Mesothelioma Trends in the US: Age-Period-Cohort Analysis
Updated Age-Period-Cohort Analysis of Mesothelioma Trends in the United States from 1973 to 2022
Alesia Jung¹, Nnaemeka Odo², Anne E. Loccisano³, and Brent D. Kerger⁴
- Exponent Inc., Menlo Park, CA USA
- Optum, Atlanta, GA USA
- Exponent Inc., Alexandria, VA USA
- Exponent Inc., Tampa, FL USA
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Jung, A., et al., 2026. Updated Age-Period-Cohort Analysis of Mesothelioma Trends in the United States from 1973 to 2022. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7203
ISSN 2375-1924
ABSTRACT
Background: Malignant mesothelioma is a rare cancer primarily associated with occupational asbestos exposure impacting mesothelial cells. Despite regulations against asbestos use, this cancer continues to be a public health concern given its long latency and shifting age cohorts older populations with higher risk historical exposures and younger cohorts with lower rates, potentially distorting the true picture of population incidence risk and trends. The objective of this study was to assess the current role of age, period, and cohort (APC) effects on malignant mesothelioma incidence. This study extends previous work which examined cases reported up until 2013 through 2022.
Methods: Incident mesothelioma cases (pleural and total) were identified using SEER-8 cancer registry data (1975-2022). Effects of mesothelioma incidence trends were described utilizing the US National Cancer Institute’s APC analysis web tool, estimating 5-year age intervals, calendar periods, and birth cohorts, stratified by age and gender groups (male and female; age 0-74 and 75+ years old).
Results: Annual incidence of mesothelioma among younger males (aged 0-74 years) has significantly decreased: -1.8%/year for all mesothelioma (95% CI: -3.3, -0.3) and -2.2%/year for pleural mesothelioma (95% CI: -4.0, -0.4). Comparatively, net drifts for mesothelioma incidence among older adults (75+ years) increased. Birth-cohort-adjusted incidence among younger males declined following a peak during the period of 1980-1984. Meanwhile, rates have continued to increase among older adults but remained mostly unchanged among younger females. Temporal patterns in mesothelioma incidence varied across age groups, suggesting birth cohort effects. The peak birth cohort for pleural and total mesothelioma incidence among younger males was 1917-1921, while flatter patterns of change among females and older males made the identification of peaks less definitive.
Conclusions: Results support an association between 1970s asbestos regulations with declining occupational asbestos exposures post-World War II, and declining mesothelioma incidence among younger males, suggesting disappearing occupational asbestos causal risk for malignant mesothelioma. Increasing rates in the older age group may be attributable to longevity-related factors influencing later onset risk, including disease latency, aging and its associated decline in immune function, radiotherapy and chemotherapy from prior cancer treatments, and genetic susceptibility.
Keywords
epidemiology, total mesothelioma, pleural mesothelioma, APC analysis web tool, longevity effect, asbestos, chemotherapy, radiotherapy
INTRODUCTION
Mesothelioma is a rare and aggressive malignancy which primarily occurs in the lungs (pleural mesothelioma PM) and has been mainly associated with historical occupational asbestos exposure in the United States (US) and globally. Other notable sites of this malignancy include the peritoneum and testis. Despite the US Occupational Safety and Health Act (OSHA) in 1970 to address high workplace asbestos exposures, and associated regulations to limit asbestos use, new cases have continued to emerge decades after this era of high exposures due in part to the long latency period for developing associated malignancy. Better understanding of cohort trends, and risk factors associated with malignant mesothelioma is important for continued effective public health prevention and management. The Surveillance, Epidemiology, and End Results (SEER) program is a cancer registry maintained by the National Cancer Institute which provides comprehensive cancer incidence and survival estimates in the US. Age-period-cohort (APC) analysis allows for the delineation of the effects of age, calendar period, and birth cohort on cancer trends.
Globally, pleural mesothelioma incidence trends vary widely. In Europe, the EU Directive 2023/2668 lowered asbestos exposure limits and mandated electron microscopy for fiber detection a significant improvement over prior optical methods. Italy’s Lombardy region is especially noteworthy for its mesothelioma registry and research efforts, documenting over 7,900 cases between 2000 and 2020. Globally, these cases frequently relate to occupational exposure in shipbuilding, agriculture, and construction, and additional insights from Asia, Africa, Australia, and South America further contextualize the United States experience, as global data show discrepancies shaped by regulation, diagnostics, and industrial practices.
This report updates prior APC analyses through 2013 of incident malignant mesothelioma trends in the United States using data from the SEER cancer database extending the results through 2022. It aims to further elucidate cohort patterns in the US workforce with potential exposure to asbestos and age-related mesothelioma incidence trends among persons with later onset (age 75+) that may be attributable to longevity-related factors.
METHODS
We analyzed incident pleural (PM) and total mesothelioma cases identified using data published by the US SEER-8 cancer registry database for cases diagnosed between 1975-2022. All cases of primary malignant mesothelioma (International Classification of Diseases for Oncology, 3rd edition (ICD-O-3) histology codes 9050-9055) and pleural mesothelioma (ICD-O-3 histology codes 9050-9055 and ICD-O-3 site codes C38.4-38.8) were considered. Incident cases were stratified by sex (male, female) and age categories (0-74 years, 75+ years) for analyses. Age categories were selected based on demographics related to post-retirement longevity and age-related PM trends associated with occupational asbestos exposure. Our prior study utilized data from SEER-9 (1973-2013) but the recent discontinuation of reporting for certain registries determined the use of SEER-8 for similar coverage of the study period for the purposes of evaluating longevity and occupational trends. The older cohort (aged 75+ years) was of particular interest due to rising malignant mesothelioma rates in aging populations and potential links to longevity factors such as immune decline and prior cancer therapies.
Stratification by age and gender was intended to help isolate occupational exposure patterns and identify non-occupational contributors to disease burden. Data from SEER were accessed and analyzed using SEER*Stat statistical software (version 9.0.41). Data were further stratified by 5-year age group (0-4, 5-9, 10-14 80-84, 85-89, 90+) and 5-year calendar year of diagnosis (1975-1979, 1980-1984, 2010-2014, 2015-2019) for APC analyses. The years 2020-2022 were excluded from APC analyses due to the sparsity of data required to evaluate a 5-year calendar period (2020-2024). Analyses were performed using the National Cancer Institute’s (NCI’s) publicly available APC analysis web tool, estimating 5-year age intervals, calendar period, and birth cohort stratification by age and sex groups (younger males aged 0-74 years; younger females aged 0-74 years; older males aged 75+ years; older females aged 75+ years).
The NCI’s APC web tool provides researchers with an interface to access a panel of interpretable and estimable APC functions and corresponding Wald tests to investigate emerging patterns and trends related to cancer incidence and mortality. Parameters and functions are estimated using weighted least squares and the assumption that the count data follows a Poisson distribution and allowing for extra-Poisson variation. The APC web tool was used to determine estimates and corresponding 95% confidence intervals (CIs) for parameters including net drift (overall annual percentage change), local drifts (annual percentage change in each age group), period rate ratios (age-specific rates in each calendar period compared to the reference period), cohort rate ratios (age-specific rates in each birth cohort compared to the reference cohort), fitted temporal trends (expected rates over time in the reference age group adjusted for cohort effects), and cross-sectional age trend (CAT, how quickly rates change with age among the reference period, adjusting for cohort effects), and longitudinal age trend (LAT, how quickly rates change with age in the reference birth cohort, adjusting for period effects) among the sex and age groups of interest.
Age-period cohort functions were also utilized to assess the stability, log-linearity, and equality of observed trends using corresponding Wald tests (p-value<0.05 was considered statistically significant). The calendar period 1990-1994, the birth cohort periods of 1932-1936 for those aged 0-74 years and 1908-1912 for those aged 75+ years, and the age groups 55-59 among those aged 0-74 years and 80-84 among those aged 75+ years were selected as reference groups in APC analyses.
RESULTS
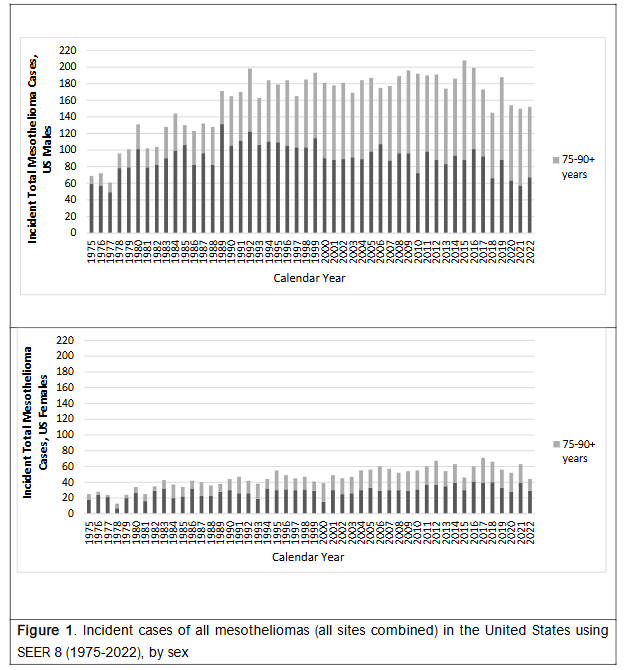
Between 1975-2022, the total number of incident cases of all mesothelioma cases diagnosed annually generally increased in men, particularly among men aged 75-90+ years in the United States, and remained mostly similar among women (Figures 1).
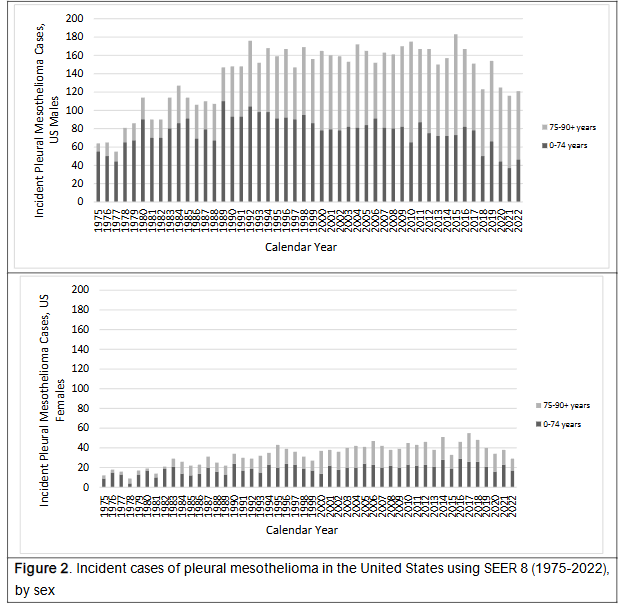
Similarly, incidence of pleural mesothelioma increased over the study period, with a slight flattening to drop in the last five years, with this trend more demonstrable among men especially 75-90+ years. In the same period, the incidence of pleural mesothelioma among women remained generally stable (Figure 2).
Estimates of the net drift (Table 1) between 1975-2022, indicate that net drift for mesothelioma incidence varied by sex and age groups. The annual incidence of mesothelioma among males aged 0-74 years has significantly decreased; -1.8% per year for all mesothelioma (95% CI -3.3, -0.3) and -2.2% per year for pleural mesothelioma (95% CI -4.0, -0.4). Among women aged 0-74 years, non-significant decreases in net drift were observed. In comparison, the net drifts for mesothelioma incidence among both men and women in the older age group were significantly increased. For men aged 75+ years the net drift was 3.7% (95% CI 0.8, 6.5) for total mesothelioma and 3.6% (95% CI 0.9, 6.4) for pleural mesothelioma. For women aged 75+ years the net drift was 2.8% (95% CI 1.8, 4.3) for total mesothelioma and 4.8% (95% CI 1.3, 7.4) for pleural mesothelioma.
| Males, 0-74 years | Males, 75+ years | Females, 0-74 years | Females, 75+ years | |
|---|---|---|---|---|
| Total All Mesothelioma | -1.8% (-3.3, -0.3) | +3.7% (+0.8, +6.5) | -0.6% (-2.1, +1.0) | +2.8% (+1.3, +4.3) |
| Pleural Mesothelioma | -2.2% (-4.0, -0.4) | +3.6% (+0.9, +6.4) | -1.8% (-3.9, +0.3) | +4.3% (+1.3, +7.4) |
Age effects on mesothelioma incidence after adjusting for either period or birth cohort effects, measured with CATs and LATs, showed variation by age and sex. For total mesothelioma, among the younger age group (0-74 years), the age effect was greater in males [CAT 12.6% (95% CI 11.0, 14.3); LAT 10.8% (95% CI 8.7, 12.9)] than in females [CAT 9.6% (95% CI 8.0, 11.1); LAT 9.0% (95% CI 6.7, 11.2)]. However, the age effect was also greater among the younger age group compared to the older age group, for example, CAT of males aged 75+ years was -3.1% (95% CI -6.2, 0.01) and LAT was 0.6% (95% CI -0.7, -1.8). Similar estimates and trends were observed for pleural mesothelioma (Tables 2 and 3).
The trend in the ratio of LAT vs. CAT, representing the influence of net drift on age-associated natural history, was negative among younger males and females (0-74 years) and positive among older males and females (75+ years), except for all mesothelioma among older females, where the trend was flat/positive.
Wald tests for heterogeneity indicated that temporal trends adjusted for birth cohort effects were not stable among males (0-74 and 75+ years) and older females (75+ years) but stable among younger females (0-74 years). This suggested that there were significant period effects among all males and older females, but not younger females. Birth cohort effects-adjusted analyses showed declining incidence rates of pleural and total mesothelioma in males aged 0-74 years over time following a peak in incidence during the calendar period of 1980-1984. Meanwhile, increasing rates were observed in 75+ year old males. Among females, there were no appreciable changes observed in incidence rates over time in females 0-74 years after adjustment for birth cohort effects, while significantly increasing rates were observed for females aged 75+ years.
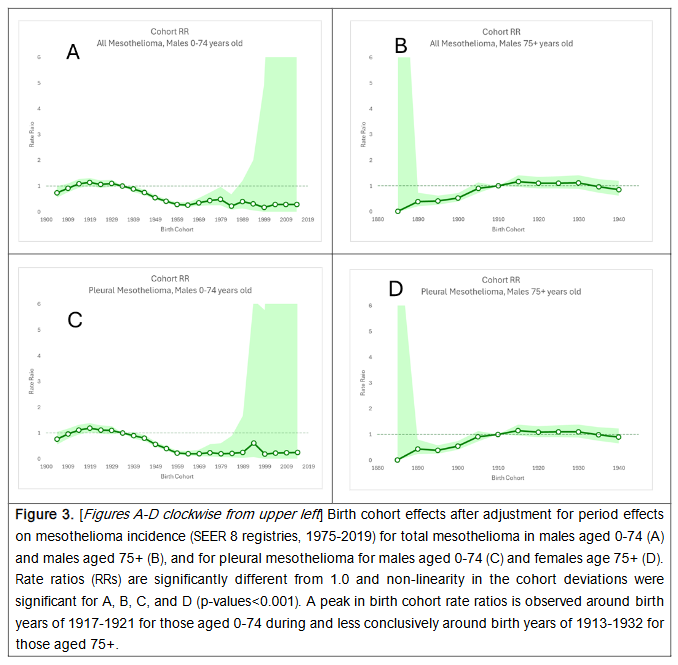
Statistical tests comparing the difference between local drifts and net drift were significant across all age and sex groups considered indicating that temporal patterns in mesothelioma incidence varied across age groups and that significant birth cohort effects were present. The respective peak birth cohorts for pleural and total mesothelioma incidence among males were identified as 1917-1921 (0-74 years) and 1913-1917 (75+ years) for males based on birth cohort rate ratios. These birth years correspond to birth cohorts who potentially entered the US workforce during the World War II era (age 18+ years). However, it should be noted that while the peak birth cohort was clear among males aged 0-74 years, the distinction among those aged 75+ was less clear, as rate ratios associated with birth years ranging from 1913-1932 were similar.
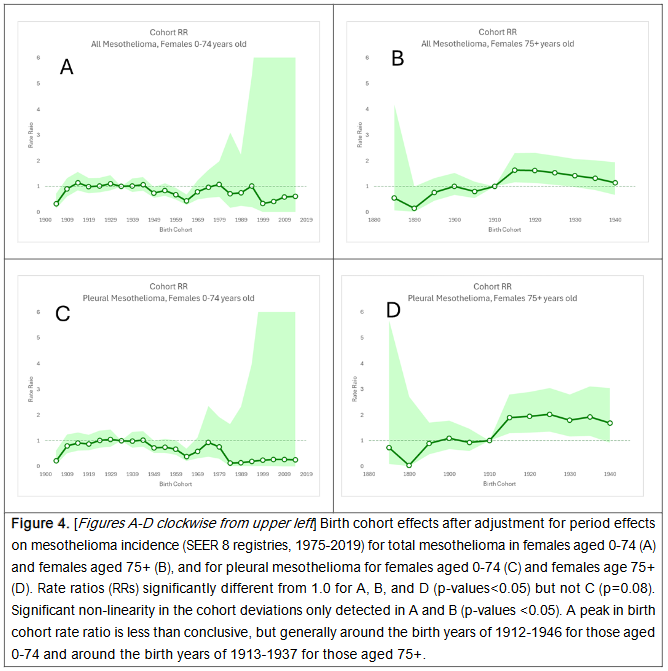
Peak birth cohorts for females aged 0-74 years differed for total mesothelioma (1912-1916) and pleural mesothelioma (1927-1931) and similarly differed for females aged 75+ years (1913-1917 for total mesothelioma and 1923-1927 for pleural mesothelioma). However, the identification of a birth cohort of peak incidence, adjusting for period effects, lacked clear distinction, as similar period-adjusted incidence rates were observed generally around the birth cohort years of 1912-1946 among females aged 0-74 years and around 1913-1937 for females aged 75+ years.
| APC Parameters | Males, 0-74 yrs | Males, 75+ yrs | Females, 0-74 yrs | Females, 75+ yrs |
|---|---|---|---|---|
| Age Effects | Cross-sectional age trend (95% CI) 12.6% (11.0%, 14.3%) | -3.1% (-6.2%, 0.01%) | +9.6% (+8.0%, +11.1%) | -4.5% (-6.7%, -2.2%) |
| Longitudinal age trend (95% CI) 10.8% (8.7%, 12.9%) | +0.6% (-0.7%, +1.8%) | +9.0% (+6.7%, +11.2%) | -1.7% (-3.6%, +0.2%) | |
| Longitudinal vs. cross-sectional RR trend | Negative | Positive | Negative | Positive/Flat |
| Age deviation is non-linear? | Yes | Yes | No | Yes |
| p-value | <0.05 | <0.001 | 0.47 | <0.05 |
| Period Effects | Calendar period with peak incidence after adjustment for cohort effects 1980-1984 | 2015-2019 | 1980-1984 | 2015-2019 |
| Net drift=0 (Temporal trends stable over time?) | No | No | Yes | No |
| p-value | 0.02 | 0.01 | 0.5 | <0.001 |
| Period RRs different from reference period? | Yes | No | No | Yes |
| p-value | <0.05 | 0.1 | 0.65 | <0.01 |
| Period deviation is non-linear? | Yes | No | No | No |
| p value | <0.05 | 0.42 | 0.59 | 0.3 |
| Cohort Effects | Birth cohort with peak incidence after adjustment for period effects 1917-1921 | 1913-1917 | 1912-1916 | 1913-1917 |
| All Local Drifts=Net Drift (Temporal patterns same across age groups?) | No | No | No | No |
| p-value | <0.001 | <0.001 | <0.05 | <0.05 |
| Cohort RRs different from reference cohort? | Yes | Yes | Yes | Yes |
| p-value | <0.001 | 0.001 | <0.05 | <0.001 |
| Cohort deviation is non-linear? | Yes | Yes | Yes | Yes |
| p-value | <0.001 | <0.01 | <0.05 | <0.05 |
DISCUSSION
Results presented here generally support findings from previous similar analyses of mesothelioma incidence using SEER data to explore mesothelioma trends in the US population. We observed a significant decline in PM incidence around 1980-1984 among males in the United States aged 0-74 years that was not seen in other groups (older males, females of any age) with peak incidence occurring among males born around 1917-1921. These strong period and birth cohort effects among younger males could not only reflect potential exposures to asbestos around the start of US involvement in WWII, when US consumption of asbestos in manufacturing significantly increased, but also correlates with regulatory milestones in the US, such as the introduction of Occupational Safety and Health Administration (OSHA) standards during the 1970s, which substantially limited asbestos exposure in construction and manufacturing, including setting permissible and peak exposure limits in the workplace. On the other hand, rising rates in adults (male and female) aged 75+ likely reflect mesothelioma’s long latency, as well as longevity-associated vulnerabilities, including genetic susceptibility, immunesenescence, and other causes such as cancer-related chemotherapies and radiotherapies.
As in the United States, pleural mesothelioma incidence trends in Europe have been strongly influenced by industrial and regulatory changes, along with political and population demographic shifts. Notably, western Europe reported extensive asbestos utilization in various industries through the mid-20th century, peaking between the 1950s to 1980s. The United Kingdom, France, Germany, Italy, and the Netherlands were the highest consumers with asbestos utilization in the shipbuilding, construction and manufacturing sectors. Peto et al. estimated that male mesothelioma deaths in Western Europe would double from 5,000 to about 9,000 between 1998 to 2018, suggesting gradual decline of aged-out exposed cohorts. These authors based their projections on cancer mortality data from six countries that represented 75% of the western Europe population.
Pleural mesothelioma incidence rates in the United Kingdom have consistently been the highest in Europe, exceeding 8 in 100,000 annually among men aged 40-74 at peak. This can be attributed to the extensive use of amphibole asbestos, notably crocidolite as well as amosite, in naval shipbuilding and industrial insulation. Though banned in 1999, the long often 30 to 50 years latency period of mesothelioma means that cases continued to emerge in the 1940-1950 birth cohorts. In Italy, Germany and France, where asbestos use was banned between 1992 and 1997, similar patterns have been reported with national registries documenting rising PM incidence rates through the early 2000s, with a subsequent plateauing or decline in more recent years. Other EU investigators have reported similar expansion of asbestos use post-WWII outside of shipbuilding to industries such as plumbing, railroad engineers, sheet metal workers, and asbestos-cement manufacturing.
The Lombardy region in Italy has been central to epidemiological surveillance reports nationally via its mesothelioma registry. This mesothelioma registry has identified thousands of cases since its start in 2000, with Ferrante et al. estimating occupational exposure in over 70% of male cases, including indirect exposure among agricultural workers via contaminated jute sacks and asbestos filters in wine production reportedly contributing to cases outside of traditional industrial settings. The Lombardy Mesothelioma Registry impact in tracking PM incidence particularly emphasizes the value of comprehensive exposure assessment and reporting by regional registries in capturing subtle epidemiologic patterns in cancer incidence. Importantly, more recent studies of this Italian registry report plateauing of mesothelioma incidence, similar to what is being identified in the current study analyses of United State mesothelioma incidence trends.
Countries which experienced comparatively later industrialization, or lower asbestos consumption, such as Spain, Poland, Estonia, and former Yugoslavian countries, have reported lower PM incidence rates, often below 1 in 100,000 among men. However, newer data suggests rising incidence in some of these regions with better diagnostic capabilities, and with possible migration impacts. For instance, increased PM rates have been reported in Ireland, suggesting an association with retirees from asbestos-intensive industries in the UK returning home. These kinds of demographic shifts, along with better diagnosis using newer tools like medical thoracoscopy, has likely resulted in better case ascertainment and more accurate mesothelioma incidence rates estimated in regions with previously underreported cancer incidence.
Asia currently reports the highest of global asbestos consumption, e.g., from 2001 to 2007, nearly 64% of all asbestos used globally was consumed in Asia. China, India, Indonesia, and Thailand have extensive legacy use, though mesothelioma rates may be underreported due to diagnostic limitations. In Japan and South Korea, centralized registries and surveillance programs reveal increasing mesothelioma incidence. In China, industrial asbestos use persists, and there is no current asbestos use regulation. Russia has also continued to produce high volumes of asbestos, with a recent study reporting increased mesothelioma risk related to high asbestos burden in asbestos mining and milling operators.
Mesothelioma reporting in Africa is sparse, though sentinel studies have emerged from South Africa, Zimbabwe, and more recently, the Democratic Republic of Congo (DRC). In the DRC, cases reportedly linked to asbestos cement plants and mining operations were initially misdiagnosed as tuberculosis due to similar respiratory symptoms and limited diagnostic capabilities. On the other hand, Australia maintains one of the highest per capita mesothelioma incidence rates globally, with 700-800 cases annually. Despite a full asbestos ban in 2003, risk from historical occupational exposure may persist, along with potential risks associated with asbestos-containing materials being present in an estimated one in three homes. In South America, Brazil, historically a leading asbestos producer, banned the substance in 2017, but loopholes still permit production and export. Comparative analyses show patterns similar to European trends, though underreporting and inadequate infrastructure obscure the full disease burden.
In this updated analysis we utilized SEER program cancer incidence data to capture mesothelioma trends across the United States. The SEER program represents the largest source of cancer incidence and survival data in the US. The SEER-8 database, which has the most robust longitudinal coverage of cancer cases in the United States (1975-2022), was utilized to provide a dataset suitable to examine US historical trends in mesothelioma. However, there are some limitations to consider, including the fact that the geographical coverage of the SEER-8 database extends to only approximately 8.3% of the population of the United States. SEER oversamples specific demographics to be more representative of the US population across racial and ethnic groups. Still, SEER-8 does include reporting from a number of port/shipbuilding regions, including the metropolitan areas of San Francisco-Oakland (California) and Seattle-Puget Sound (Washington state), and the state of Hawaii, which may enhance detection of historical asbestos-related cancer incidence.
It should also be noted that cancer surveillance data exists at the state-level in the US and participation and reporting to SEER is not universal across state cancer registries. This is particularly important because mesothelioma is a rare cancer, correlated with regions with historically high occupational exposures. Over time, however, state registries may join or leave/discontinue the SEER Program. In the current study, the Detroit registry, which covers the metropolitan area of Detroit, Michigan, discontinued submission of data in 2020 (resulting in a final year of data submission for 2018) and led us to rely on SEER-8 data than SEER-9, which was used in our previous analyses. Though it is possible that this may have impacted our results, given the historical presence of automotive and manufacturing industries that involved asbestos exposures in the Detroit region, the fact that our current analyses generally support findings from our previous APC analysis using SEER-9 and the reporting of current registries that represent regions with strong port/shipbuilding activities in SEER-8, suggest that the use of SEER-8 data to examine mesothelioma trends in the US is consistent with trends observed using SEER-9.
The SEER program data lacks detailed exposure data, limiting the ability to assess the relationship between mesothelioma incidence and individual-level asbestos exposure. The unavailable exposure data may include occupational history, exposure quantities, characteristics, duration, or intensity of exposure to asbestos-containing materials. Therefore, the APC modeling relies on correlates of exposure such as birth cohort, sex, and age group, with the attendant risk of ecological fallacy. Absence of direct exposure estimates limits the ability to discern specific contributors to mesothelioma incidence using SEER data.
Diagnostic misclassification and coding inconsistencies may affect SEER program data quality. Mesothelioma subtypes such as epithelioid, sarcomatoid, and biphasic, along with anatomical sites such as peritoneal, pericardial, or tunica vaginalis mesothelioma, are not consistently coded across contributing registries. Additionally, as diagnostic technology improves over time, there may be an overestimation of incidence in later periods due to higher case ascertainment rather than true increases in disease rates. These factors complicate the interpretation of cancer trends over time and may conceal slight shifts in incidence. One must also consider the consistent trend of plateauing of pleural mesothelioma incidence with older age cohorts seen in the SEER and European registries; this may be influenced by chronic environmental exposures to other known causes of mesothelioma (e.g., ionizing radiation, chemotherapy, and non-asbestos fibers like erionite). Importantly, such longevity-related causes may increasingly influence mesothelioma incidence trends in regions where subpopulations with meaningful occupational asbestos exposures have diminished by attrition over time.
Despite these challenges, the SEER data provides a relatively robust representation of cancer incidence trends in the US over time. The generated estimates reinforce meso-level insights into malignant mesothelioma trends in the United States and situates them within a broader global framework of mesothelioma epidemiology and population burden of disease. Importantly, it remains broadly representative of the cancer incidence of the US population across different demographics, and follows rigorous standards for data quality, including diagnosis ascertainment and data completeness for malignancies, compared to the limited generalizability of international registries (such as in Italy, Australia, France, or the UK) that may be subject to inconsistent methods for data collection and reporting, and variations in asbestos regulations, clinical practice, and diagnostic capabilities for case ascertainment and completeness of reporting.
CONCLUSIONS
Our updated analyses continue to suggest an association between declining occupational asbestos exposures post-World War II, along with US regulations limiting asbestos use/exposures since the early 1970s, and the steady decline in PM incidence rates observed among younger (age 0-74) males potentially at risk of earlier PM onset from occupational exposures to asbestos. Increasing PM rates in the 75+ subgroup of both genders may also be attributable to longevity-related factors influencing later onset PM risk, e.g., aging, prior cancer treatments, or genetic susceptibility.
Conflict of Interest Statement:
BK, AL, and AJ are employed by an engineering and health sciences consulting firm with diverse professional staff providing assistance to governmental and private sector clients including litigation support and expert witness testimony. NO is employed by a life sciences consulting firm. The authors declare no conflict of interest.
References:
- Roggli VL, Sharma A, Butnor KJ, Sporn T, Vollmer RT. Malignant mesothelioma and occupational exposure to asbestos: a clinicopathological correlation of 1445 cases. Ultrastruct Pathol. 2002;26(2):55-65.
- Lacourt A, Gramond C, Rolland P, Ducamp S, Audignon S, Astoul P, Ilg AG, Rinaldo M, Raherison C, Galateau-Salle F, Imbernon E. Occupational and non-occupational attributable risk of asbestos exposure for malignant pleural mesothelioma. Thorax. 2014;69(6):532-539.
- Spirtas R, Heineman EF, Bernstein L, Beebe GW, Keehn RJ, Stark A, Harlow BL, Benichou J. Malignant mesothelioma: attributable risk of asbestos exposure. Occup Environ Med. 1994;51(12):804-811.
- Ferrante D, Mirabelli D, Tunesi S, Terracini B, Magnani C. Pleural mesothelioma and occupational and non-occupational asbestos exposure: a case-control study with quantitative risk assessment. Occup Environ Med. 2016;73(3):147-153.
- Bartrip PW. History of asbestos related disease. Postgrad Med J. 2004;80(940):72-76.
- Friedman S, Negoita S. History of the surveillance, epidemiology, and end results (SEER) program. JNCI Monogr. 2024;2024(65):105-109.
- Bell A. Age period cohort analysis: a review of what we should and shouldn’t do. Ann Hum Biol. 2020;47(2):208-217.
- European Commission. EU Directive 2023/2668 on the protection of workers from the risks related to exposure to asbestos at work. Brussels, Belgium: European Commission; 2023.
- Marinaccio A, Binazzi A, Marzio DD, Scarselli A, Verardo M, Mirabelli D, Gennaro V, Mensi C, Riboldi L, Merler E, Zotti RD. Pleural malignant mesothelioma epidemic: incidence, modalities of asbestos exposure and occupations involved from the Italian National Register. Int J Cancer. 2012;130(9):2146-2154.
- Consonni D, Groppi A, Pesatori AC, Dallari B, Mensi C, Bonzini M, Rugarli S, Stella S. Incidence of malignant mesothelioma in Lombardy, Italy: accuracy of predictive models 2013-2020. Occup Environ Med. Conference Abstract.
- Takahashi K, Landrigan PJ, Ramazzini C. The global health dimensions of asbestos and asbestos-related diseases. Ann Glob Health. 2016;82(1):209-213.
- Rees D, Goodman K, Fourie E, Chapman R, Blignaut C, Bachmann MO, Myers J. Asbestos exposure and mesothelioma in South Africa. S Afr Med J. 1999;89(6): .
- Huang J, Chan SC, Pang WS, Chow SH, Lok V, Zhang L, Lin X, Lucero-Prisno DE III, Xu W, Zheng ZJ, Elcarte E. Global incidence, risk factors, and temporal trends of mesothelioma: a population-based study. J Thorac Oncol. 2023;18(6):792-802.
- Kerger BD. Longevity and pleural mesothelioma: age-period-cohort analysis of incidence data from the Surveillance, Epidemiology, and End Results (SEER) Program, 1973-2013. BMC Res Notes. 2018;11(1):337.
- Surveillance, Epidemiology, and End Results (SEER) Program. SEER*Stat Database: Incidence SEER Research Data, 8 Registries, Nov 2024 Sub (1975-2022) Linked to County Attributes Time Dependent (1990-2023) Income/Rurality, 1969-2023 Counties. National Cancer Institute, DCCPS, Surveillance Research Program. Released April 2025, based on the November 2024 submission. Available at: www.seer.cancer.gov.
- Xie GD, Liu YR, Jiang YZ, Shao ZM. Epidemiology and survival outcomes of mucinous adenocarcinomas: a SEER population-based study. Sci Rep. 2018;8(1):6117.
- Pavlisko EN, Li H, Pina-Oviedo S, Nel A, Glass CH, Sporn TA. Mesothelioma. In: Pathology of Asbestos-Associated Diseases. Cham, Switzerland: Springer Nature; 2025:111-205.
- Rosenberg PS, Check DP, Anderson WF. A web tool for age period cohort analysis of cancer incidence and mortality rates. Cancer Epidemiol Biomarkers Prev. 2014;23(11):2296-2302.
- Kerger BD, Odo NU, Loccisano AE. Age-period-cohort analysis of mesothelioma: flat incidence trends for males entering US workforce after 1972. Ann Clin Pathol. 2023;10(1): .
- Price B, Ware A. Time trend of mesothelioma incidence in the United States and projection of future cases: an update based on SEER data for 1973 through 2005. Crit Rev Toxicol. 2009;39(7):576-588.
- Teta MJ, Mink PJ, Lau E, Sceurman BK, Foster ED. US mesothelioma patterns 1973-2002: indicators of change and insights into background rates. Eur J Cancer Prev. 2008;17(6):525-534.
- Franke K, Paustenbach D. Government and Navy knowledge regarding health hazards of asbestos: a state of the science evaluation (1900 to 1970). Inhal Toxicol. 2011;23(suppl 3):1-20. doi:10.3109/08958378.2011.643417
- Barlow CA, Sahmel J, Paustenbach DJ, Henshaw JL. History of knowledge and evolution of occupational health and regulatory aspects of asbestos exposure science: 1900-1975. Crit Rev Toxicol. 2017;47(4):286-316.
- Peto J, Decarli A, La Vecchia C, Levi F, Negri E. The European mesothelioma epidemic. Br J Cancer. 1999;79(3):666-672.
- Montanaro F, Bray F, Gennaro V, Merler E, Tyczynski JE, Parkin DM. Pleural mesothelioma incidence in Europe: evidence of some deceleration in the increasing trends. Cancer Causes Control. 2003;14(8):791-803.
- Rushworth DH. The Navy and asbestos thermal insulation. Nav Eng J. 2005;117(2):35-48.
- Mirabelli D, Somigliana AB, Azzolina D, Consonni D, Barbieri PG. Lung fibre burden and risk of malignant mesothelioma in shipyard workers: a necropsy-based case control study. Ann Work Expo Health. 2024;68(5):476-485.
- Van den Borre L, Deboosere P. Asbestos in Belgium: an underestimated health risk. The evolution of mesothelioma mortality rates (1969-2009). Int J Occup Environ Health. 2014;20(2):134-140.
- Plato N, Martinsen JI, Sparén P, Hillerdal G, Weiderpass E. Occupation and mesothelioma in Sweden: updated incidence in men and women in the 27 years after the asbestos ban. Epidemiol Health. 2016;38:e2016039.
- Ahmed M, Flannery A, Mujammil I, Breen D. Variation in incidence trends of malignant pleural mesothelioma in Europe. Eur Respir J. 2018;51(2): .
- Ferrante D, Chellini E, Merler E, Pavone V, Silvestri S, Miligi L, Gorini G, Bressan V, Girardi P, Ancona L, Romeo E. Italian pool of asbestos workers cohorts: mortality trends of asbestos-related neoplasms after long time since first exposure. Occup Environ Med. 2017;74(12):887-898.
- Ferrante D, Angelini A, Barbiero F, Barbone F, Bauleo L, Binazzi A, Bovenzi M, Bruno C, Casotto V, Cernigliaro A, Ceppi M. Cause specific mortality in an Italian pool of asbestos workers cohorts. Am J Ind Med. 2024;67(1):31-43.
- Frank AL, Joshi TK. The global spread of asbestos. Ann Glob Health. 2014;80(4):257-262.
- Stayner L, Welch LS, Lemen R. The worldwide pandemic of asbestos-related diseases. Annu Rev Public Health. 2013;34:205-216.
- Schüz J, Kovalevskiy E, Olsson A, Moissonnier M, Ostroumova E, Ferro G, Feletto E, Schonfeld SJ, Byrnes G, Tskhomariia I, Straif K. Cancer mortality in chrysotile miners and millers, Russian Federation: main results (Asbest Chrysotile Cohort-Study). JNCI: J Natl Cancer Inst. 2024;116(6):866-875.
- Pyana Kitenge J, Dubbeldam A, Said-Hartley Q, Ronsmans S, Jeebhay M, Nemery B. Asbestos-related diseases in Africa: sentinel cases of mesothelioma and asbestosis from DR Congo. Pulmonology. 2025;31(1):2449268.
- Walker-Bone K, Goodwin MA, Bufton B, Davis BA, Wong H, Harvey J, Barker S, Chalker E, Klebe S, Prabhakaran S, Brims FJ. Mesothelioma incidence rates in Australia since 1982: exploring age, period, and cohort effects and future projections. Cancer Epidemiol Biomarkers Prev. 2025;34(7):1149-1155.
- Marsh GM, Riordan AS, Keeton KA, Benson SM. Non-occupational exposure to asbestos and risk of pleural mesothelioma: review and meta-analysis. Occup Environ Med. 2017;74(11):838-846.
- US Geological Survey (USGS). Mineral Commodity Summaries 2022. Reston, VA: US Geological Survey; 2022. doi:10.3133/mcs2022.
- Gariazzo C, Gasparrini A, Marinaccio A. Asbestos consumption and malignant mesothelioma mortality trends in the major user countries. Ann Glob Health. 2023;89(1):11.
- National Cancer Institute SEER Program. Number of persons by race and Hispanic ethnicity for SEER participants (2020 Census Data). Available at: https://seer.cancer.gov/registries/data.html.
- Kolonel LN, Yoshizawa CN, Hirohata T, Myers BC. Cancer occurrence in shipyard workers exposed to asbestos in Hawaii. Cancer Res. 1985;45(8):3924-3928.
- Howlader N, Noone AM, Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA, eds. SEER Cancer Statistics Review, 1975-2017. Bethesda, MD: National Cancer Institute; 2020. Available at: https://seer.cancer.gov/csr/1975_2017/.
- Lawrence Alexander Santhi J, Ali H, Vegivinti CTR, Narra SA, Prabhakaran SY, Mehta M, Boc A, Varghese RS, Thirumaran R. A SEER stat analysis of epidemiological and anatomical prognostic indicators of mesothelioma. J Clin Oncol. 2025;43:e20091.
- Marinaccio A, Binazzi A, Bonafede M, Corfiati M, Di Marzio D, Scarselli A, Verardo M, Mirabelli D, Gennaro V, Mensi C, Schallemberg G. Malignant mesothelioma due to non-occupational asbestos exposure from the Italian national surveillance system (ReNaM): epidemiology and public health issues. Occup Environ Med. 2015;72(9):648-655.
- Attanoos RL, Churg A, Galateau-Salle F, Gibbs AR, Roggli VL. Malignant mesothelioma and its non-asbestos causes. Arch Pathol Lab Med. 2018;142(6):753-760.
- Alpert N, van Gerwen M, Taioli E. Epidemiology of mesothelioma in the 21st century in Europe and the United States, 40 years after restricted/banned asbestos use. Transl Lung Cancer Res. 2020;9(suppl 1):S28.
- Roy P, Raheja A, Prajapati K, Roy S, Bardhan M, Frank AL. Challenges in identifying and diagnosing asbestos-related diseases in emerging economies: a global health perspective. Ann Glob Health. 2025;90(1):65.
- Penberthy L, Friedman S. The SEER Program’s evolution: supporting clinically meaningful population-level research. JNCI Monogr. 2024;2024(65):110-117.