Long-Term Survival in NSCLC with Dual EGFR Mutations
Exceptionally Long-Term Survival in Non-Small Cell Lung Cancer with Dual EGFR Mutations: Exon 19 Deletion and G719X — A Case Report
Leylakhanim A. Melikova¹,², Carla Clemente³, Sabina G. Mehdizadeh¹, Javid E. Aliyev¹, Ilqar Sh. Gasinzadeh¹, Fuad A. Novruzov¹, Rufa A. Huseynova¹, Elnara E. Aliyeva¹, Jamil A. Aliyev¹
- National Oncology Center, Ministry of Health of the Republic of Azerbaijan, Baku, Azerbaijan
- Institute of Biophysics, Ministry of Science and Education of the Republic of Azerbaijan, Baku, Azerbaijan
- Stab Vida Investigacao e Servicos em Ciencias Biologicas Lda, Portugal
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Melikova, LA., Clemente, C., et al., 2025. Exceptionally Long-Term Survival in Non-Small Cell Lung Cancer with Dual EGFR Mutations: Exon 19 Deletion and G719X — A Case Report. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6681
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i6.6681
ISSN 2375-1924
ABSTRACT
Non-small cell lung cancer (NSCLC) remains one of the leading causes of cancer-related mortality worldwide. The discovery of activating mutations in the epidermal growth factor receptor (EGFR) gene has transformed targeted therapy, providing significant clinical benefit in selected patient populations. Nevertheless, survival beyond 10 years remains exceptionally rare. Although compound EGFR mutations are uncommon, emerging evidence suggests that such combinations can profoundly affect therapeutic efficacy, resistance patterns, and clinical outcomes.
We report the case of an Azerbaijani male who was diagnosed at the age of 69 years with stage IIIA lung adenocarcinoma, and who is currently 79 years old after achieving long-term survival. Following multimodal treatment—including EGFR tyrosine kinase inhibitor (TKI) therapy, chemotherapy, radiotherapy, and surgery—durable disease control has been achieved for over a decade. Serial ^18F-FDG PET-CT imaging and circulating tumor DNA (ctDNA) monitoring have confirmed sustained remission and molecular stability, with no evidence of emergent resistance mutations.
This case underscores the potential for exceptionally long-term survival in NSCLC patients with compound EGFR mutations managed with a comprehensive, molecularly guided therapeutic strategy. It also highlights the importance of evaluating hereditary cancer predisposition in such clinical contexts.
Keywords: Non-small cell lung cancer; EGFR mutation; exon 19 deletion; G719X; tyrosine kinase inhibitor; Erlotinib; long-term survival; cfDNA; PET-CT; hereditary lung cancer
INTRODUCTION
Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases and remains the leading cause of cancer-related mortality worldwide. Despite advances in screening and treatment, the 5-year overall survival (OS) rate remains below 25% in most populations. The therapeutic landscape of NSCLC has been fundamentally transformed by the discovery of actionable driver mutations, particularly those in the epidermal growth factor receptor (EGFR) gene.
Classical EGFR mutations—such as exon 19 deletions and the L858R point mutation in exon 21—predict favorable responses to EGFR tyrosine kinase inhibitors (TKIs), significantly improving progression-free survival (PFS) and overall outcomes. The development of first-, second-, and third-generation TKIs has extended the treatment options available to patients with EGFR-mutated NSCLC. However, the advent of next-generation sequencing (NGS) has further expanded our understanding of EGFR mutation heterogeneity, revealing a spectrum of rare and compound variants.
Compound EGFR mutations, defined as the coexistence of two or more EGFR alterations within the same tumor, constitute approximately 4–14% of EGFR-mutated NSCLC cases. These mutations exhibit variable clinical behavior and therapeutic sensitivity, and their optimal management remains an area of ongoing research. Certain combinations of compound mutations demonstrate enhanced sensitivity to EGFR-TKIs, whereas others are associated with primary resistance or rapid emergence of secondary resistance mechanisms.
Additionally, emerging evidence suggests that germline EGFR variants may contribute to familial lung cancer predisposition, particularly in cases exhibiting familial clustering. Germline T790M mutations, as well as polygenic and epigenetic factors, are being investigated as contributors to hereditary lung cancer syndromes. However, the clinical relevance of germline EGFR alterations and their impact on therapeutic response remain incompletely understood.
This case report contributes to the growing body of literature on compound EGFR-mutated NSCLC by presenting an exceptional instance of long-term survival exceeding 10 years. It underscores the critical importance of comprehensive molecular profiling, vigilant long-term monitoring, and personalized treatment strategies in optimizing outcomes for patients with complex EGFR mutational profiles.
CASE PRESENTATION
PATIENT DEMOGRAPHICS AND INITIAL PRESENTATION
A 69-year-old Azerbaijani male, born in 1946, presented to the National Oncology Center (NOC) in Baku in April 2015 with a one-month history of hemoptysis. He reported no other respiratory symptoms such as dyspnea, chest pain, or persistent cough. The patient had a prior history of tobacco use spanning over 25 years but had successfully quit smoking nine years before presentation. He denied alcohol consumption and had no other significant lifestyle-related risk factors. A notable familial predisposition to lung cancer was identified, as both his father and older brother had died from this disease. Given the patient’s strong family history of lung cancer, the pedigree of the family was documented to assess potential hereditary risk factors.
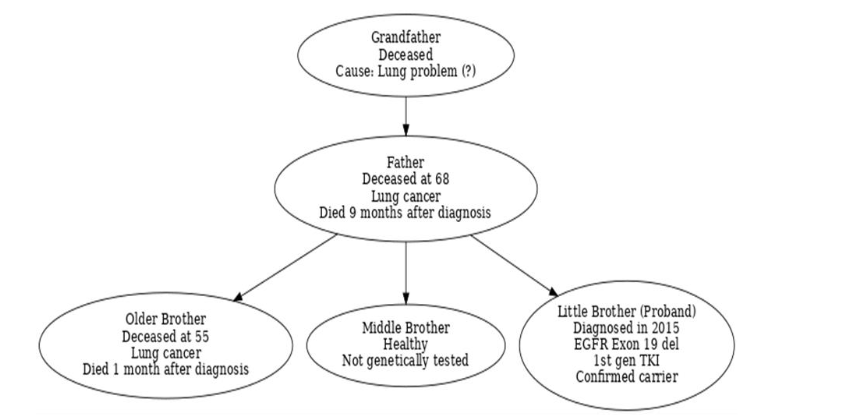
In light of this strong family history, genetic counseling was provided to evaluate potential hereditary cancer susceptibility. Broad-panel next-generation sequencing (NGS) was proposed for both the patient and at-risk family members; however, due to psychological concerns, the family declined germline testing and opted instead for regular clinical surveillance and long-term follow-up.
INITIAL DIAGNOSTIC WORKUP
Thoracic computed tomography (CT) revealed a solid mass in the lower lobe of the right lung, measuring 70 × 67 × 67 mm. Enlarged metastatic lymph nodes were identified in the subcarinal (30 × 20 mm), right paraesophageal (23 × 13 mm), and right hilar (22 × 19 mm) regions. Laboratory findings included: hemoglobin 110 g/L, platelets 140 × 10⁹/L, white blood cells 2.5 × 10³/μL, creatinine 94 μmol/L, AST 11.9 U/L, ALT 21.7 U/L.
On April 8, 2015, a percutaneous core needle biopsy confirmed moderately differentiated pulmonary adenocarcinoma (G2). Based on clinical, radiological, and histopathological data, the tumor was staged as cT3N2M0, corresponding to stage IIIA (AJCC 7th edition).
MOLECULAR PROFILING AND INITIAL TREATMENT
Comprehensive molecular profiling of the tumor tissue using NGS targeting EGFR, ALK, ROS1, MET, and RET driver mutations identified two pathogenic EGFR variants: an exon 19 deletion and a G719X missense mutation in exon 18. No actionable alterations were found in ALK, ROS1, MET, or RET.
The patient was started on first-line Erlotinib (Tarceva®) at a standard oral dose of 150 mg/day, combined with three cycles of platinum-based chemotherapy (cisplatin and pemetrexed). Follow-up imaging demonstrated disease stability.
DISEASE PROGRESSION AND SUBSEQUENT MANAGEMENT
Given radiologic evidence of disease stability, systemic therapy was transitioned to carboplatin (270 mg) combined with paclitaxel (140 mg). No infusion-related adverse events were observed. Concurrent thoracic radiotherapy was administered in 30 fractions over 30 days, for a total dose of 54 Gy.
In January 2016, the patient underwent right lower lobectomy with lymph node dissection. In 2017, a second surgical intervention was performed to address a loco regional recurrence with curative intent.
DEVELOPMENT OF SECONDARY MALIGNANCY
In 2020, imaging detected a new intravesical lesion. The patient underwent transurethral resection of the bladder tumor (TURBT), which revealed urothelial carcinoma. He subsequently received intravesical Bacillus Calmette–Guérin (BCG) therapy from January 29 to February 27, 2020. Post-treatment ^18F-FDG PET/CT demonstrated no evidence of residual disease.
FOLLOW-UP AND LONG-TERM OUTCOME
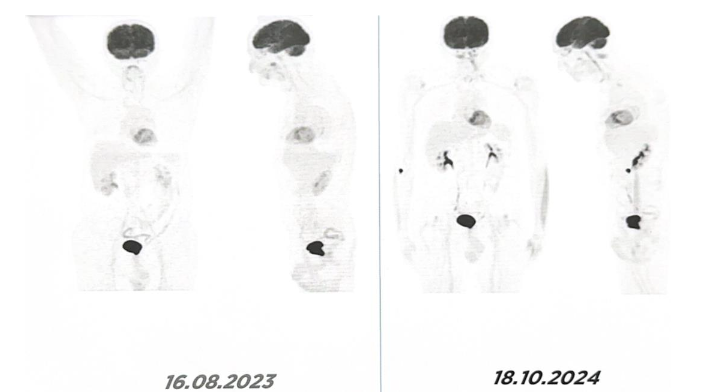
Since completing definitive therapy, the patient has been monitored annually with whole-body PET/CT.
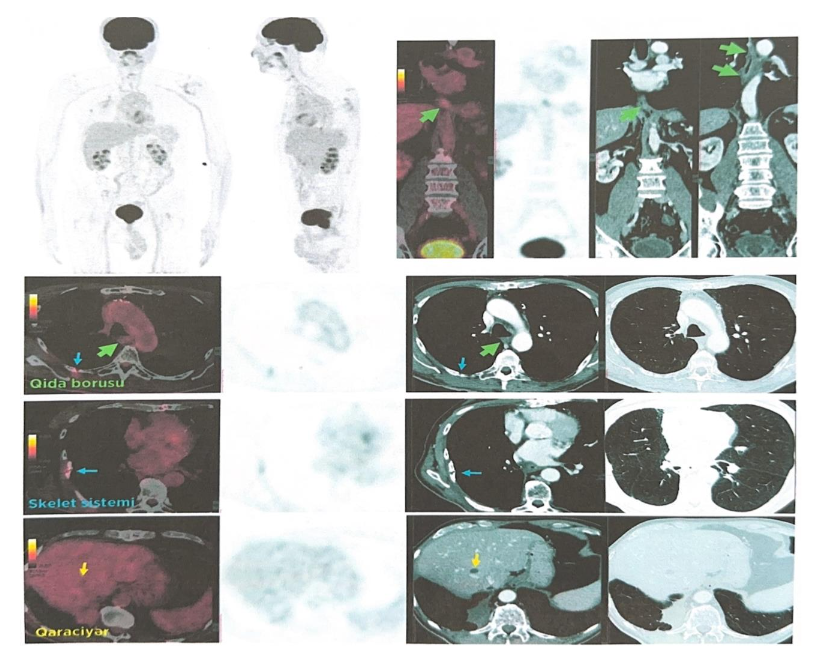
During follow-up, a secondary malignancy was detected and treated successfully. Subsequent imaging confirmed the absence of residual or recurrent bladder disease.
To date, there is no evidence of recurrence of either the primary pulmonary adenocarcinoma or the secondary bladder carcinoma. More than 10 years after initial diagnosis, the patient remains clinically stable and continues maintenance therapy with Erlotinib (150 mg/day). Annual whole-body PET/CT imaging continues to demonstrate complete remission with no evidence of disease recurrence or progression.
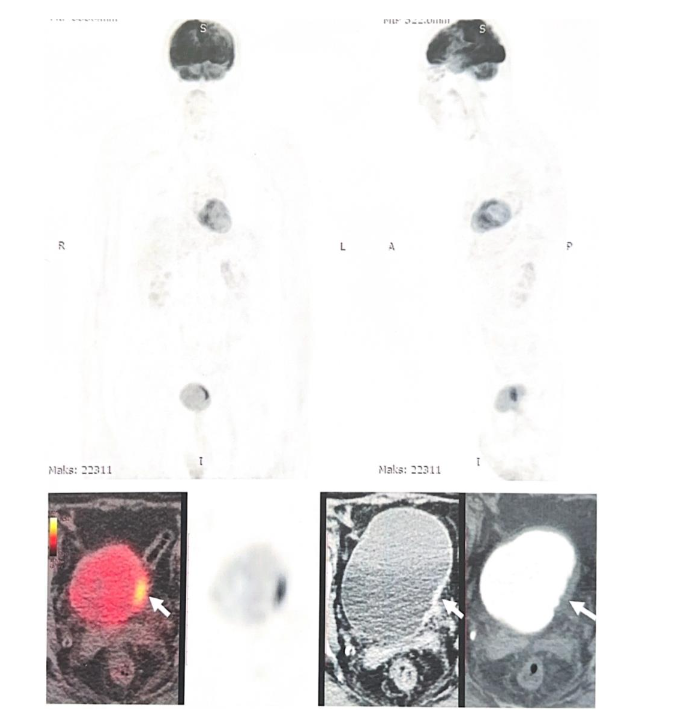
Importantly, the persistence of the EGFR exon 19 deletion was confirmed twice (between 2020 and 2024) through circulating tumor DNA (ctDNA) analysis of peripheral blood using real time PCR and a portable LAMP system, the Doctor Vida® device (STAB VIDA, Caparica, Portugal). No emergent resistance mutations have been detected, supporting the continued efficacy of long-term EGFR-targeted therapy.
DISCUSSION
Long-term survival exceeding ten years in patients with non-small cell lung cancer (NSCLC) remains exceptionally rare, even among those harboring sensitizing epidermal growth factor receptor (EGFR) mutations. Despite the remarkable progress achieved with EGFR tyrosine kinase inhibitors (TKIs), most patients ultimately experience disease progression, driven by the development of acquired resistance and tumor heterogeneity.
In this case, several clinical and molecular factors likely contributed to the exceptional outcome. The coexistence of two sensitizing EGFR mutations—an exon 19 deletion and a G719X point mutation in exon 18—may have provided synergistic sensitivity to EGFR-targeted therapy. Previous studies suggest that certain combinations of compound EGFR mutations exhibit enhanced responsiveness to TKIs, although clinical behavior remains heterogeneous across different mutation patterns. In particular, exon 19 deletions are well-established predictors of favorable response, while uncommon mutations such as G719X, when co-occurring with sensitizing mutations, may further modulate therapeutic efficacy.
Additionally, the relatively low tumor mutational burden (TMB) and limited intratumoral heterogeneity observed in this patient may have played a pivotal role in achieving durable disease control. Low TMB has been associated with sustained sensitivity to targeted therapies, while high TMB and clonal diversity promote the emergence of resistant subclones. In this patient, longitudinal analysis of circulating tumor DNA (ctDNA) consistently revealed persistence of the EGFR exon 19 deletion without detection of resistance mutations such as T790M or MET amplification. The integration of ctDNA monitoring into clinical practice enables real-time tracking of tumor evolution and supports timely therapeutic adjustments.
Furthermore, the comprehensive multimodal treatment strategy—including systemic chemotherapy, thoracic radiotherapy, curative-intent surgery, and long-term EGFR-TKI maintenance—likely contributed significantly to the exceptional outcome. Combining targeted therapy with local interventions has been shown to enhance disease control, delay progression, and suppress the emergence of resistant clones. Recent studies highlight the value of such integrative approaches in improving outcomes for patients with complex EGFR mutation profiles.
An additional noteworthy aspect of this case is the strong familial history of lung cancer involving first-degree relatives. While germline testing for EGFR T790M and other mutations was not performed, the clustering of lung cancer cases suggests a potential hereditary predisposition. Germline EGFR alterations, including T790M, have been reported in familial lung cancer and may influence both disease biology and treatment outcomes. Moreover, emerging evidence implicates epigenetic mechanisms—such as aberrant DNA methylation, histone modifications, and non-coding RNA dysregulation—in modulating hereditary cancer risk. Comprehensive genomic and epigenomic profiling may enhance risk stratification and inform surveillance strategies in families exhibiting clustering of lung cancer.
Finally, this case underscores the critical importance of comprehensive molecular profiling and personalized oncology care in NSCLC. Incorporating serial ctDNA monitoring and individualized treatment strategies informed by evolving molecular data can optimize outcomes and extend survival in patients with complex EGFR-mutated NSCLC. The exceptional long-term survival observed in this patient exemplifies the transformative potential of precision oncology when combined with vigilant long-term follow-up and multidisciplinary care.
CONCLUSION
This case highlights how precision oncology, when integrated with a comprehensive multimodal treatment approach and vigilant long-term surveillance, can achieve exceptionally prolonged survival in patients with non-small cell lung cancer (NSCLC) harboring compound EGFR mutations. The synergistic effects of targeted therapy, surgery, chemotherapy, and radiotherapy—guided by detailed molecular profiling and continuously supported by maintenance EGFR inhibition—underscore the transformative potential of personalized cancer care. The durable disease control observed in this patient, despite high-risk clinical features and a strong familial predisposition to lung cancer, emphasizes the importance of thorough genomic assessment and long-term molecular monitoring. Notably, the persistent sensitivity to EGFR-TKI and absence of resistance mutations suggest that certain compound EGFR mutation profiles may confer distinct therapeutic advantages. Furthermore, this case illustrates the clinical relevance of exploring hereditary and epigenetic factors in familial lung cancer syndromes, which may influence disease behavior and treatment outcomes. As our understanding of the molecular and genetic heterogeneity of NSCLC deepens, real-world cases such as this one provide valuable insights into optimizing individualized treatment strategies and identifying novel biomarkers for predicting therapeutic response and ensuring long-term disease control.
REFERENCES
- Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide. CA Cancer J Clin. 2021;71(3):209-249.
- Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553(7689):446-454.
- Soria JC, Ohe Y, Vansteenkiste J, et al. Osimertinib in untreated EGFR-mutated advanced NSCLC. N Engl J Med. 2018;378(2):113-125.
- Mok TS, et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947-957.
- Mok TS, Wu YL, Ahn MJ, et al. Osimertinib or platinum–pemetrexed in EGFR T790M–positive lung cancer. N Engl J Med. 2017;376(7):629-640.
- Ramalingam SS, et al. Osimertinib in EGFR-mutated advanced NSCLC. N Engl J Med. 2020;382(1):41-50.
- Robichaux JP, Le X, Vijayan RSK, et al. Structure-based classification predicts drug response in EGFR-mutant NSCLC. Nature. 2021;597(7878):732-737.
- Gristina V, Malapelle U, Galvano A, et al. The significance of complex EGFR mutations in non-small cell lung cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol. 2020;147:102858.
- Beau-Faller M, Prim N, Ruppert AM, et al. Rare EGFR exon 18 and exon 20 mutations in non–small-cell lung cancer on 10 years of molecular testing: a nationwide study of 1257 mutations. Ann Oncol. 2014;25(1):126-131.
- Yang JC, Sequist LV, Geater SL, et al. Clinical activity of afatinib in patients with advanced non–small-cell lung cancer harboring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. 2015;16(7):830-838.
- Liu Y, Li Y, Ou Q, et al. Acquired T790M and C797S mutations after first-line treatment with EGFR tyrosine kinase inhibitors. Thorac Cancer. 2021;12(5):760-767.
- Zhang Y, He J, Shi X. Clinical characteristics and prognostic analysis of germline EGFR T790M mutation in familial lung cancer. J Thorac Oncol. 2018;13(9):1353-1361.
- Bell DW, Gore I, Okimoto RA, et al. Inherited susceptibility to lung cancer may be associated with the T790M drug resistance mutation in EGFR. Nat Genet. 2005;37(12):1315-1316.
- Feinberg AP, Irizarry RA, Fradin D, et al. The epigenetic basis of common human disease: from local to global effects in cancer. Cancer Res. 2023;83(5):845-858.
- Liu Q, Liu Y, Wang Y, et al. Epigenetic heterogeneity in lung cancer: mechanisms and therapeutic opportunities. Genomics Proteomics Bioinformatics. 2023;21(4):460-475.
- Yu HA, Arcila ME, Rekhtman N, et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients. Clin Cancer Res. 2013;19(8):2240-2247.
- McGranahan N, Swanton C. Clonal heterogeneity and tumor evolution: past, present, and the future. Cell. 2017;168(4):613-628.
- Sequist LV, Waltman BA, Dias-Santagata D, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med. 2011;3(75):75ra26.
- Fang W, Huang Y, Hong S, et al. EGFR exon 20 insertion mutations and response to osimertinib in non–small-cell lung cancer. Onco Targets Ther. 2021;14:415-422.
- Peters S, et al. Alectinib versus crizotinib in untreated ALK-positive NSCLC. N Engl J Med. 2017;377(9):829-838.
- Zhao S, Fang W, Lu Y, et al. Clinical outcomes of NSCLC patients with uncommon EGFR mutations. Lung Cancer. 2019;127:53-58.
- Oxnard GR, Lo PC, Nishino M, et al. Natural history and molecular characteristics of lung cancers harboring EGFR exon 20 insertions. J Thorac Oncol. 2013;8(2):179-184.
- Kukita Y, Uchida J, Oba S, et al. Quantitative identification of mutant alleles derived from lung cancer in plasma cell-free DNA via anomaly detection using deep sequencing data. PLoS One. 2015;10(10):e0139280.
- Rolfo C, Mack PC, Scagliotti GV, et al. Liquid biopsy for advanced non–small cell lung cancer (NSCLC): a statement paper from the IASLC. J Thorac Oncol. 2018;13(9):1248-1268.
- Wu YL, et al. Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma. Lancet Oncol. 2014;15(2):213-222.
- Planchard D, et al. Dabrafenib plus trametinib in patients with previously untreated BRAF(V600E)-mutant metastatic NSCLC: an open-label, phase 2 trial. Lancet Oncol. 2017;18(10):1307-1316.
- Kris MG, Johnson BE, Berry LD, et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. JAMA. 2014;311(19):1998-2006.
- Toyooka S, Kiura K, Mitsudomi T. EGFR mutation and response of lung cancer to gefitinib. N Engl J Med. 2003;348(26):2570-2572.
- Mok TS, et al. Efficacy of osimertinib in Asian patients with NSCLC. J Thorac Oncol. 2019;14(8):1453-1463.
- Wu YL, et al. CNS efficacy of osimertinib in EGFR T790M-positive NSCLC: pooled analysis. J Thorac Oncol. 2017;12(9):1374-1382.
- Liu Y, et al. The impact of EGFR compound mutations on resistance mechanisms. Thorac Cancer. 2022;13(2):190-198.
- Zhang X, et al. Epigenetic regulation and therapeutic implications in lung cancer. Cancer Lett. 2022;545:215822.
- Rosell R, et al. Screening for EGFR mutations in lung cancer. N Engl J Med. 2009;361(10):958-967.

