Minimally Invasive Treatment for Achilles Tendon Tears
Debride, Refill, Fertilize: Technique Description with Corresponding Case Report of a Novel, Minimally Invasive Treatment for Achilles Tendon Partial Tears
Michael Khadavi1,2,3*, Ann Wingard3, Alexandre Lavigne1,2,4, Kirk McCullough1
- Kansas City Orthopedic Alliance, 10777 Nall Ave Suite 300, Overland Park, KS, USA
- Department of Physical Medicine and Rehabilitation, University of Missouri, Columbia, MO, USA
- Department of Physical Medicine and Rehabilitation, University of Kansas Medical Center, Kansas City, KS, USA
- Department of Physical Medicine and Rehabilitation, Centre hospitalier de Montréal, QC, Canada
*Email: [email protected]
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION: Khadavi, M., Wingard, A., et al., 2025. Debride, Refill, Fertilize: Technique Description with Corresponding Case Report of a Novel, Minimally Invasive Treatment for Achilles Tendon Partial Tears. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6379
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i3.6379
ISSN: 2375-1924
ABSTRACT
Treatment of midportion Achilles tendinopathy and partial tear pathology is particularly challenging, with surgical treatments characterized by prolonged recoveries, modest outcomes, and up to 11% complication rate, while more recent minimally invasive strategies including percutaneous tenotomy, platelet-rich plasma injection, and tendon hydrodissection have at best demonstrated mixed results. We hypothesize that failures from available surgical and minimally invasive treatments occur because they address only part of the pathology (e.g., orthobiologics focused on promoting tendon growth without addressing the burden of diseased, degenerative tendon tissue). Similarly, percutaneous or surgical debridement while removing pathologic tissue unfortunately leaves large voids in the tendon with hopes of creating a vascular and healing response that will fill the void. Herein, we present a novel multifaceted approach with a corresponding case report of three sequential, minimally invasive therapies utilizing unique yet complimentary mechanisms of action: debridement with percutaneous ultrasonic tenotomy, tissue void grafting with allograft connective tissue matrix, and promotion of tendon repair/regeneration with harvested bone marrow concentrate. This case is of a 39-year-old male recreational athlete who failed two years of conservative care and orthobiologic injections for his midportion Achilles tendinopathy and partial tear. Following treatment with percutaneous ultrasonic tenotomy and injection of allograft connective tissue matrix along with autologous bone marrow concentrate, he returned to most athletic activities by three months and ultimately achieved a full asymptomatic and functional recovery by six months with complete healing of his partial tendon tear on follow-up magnetic resonance imaging.
Keywords: Achilles tendinopathy; tenotomy; bone marrow concentrate; connective tissue matrix; orthobiologics; ultrasound.
INTRODUCTION
The Achilles tendon is one of the largest tendons in the body. Its macro- and micro-structure, coupled with its physiology, make it unique. During loading activity such as running, the Achilles tendon can transmit forces that are 12.5 times a person’s body weight. Studies have shown this ability was mainly due to the collagen fibers orientation, alignment and dispersion, and the mineralized fibrocartilage anchoring the tendon to the bone. The Achilles tendon needs eccentric forces to promote collagen turnover and remodeling that result in a more durable and resilient tissue architecture. The alignment of collagen fibers is a large factor in the Achilles loading. Tendon injury leads to misalignment of collagen fibers, and inflammatory destruction of the extracellular matrix that compromises the tensile architecture. This complex structure and fragile balance between loading and remodeling, combined with suboptimal vasculature within the mid-substance, may be the reason Achilles tendinopathies are so difficult to treat and subsequently heal.
Incidence of tendon injuries are 0.2% in the general population. In a focused analysis looking at a group of 3379 runners, Achilles tendinopathy affected 4.2%, with 23.7% localized to the insertion and 63.8% occurring at the mid-substance. Achilles tendon injuries are typically overuse injuries thought to be due to small microtears that accumulate due to suboptimal healing with poor blood supply in a watershed area 2-6 cm proximal to the tendon insertion on the calcaneus, that can ultimately result in partial tears or possible complete rupture. The nutrition for healing relies on the paratenon to create the environment for healing. Depending on the microenvironment, attempted tendon healing can result in non-functional fibrous scar tissue that disrupts the normal architecture of the tendon making this area mechanically weaker and more prone to re-injury.
The role of macrophages in modulating this reparative process is pivotal. Their polarization to inflammatory pathways mediated by interferons and steroid receptor activation could lead to a failed fibrotic healing response. When this cascade leads to macrophage dysfunction, it is crucial to debride the tissue to preserve the tendon’s functional architecture and promote a regenerative microenvironment through essential cell signaling molecules.
Historically, first-line treatment for Achilles tendinopathy has been a multimodal conservative approach that includes activity modification with relative rest, shoewear modifications such as heel lifts, and a physical therapy program. Physical therapy includes progressive concentric and eccentric strengthening exercises, generally over 2-3 months. When those treatments fail to restore Achilles tendon function, patients typically turn to potential procedural/surgical options. Surgery in these patients has been shown to be effective in 70%-85% of cases, however length of recovery and adverse events need to be considered. In cases of partial tear pathology, the surgery consists of debridement and side-to-side or end-to-end repair of the tendon. Patients are typically managed with protective weightbearing (e.g. CAM boot) for 6-8 weeks depending on the volume of debrided tendon and extent of repair, while maintaining range of motion at the ankle with care to avoid excess tension with dorsiflexion past neutral during the early healing phases. Activity tolerance is then measured by pain, and the patient is slowly weaned back into regular activity with physical therapy for a total of 12-14 weeks of recovery. The treatment timeline for surgery tends to be about two weeks to one month longer than conservative treatments, with more chances for complications. Open surgical intervention has a 10.5% risk of complications as compared to minimally invasive procedures, which have a 5.3% risk. When looking at open repairs of 432 mid/insertional tendinopathy, complications for open repairs occurred about 11% of cases. These complications included wound necrosis (3%), superficial infection (2.5%), sural nerve injury (1%), seroma (1%), hematoma (1%), and hypertrophic scar (1%). Overall, the reoperation rate was 3%.
A common minimally invasive procedure is the endoscopic Achilles tendon repair. This technique uses an arthroscopic cannula and small incisions made on either side of the tendon. Proposed surgical advantages are better evaluation of the tendon, debridement, and mobilization of the tendon ends, accurate passage of needles and guided approximation of tendon ends. Treatment advantages are mainly the decrease in infection rates. Though this treatment is considered minimally invasive, it still requires multiple incisions as compared to the method discussed next.
The use of percutaneous ultrasonic tenotomy for Achilles tendinopathy has not been well studied. However, it has shown promising results in chronic elbow tendinopathies. Though the results are not statistically significant, debridement of dead tissue in conjunction with regenerative substances could provide better outcomes.
Orthobiologic treatments utilize naturally occurring cells and growth factors that promote healing. These critical components can be concentrated and then introduced into diseased tissue such as cartilage, bone, ligament, or tendon to initiate the healing cascade. These include cells, growth factors, and proteins whose functions are to promote the healing of these tissues. Two such growth factors in tendon healing are vascular endothelial growth factor (VEGF) and insulin-like growth factor 1 (IGF-1). VEGF and IGF-1 are highly expressed during tendon healing and have been shown to be associated with upregulation of angiogenesis and collagen synthesis, respectively. VEGF, in particular, peaks in the early stages of tendon healing, and high levels are associated with pericytes (tendon progenitor cells) and matrix metalloproteinases that can influence tendon tensile strength. Though these molecules have been found in the microenvironments of healing tendons, understanding how to harness these molecules for healing is still debated.
Two orthobiologic treatment interventions that have been researched for Achilles tendinopathy are platelet-rich plasma and bone marrow concentrate. When comparing the composition of these substances, studies show significant differences that could be influential in clinical management. Platelet-rich plasma houses substances such as alpha granules that contain growth factors such as VEGF and IGF-1 that initiate healing cascades through the paracrine effect. Paracrine signaling, in this instance, allows these growth factors to promote the regenerative cascade directly through diffusion to adjacent cells. Several randomized controlled trials and meta-analyses have studied the efficacy of platelet-rich plasma in Achilles tendinopathy but these studies have not shown statistically and clinically significant benefit.
The clinical outcomes of bone marrow concentrate in Achilles tendinopathy have not been studied as much as those of platelet-rich plasma, but several theoretical benefits of bone marrow concentrate exist compared to platelet-rich plasma in tendinopathy. First and foremost, bone marrow concentrate contains a significantly greater amount of Interleukin-1 receptor antagonist protein, a potent anti-inflammatory cytokine that reduces matrix degradation and enhances collagen deposition. Also, platelets in bone marrow concentrate are younger and demonstrate a higher concentration of interleukin-4, interleukin-10, and interferon gamma that may have greater anti-inflammatory potential. Lastly, bone marrow concentrate contains more mesenchymal/stromal stem cells that carry extracellular vesicles promoting a more robust regenerative healing through the paracrine effect.
Recent studies have shown that the addition of an acellular scaffold connective tissue matrix could help to replenish the extracellular matrix frequently damaged or absent in areas of tissue disruption and chronic inflammatory changes, and aid in the growth and maturation of healthy tendon tissue through improved cell adhesion and local cell signaling. Connective tissue matrix can be sourced from various tissues and with resultant variabilities in content and connective tissue matrix concentration. One such commercially available product is Interfyl (Celularity, Mahwah, NJ), a decellularized flowable allograft particulate derived from placental tissue. Interfyl has shown numerous benefits in cell adhesion/proliferation studies and wound healing compared to other matrices.
Current invasive treatment options for Achilles tendinopathy focus on only one mechanism of healing and as such, they address only part of the pathology. For example, orthobiologics focus on promoting tendon growth without addressing the burden of diseased, degenerative tendon tissue. Meanwhile, percutaneous or surgical debridement leaves large voids in the tendon with hopes to create a vascular and healing response without an appropriate local environment or scaffold to facilitate appropriate cell binding for healing cells recruited to the area.
Herein, we present a novel combinatorial concept with a corresponding case report of three sequential, minimally invasive therapies that utilize unique, complimentary mechanisms of action: debridement with percutaneous ultrasonic tenotomy to remove damaged tissue, filling the void with connective tissue matrix, and promoting tendon regrowth with bone marrow concentrate.
CASE DESCRIPTION
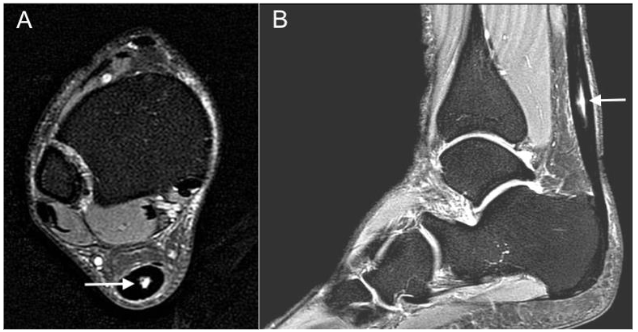
A 40-year-old male avid hiker presented to an outpatient sports medicine clinic with 18 months of persistent right posterior ankle pain. Symptoms were insidious without a particular inciting event, worse with long walks, hiking, and jumping. He had failed conservative care with four months of formal physical therapy, relative rest, and a heel lift, and showed no improvement within 12 months after an ultrasound-guided intratendinous leukocyte-rich platelet-rich plasma injection. Physical exam showed swelling with tenderness to palpation at midportion of the Achilles tendon. Radiographs were without any obvious calcific changes or other skeletal irregularity, and diagnostic ultrasound (US) showed tendinosis and a partial longitudinal tear in the midportion Achilles tendon.
Magnetic resonance imaging (MRI) confirmed the US findings, with midportion Achilles tendinosis and a large partial-thickness interstitial tear (18 x 4 x 3mm). Pathology and treatment options were discussed, and he decided on a sequential, staged treatment approach that included percutaneous ultrasonic tenotomy, connective tissue matrix injection, and a bone marrow concentrate procedure.
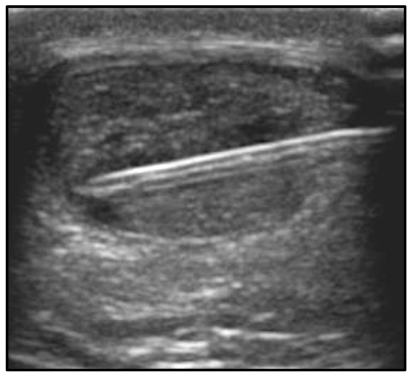
Consent was obtained for all procedures. With the patient in a prone position for the entire procedure, a US-guided popliteal block was performed with 12 ml of 1% lidocaine by the senior author (MK) for peri-procedural anesthesia. Using US guidance, a medial-to-lateral approach to the tendon was utilized to avoid the sural nerve, and a 4mm incision was made with an 11-blade in the skin at two locations along the tendon approximately 15 mm apart. Subsequently, an 18-gauge 1.5 inch needle was passed through the area of tendinosis from medial to lateral with approximately 15 passes at each incision site for initial tenotomy. The needle was withdrawn, and Tenex TX-1 needle probe (Tenex Health, Lake Forest, CA, USA) was used to debride and tenotomize the area of tendinosis with a total cut time of 150 seconds. Then, 1.5-ml Interfyl mixed with 1.5-ml normal saline was injected with US guidance into the area of tendinosis and tear. Bone marrow concentrate and platelet-rich plasma procedures were performed ten days after the tenotomy, and connective tissue matrix injection was performed to allow for the expected post-procedural edema to decrease. Each of bilateral posterior iliac crests were identified under ultrasound and anesthetized with 10ml lidocaine 1% with epinephrine, to minimize post-procedural bleeding. Then, a popliteal block, as described above, was performed. Ten minutes after periosteal lidocaine injection, an 11-gauge jamshidi was utilized to penetrate beneath the left posterior iliac crest boney cortex, and 2ml bone marrow was aspirated into a 10ml syringe preloaded with 1ml A-CDA and 0.25ml heparin 1:10,000. Jamshidi was advanced 2-5 mm deeper before another 2ml bone marrow was aspirated. This procedure was repeated until 30ml bone marrow was aspirated into three 10ml syringes, and then repeated on the contralateral posterior iliac crest. Small volumes of bone marrow were aspirated in this manner to achieve higher mesenchymal stem cell counts and lower peripheral blood contamination size. 60ml antecubital blood was aspirated with a 17-gauge needle and large-bore tubing to decrease premature platelet activation. Bone marrow and blood were centrifuged separately under leukocyte-rich protocols with an Arthrex Angel centrifuge (Arthrex, Naples, FL). Then with a 22 gauge 1 ½ inch needle and US guidance, 4ml platelet-rich plasma and 4ml bone marrow concentrate was peppered into the area of Achilles tendinosis and partial tear as shown in Figure 2.
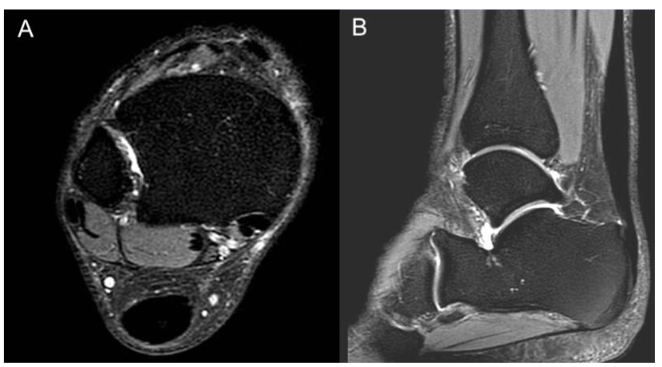
Post-procedural care included limited partial weight bearing in a full-length boot day and night for two weeks after the percutaneous ultrasonic tenotomy, then full weight bearing in the boot for the next 2-3 weeks until follow-up. Heat was utilized for post-procedural pain control as well as oral oxycodone only for the two days following the bone marrow concentrate and acetaminophen. Non-steroidal anti-inflammatory drugs were avoided for two weeks before and two weeks after the bone marrow concentrate procedure. Physical therapy began one week after the bone marrow concentrate, beginning with range of motion and isometric exercises, progressing to concentric calf strengthening between 2-4 weeks post-procedure. By 6 weeks, eccentric calf strengthening began and progressed until 3 months post-procedure. By three months post-procedure, he was completely pain-free and had returned to long and fast walking on even surfaces but had not returned to hiking. At his six-month follow-up he remained 100% pain-free and achieved a full functional recovery, including jumping sports and day hikes of 20 miles with over 5,000 feet of elevation gain. At his one and two-year follow-up examinations, he remained pain-free and functioned at his prior level. Interestingly, there was still focal swelling and tenderness at both the three and six-month follow-up examinations, both of which had resolved by one year. Repeat MRI at seven months showed complete healing of previous Achilles tendon interstitial tear, as shown in Figure 3.
DISCUSSION
Achilles tendinosis and partial tears have been a clinical challenge for foot and ankle surgeons and sports medicine physicians. Surgical interventions carry risks, including potential prolonged recovery time frames and increased potential for wound healing issues, all of which cause hesitation for many patients. At the same time, arrested and incomplete healing is not uncommon with standard-of-care conservative treatments, resulting in continued pain and functional limitations. While orthobiologic treatments have shown tremendous success for most tendons, the majority of studies are negative for the Achilles tendinopathy. Potential reasons for this include physiology and stretching of the Achilles tendon, unique mechanical load based on the Achilles tendon with normal daily activities and athletics, and perhaps more importantly the propensity to develop excessive scar formation following minor injury. When looking at prior studies of just platelet-rich plasma or bone marrow concentrate for Achilles tendinopathy, the data fails to show a significant difference. The patellar tendon, similar in physiology to the Achilles tendon, also fails to show significant improvement when using platelet-rich plasma or bone marrow concentrate in isolation. Despite this, there are studies that show the success of platelet-rich plasma and bone marrow concentrate in physiologically different tendons. The most successful tendon with orthobiologics is currently the common extensor tendon. When compared to steroid injections, platelet-rich plasma improves the Disabilities of the Arm, Shoulder and Hand questionnaire score and visual analog scale significantly for up to 2 years. Similar results are seen in plantar fasciopathy, with significant improvement in multiple randomized controlled trials.
Furthermore, Kirschner et al assessed the efficacy of combinatorial regenerative treatments for tendinopathy. In this randomized controlled trial, they compared percutaneous needle tenotomy combined with platelet-rich plasma versus percutaneous needle tenotomy alone. They included all tendinopathies and found no significant difference between the two groups at the 1-year and 2-year follow-ups.
Percutaneous ultrasonic tenotomy provides a minimally invasive mechanism of increasing blood flow, breaking up and debriding fibrotic and diseased tendinopathic tendon tissue, and resetting the neovascularization process. Tiny voids are left in a tendon following both a tear, as well as following a tenotomy or debridement procedure. Interfyl is an acellular scaffold connective tissue matrix that provides a cell-friendly healing and microstructural environment through enhanced cell binding potential for new cellular and collagen growth following either a tear or a debridement procedure. It may also enhance tendon progenitor cells, as well as tenocyte growth and differentiation. One study has assessed the clinical outcomes of percutaneous ultrasonic tenotomy alone for Achilles tendinopathy. Chimenti et al looked at functional recovery post-percutaneous ultrasonic tenotomy for 34 patients (40 tendons total) with chronic insertional Achilles tendinopathy. The results of this study showed most patients with moderate to severe Achilles tendon pain noted a significant pain improvement, but the majority of patients remained with persistent mild pain at long-term follow-up. Despite this shortcoming, this study did showcase the safety of the percutaneous ultrasonic tenotomy procedure with only one mild superficial skin infection among 40 procedures completed.
Once the fibrotic and diseased Achilles tendon tissue has been debrided and the voids following the debridement and tear have been filled with extracellular scaffold connective tissue matrix such as Interfyl, growth factors and mesenchymal stem cells may facilitate growth and remodeling of new tendon tissue. Multiple methods exist through which bone marrow concentrate and platelet-rich plasma facilitate this healing, including the release of growth factors from the alpha granules of platelets that initiate the natural healing cascade, the paracrine effect from mesenchymal stem cells, and the enhanced growth differentiation of native tendon progenitor cells and tenocytes. Two other clinical studies have used bone marrow concentrate to treat Achilles tendon pathologies. Thueakthong et al published a retrospective case series of 15 patients with Achilles tendinopathy (five insertional, eight midportion, and two combined) treated with bone marrow concentrate. The average pain improvement at the 48-week mark was 3.22/10 according to the numeric rating scale, and there were no complications. Stein et al published a retrospective case series of 27 patients with Achilles tendon rupture treated with a surgical repair combined with a bone marrow concentrate augmentation, averaging a follow-up length of 30 months. The average time to return to light activities was 3.4 months, and 90% of the cohort were fully returned to their respective sport by 5.9 months. There was no Achilles tendon re-rupture among patients in this cohort.
This case is a high-level recreational athlete with Achilles tendinopathy and partial tear who had previously been unresponsive to physical therapy, activity modification, rest, anti-inflammatory medications, and leukocyte-rich platelet-rich plasma injection. Many potential explanations exist, both for why his Achilles tendon did not heal initially with conservative measures, and for why profound symptomatic and advanced imaging improvement occurred following a combined procedure of percutaneous ultrasonic tenotomy, Interfyl injection, and bone marrow concentrate injection. We hypothesize that the chronic tendinosis in this Achilles tendon necessitated tenotomy and debridement of fibrotic and diseased tendinopathic tissue before re-initiation of the healing cascade could sufficiently occur. Bone marrow concentrate may be more powerful than platelet-rich plasma, which is why we chose it as our major orthobiologic treatment for this case involving the Achilles tendon–one of the most poorly responsive tendons to orthobiologic treatments in the body. It is because of this recalcitrant nature and extent of tissue disruption that an allogenic extracellular matrix product was added following the percutaneous ultrasonic tenotomy.
The purpose of this report is to describe a novel, minimally invasive treatment combination for a historically challenging and recalcitrant diagnosis treatment of midportion Achilles tendinopathy and partial tearing refractory to exhaustive conservative care and leukocyte-rich platelet-rich plasma. Indeed, this combination approach with percutaneous ultrasonic tenotomy, Interfyl injection, and bone marrow concentrate injection led to a complete symptomatic and functional recovery and resolution of the tear on follow-up MRI. Limitations include those typical of a case report most importantly a single case and no external control however this patient serves as his own control as he had already failed conservative treatments including relative rest, physical therapy, and a leukocyte-rich platelet-rich plasma injection. Further basic science and clinical studies of this treatment combination are necessary to optimize the management of Achilles tendinopathy associated with a partial tear. The commonness of Achilles tendinopathy, the high frequency of conservative treatment failures, and the risks and recovery time of surgical treatments highlights the necessity of this further research.
CONCLUSION
Combined treatment with percutaneous ultrasonic tenotomy, injection of allogenic connective tissue matrix, and US-guided bone marrow concentrate injection achieved complete symptomatic and functional recovery with MRI resolution in this case of midportion Achilles tendinopathy with a large partial-thickness interstitial tear.
Conflict of Interest: None.
Funding Statement: None.
Acknowledgements: None.
ORCID:
Michael Khadavi: 0009-0002-2304-5917
Ann Wingard: 0009-0001-0041-3564
Alexandre Lavigne: 0000-0003-4668-4543
Kirk McCullough: 0000-0001-8535-6230
REFERENCES
- Del Buono A, Chan O, Maffulli N. Achilles tendon: functional anatomy and novel emerging models of imaging classification. Int Orthop. 2013;37(4):715-721.
- Komi PV, Fukashiro S, Jarvinen M. Biomechanical loading of Achilles tendon during normal locomotion. Clin Sports Med. 1992;11(3):521-531.
- Sadeghi S, Taghizadeh H. Microstructural modeling of Achilles Tendon biomechanics focusing on bone insertion site. Med Eng Phys. 2020;78:48-54.
- Tits A, Blouin S, Rummler M, et al. Structural and functional heterogeneity of mineralized fibrocartilage at the Achilles tendon-bone insertion. Acta Biomater. 2023;166:409-418.
- Palmes D, Spiegel HU, Schneider TO, et al. Achilles tendon healing: long-term biomechanical effects of postoperative mobilization and immobilization in a new mouse model. J Orthop Res. 2002;20(5):939-946.
- Yasuda T, Kinoshita M, Abe M, Shibayama Y. Unfavorable effect of knee immobilization on Achilles tendon healing in rabbits. Acta Orthop Scand. 2000;71(1):69-73.
- Reddy GK, Stehno-Bittel L, Enwemeka CS. Matrix remodeling in healing rabbit Achilles tendon. Wound Repair Regen. 1999;7(6):518-527.
- de Jonge S, van den Berg C, de Vos RJ, et al. Incidence of midportion Achilles tendinopathy in the general population. Br J Sports Med. 2011;45(13):1026-1028.
- Chen W, Cloosterman KLA, Bierma-Zeinstra SMA, van Middelkoop M, de Vos RJ. Epidemiology of insertional and midportion Achilles tendinopathy in runners: A prospective cohort study. J Sport Health Sci. 2023.
- Allenmark C. Partial Achilles tendon tears. Clin Sports Med. 1992;11(4):759-769.
- Jarvinen TA, Kannus P, Paavola M, Jarvinen TL, Jozsa L, Jarvinen M. Achilles tendon injuries. Curr Opin Rheumatol. 2001;13(2):150-155.
- von Rickenbach KJ, Borgstrom H, Tenforde A, Borg-Stein J, McInnis KC. Achilles Tendinopathy: Evaluation, Rehabilitation, and Prevention. Curr Sports Med Rep. 2021;20(6):327-334.
- Fenwick SA, Hazleman BL, Riley GP. The vasculature and its role in the damaged and healing tendon. Arthritis Res. 2002;4(4):252-260.
- Yang G, Rothrauff BB, Tuan RS. Tendon and ligament regeneration and repair: clinical relevance and developmental paradigm. Birth Defects Res C Embryo Today. 2013;99(3):203-222.
- Arvind V, Huang AH. Reparative and Maladaptive Inflammation in Tendon Healing. Front Bioeng Biotechnol. 2021;9:719047.
- Gatz M, Spang C, Alfredson H. Partial Achilles Tendon Rupture-A Neglected Entity: A Narrative Literature Review on Diagnostics and Treatment Options. J Clin Med. 2020;9(10).
- Medeiros DM. Conservative treatment of Achilles tendon partial tear in a futsal player: A case report. Physiother Theory Pract. 2021;37(10):1158-1165.
- Tallon C, Coleman BD, Khan KM, Maffulli N. Outcome of surgery for chronic Achilles tendinopathy. A critical review. Am J Sports Med. 2001;29(3):315-320.
- Maffulli N, Kader D. Tendinopathy of tendo achillis. J Bone Joint Surg Br. 2002;84(1):1-8.
- Lohrer H. Minimum 3.5-year outcomes of operative treatment for Achilles tendon partial tears in the midportion and retrocalcaneal area. J Orthop Surg Res. 2020;15(1):395.
- Lohrer H, David S, Nauck T. Surgical treatment for achilles tendinopathy – a systematic review. BMC Musculoskelet Disord. 2016;17:207.
- Paavola M, Orava S, Leppilahti J, Kannus P, Jarvinen M. Chronic Achilles tendon overuse injury: complications after surgical treatment. An analysis of 432 consecutive patients. Am J Sports Med. 2000;28(1):77-82.
- Phisitkul P. Endoscopic surgery of the Achilles tendon. Curr Rev Musculoskelet Med. 2012;5(2):156-163.
- Carreira D, Ballard A. Achilles tendoscopy. Foot Ankle Clin. 2015;20(1):27-40.
- Vajapey S, Ghenbot S, Baria MR, Magnussen RA, Vasileff WK. Utility of Percutaneous Ultrasonic Tenotomy for Tendinopathies: A Systematic Review. Sports Health. 2021;13(3):258-264.
- Wang H, Yu R, Wang M, et al. Insulin-like growth factor binding protein 4 loaded electrospun membrane ameliorating tendon injury by promoting retention of IGF-1. J Control Release. 2023;356:162-174.
- Liu X, Zhu B, Li Y, et al. The Role of Vascular Endothelial Growth Factor in Tendon Healing. Front Physiol. 2021;12:766080.
- Blair P, Flaumenhaft R. Platelet alpha-granules: basic biology and clinical correlates. Blood Rev. 2009;23(4):177-189.
- Everts P, Onishi K, Jayaram P, Lana JF, Mautner K. Platelet-Rich Plasma: New Performance Understandings and Therapeutic Considerations in 2020. Int J Mol Sci. 2020;21(20).
- Miller LE, Parrish WR, Roides B, Bhattacharyya S. Efficacy of platelet-rich plasma injections for symptomatic tendinopathy: systematic review and meta-analysis of randomised injection-controlled trials. BMJ Open Sport Exerc Med. 2017;3(1):e000237.
- de Vos RJ, Weir A, van Schie HTM, et al. Platelet-Rich Plasma Injection for Chronic Achilles Tendinopathy: A Randomized Controlled Trial. JAMA. 2010;303(2):144-149.
- Barreto ESR, Antunes Junior CR, Silva IC, Alencar VB, Faleiro TB, Kraychete DC. Is Platelet-rich Plasma Effective in Treating Achilles Tendinopathy? A Meta-analysis of Randomized Clinical Trials. Clin Orthop Relat Res. 2024.
- Cassano JM, Kennedy JG, Ross KA, Fraser EJ, Goodale MB, Fortier LA. Bone marrow concentrate and platelet-rich plasma differ in cell distribution and interleukin 1 receptor antagonist protein concentration. Knee Surg Sports Traumatol Arthrosc. 2018;26(1):333-342.
- Yuksel S, Gulec MA, Gultekin MZ, et al. Comparison of the early period effects of bone marrow-derived mesenchymal stem cells and platelet-rich plasma on the Achilles tendon ruptures in rats. Connect Tissue Res. 2016;57(5):360-373.
- Thampatty B, Li H, Im H, Wang J. EP4 receptor regulates collagen type-I, MMP-1, and MMP-3 gene expression in human tendon fibroblasts in response to IL-1β treatment. Gene. 2007;386(1-2):154-161.
- Ziegler CG, Van Sloun R, Gonzalez S, et al. Characterization of Growth Factors, Cytokines, and Chemokines in Bone Marrow Concentrate and Platelet-Rich Plasma: A Prospective Analysis. Am J Sports Med. 2019;47(9):2174-2187.
- Machlus KR, Italiano JE, Jr. The incredible journey: From megakaryocyte development to platelet formation. J Cell Biol. 2013;201(6):785-796.
- Gissi C, Radeghieri A, Antonetti Lamorgese Passeri C, et al. Extracellular vesicles from rat-bone-marrow mesenchymal stromal/stem cells improve tendon repair in rat Achilles tendon injury model in dose-dependent manner: A pilot study. PLoS One. 2020;15(3):e0229914.
- Yin Z, Chen X, Zhu T, et al. The effect of decellularized matrices on human tendon stem/progenitor cell differentiation and tendon repair. Acta Biomater. 2013;9(12):9317-9329.
- Mao Y, John N, Protzman NM, et al. A decellularized flowable placental connective tissue matrix supports cellular functions of human tenocytes in vitro. J Exp Orthop. 2022;9(1):69.
- Merry K, Napier C, Waugh CM, Scott A. Foundational Principles and Adaptation of the Healthy and Pathological Achilles Tendon in Response to Resistance Exercise: A Narrative Review and Clinical Implications. J Clin Med. 2022;11(16).
- Khadavi M, Rehor D, Roney A, Podesta L, Smith DR. Regional Anesthesia for Orthobiologic Procedures. Phys Med Rehabil Clin N Am. 2023;34(1):291-309.
- Hernigou P, Homma Y, Flouzat Lachaniette CH, et al. Benefits of small volume and small syringe for bone marrow aspirations of mesenchymal stem cells. Int Orthop. 2013;37(11):2279-2287.
- Abadin AA, Orr JP, Lloyd AR, Henning PT, Pourcho A. An Evidence-Based Approach to Orthobiologics for Tendon Disorders. Phys Med Rehabil Clin N Am. 2023;34(1):83-103.
- Peerbooms JC, Sluimer J, Bruijn DJ, Gosens T. Positive effect of an autologous platelet concentrate in lateral epicondylitis in a double-blind randomized controlled trial: platelet-rich plasma versus corticosteroid injection with a 1-year follow-up. Am J Sports Med. 2010;38(2):255-262.
- Mishra AK, Skrepnik NV, Edwards SG, et al. Efficacy of platelet-rich plasma for chronic tennis elbow: a double-blind, prospective, multicenter, randomized controlled trial of 230 patients. Am J Sports Med. 2014;42(2):463-471.
- Peerbooms JC, Lodder P, den Oudsten BL, Doorgeest K, Schuller HM, Gosens T. Positive Effect of Platelet-Rich Plasma on Pain in Plantar Fasciitis: A Double-Blind Multicenter Randomized Controlled Trial. Am J Sports Med. 2019;47(13):3238-3246.
- Kirschner JS, Cheng J, Hurwitz N, et al. Ultrasound-guided percutaneous needle tenotomy (PNT) alone versus PNT plus platelet-rich plasma injection for the treatment of chronic tendinosis: A randomized controlled trial. PM R. 2021;13(12):1340-1349.
- Chimenti RL, Stover DW, Fick BS, Hall MM. Percutaneous Ultrasonic Tenotomy Reduces Insertional Achilles Tendinopathy Pain With High Patient Satisfaction and a Low Complication Rate. J Ultrasound Med. 2019;38(6):1629-1635.
- Thueakthong W, de Cesar Netto C, Garnjanagoonchorn A, et al. Outcomes of iliac crest bone marrow aspirate injection for the treatment of recalcitrant Achilles tendinopathy. Int Orthop. 2021;45(9):2423-2428.
- Stein BE, Stroh DA, Schon LC. Outcomes of acute Achilles tendon rupture repair with bone marrow aspirate concentrate augmentation. Int Orthop. 2015;39(5):901-905.

