1016/s0002-9394(00)00472-4.
- Yanyali A, Horozoglu F, Celik E, Ercalik Y, Nohutcu A. Pars plana vitrectomy and removal of the internal limiting membrane in diabetic macular edema unresponsive to grid laser photocoagulation. Eur J Ophthalmol. 2006;16(4):573 -581. doi:10.1177/112067210601600412.
- Landers MB III, Kon Graversen VA, Stewart MW. Early vitrectomy for DME: What makes it a viable treatment? Retinal Physician. 2013;10:56-60.
- Flaxel CJ, Edwards AR, Aiello LP, Arrigg PA, Beck RW, Bressler NM, et al. Factors associated with visual acuity outcomes after vitrectomy for diabetic macular edema: Diabetic Retinopathy Clinical Research network. Retina. 2010;30(9):1488-1495. doi:10.1097/IAE.0b013e3181e7974f.
- Zhou H, Zhang J , Guo B, Lin J, Mei J, Deng C, et al. Effect of anti-VEGF on early-stage post-vitrectomy macular edema in patients with proliferative diabetic retinopathy. BMC Ophthalmol. 2024;24(1):398. doi: 10.1186/s12886-024-03634-z.
- Liao M, Huang Y, Wang J, Meng X, Liu Y, Yu J, et al. Long-term outcomes of administration of intravitreal triamcinolone acetonide after posterior vitreous detachment during pars plana vitrectomy for proliferative diabetic retinopathy. Br J Ophthalmol. 2023;107(4):560-564. doi: 10.1136/bjophthalmol-2021-320332.
- Kim J, Kang SW, Shin DH, Kim SJ, Cho GE. Macular ischemia and outcome of vitrectomy for diabetic macular edema. Jpn J Ophthalmol. 2015;59(5):295-304. doi: 10.1007/s10384-015-0402-4.
- Hatano M, Higashijima F, Yoshimoto T, Ogata T, Ohta M, Kobayashi Y, et al. Evaluation of microaneurysms as predictors of therapeutic response to anti-VEGF therapy in patients with DME. Plos One. 2022;17(11):e0277920. doi:10.1371/journal.pone.0277920.
- Watanabe A, Takashima H, Nakano T. Effect of microaneurysms on anti-VEGF treatment for diabetic macular edema: A retrospective cross-sectional study. Medicine (Baltimore). 2023;102(44):e35888. doi:10.1097/MD.000000000000035888.
- Nozaki M, Kato A, Yasukawa T, Suzuki K, Yoshida M, Ogura Y. Indocyanine green angiography-guided focal navigated laser photocoagulation for diabetic macular edema. Jpn J Ophthalmol. 2019;63(3):243–254. doi: 10.1007/s10384-019-00662-x.
- Figueira J, Khan J, Nunes S, Sivaprasad S, Rosa A, de Abreu JF, et al. Prospective randomised controlled trial comparing sub-threshold micropulse diode laser photocoagulation and conventional green laser for clinically significant diabetic macular oedema. Br J Ophthalmol. 2009;93(10):1341–1344. doi: 10.1136/bjo.2008.146712.
- Tanaka H, Kojima K, Miyatani T, Kusada N, Terao N, Nagata K, et al. A new surgical approach for the treatment of a refractory foveal microaneurysm: A case report. Am J Ophthalmol Case Rep. 2024 Mar 1;34:102034. doi:10.1016/j.ajoc.2024.102034.
- Takamura Y, Yamada Y, Morioka M, Gozawa M, Matsumura T, Inatani M. Turnover of Microaneurysms after intravitreal injections of faricimab for diabetic macular edema. Invest Ophthalmol Vis Sci. 2023 Oct 3;64(13):31. doi: 10.1167/iovs.64.13.31.
- Rush RB. One year results of faricimab for aflibercept-resistant diabetic macular edema. Clin Ophthalmol 2023 Aug 16;17:2397-2403. Doi: 10.2147/OPTH.S424314.
- Takamura Y, Yamada Y, Inatani M. Role of microaneurysms in the pathogenesis and therapy of diabetic macular edema: A descriptive review. Medicina (Kaunas). 2023;59(3):435. doi: 10/3390/medicina59030435
- Sawa M, Nakagawa N, Shunto T, Nishiyama I. Two cases of diabetic macular edema with diminished areas of retinal non-perfusion and microaneurysms after intravitreal faricimab injections. Am J Ophthalmol Case Rep. 2023 Dec 7;4;33:101973. doi: 10.1016/j.ajoc.2023.101973.
- Browning DJ, Altaweel MM, Bressler NM, Bressler SB, Scott IU. Diabetic macular edema: What is focal and what is diffuse? Am J Ophthalmol. 2008;146(5):649–655. doi: 10.1016/j.ajo.2008.07.013.
- Yamada Y, Suzuma K, Ryu M, Tsuiki E, Fujikawa A, Kitaoka T. Systemic factors influence the prognosis of diabetic macular edema after pars plana vitrectomy with internal limiting membrane peeling. Curr Eye Res. 2013;38(12):1261-1265. doi: 10.3109/02713683.2013.820327?

