Mesenchymal Stem Cell Secretome for Chronic Wound Healing
Adjunctive Mesenchymal Stem Cell Secretome Therapy for a Chronic Hard to Heal Wound in a Patient with Multiple Comorbids: A Case Report
Nurul Hamizah Amran 1, Syafiqah Mohd Satali 1, Hamishatul Shamsiah Abdul Manaf 1, Norhasliza Zaina Abidin 1, Shukur Ahmad 2, Angelina Tiah 3
- Wound Care Unit Hospital Teluk Intan, Teluk Intan, Perak, Malaysia
- Orthopaedic Department Hospital Teluk Intan, Teluk Intan, Perak, Malaysia
- 23 Century International Life Science Centre, Seri Kembangan, Selangor, Malaysia
OPEN ACCESS
PUBLISHED: 31 December 2025
CITATION: Amran, NH., Satali, SM., et al., 2025. Adjunctive Mesenchymal Stem Cell Secretome Therapy for a Chronic Hard to Heal Wound in a Patient with Multiple Comorbids: A Case Report. Medical Research Archives, [online] 13(12). https://doi.org/10.18103/mra.v13i12.0000
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i12.0000
ISSN 2375-1924
ABSTRACT
Introduction: Chronic wounds in patients with complex comorbidities such as diabetes mellitus (DM), hypertension (HPT), and chronic kidney disease (CKD) are notoriously difficult to heal due to impaired angiogenesis, persistent inflammation, and fibroblast dysfunction which are frequently refractory to standard wound care. Mesenchymal stem cell (MSC) secretome, acellular biologic rich in growth factors, cytokines, and extracellular vesicles, has emerged as a promising therapeutic strategy demonstrated regenerative, angiogenic, and immunomodulatory properties capable of reactivating stalled wound healing processes.
Objectives: To evaluate the effectiveness of adjunctive MSC secretome therapy in a patient with a 7-year history of chronic, non-healing wound with multiple comorbidities who failed to respond to conventional and advanced wound care modalities.
Case Summary: Case report was conducted on a 67-year-old male with a stable DM, HPT, and CKD with a 7-year history of chronic wounds on the right lateral leg. MSC secretome (5 mL) was administered via subcutaneous injection twice weekly at the wound edges, alongside standard wound care based on the TIME framework. Wound surface area was calculated as length × width (cm2) and photographic progression were documented over 126 days.
Results: The total wound surface area initially measured 19.65 cm2. A transient increase to 33.32 cm2 was observed on Day 32 due to coalescence of wound margins, followed by steady and progressive reduction. By Day 126, the area had decreased to 2.4 cm2. Despite an initial increase in wound area due to margin coalescence, a steady reduction followed, culminating in an 87.79% total wound surface area reduction (from 19.65 cm² to 2.4 cm²). Clinical improvement in granulation tissue formation and partial to complete epithelialization was observed.
Discussion: In this case report, wound healing progressed steadily despite pale granulation, indicating that MSC secretome may exert its effects by activating cellular repair pathways through paracrine signalling before structural tissue changes are clinically apparent. MSC secretome components including growth factors, cytokines, and extracellular vesicles such as exosomes are believed to modulate inflammation, promote angiogenesis, and stimulate fibroblast activity, which may help overcome the impaired healing typical in patients with multiple comorbidities.
Conclusion: MSC secretome therapy demonstrated substantial benefit in accelerating wound healing in a complex, multimorbid patient with long standing hard to heal chronic wounds. Its multimodal actions, anti-inflammatory, pro-angiogenic, and regenerative, may help shift chronic wounds into an active healing phase and hold promise as a safe, effective adjunct in chronic wound management.
Keywords
Mesenchymal stem cell, secretome, chronic wound, diabetes mellitus, hypertension, chronic kidney disease, wound healing
Introduction
Chronic wounds remain a major therapeutic challenge, particularly among individuals with diabetes mellitus, hypertension, and chronic kidney disease. These systemic conditions adversely affect key processes involved in wound healing, including angiogenesis, neural integrity, microvascular perfusion, inflammatory regulation, and extracellular matrix synthesis, which ultimately impair normal tissue repair 1-3. As a result, chronic wounds remain stalled in the inflammatory phase for extended periods, failing to respond to standard or advanced wound care despite adherence to established principles and the use of advanced dressings 1,2.
Current evidence indicates that non-healing diabetic and chronic wounds arise from dysregulated cellular responses, oxidative stress, persistent inflammatory signalling, and inadequate activation of pro-healing molecular pathways3,4. These abnormalities impair the wound’s ability to progress into the proliferative phase. Conventional wound therapies do not sufficiently address or reverse these underlying pathophysiologic disturbances, emphasizing the need for regenerative strategies capable of restoring a more favourable wound microenvironment.
Mesenchymal stem cell (MSC)–based therapies have emerged as an important therapeutic modality in regenerative medicine due to their immunomodulatory, pro-angiogenic, and tissue-repair properties 5-7. Clinical studies and meta-analyses indicate that MSC-related therapies are generally safe, with adverse events typically mild and self-limiting 8. Increasing evidence suggests that the therapeutic benefits of MSCs are mediated predominantly through paracrine signalling rather than direct cellular engraftment 6. This has shifted scientific interest toward the MSC secretome, a cell-free biologic containing a complex mixture of cytokines, chemokines, growth factors, and extracellular vesicles, including exosomes 5,6.
Secretome-based interventions have been shown to enhance keratinocyte migration, promote endothelial repair, modulate macrophage phenotypes, regulate inflammatory activity, and facilitate extracellular matrix remodelling 5,6. In diabetic wound models, MSC-derived secretome activates key intracellular signalling pathways, most notably PI3K/AKT, which promote cell survival, angiogenesis, and granulation tissue formation 4. Compared with cell-based MSC therapy, secretome-based approaches offer several practical advantages, including lower immunogenicity, greater stability during storage, and improved ease of standardisation, handling, and administration. These attributes make the MSC secretome particularly suitable for outpatient care and resource-limited clinical environments.
Given these therapeutic attributes, MSC secretome has emerged as a valuable adjunct for chronic wounds that remain refractory to conventional management. This case report describes the clinical application of adjunctive MSC secretome therapy in a patient with a long-standing, non-healing leg wound complicated by multiple comorbidities.
Case Presentation
Mr. AA, a 67-year-old gentleman with Type II diabetes mellitus (DM), hypertension, and Stage 4 chronic kidney disease (CKD) secondary to diabetic nephropathy, was referred to the Wound Care Clinic (WCC), Hospital Teluk Intan for management of a chronic non-healing wound over the right lateral leg. The wound originated following surgical debridement for necrotizing fasciitis in 2018. Despite 7 years of continuous outpatient wound care at a community clinic including intermittent sharp debridement and application of multiple advanced dressings (silver hydrofiber, alginate, hydrocolloid, foam dressings), the ulcer failed to achieve closure.
His DM was moderately controlled on oral hypoglycemic agents, with recent HbA1c 5.7. CKD remained stable, and he was not on renal replacement therapy. Baseline investigations at presentation included haemoglobin 10.8 g/dL, serum albumin 36 g/L, and eGFR 24 mL/min/1.73 m2. His ankle–brachial systolic index was 1.2 bilaterally, indicating adequate arterial inflow.
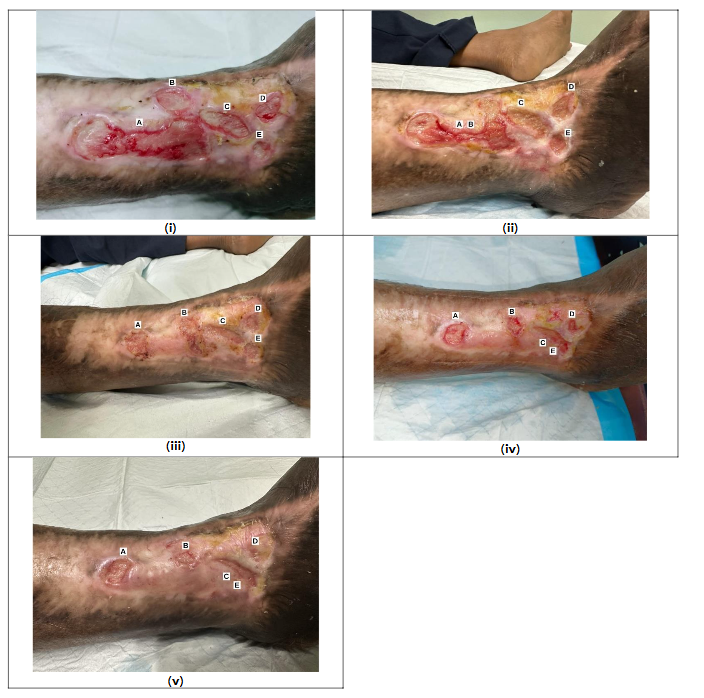
In March 2025, MSC secretome injections were started as an adjunctive therapy. At baseline, five irregular chronic ulcers (Wounds A–E) were noted along the right lateral lower leg. The combined wound surface area measured 19.65 cm2. Wound beds contained approximately 40% healthy red granulation tissue and 60% pale granulation. The edges were flat without epibole or undermining. Exudate was moderate, serous, and odorless. Periwound skin was intact with no maceration. There were no clinical signs of local infection.
Given the wound chronicity, persistent pale granulation despite adequate perfusion, limited response to years of optimized standard therapy, and stable systemic parameters, the patient was considered an appropriate candidate for adjunctive MSC secretome therapy. The off-label nature, potential benefits, and risks of the treatment were explained, and written informed consent was obtained.
Treatment Protocol
The patient received treatment in the outpatient setting of the Wound Care Clinic (WCC), Hospital Teluk Intan, Malaysia. Wound management was conducted according to the TIME (Tissue, Inflammation/Infection, Moisture, Edge) framework. For each wound, surface area was calculated as a product of length × width (in cm2) and photographic documentation were obtained and recorded at each WCC visit to monitor healing progression.
At each session, wounds were cleansed with a surfactant-based antimicrobial solution. Following cleansing, 5 mL of MSC secretome was administered via subcutaneous injection at the wound edges, performed twice weekly under aseptic technique. Wound-edge injections were used to target the zone of epithelial advancement and stimulate fibroblast activation. Moisture balance was maintained using secondary absorbent dressing.
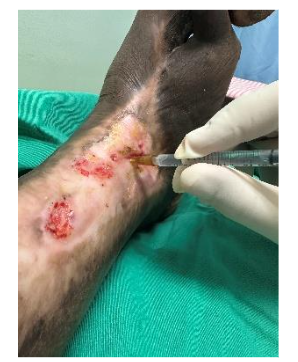
The patient was observed immediately after each session and at subsequent visits for adverse events including injection-site pain, hypersensitivity reactions, erythema, hematoma, or systemic symptoms. Apart from brief, self-limiting pain during injection, no adverse events were recorded throughout the treatment course.
Results
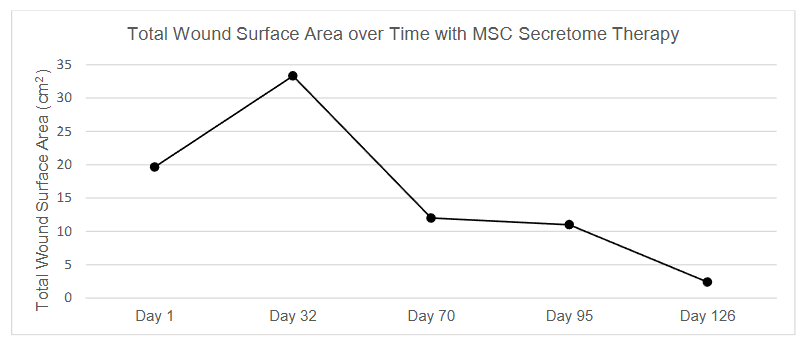
Figure 2 shows serial wound measurements recorded over 126 days to assess the wound healing response following adjunctive MSC secretome therapy. Total wound surface area reduced by 87.79% from 19.65cm2 to 2.4cm2 throughout this study.
At baseline (Day 1), five wounds (A–E) were present on the patient’s right lateral leg, with total wound surface area of 19.65 cm2. The wound beds consisted of 40% healthy granulation tissue and 60% pale granulation tissue, with moderate exudate and healthy periwound skin.
By Day 32, the total wound surface area increased to 33.32 cm2, due to the coalescence of wounds A and B, rather than clinical deterioration. Despite this temporary increase total wound surface, a progressive reduction in wound size was observed afterwards. By Day 70, the total area had reduced to 12.0 cm2, and by Day 95, it further decreased to 11.0 cm2. On Day 126, the total wound surface area was 2.4 cm2. Figure 3 shows reduction of total wound surface area over time.
Wound D achieved complete epithelialization, Wound A and B demonstrated 89.09% and 80% reduction, respectively. Although some wound beds remained pale throughout the treatment period, all wounds showed progressive reduction in size, and the periwound skin remained intact and healthy.
The only adverse event reported was transient self-limiting pain at the injection site during the subcutaneous injection. The pain resolved immediately after each injection, no additional analgesia was required, and no local or systemic allergic reactions or other adverse events were observed during the 126-day follow-up.
Discussion
This case demonstrated a significant wound healing response after the application of MSC secretome therapy in a patient with chronic hard to heal wounds with comorbidities. Significantly, healing progressed even in the presence of pale granulation tissue, an observation suggesting that biochemical signalling from the secretome may reactivate stalled repair processes before visible vascularization or granulation happens. The consistent integrity of the periwound skin throughout also reflects a stable local wound environment during treatment.
This observation indicates that biochemical signalling from MSC secretome may trigger cellular repair prior to the onset of visible vascularization or significant granulation. MSC secretomes contain growth factors (e.g., VEGF, PDGF, TGF-β), anti-inflammatory cytokines (e.g., IL-10), and extracellular vesicles including exosomes that activate fibroblasts, modulate inflammation, and promote angiogenesis through paracrine pathways 5,7.
Ibrahim R. et al. noted that such components can stimulate wound healing even under ischemic or metabolically compromised conditions. This may explain the clinical finding that wound contraction and epithelialization occurred despite the pale wound bed. These components collectively orchestrate the transition of chronic wounds from an inflammatory phase to a proliferative and remodeling phase by engaging multiple biological pathways:
- Immunomodulation: The MSC secretome modulates chronic inflammation by downregulating TNF-α and IL-6 and upregulating IL-10. This immunoregulatory effect is particularly important in chronic diabetic wounds, which usually stalled in a prolonged inflammatory phase. Clinical and preclinical studies in diabetic foot ulcer models showed that MSC therapy lowered TNF-α and IL-6 levels while increasing IL-10, which helped resolve inflammation, control infection, and promote tissue repair 8. Findings from umbilical cord-derived MSC secretome research further support these immunomodulatory properties, showing suppression of inflammatory pathways and a shift toward a pro-regenerative microenvironment 9.
- Fibroblast Activation and Extracellular Matrix (ECM) Remodeling: Fibroblast proliferation and migration are promoted by secreted growth factors, leading to increased collagen synthesis and matrix remodeling. TGF-β in the secretome aids in ECM deposition and maturation, which is essential for wound closure and tensile strength restoration 7.
- Angiogenesis and Neovascularization: Angiogenesis and microvascular repair likely played a central role in the patient’s wound progression, particularly given the early presence of pale granulation. MSC secretome contains angiogenic factors such as vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF) and hepatocyte growth factor (HGF), along with microRNA-rich exosomes that stimulate endothelial proliferation and vascular regeneration 6,10-12. These biochemical processes are known to occur before visible granulation develops. Preclinical studies have shown that MSC-derived exosomes increase endothelial tube formation and capillary density 3,13, supporting the clinical observation that wound contraction and epithelial advancement continued despite the initial lack of granulation colour. Collectively, these angiogenic mechanisms provide a biologically coherent explanation for the patient’s sustained healing trajectory despite hypoxic, chronic wound environment.
- Cell Recruitment and Proliferation: Chemokines within the secretome, such as SDF-1 (stromal cell-derived factor 1), attract endothelial progenitor cells, macrophages and fibroblast 5 which in turn support angiogenesis and ECM deposition. This aligns with the improved wound edge and consistent granulation tissue noted across serial assessment.
- Promotion of Keratinocyte Migration and Re-epithelialization: Epidermal growth factor (EGF), HGF, and other keratinocyte-stimulating factors found in the secretome accelerate re-epithelialization by enhancing keratinocyte migration and proliferation, facilitating wound closure 6,7. This mechanism likely facilitated epithelial advancement despite years of prior non-healing.
- Antioxidant and Anti-apoptotic Effects: In diabetic wounds, oxidative stress and apoptosis impair cell survival and function. MSC-derived secretome contains anti-oxidative enzymes (e.g., SOD, catalase) and anti-apoptotic factors that protect local cells and enhance tissue survival under ischemic and inflammatory conditions 6,8. These cytoprotective actions likely supported the uninterrupted healing trajectory and absence of regression throughout the course of treatment period.
The clinical observation of consistent wound healing and epithelialization despite minimal early visible granulation highlights the early biochemical influence of MSC secretome, likely reprogramming the wound microenvironment toward regeneration. This biologic shift may be particularly valuable in patients with diabetes and CKD, where vascular insufficiency, cellular senescence, and immune dysfunction hinder healing.
Importantly, the patient’s diabetes was well controlled (HbA1c 5.7), a systemic condition known to enhance the likelihood of timely wound repair. Adequate glycaemic control mitigates hyperglycemia-induced impairments in leukocyte function, endothelial responsiveness, and extracellular matrix deposition, thereby reducing risks of delayed healing, microbial proliferation, and secondary infection 14-16. Patients with near-normoglycemic profiles are also less likely to exhibit functional immunocompromise, enabling a more coordinated inflammatory and proliferative response 14. Within this physiologic context, a more predictable healing trajectory was anticipated, allowing MSC secretome therapy to exert its regenerative and immunomodulatory effects without interruption from infectious complications 15,17.
The only adverse effect observed was transient injection-site pain, which resolved spontaneously and is consistent with the mild, self-limiting local reactions most frequently reported in clinical studies of MSC-based therapies 18, while controlled safety data for secretome-specific preparations remain limited 19.
Although this single-case study without a control group cannot confirm a direct effect of MSC secretome, the consistency of wound size reduction, following years of failed conventional therapy, suggests that MSC secretome may serve as a biological adjunct that helps shift chronic wounds into an active healing state. Further clinical studies are needed to determine optimal dosing, treatment intervals, and patient selection criteria.
Conclusion
MSC secretome therapy demonstrated substantial benefit in accelerating wound healing in a complex, multimorbid patient with long-standing chronic wounds. Its multimodal actions, anti-inflammatory, pro-angiogenic, and regenerative, may help shift chronic wounds into an active healing phase and hold promise as a safe, effective adjunct in chronic wound management.
Ethics Statement
Written informed consent was obtained from the patient for both treatment and publication of anonymised clinical data and images. The MSC secretome therapy was provided on an off-label basis as part of clinical care, and no formal research protocol was undertaken. Case reports do not generally require prospective ethics approval when patient consent is documented.
Sponsorship
The mesenchymal stem cell secretome used in this study were provided by 23 Century International Life Science Centre. The sponsor had no role in study design, data collection, data analysis, or manuscript preparation. All other aspects of the study were conducted independently by the authors without financial support.
References
- Nunan R, Harding KG, Martin P. Clinical challenges of chronic wounds: searching for an optimal animal model to recapitulate their complexity. Dis Model Mech. Nov 2014;7(11):1205-13. doi:10.1242/dmm.016782
- Guo S, Dipietro LA. Factors affecting wound healing. J Dent Res. Mar 2010;89(3):219-29. doi:10.1177/0022034509359125
- Wu S, Zhou Z, Li Y, Jiang J. Advancements in diabetic foot ulcer research: Focus on mesenchymal stem cells and their exosomes. Heliyon. Sep 15 2024;10(17):e37031. doi:10.1016/j.heliyon.2024.e37031
- Huang J, Deng Q, Tsang LL, et al. Mesenchymal stem cells from perinatal tissues promote diabetic wound healing via PI3K/AKT activation. Stem Cell Res Ther. Feb 8 2025;16(1):59. doi:10.1186/s13287-025-04141-8
- Ibrahim R, Mndlovu H, Kumar P, Adeyemi SA, Choonara YE. Cell Secretome Strategies for Controlled Drug Delivery and Wound-Healing Applications. Polymers. Jul 20 2022;14(14) doi:10.3390/polym14142929
- Vizoso FJ, Eiro N, Cid S, Schneider J, Perez-Fernandez R. Mesenchymal Stem Cell Secretome: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. Int J Mol Sci. Aug 25 2017;18(9) doi:10.3390/ijms18091852
- Ferreira JR, Teixeira GQ, Santos SG, Barbosa MA, Almeida-Porada G, Gonçalves RM. Mesenchymal Stromal Cell Secretome: Influencing Therapeutic Potential by Cellular Pre-conditioning. Front Immunol. 2018;9:2837. doi:10.3389/fimmu.2018.02837
- Mahmoudvand G, Karimi Rouzbahani A, Razavi ZS, Mahjoor M, Afkhami H. Mesenchymal stem cell therapy for non-healing diabetic foot ulcer infection: New insight. Front Bioeng Biotechnol. 2023;11:1158484. doi:10.3389/fbioe.2023.1158484
- Li X, Zhang D, Yu Y, Wang L, Zhao M. Umbilical cord-derived mesenchymal stem cell secretome promotes skin regeneration and rejuvenation: From mechanism to therapeutics. Cell Prolif. Apr 2024;57(4):e13586. doi:10.1111/cpr.13586
- Marofi F, Alexandrovna KI, Margiana R, et al. MSCs and their exosomes: a rapidly evolving approach in the context of cutaneous wounds therapy. Stem Cell Res Ther. Dec 4 2021;12(1):597. doi:10.1186/s13287-021-02662-6
- Kishta MS, Hafez AM, Hydara T, et al. The transforming role of wharton’s jelly mesenchymal stem cell-derived exosomes for diabetic foot ulcer healing: a randomized controlled clinical trial. Stem Cell Res Ther. Oct 13 2025;16(1):559. doi:10.1186/s13287-025-04690-y
- Zi Y, Li J, Qian X, et al. Human umbilical cord mesenchymal stem cell exosomes promote elastin production and acute skin wound healing via TGFbeta1-Smad pathway. Mol Cell Biochem. Jul 2025;480(7):4499-4511. doi:10.1007/s11010-025-05264-5
- Shabbir A, Cox A, Rodriguez-Menocal L, Salgado M, Van Badiavas E. Mesenchymal Stem Cell Exosomes Induce Proliferation and Migration of Normal and Chronic Wound Fibroblasts, and Enhance Angiogenesis In Vitro. Stem Cells Dev. Jul 15 2015;24(14):1635-47. doi:10.1089/scd.2014.0316
- Lecube A, Pachon G, Petriz J, Hernandez C, Simo R. Phagocytic activity is impaired in type 2 diabetes mellitus and increases after metabolic improvement. PLoS One. 2011;6(8):e23366. doi:10.1371/journal.pone.0023366
- Dasari N, Jiang A, Skochdopole A, et al. Updates in Diabetic Wound Healing, Inflammation, and Scarring. Semin Plast Surg. Aug 2021;35(3):153-158. doi:10.1055/s-0041-1731460
- Huang Y, Kyriakides TR. The role of extracellular matrix in the pathophysiology of diabetic wounds. Matrix Biol Plus. May 2020;6-7:100037. doi:10.1016/j.mbplus.2020.100037
- Dronge AS, Perkal MF, Kancir S, Concato J, Aslan M, Rosenthal RA. Long-term Glycemic Control and Postoperative Infectious Complications. Archives of Surgery. 2006;141(4):375-380. doi:10.1001/archsurg.141.4.375
- Lalu MM, McIntyre L, Pugliese C, et al. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoS One. 2012;7(10):e47559. doi:10.1371/journal.pone.0047559
- Thompson M, Mei SHJ, Wolfe D, et al. Cell therapy with intravascular administration of mesenchymal stromal cells continues to appear safe: An updated systematic review and meta-analysis. EClinical Medicine. Feb 2020;19:100249. doi:10.1016/j.eclinm.2019.100249

