Mast Cell Tumor Treatment in Dogs: Viscum album Therapy
Treatment of a Grade II Mast Cell Tumor in a Young Dog Using Injectable Viscum album Therapy
Ana Catarina Viana Valle¹²*; Maria Luiza Pereira Viana Valle³
- IMVN – Natural Veterinary Medicine Institute, Brasília – Brazil
- IDIS – Dr. Izao Soares Institute, Ribeirão Preto, Brazil
- UnB – Brasília University, Brasília – Brazil
OPEN ACCESS
PUBLISHED 30 April 2026
CITATION Viana Valle, A.C., Pereira Viana Valle, M.L., 2026. Treatment of a Grade II Mast Cell Tumor in a Young Dog Using Injectable Viscum album Therapy. Medical Research Archives, [online] 14(4). https://doi.org/10.18103/mra.v14i4.7371
DOI https://doi.org/10.18103/mra.v14i4.7371
ISSN 2375-1924
Abstract
Mast cell tumor (MCT) is a common skin neoplasm in dogs, and its conventional treatment may include surgery, chemotherapy, and radiotherapy. This study presents a case of a grade II MCT in a young dog treated with injectable homeopathic Viscum album. The diagnosis was confirmed by histopathological examination in December 2022, and immunohistochemistry was performed in March 2023. The patient was treated with multiple integrative therapeutic protocols, and the frequency and potency of the medicines were changed every 60 – 90 days over a 20-month period, with no adverse effects observed. A follow-up biopsy performed in January 2025 revealed the absence of tumor cells at the site of the initial incision, indicating complete tumor regression. Although multiple therapies were used throughout the treatment period, the consistent inclusion of Viscum album suggests a potential role for this therapy as part of the integrative approach. The results indicate a possible therapeutic benefit, especially when initiated early, although further controlled studies are required to confirm efficacy and safety.
Keywords
Mast cell tumor, Viscum album, homeopathy, veterinary oncology, integrative therapy
Introduction
Mast cell tumor (MCT) is a malignant skin neoplasm originating from the proliferation of mast cells. It is the most common skin tumor in dogs and can affect any organ, although it predominantly affects the skin. MCTs can develop at any age, with a higher incidence between 8 and 9 years of age, with no clear sex predisposition. Certain breeds, including Boxers, Boston Terriers, Bull Terriers, Beagles, Labradors, and Golden Retrievers, are at higher risk.
The histopathological classification of MCTs is fundamental in determining prognosis and guiding treatment decisions, and these tumors are classified into three grades: Grade I (well-differentiated), Grade II (moderately differentiated), and Grade III (poorly differentiated). Grade II MCTs exhibit intermediate biological behavior with variable malignant potential.
Conventional MCT treatment includes surgery, chemotherapy, and radiotherapy. However, treatment decisions may be challenging in cases with incomplete surgical margins or when owners decline aggressive therapies, leading to increased interest in complementary approaches.
Viscum album, a medicinal plant, has been investigated for its immunomodulatory and antitumor properties. Studies indicate that extracts from Viscum album contain bioactive compounds, such as lectins and viscotoxins, which promote cell apoptosis and inhibit tumor growth. Additionally, research suggests that Viscum album enhances immune response and improves the quality of life in cancer patients.
Despite growing interest, evidence supporting its use in veterinary oncology remains limited, particularly in controlled clinical settings. This study presents a case of a grade II MCT in a young dog treated with injectable homeopathic Viscum album. The aim is to describe the clinical evolution and to contribute to the generation of hypotheses for future studies evaluating integrative oncologic approaches. This report contributes to the ongoing discussion regarding the therapeutic potential of Viscum album in managing canine MCT and underscores the need for further research to validate its efficacy and safety.
Case Report:
A 1-year-and-11-month-old male Teckel, weighing 6.7 kg, was evaluated in March 2023 with a history of a small, rapidly growing nodule located in the right hip region. The lesion first appeared in November 2022 and was removed via incisional biopsy in December 2022. The tissue sample was submitted for histopathological analysis, confirming a diagnosis of cutaneous MCT, Grade II (low grade) with the following characteristics: a) Mitotic index (FN22 – 0.237 mm²); b) Invasion/infiltration depth: deep dermis; c) Presence of multinucleated/bizarre cells (FN22 – 0.237 mm²); d) Estimated histological margins (lateral and deep): compromised.
Following this diagnosis, no immediate treatment was initiated between January and March. Upon physical examination, the patient exhibited an excellent overall condition, with normal-colored mucous membranes, adequate body condition, good hydration, and normal heart and respiratory rates for his age and species. There were no signs of abdominal discomfort, and feeding, drinking, urination, and defecation patterns were all normal.
At that time, an immunohistochemical profile was requested and referred to an oncologist. Blood samples were collected (Table I) for a complete blood count and biochemical analysis, and additional imaging, including abdominal ultrasound (Figure I) and thoracic radiographs (Figure II), was performed. The following oral medications were prescribed: Vitamin C 500 mg, 1 tablet, SID for 30 days; Vitamin D3 1000UI/drop, 5 drops, SID for 30 days; Curcumin 90 mg + Piperine 90 mcg + Ganoderma lucidum 80 mg, 1 capsule, SID for 60 days; Phosphorus 30CH, 3 drops, SID for 30 days. For subcutaneous administration, the following were prescribed: Viscum album D2, D3, D6, D9, D12, and D30 (Injectcenter, Brazil), 1 ampoule (1.1 mL), in combinations of 2 ampoules in the same application, SID for 60 days; Histaminum D35 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, on alternate days for 60 days.

Results
The immunohistochemistry results were as follows: a) C-kit: stained positively for C-kit with a membranous pattern of reactivity (C-kit pattern I); b) Vascular Endothelial Growth Factor (VEGF): positive; c) Mean positive cells for the Ki-67 proliferation marker (400x field, 10 x 10mm reticulum): 8.
Following these results, the owners consulted a conventional oncologist and decided against chemotherapy, considering it to be too aggressive since the animal was clinically stable and already undergoing complementary therapy. No significant abnormalities were reported in the ultrasound (US) and X-ray examinations.
Follow-up I – May 2023: The owners reported that the animal was in excellent physical condition, with no notable changes. Blood samples were collected for additional biochemical assessments and a complete blood count (Table I). Chest X-ray and ultrasound imaging revealed no significant abnormalities. The following oral medications were prescribed: Histaminum 30CH + Apis 30CH + Graphites 30CH, 30 mL, 3 drops, SID for 60 days. For subcutaneous administration, the prescribed medications were: Viscum album D2, D3, D6, D9, D12, and D30 (Injectcenter, Brazil), 1 ampoule (1.1 mL), in combinations, SID for 60 days; Histaminum D35 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, every other day, for 60 days.
Follow-up II – July 2023: The owners reported that the animal remained in excellent physical condition, with no significant changes. Blood samples were collected for additional biochemical assessments and a complete blood count (Table I). Chest X-ray and ultrasound imaging revealed no significant abnormalities. A new protocol was established for oral administration: Histaminum 30CH + Apis 30CH + Graphites 30CH, 30 mL, 3 drops, SID for 60 days; Vitamin C 500 mg, 1 tablet, SID for 30 days. For subcutaneous administration, the following medications were prescribed: Viscum album D3 (Injectcenter, Brazil), 1 ampoule (1.1 mL), every other day, SID for 60 days; Histaminum D35 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, three times a week, for 60 days.
Follow-up III – September 2023: The owners reported that the animal remained in good physical condition but developed pasty stools that progressed to gastroenteritis. Blood samples were collected for biochemical assessments and a complete blood count (Table I). Chest X-ray and ultrasound imaging revealed no significant abnormalities. In response to the gastroenteritis episode, the treatment protocol was adjusted as follows: for oral administration, Staphysagria 30CH + Arsenicum album 30CH + Antimonium crudum 30CH, 30 mL, 3 drops, SID for 60 days; Vitamin D3 1000UI/drop, 4 drops, SID for 60 days; Enterogermin Plus, 1 blister, SID for 30 days. The patient underwent four intestinal microbiota transplantation procedures performed once a week. For subcutaneous administration, the following medications were prescribed: Viscum album D3 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, three times a week, for 90 days; Mercurius vivus D12 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID for 15 days.
Follow-up IV – December 2023: The owners reported that the animal was in excellent physical condition, with no notable changes. Blood samples were collected for biochemical assessments and a complete blood count (Table I). Chest X-ray and ultrasound imaging revealed no significant abnormalities. A new protocol was established for oral administration: Phosphorus 30CH, 30 mL, 3 drops, SID for 60 days; Vitamin C 500 mg, 1 tablet, SID for 60 days. For subcutaneous administration: Viscum album D3 (Injectcenter, Brazil), 1 ampoule (1.1mL), SID, three times a week, for 90 days; Thuya D9 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, three times a week, for 90 days.
Follow-up V – March 2024: The owners reported that the animal exhibited pruritus in the axillary region following the ingestion of a commercial snack. Blood samples were collected for biochemical assessments and a complete blood count (Table I). Chest X-ray and ultrasound remained unremarkable. A new protocol was established for oral administration: Histaminum 30CH + Apis 30CH + Rhus tox 30CH, 30 mL, 3 drops, BID for 30 days; Loratadine 10 mg, 1 tablet, SID for 3 days; Enterogermin Plus, 1 blister, SID for 30 days; Vitamin C 500 mg, SID for 30 days. Additionally, injectable Viscum album D2 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, twice a week, for 90 days.
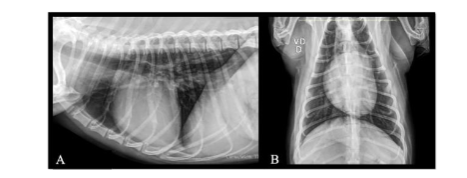
Follow-up VI – August 2024: The owners reported that the animal remained in excellent physical condition with no significant clinical changes. Blood samples were collected for biochemical assessments and a complete blood count. Chest X-ray and ultrasound imaging revealed no significant abnormalities. A new protocol was established for oral administration: Vitamin C 500 mg, 1 tablet, SID for 30 days; Vitamin D3 1000UI/drop, 4 drops, SID for 60 days; Macrogard Stick, 1 unit, SID for 60 days; Enterogermin Plus, 1 blister, SID, 3 times a week. For subcutaneous administration: Viscum album D3 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID, once a week, for 60 days.
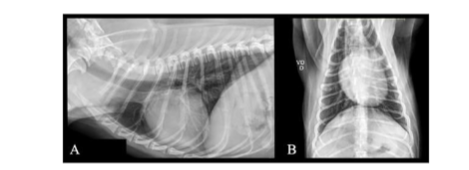
Follow-up VII – January 2025: The owners reported that the animal received an anti-rabies vaccination in December 2024. Twenty days post-vaccination, they observed a small nodule in the right axillary region. Consequently, a new protocol was prescribed: Arnica D12 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID for 10 days; Apis D4 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID for 10 days; Viscum album D3 (Injectcenter, Brazil), 1 ampoule (1.1 mL), SID for 10 days. In the same week, preoperative exams were conducted, including blood collection for complete blood count and biochemical assessments, chest X-ray, abdominal ultrasound, and echocardiography. No significant abnormalities were observed in the hematological, biochemical, chest X-ray, ultrasound, and echocardiographic findings (Table I). Subsequently, the patient underwent a surgical procedure (incisional biopsy) of the apparent nodule. Also, a puncture was performed in two places from where a fragment diagnosed with a Grade II MCT was removed in December 2022.
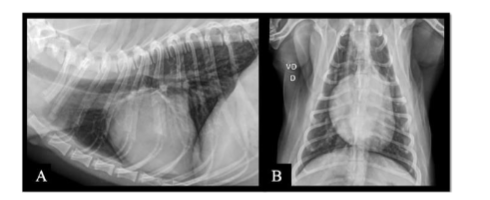
Follow-up VIII – February 2025: The owners reported that the animal was in excellent physical condition and demonstrated proper wound healing, with no other notable changes. The histopathological analysis of the local scar biopsy revealed a moderate focally extensive suppurative perianexal dermatitis, associated with edema and comedones. The nodule was diagnosed as a histiocytoma with free margins. Following this result, the patient was officially discharged from the initial diagnosis in December 2022. However, the animal remained under continuous follow-up every six months for medication to maintain overall health, treatment of dermatitis resulting from the biopsy, and ongoing care for the histiocytoma, which, despite free surgical margins, required an additional six months of treatment.
| Parameter | March/23 | July/23 | August/23 | November/23 | March/24 | August/24 | January/25 |
|---|---|---|---|---|---|---|---|
| Erythrocytes (uL) | 10,400,000 | 9,930,000 | 9,950,000 | 9,510,000 | 8,750,000 | 9,090,000 | 7,200,000 |
| Hemoglobin (g/dL) | 20.8 | 22.3 | 21.3 | 21.1 | 19.6 | 19.1 | 17.3 |
| Hematocrit (%) | 63.8 | 65 | 65 | 63.1 | 56.2 | 58.1 | 49 |
| Leukocytes (uL) | 10,600 | 10,300 | 8,000 | 9,600 | 15,200 | 7,800 | 12,200 |
| Lymphocytes (uL) | 1,908 | 2,780 | 1,200 | 1,632 | 2,430 | 2,262 | 1,708 |
| Platelets (uL) | 361,000 | 259,000 | 290,000 | 257,000 | 364,000 | 285,000 | 424,000 |
| ALT (U/L) | 105 | 468 | 161 | 99 | 93 | 107 | 57 |
| Creatinine (mg/dL) | 0.51 | 0.48 | 0.87 | 0.75 | 0.93 | 0.87 | 0.80 |
| Alkaline Phosphatase (U/L) | 12 | 28 | 75 | 26 | 20 | 26 | 24 |
| Urea (mg/dL) | 37 | 44 | 45 | 31 | 29 | 32 | 30 |
Discussion
Mast cell tumor (MCT) is a common skin neoplasm in dogs, classified into different grades based on histopathological features and disease prognosis. The present study describes a case of a grade II MCT in a young dog treated with an injectable homeopathic formulation of Viscum album. The tumor evolved favorably and did not recur in 24 months, despite the compromised lateral and deep margins of the removed tissue. Given that incomplete excision of MCTs is often associated with higher recurrence rates, the complete regression observed in this case highlights the possible role of Viscum album in modulating tumor progression.
Studies indicate that Viscum album, commonly known as mistletoe, possesses antitumor, immunomodulatory, and apoptotic properties. The use of this therapy in veterinary oncology has been explored as a less invasive alternative with fewer side effects compared to conventional treatments such as chemotherapy.
In the case presented, treatment with Viscum album resulted in complete regression of local tumor cells, as demonstrated in the new histopathology result performed in January 2025, suggesting a positive impact of therapy on MCT progression. The absence of tumor recurrence over 24 months, despite compromised surgical margins, supports a potential long-term benefit of Viscum album therapy in MCT management. While the use of Viscum album in veterinary oncology has been explored, reports on its efficacy in MCT cases remain limited, highlighting the novelty of this case. Similar findings have been reported in another clinical case involving a dog with grade II mast cell tumor treated with Viscum album, in which no recurrence was observed despite incomplete surgical margins, suggesting a potential role of this therapy as an adjuvant approach in MCT management.
A systematic review evaluating the use of Viscum album in companion animals reported that, although experimental and clinical studies suggest potential antitumor effects – particularly in melanomas, sarcomas, and mammary tumors – the number and quality of available studies remain limited. Therefore, current evidence is not sufficient to draw definitive conclusions regarding its efficacy, reinforcing the need for further controlled investigations in veterinary oncology.
This highlights an important gap in the literature, as most available data are derived from in vitro studies or small-scale clinical observations, with limited controlled trials in canine patients. Consequently, well-documented clinical cases such as the present report may contribute to hypothesis generation and guide the design of future prospective studies, particularly those investigating tumor microenvironment interactions in mast cell tumors.
From a clinical perspective, the management of mast cell tumors with incomplete surgical margins remains a challenge, as recurrence rates can vary depending on tumor grade and biological behavior. In such scenarios, adjunctive or integrative therapies may be considered, particularly when conventional treatments are declined or contraindicated. The present case illustrates a situation in which an integrative approach, consistently including Viscum album, was associated with long-term disease control, warranting further investigation.
Extracts from Viscum album contain viscotoxins and lectins, which play a key role in modulating the immune response and inducing apoptosis in cancer cells. Valle & Valle et al. emphasize that this therapy can be an effective adjuvant in controlling tumor growth, reinforcing its potential efficacy and safety in treating neoplasms. Additionally, studies by Biegel et al. suggest that Viscum album therapy can enhance the antitumor immune response, ultimately improving survival rates and quality of life in cancer patients.
In human oncology, Viscum album preparations have also been associated with improvements in quality of life parameters, including reduction of treatment-related adverse effects and enhancement of general well-being, further supporting its potential role as an adjunctive therapy. Although direct extrapolation to veterinary patients should be made with caution, these findings provide a relevant translational perspective.
Furthermore, the absence of adverse effects throughout the treatment period reinforces its safety, an essential consideration in the clinical management of canine neoplasms. The literature suggests that Viscum album may stimulate the immune system and inhibit tumor cell proliferation, which may help explain the favorable results observed in this case.
It is important to note that multiple therapeutic interventions were administered throughout the follow-up period, including supportive and homeopathic treatments. Therefore, the observed clinical outcome cannot be attributed exclusively to Viscum album. However, Viscum album was consistently included in the treatment protocols, which supports its potential contribution within an integrative therapeutic context.
The findings of this study align with previous research demonstrating the efficacy of Viscum album as either a complementary or primary therapy in veterinary cancer treatment. The young age of the patient may have been a decisive factor in the positive response observed. The dog was considerably younger than the average age of 8-9 years, as described by Kiupel et al., when MCT incidence is higher. Younger patients may exhibit a more robust immune response, potentially enhancing the therapeutic effects of Viscum album. In this case, 26 months after the appearance of the nodule, the patient showed no evidence of neoplastic cells at the original lesion site, where the excised tumor had initially displayed compromised margins. Additionally, the biological variability of grade II MCTs should be considered, as some tumors may present less aggressive behavior, which could have influenced the outcome. Therefore, further investigation in controlled clinical settings is essential to better understand the role of Viscum album in canine MCTs.
Future studies should aim to standardize treatment protocols, evaluate dose-response relationships, and investigate the specific mechanisms of action of Viscum album in canine mast cell tumors, in order to establish clearer clinical guidelines for its use.
Conclusion
The present study demonstrated the potential efficacy of injectable homeopathic Viscum album as an integrative therapeutic for treating MCT in a young dog. The initial histopathological analysis confirmed the diagnosis of MCT. After two years of treatment, a follow-up biopsy revealed significant tumor regression, with no clinical signs of recurrence. However, due to the use of multiple concurrent therapies and the single-case design, these findings should be interpreted with caution and should be considered hypothesis-generating. These findings suggest that this therapeutic approach may be a viable option, particularly in cases where surgery or chemotherapy pose significant risks, limitations, or adverse effects. Additionally, the results underscore the necessity for more controlled studies with larger sample sizes to further elucidate the mechanisms of action of Viscum album. When used judiciously and supported by clinical evidence, homeopathy represents a valuable complementary approach in veterinary oncology. It offers less invasive yet potentially effective alternatives for animal health and welfare. The continued exploration and expansion of this therapy could contribute to a broader range of treatment options in veterinary medicine.
Conflict of Interest Statement:
None.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Kiupel M, Webster JD, Bailey KL, et al. Proposal of a 2-tier histologic grading system for canine cutaneous mast cell tumors to more accurately predict biological behavior. Vet Pathol. 2011;48(1): 147-155. doi:10.1177/0300985810386469
- Patnaik AK, Ehler WJ, Macewen EG. Canine cutaneous mast cell tumors: morphologic grading and survival time in 83 dogs. Vet Pathol. 1984;21(5):469-474.
- Biegel U, von Bodungen U, Ruess K, et al. Mistletoe in adjuvant cancer treatment of companion animals. Planta Med. 2019;85:1392.
- Gardin NE. Immunological response to mistletoe (Viscum album L.) in cancer patients: a four-case series. Phytother Res. 2009;23(3):407-411. doi:10.1002/ptr.2643
- Wright A, Watanabe R, Koehler JW. European mistletoe (Viscum album) extract is cytotoxic to canine high-grade astrocytoma cells in vitro and has additive effects with mebendazole. Vet Sci. 2022;9(1):31. doi:10.3390/vetsci9010031
- Klingemann H. Viscum album (mistletoe) extract for dogs with cancer?. Front Vet Sci. 2024;10:1285354. Published 2024 Jan 3. doi:10.3389/fvets.2023.1285354
- Valle ACV, Valle MLP. Treatment and survival of a dog diagnosed with B-cell lymphoma using injectable Viscum album therapy: case report. Med Res Arch. 2025;13(2). doi:10.18103/mra.v13i2.6360
- Damiani GT, Bonamin LV, Pérez Hurtado EC. Use of Viscum album 5 LM as an adjuvant in treating a dog with cutaneous mastocytoma: a case report. Int J High Dilution Res. 2025;25(1):47-48. doi:10.51910/ijhdr.v25i1.1669
- Biegel U, Mevissen M, Schuller S, et al. Viscum album L., a therapeutic option for neoplastic diseases in companion animals? A systematic review. Complement Med Res. 2022;29(6):465-482. doi:10.1159/000525035
- Aponte-López A, Muñoz-Cruz S. Mast Cells in the Tumor Microenvironment. Adv Exp Med Biol. 2020;1273:159-173. doi:10.1007/978-3-030-49270-0_9
- Komi DEA, Redegeld FA. Role of Mast Cells in Shaping the Tumor Microenvironment. Clin Rev Allergy Immunol. 2020;58(3):313-325. doi:10.1007/s12016-019-08753-w
- Büssing A, Schietzel M. Apoptosis-inducing properties of Viscum album L. extracts from different host trees, correlate with their content of toxic mistletoe lectins. Anticancer Res. 1999;19(1A):23-28.
- Thronicke A, Schad F, Debus M, Grabowski J, Soldner G. Viscum album L. therapy in oncology: an update on current evidence. Complement Med Res. 2022;29(4):362-368. doi:10.1159/000524184
- Yosri N, Kamal N, Mediani A, et al. Immunomodulatory Activity and Inhibitory Effects of Viscum album on Cancer Cells, Its Safety Profiles and Recent Nanotechnology Development. Planta Med. 2024;90(14):1059-1079. doi:10.1055/a-2412-8471
- Valle ACV, Carvalho AC. Integrative oncology using Viscum album therapy improves quality of life in a dog diagnosed with oral fibrosarcoma: case report. Braz J Dev. 2022;8(3):17732-17754. doi:10.34117/bjdv8n3-149

