Multidisciplinary Approaches in Heart Failure Management
Heart failure management – A multidisciplinary approach: Literature review and single center experience
Radu Darciuc¹, Irina Boiciuc¹, Hakan Eraslan¹, Mihail Tașnic¹, Constantin Cozma¹, Daniela Ivanov¹, Sergiu Voitov², Erdem Diker¹˒³, Aureliu Batrînac²
- Department of Cardiology, Medpark International Hospital, Chisinau, Republic of Moldova
- Department of Cardiothoracic Surgery, Medpark International Hospital, Chisinau, Republic of Moldova
- Department of Cardiology, Bayindir Sogutozu Hospital, Ankara, Turkey
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Darciuc, R., Boiciuc, I., et al., 2024. Heart failure management – A multidisciplinary approach: Literature review and single center experience. Medical Research Archives, [online] 12(11).
https://doi.org/10.18103/mra.v12i11.5918
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.5918
ISSN 2375-1924
Abstract
Introduction: Heart failure is a severe chronic disease affecting millions of people worldwide, decreasing their quality of life and having a high burden of mortality. The treatment of the heart failure evolved in a spectacular way in the last years with the development of the new medical therapies, interventions and devices. Unfortunately, outcomes remain poor and need proper implementation of the current knowledge. A multidisciplinary team approach is considered the best way to deliver care for heart failure patients.
Objective: To make a short review of recent data provided by the trials and to describe why the multidisciplinary team approach is necessary during heart failure care. To describe how is the heart failure management performed in our center.
Literature review and our center experience: We analyzed the European Society of Cardiology Guidelines for the diagnosis and treatment of acute and chronic heart failure, made a literature search in Pubmed database and selected the most important articles related to the heart failure management, giving special attention to the evidence provided by the clinical trials. We describe how we implemented the recommendation and multidisciplinary team approach in Medpark International Hospital, Chisinau, Republic of Moldova. There are pharmacological management and a non-pharmacological management available. Non-pharmacological management of the heart failure includes cardiac surgery, interventional cardiology, electrophysiology, sleep-disordered breathing treatment, physical therapy, patient education, etc. Many cases of HF patients need a Heart Team approach that includes a cardiac surgeon, an interventional cardiologist, an electrophysiologist and a general cardiologist. We implemented the multidisciplinary approach providing better care for our patients.
Conclusions: The care of heart failure is multidisciplinary and needs some super-specialized team members. A multidisciplinary team approach is considered the best way to deliver care for HF patients. There are lot of tricky situations when a decision made without the team support could be a wrong decision. To avoid such situations is important to implement the MDT approach and to continually educate the team members, keeping the pace with the HF management updates.
Keywords
Cardiac surgeon, cardiologist, electrophysiologist, interventional cardiologist, heart failure, Heart Team, multidisciplinary team, optimal medical treatment.
1. Introduction
Heart failure (HF) is a severe chronic disease affecting millions of people worldwide, decreasing their quality of life (QOL) and having a high burden of mortality. The incidence and prevalence vary widely between geographic regions. One-year HF mortality has an average of 33% across all adult ages. Huge efforts are made to develop new strategies in the management of the HF. To exemplify let’s think about the last European Society of Cardiology (ESC) guidelines for the diagnosis and treatment of the acute and chronic HF. The guidelines were published in 2021 and in just 2 years, in 2023, a focused update of the guidelines was necessary to be released taking into consideration the new data provided by several trials. Is just an example how quickly is changing the field of the HF management. It evolved in a spectacular way in the last years with the development of the new medical therapies, interventions and devices. Unfortunately, outcomes remain poor and need proper implementation of the current knowledge. The are many possibilities how we could deal with the challenges in the management of HF patients. A multidisciplinary team (MDT) approach is considered the best way to deliver care for HF patients.
2. Objective
To make a short review of recent data provided by the trials related to the management of the HF and to describe why the MDT approach is necessary. Another objective of our paper is to describe how is the HF management performed in our center, in Medpark International Hospital, Chisinau, Republic of Moldova.
3. Literature review and our center experience
The main reference papers for our review were 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic HF and 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic HF. We made a literature search in Pubmed database and selected the most important articles related to the HF management, giving special attention to the evidence provided by the clinical trials. When analyzing the recent evidence about the HF management we describe how we implemented the recommendation in our center, describing the challenges, the obstacles and the solutions. We gave special attention to the description of the MDT approach of our center.
The 2021 ESC guidelines for the diagnosis and treatment of the acute and chronic HF provide information of the full spectrum of the tools available for the management of the HF. The guidelines make clear distinction of the terminology describing 3 types of HF based of the measurements of the left ventricular ejection fraction (LVEF):
- HF with reduced ejection fraction (HFrEF). The LVEF is ≤ 40%.
- HF with mildly reduced ejection fraction (HFmrEF) have a LVEF between 41% and 49%
- HF with preserved ejection fraction (HFpEF) have LVEF ≥ 50%.
According to the type of the HF the proper management pathway is recommended. The current ESC guidelines underline the importance of the MDT approach with the next statement: It is recommended that HF patients are enrolled in a multidisciplinary HF management program to reduce the risk of HF hospitalization and mortality.
3.1. Pharmacological management
A lot of drugs have been developed and currently used in the management of HF. There are angiotensin converting enzyme inhibitors (ACEI), angiotensin-receptor blockers (ARB), an angiotensin receptor-neprilysin inhibitor (ARNI), beta-blockers (BB), mineralocorticoid receptor antagonists (MRA), loop diuretics, sodium-glucose co-transporter 2 (SGLT2) inhibitors, If-channel inhibitor, antiarrhythmic drugs, inotropes, myosin inhibitors, intravenous iron, etc. The 2021 ESC guidelines for the diagnosis and treatment of the acute and chronic HF were mentioning the evolution of the treatment of the HF therapy. The triad of ACEI or ARB, BB and MRA is still recommended as cornerstone therapy in patients with HFrEF taking into consideration the improve in survival, QOL and reduction in the hospitalization, evidence provided by studies as MERIT-HF, COPERNICUS, CIBIS-II, SENIORS, CONSENSUS, SOLVD, ATLAS, RALES, EMPHASIS-HF, CHARM-Added, CHARM-Alternative and Val-HeFT.
Taking into consideration the results of PARADIGM-HF study, the 2021 ESC guidelines recommend the use of ARNI as a replacement to ACEI in patients with HFrEF who remain symptomatic on the treatment with ACEI, BB and MRA. In some patients ARNI can be considered as first line drug instead of ACEI. In 2021, the ESC guidelines recommended to use an SGLT2 inhibitor in all patients with HFrEF, evidence provided by EMPEROR-Reduced and DAPA-HF trials. Later, the information provided by new trials as EMPEROR-Preserved and DELIVER showed clear benefits of the SGLT2 inhibitor use in the treatment of also HFmrEF and HFpEF leading to the focused update of the guidelines in 2023. Some patients may benefit from other drugs as If-channel inhibitor ivabradine, data provided by SHIFT trial.
The trials PIVOTAL and IRONMAN showed benefits of the intravenous iron therapy in patients with iron deficiency and HFrEF and HFmrEF. Thereby, the intravenous iron supplementation is now recommended to improve symptoms, QOL and reduction of HF hospitalization. Another important area of the pharmacological management is the antiarrhythmic therapy. The BB have a potent antiarrhythmic effect, being Class II antiarrhythmic drugs according to Vaughan Williams’s classification. Unfortunately, in some patients with HF, the antiarrhythmic effect of the BB is not enough to control the arrhythmia, either supraventricular or ventricular. Is necessary to know that amiodarone is effective for suppression of supraventricular and ventricular arrhythmias. However, it does not reduce the incidence of sudden cardiac death or overall mortality, as was showed by Bardy et al. Thereby we need to be precautions when managing patients with HFrEF, taking into consideration the indications for implantable cardioverter-defibrillator (ICD) and for catheter ablation, the topic that will be covered later in our article.
Some words have to be said about the therapies used for HF patients with specific conditions. In patients with an obstructive hypertrophic cardiomyopathy (OHCM) the treatment with mavacamten, a myosin inhibitor, showed clear benefits improving symptoms and decreasing the obstruction of the LV as was showed in Explorer-HCM trial. Also, the patients with HF have comorbidities as lung disease, kidney disease, diabetes, neurological disease, depression or cancer. All these topics are very well underlined in the 2021 ESC guidelines for the diagnosis and treatment of the acute and chronic HF. It is another reason why the MDT approach is so important. For example, the current data show that depression affects 20-30% of the patients with HF. The preferred pharmacological therapies are serotonin reuptake inhibitors. As was shown by SADHART-CHF and MOOD-HF trials, both sertraline and escitalopram are safe in the treatment of HF patients, but did not provide greater reduction in depression or improved cardiovascular status compared to placebo.
When the patient is taking all recommended drugs, we can say is on optimal medical treatment (OMT).
3.2. Non-pharmacological management
When we speak about non-pharmacological management of the HF we mean cardiac surgery, interventional cardiology, electrophysiology, sleep-disordered breathing treatment, physical therapy, patient education, etc. Many cases of HF patients need a Heart Team approach that includes a cardiac surgeon, an interventional cardiologist, an electrophysiologist and a general cardiologist. The Heart Team will elaborate the best strategy taking into considerations the risks, benefits and the patient preferences.
3.2.1. Cardiac surgery
What can a cardiac surgeon offer as a member of the Heart Team and MDT? First of all, many patients with advanced HF patients will need heart transplantation at some moment. It remains the gold standard for refractory HF cases. As a bridge to heart transplantation some patients will need left ventricular assist device (LVAD) implantation. Some patients with HF have indications for coronary artery by-pass graft (CABG) surgery, others have indications for valvular surgery, some patients with OHCM need myectomy with mitral valve repair. The data provided by the recent trials and meta-analysis have refined the guidelines for Heart Team and continue to provide recommendations. For example, as was showed by Wu et al. the surgery to repair mitral valves in patients with severe left ventricular dysfunction do not improve mortality. This is why in secondary (functional) mitral regurgitation the Heart Team will avoid the surgery if possible. The team will verify if the patient with HFrEF is on OMT taking all recommended drugs as ARNI, BB, MRA, SGLT2; will evaluate the coronary arteries, will see if there are indications for cardiac resynchronization therapy (CRT) and will take the best decision. Some patients will need surgical valve repair. Patients with LV aneurysm refractory to the OMT may need surgical aneurysmectomy performed using Dor or another technique with improve in LV volumes, systolic function leading to symptomatic improvement. Cardiac surgeons will manage surgically the patients with OHCM. As was mentioned by Maron et al. the myectomy provides permanent abolition of outflow gradients with reversal of HF and highly favorable long term survival. Unfortunately, as was mentioned by Pelliccia et al. not all centers have the necessary experience performing very low number of myectomy procedures. And last, but not the least, in all the patients with atrial fibrillation (AF), when a cardiac surgery is planned the Heart Team need to discuss the indications for surgical AF ablation. The Maze procedure was refined during last years. In 2004 Levy described the use of Cardioblate (Medtronic) radiofrequency (RF) device to perform surgical ablation of the AF. In the recent review and meta-analysis was shown that surgical AF ablation during cardiac surgery improves freedom from AF. The 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery provide clear recommendation to consider surgical AF ablation during mitral valve surgery and in some non-mitral valve surgeries.
3.2.2. Interventional cardiology
As a valuable member of the MDT and Heart Team, the interventional cardiologist will have the opportunity to help the HF with minimally invasive techniques such a percutaneous coronary intervention (PCI), endovascular valvular procedures in selected patients. Bista et al. have published evidence of no apparent benefit of PCI in severe ischemic cardiomyopathy as compared to OMT, but that CABG improves outcomes in this patient population. Taking into consideration the gap in evidence, is important to follow the last updates related to the revascularization strategy. For example, Gallinoro et colleagues have found that in patients with HFrEF deferring revascularization of intermediate coronary stenoses based on fractional flow reserve is associated with a lower incidence of death and major adverse cardiovascular and cerebrovascular events at 10 years, DeVore at al. published data that in patients with stable coronary artery disease amenable to PCI and moderate or severe LV systolic dysfunction, the addition of PCI to medical therapy did not improve long-term mortality, or the composite of mortality or cardiovascular hospitalization. Transcatheter aortic valve implantation (TAVI) and surgical aortic replacement (SAR) in high risk patients was shown to have similar rates of survival at 1 year, although there were important differences in periprocedural. Later, the PARTNER 3 trial showed evidence that among patients with severe aortic stenosis who were at low surgical risk, the rate of the composite of death, stroke, or rehospitalization at 1 year was significantly lower with TAVI than with SAR. There are contradictory data about the endovascular mitral valve repair with the use of MitraClip device (Abbott), provided by MITRA-FR and COAPT randomized trials. The 2021 ESC guidelines for the diagnosis and treatment of the acute and chronic HF mention that the difference in the results could be explained by differences in trial design and recommends to perform percutaneous edge-to-edge mitral valve repair only in carefully selected patients who remain symptomatic (NYHA class II-IV) despite OMT, with moderate-to-severe or severe secondary mitral regurgitation and, favorable anatomical conditions.
3.2.3. Cardiac electrophysiology
In the last years an important role in the management of HF patients is played by the cardiac electrophysiologists (EP). What can an EP do for the management of HF? We can divide the task in two fields: cardiac implantable electronic devices (CIED) and catheter ablation (CA).
3.2.3.1. Cardiac implantable electronic devices
Let start with the fact that many patients with HF have an CIED. The interrogation of the device can provide important data about AF burden, thoracic impedance or ventricular arrhythmia. Using that information, we can better understand the efficiency of AF or ventricular arrhythmia treatment. We can use pacing maneuvers to perform conversion to sinus rhythm in patients with atrial flutter. In some HFrEF patient who developed pacemaker induced cardiomyopathy we will upgrade the device to CRT. The patients with HFrEF will need an ICD or CRTD for primary prevention of sudden cardiac death and we will consider ICD or CRTD implantation in all patient for secondary prevention. The importance of the ICD vs antiarrhythmic therapy was clearly demonstrated by randomized trials as AVID, CASH, CIDS. There is a 28% reduction in the relative risk of death with the ICD that is due almost entirely to a 50% reduction in arrhythmic death. In patients with non-ischemic cardiomyopathy (NICM) with HFrEF the DANISH trial did not show a significant benefit from ICD therapy, but the 2021 ESC guidelines for the diagnosis and treatment of the acute and chronic HF underlines the importance to understand that NICM is a heterogeneous condition, therefore careful evaluation of the indications should be done.
3.2.3.2. Catheter ablation
A huge area where an EP can provide important support in the management of HF patient is the management of the arrhythmias using CA. HF and AF are two disorders that frequently coexist and complicate one another. There is rate control strategy that can be achieved with medication, but also rhythm control strategy that can be achieved either with the use of antiarrhythmic drugs or CA. The CASTLE-AF trial showed that CA of AF improves the prognosis in patients with HFrEF, but the CABANA trial showed the improve in QOL only and not the prognosis in patient with CA vs. medical therapy. Another option in the management of the HF in patients with AF is ablate and pace strategy, when atrioventricular junction ablation is performed together with CRT implantation. The beneficial effect of such a strategy was provided by APAF-CRT trial. The trial showed that ablation plus CRT was superior to pharmacological therapy in reducing HF and hospitalization and improving QOL in elderly patients with permanent AF and narrow QRS. Ventricular arrhythmias need special attention in patients with HF. They can be a complication of the HF and present a significant risk for sudden cardiac death. But there can be also HF as a consequence of a ventricular arrhythmias as frequent premature ventricular contractions, idioventricular rhythm, etc. As we early mentioned, Bardy et al. showed that amiodarone is effective for suppression of supraventricular and ventricular arrhythmias, however, it does not reduce the incidence of sudden cardiac death or overall mortality. So, after considering ICD or CRTD implantation, a catheter ablation can be performed to avoid shocks or to avoid the need in amiodarone therapy.
3.3. Our center approach
Republic of Moldova is a country in eastern Europe with about 3 million inhabitants. Moldova has a range of public and private medical facilities, public agencies, and authorities. The health care institutions could be primary, secondary, or tertiary level. In order to obtain free access to the specialized care, health care providers must be contracted with the National Health Insurance Company. Our center provides the full spectrum of services for patients with HF from ambulatory consultations to invasive open-heart surgery and interventional procedures.
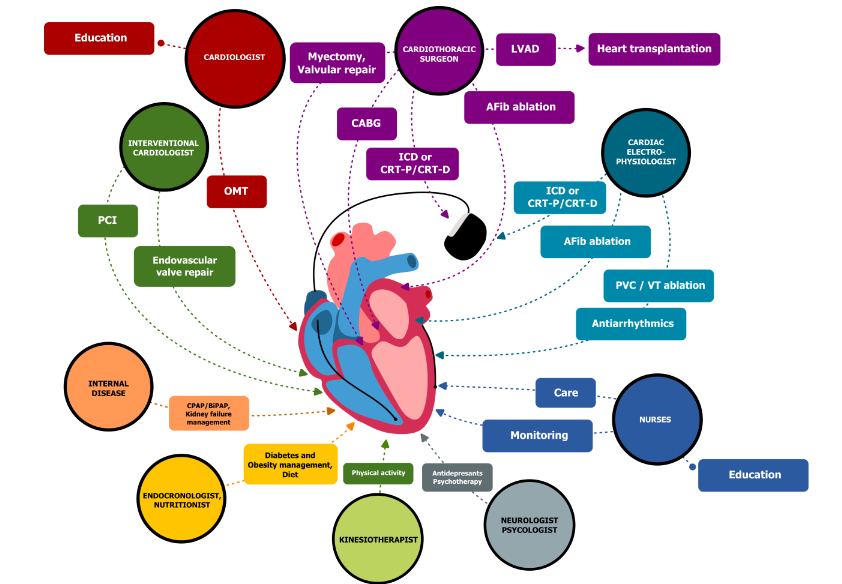
As is schematically shown in Figure 1, the MDT that is providing care for HF patients in our center is led by general cardiologist and includes cardiac surgeons, interventional cardiologists, EP, endocrinologists/nutritionist, internal disease physicians, neurologist, psychologist, kinesiotherapist and nurses. Every member of the team has its specific role in HF care. The leading role in the patient education have general cardiologists and nurses. Some more specific decisions about the interventional management are made by Heart Team, that includes a cardiac surgeon, an interventional cardiologist, an electrophysiologist and a general cardiologist. Our cardiac surgery team is one of the most experienced teams in the region. The cardiac surgeons of our center perform open heart procedures via classical sternotomy and via minimally invasive techniques in valvular pathologies, perform on-pump and off-pump CABG surgeries using both arterial and venous grafts, perform complex surgical interventions for the treatment of OHCM, LV aneurysms. We early mentioned the article of Pelliccia et al. about the importance of high number of myectomy procedures to provide good quality of care for patients with OHCM. We want to mention that our surgical team performs about 10-17 surgeries per year using transmitral myectomy approach with complex mitral valve repair.
In 2013 our cardiac surgeons implemented the use of RF ablation of AF during heart surgery. We are using Cardioblate (Medtronic) device and perform RF ablation especially in cases of mitral valve surgery. More that 100 procedures were performed from 2013. Additional retrospective study is necessary to provide information about the freedom from recurrence. Unfortunately, at the moment there is no legal base in our country to perform heart transplantation. Neither LVAD are implanted in our center. The interventional cardiologists perform the full amount of PCI procedures, including complex cases, but unfortunately not the full amount of endovascular valve procedures. For example, procedures as endovascular mitral valve repair or TAVI are not performed in our hospital taking into consideration the lack of NHIC financial support at the moment. Our EP team also had a lot of challenges in the treatment of the HF patients. As we published earlier, we started to perform CRT implantations in 2016, but was only 2019 when we obtained NHIC financial support and could increase the number of the cases. In the last years we implant more then 15 CRTD and more then 10 ICD per year. From 2021 we use only quadripolar LV leads for primary implants. Even there is current evidence of no clinical benefits of the use of quadripolar vs bipolar LV leads, we prefer quadripolar leads taking into consideration less issues with phrenic nerve capture, more possibilities to choose lower threshold and to increase battery longevity. We use the LBBAP in patients with HFrEF or HFmrEF and need for pacing, or in patients with indications for biventricular CRT but without suitable anatomy. Our EP team makes special efforts to properly manage the patients with CRT. The team is trained to perform the optimization of the CRT using the right ventricle – LV delay adjustment with the echocardiographic guidance. Also, in order to obtain good response to CRT in patient with AF we evaluate the percentage of the biventricular pacing and, if necessary, adjust the dosage of BB or digoxin to decrease the basal heart rate.
In 2015 we started to perform RF catheter ablations for the first time in our country. We started with conventional procedures as atrial flutter, premature ventricular contractions or ventricular tachycardia ablations in patients with HF. In 2018 we starter to perform pulmonary vein isolation using 28 mm Arctic Front Advance cryoballoon (Medtronic) and in 2024 we started to perform RF ablation with Columbus Black Suit 4.2 3D mapping system (Everpace) using FireMagic TrueForce SuperCool (Everpace) catheter for ablation. The preliminary results we obtained after cryoballoon ablation show 70% freedom from AF recurrence, that is comparable to the results from other centers. In the next years we will provide information about our results of AF management with the use of RF ablation. In our center, some of the internal disease physicians, neurologist and pulmonologist are specialized in the management of the sleep-disordered breathing as central sleep apnoea and obstructive sleep apnoea. As is mentioned in the 2021 ESC guidelines for the diagnosis and treatment of the acute and chronic HF, sleep-disordered breathing is present in more than one third of patients with HF. The patients with HFrEF and HFmrEF being considered for a sleep-disordered breathing treatment with positive pressure airway mask must undergo formal sleep study taking into consideration the fact that if sleep-disordered breathing is caused by central sleep apnoea, positive pressure airway masks are contraindicated in HFrEF patients.
Another task of the MDT includes the screening for depression. If the diagnosis of depression is established, we can either decide to use SRI as we mentioned earlier, or to use psychotherapy. Edward Everett Hale said: “Together — one of the most inspiring words in the English language. Coming together is a beginning; keeping together is progress; working together is success.” This is true also related to HF management. The care of HF is multidisciplinary and needs some super-specialized team members. Without some of them or with improper collaboration between the members there will be poor outcome for some patients. Crucial decisions about the time for CIED implantation or indications for CA need special attention. Some patients have a chance to avoid unnecessary ICD or CRTD implantation when the full potential of the OMT is used. This is where working together is success. The large real-world registry data demonstrated that the pharmacological therapy is frequently not enough optimized prior to primary prevention ICD placement. Another aspect we want to recognize is that we frequently do not have enough time for proper patient educations. A lot of improvements could be done related to communication between MDT and the HF patient. Relatives are another important player in this scenario. This is why is important to implement proper strategies to achieve the established targets.
4. Conclusions
The care of HF is multidisciplinary and needs some super-specialized team members. An MDT approach is considered the best way to deliver care for HF patients. The MDT has an important role in using the current knowledge and resources to provide the best possible care for the patient with HF. There are lot of tricky situations when a decision made without the team support could be a wrong decision. To avoid such situations is important to implement the MDT approach and to continually educate the team members, keeping the pace with the HF management updates. Edward Everett Hale said: “Together — one of the most inspiring words in the English language. Coming together is a beginning; keeping together is progress; working together is success.” This is true also related to HF management. With MDT approach we can do the care of HF patients as much as possible suitable for the patient needs.
Conflicts of interests:
None
Funding statement:
The authors have no funding to report.
Acknowledgement:
We want to express our gratitude to the entire team of the physicians and nurses providing care for the HF patient in Medpark International Hospital, Chisinau, Republic of Moldova.
References
- Savarese G, Lund LH. Global Public Health Burden of Heart Failure. Card Fail Rev. 2017;3(1):7-11. Doi:10.15420/cfr.2016:25:2
- Emmons-Bell S, Johnson C, Roth G. Prevalence, incidence and survival of heart failure: a systematic review. Heart. 2022;108(17):1351-1360. Published 2022 Aug 11. Doi:10.1136/heartjnl-2021-320131
- Authors/Task Force Members:, McDonagh TA, Metra M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2022;24(1):4-131. Doi:10.1002/ejhf.2333
- Authors/Task Force Members:, McDonagh TA, Metra M, et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2024;26(1):5-17. Doi:10.1002/ejhf.3024
- MERIT-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999;353:2001-2007.
- Hjalmarson A, Goldstein S, Fagerberg B, et al. Effects of controlled-release metoprolol on total mortality, hospitalizations, and well-being in patients with heart failure: the Metoprolol CR/XL Randomized Intervention Trial in congestive heart failure (MERIT-HF). MERIT-HF Study Group. JAMA. 2000;283(10):1295-1302. Doi:10.1001/jama.283.10.1295
- Packer M, Fowler MB, Roecker EB, et al. Effect of carvedilol on the morbidity of patients with severe chronic heart failure: results of the carvedilol prospective randomized cumulative survival (COPERNICUS) study. Circulation. 2002;106(17):2194-2199. Doi:10.1161/01.cir.0000035653.72855.bf
- CIBIS-II Investigators and Committees. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial. Lancet 1999;353:9-13.
- Flather MD, Shibata MC, Coats AJ, et al. Randomized trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS). Eur Heart J. 2005;26(3):215-225. Doi:10.1093/eurheartj/ehi115
- The CONSENSUS Trial Study Group. Enalapril for congestive heart failure. N Engl J Med. 1987;317(21):1349-1351. Doi:10.1056/NEJM198711193172112
- SOLVD Investigators, Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med. 1991;325(5):293-302. Doi:10.1056/NEJM199108013250501
- Packer M, Poole-Wilson PA, Armstrong PW, et al. Comparative effects of low and high doses of the angiotensin-converting enzyme inhibitor, lisinopril, on morbidity and mortality in chronic heart failure. ATLAS Study Group. Circulation. 1999;100(23):2312-2318. Doi:10.1161/01.cir.100.23.2312.
- Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med. 1999;341(10):709-717. Doi:10.1056/NEJM199909023411001
- Zannad F, McMurray JJ, Krum H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med. 2011;364(1):11-21. Doi:10.1056/NEJMoa1009492
- McMurray JJ, Ostergren J, Swedberg K, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-Added trial. Lancet. 2003;362(9386):767-771. Doi:10.1016/S0140-6736(03)14283-3
- Granger CB, McMurray JJ, Yusuf S, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: the CHARM-Alternative trial. Lancet. 2003;362(9386):772-776. Doi:10.1016/S0140-6736(03)14284-5
- Cohn JN, Tognoni G; Valsartan Heart Failure Trial Investigators. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345(23):1667-1675. Doi:10.1056/NEJMoa010713
- McMurray JJ, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371(11):993-1004. Doi:10.1056/NEJMoa1409077
- Packer M, Anker SD, Butler J, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N Engl J Med. 2020;383(15):1413-1424. Doi:10.1056/NEJMoa2022190
- McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N Engl J Med. 2019;381(21):1995-2008. Doi:10.1056/NEJMoa1911303
- Anker SD, Butler J, Filippatos G, et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021;385(16):1451-1461. Doi:10.1056/NEJMoa2107038
- Solomon SD, McMurray JJV, Claggett B, et al. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. 2022;387(12):1089-1098. Doi:10.1056/NEJMoa2206286
- Swedberg K, Komajda M, Böhm M, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study [published correction appears in Lancet. 2010 Dec 11;376(9757):1988. Lajnscak, M [corrected to Lainscak, M]; Rabanedo, I Roldan [corrected to Rabadán, I Roldan]; Leva, M [corrected to Ieva, M]]. Lancet. 2010;376(9744):875-885. Doi:10.1016/S0140-6736(10)61198-1
- Macdougall IC, White C, Anker SD, et al. Intravenous Iron in Patients Undergoing Maintenance Hemodialysis [published correction appears in N Engl J Med. 2019 Jan 31;380(5):502. Doi:10.1056/NEJMx180044]. N Engl J Med. 2019;380(5):447-458. Doi:10.1056/NEJMoa1810742
- Kalra PR, Cleland JGF, Petrie MC, et al. Intravenous ferric derisomaltose in patients with heart failure and iron deficiency in the UK (IRONMAN): an investigator-initiated, prospective, randomised, open-label, blinded-endpoint trial. Lancet. 2022;400(10369):2199-2209. Doi:10.1016/S0140-6736(22)02083-9
- Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure [published correction appears in N Engl J Med. 2005 May 19;352(20):2146]. N Engl J Med. 2005;352(3):225-237. Doi:10.1056/NEJMoa043399
- Olivotto I, Oreziak A, Barriales-Villa R, et al. Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): a randomised, double-blind, placebo-controlled, phase 3 trial [published correction appears in Lancet. 2020 Sep 12;396(10253):758. Doi:10.1016/S0140-6736(20)31872-9]. Lancet. 2020;396(10253):759-769. Doi:10.1016/S0140-6736(20)31792-X
- Rutledge T, Reis VA, Linke SE, Greenberg BH, Mills PJ. Depression in heart failure a meta-analytic review of prevalence, intervention effects, and associations with clinical outcomes. J Am Coll Cardiol. 2006;48(8):1527-1537. Doi:10.1016/j.jacc.2006.06.055
- Sokoreli I, de Vries JJG, Pauws SC, Steyerberg EW. Depression and anxiety as predictors of mortality among heart failure patients: systematic review and meta-analysis. Heart Fail Rev. 2016;21(1):49-63. Doi:10.1007/s10741-015-9517-4
- Sbolli M, Fiuzat M, Cani D, O’Connor CM. Depression and heart failure: the lonely comorbidity. Eur J Heart Fail. 2020;22(11):2007-2017. Doi:10.1002/ejhf.1865
- O’Connor CM, Jiang W, Kuchibhatla M, et al. Safety and efficacy of sertraline for depression in patients with heart failure: results of the SADHART-CHF (Sertraline Against Depression and Heart Disease in Chronic Heart Failure) trial. J Am Coll Cardiol. 2010;56(9):692-699. Doi:10.1016/j.jacc.2010.03.068
- Angermann CE, Gelbrich G, Störk S, et al. Effect of Escitalopram on All-Cause Mortality and Hospitalization in Patients With Heart Failure and Depression: The MOOD-HF Randomized Clinical Trial. JAMA. 2016;315(24):2683-2693. Doi:10.1001/jama.2016.7635
- Morton G, Masters J, Cowburn PJ. Multidisciplinary team approach to heart failure management. Heart. 2018;104(16):1376-1382. Doi:10.1136/heartjnl-2016-310598
- Varshney AS, Singh JP, Vaduganathan M. A Heart Team approach to contemporary device decision-making in heart failure. Eur J Heart Fail. 2022;24(3):562-564. Doi:10.1002/ejhf.2445
- Wu AH, Aaronson KD, Bolling SF, Pagani FD, Welch K, Koelling TM. Impact of mitral valve annuloplasty on mortality risk in patients with mitral regurgitation and left ventricular systolic dysfunction. J Am Coll Cardiol. 2005;45(3):381-387. Doi:10.1016/j.jacc.2004.09.073
- Ferrell BE, Jimenez DC, Ahmad D, et al. Surgical ventricular reconstruction for ischemic cardiomyopathy-a systematic review and meta-analysis of 7,685 patients. Ann Cardiothorac Surg. 2022;11(3):226-238. Doi:10.21037/acs-2021-ami-17
- Maron MS, Rastegar H, Dolan N, et al. Outcomes Over Follow-up ≥10 Years After Surgical Myectomy for Symptomatic Obstructive Hypertrophic Cardiomyopathy. Am J Cardiol. 2022;163:91-97. Doi:10.1016/j.amjcard.2021.09.040
- Pelliccia F, Seggewiss H, Cecchi F, et al. Septal Ablation Versus Surgical Myomectomy for Hypertrophic Obstructive Cardiomyopathy. Curr Cardiol Rep. 2021;23(11):165. Published 2021 Oct 1. Doi:10.1007/s11886-021-01600-5
- Levy P. Atrial fibrillation ablation during mitral valve surgery using the medtronic cardioblate® irrigated radiofrequency system. Operative Techniques in Thoracic and Cardiovascular Surgery, Vol 9, No 1 (Spring), 2004;34-42. Doi:10.1053/j.optechstcvs.2004.01.002
- McClure GR, Belley-Cote EP, Jaffer IH, et al. Surgical ablation of atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. Europace. 2018;20(9):1442-1450. Doi:10.1093/europace/eux336
- Van Gelder IC, Rienstra M, Bunting KV, et al. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. Published online August 30, 2024. Doi:10.1093/eurheartj/ehae176
- Bista R, Zghouzi M, Jasti M, et al. Outcomes of Percutaneous Revascularization in Severe Ischemic Left Ventricular Dysfunction. Curr Cardiol Rep. 2024;26(5):435-442. Doi:10.1007/s11886-024-02045-2
- Gallinoro E, Paolisso P, Di Gioia G, et al. Deferral of Coronary Revascularization in Patients With Reduced Ejection Fraction Based on Physiological Assessment: Impact on Long-Term Survival. J Am Heart Assoc. 2022;11(19):e026656. Doi:10.1161/JAHA.122.026656
- DeVore AD, Yow E, Krucoff MW, et al. Percutaneous coronary intervention outcomes in patients with stable coronary disease and left ventricular systolic dysfunction. ESC Heart Fail. 2019;6(6):1233-1242. Doi:10.1002/ehf2.12510
- Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187-2198. Doi:10.1056/NEJMoa1103510
- Mack MJ, Leon MB, Thourani VH, et al. Transcatheter Aortic-Valve Replacement with a Balloon-Expandable Valve in Low-Risk Patients. N Engl J Med. 2019;380(18):1695-1705. Doi:10.1056/NEJMoa1814052
- Iung B, Armoiry X, Vahanian A, et al. Percutaneous repair or medical treatment for secondary mitral regurgitation: outcomes at 2 years. Eur J Heart Fail. 2019;21(12):1619-1627. Doi:10.1002/ejhf.1616
- Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. N Engl J Med. 2018;379(24):2307-2318. Doi:10.1056/NEJMoa1806640
- Connolly SJ, Hallstrom AP, Cappato R, et al. Meta-analysis of the implantable cardioverter defibrillator secondary prevention trials. AVID, CASH and CIDS studies. Antiarrhythmics vs Implantable Defibrillator study. Cardiac Arrest Study Hamburg. Canadian Implantable Defibrillator Study. Eur Heart J. 2000;21(24):2071-2078. Doi:10.1053/euhj.2000.2476
- Køber L, Thune JJ, Nielsen JC, et al. Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure. N Engl J Med. 2016;375(13):1221-1230. Doi:10.1056/NEJMoa1608029
- Moss AJ, Hall WJ, Cannom DS, et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N Engl J Med. 2009;361(14):1329-1338. Doi:10.1056/NEJMoa0906431
- Cleland JG, Freemantle N, Erdmann E, et al. Long-term mortality with cardiac resynchronization therapy in the Cardiac Resynchronization-Heart Failure (CARE-HF) trial. Eur J Heart Fail. 2012;14(6):628-634. Doi:10.1093/eurjhf/hfs055
- Daubert C, Gold MR, Abraham WT, et al. Prevention of disease progression by cardiac resynchronization therapy in patients with asymptomatic or mildly symptomatic left ventricular dysfunction: insights from the European cohort of the REVERSE (Resynchronization Reverses Remodeling in Systolic Left Ventricular Dysfunction) trial. J Am Coll Cardiol. 2009;54(20):1837-1846. Doi:10.1016/j.jacc.2009.08.011
- Diaz JC, Duque M, Aristizabal J, et al. The Emerging Role of Left Bundle Branch Area Pacing for Cardiac Resynchronisation Therapy. Arrhythm Electrophysiol Rev. 2023;12:e29. Published 2023 Dec 1. Doi:10.15420/aer.2023.15
- Carlisle MA, Fudim M, DeVore AD, Piccini JP. Heart Failure and Atrial Fibrillation, Like Fire and Fury. JACC Heart Fail. 2019;7(6):447-456. Doi:10.1016/j.jchf.2019.03.005
- Marrouche NF, Brachmann J, Andresen D, et al. Catheter Ablation for Atrial Fibrillation with Heart Failure. N Engl J Med. 2018;378(5):417-427. Doi:10.1056/NEJMoa1707855
- Packer DL, Mark DB, Robb RA, et al. Effect of Catheter Ablation vs Antiarrhythmic Drug Therapy on Mortality, Stroke, Bleeding, and Cardiac Arrest Among Patients With Atrial Fibrillation: The CABANA Randomized Clinical Trial. JAMA. 2019;321(13):1261-1274. Doi:10.1001/jama.2019.0693
- Packer DL, Piccini JP, Monahan KH, et al. Ablation Versus Drug Therapy for Atrial Fibrillation in Heart Failure: Results from the CABANA Trial. Circulation. 2021;143(14):1377-1390. Doi:10.1161/circulationaha.120.050991
- Mark DB, Anstrom KJ, Sheng S, et al. Effect of Catheter Ablation vs Medical Therapy on Quality of Life Among Patients With Atrial Fibrillation: The CABANA Randomized Clinical Trial [published correction appears in JAMA. 2019 Jun 18;321(23):2370. Doi:10.1001/jama.2019.7682]. JAMA. 2019;321(13):1275-1285. Doi:10.1001/jama.2019.0692
- Brignole M, Pokushalov E, Pentimalli F, et al. A randomized controlled trial of atrioventricular junction ablation and cardiac resynchronization therapy in patients with permanent atrial fibrillation and narrow QRS. Eur Heart J. 2018;39(45):3999-4008. Doi:10.1093/eurheartj/ehy555
- Linde C. Pace and ablate better than drugs in patients with heart failure and atrial fibrillation: lessons from the APAF-CRT mortality trial. Eur Heart J. 2021;42(46):4740-4742. Doi:10.1093/eurheartj/ehab695
- Latchamsetty R, Bogun F. Premature Ventricular Complex-Induced Cardiomyopathy. JACC Clin Electrophysiol. 2019;5(5):537-550. Doi:10.1016/j.jacep.2019.03.013
- Darciuc R, Boiciuc I, Cazacu A, Eraslan H, Diker E. Complete reversal of arrhythmia-induced cardiomyopathy. Case Report. Open J Clin Med Case Rep. 2023;9(13). Doi:10.52768/2379-1039/2023
- Turcanu G, Domente S, Buga M, Richardson E. Republic of Moldova health system review. Health Syst Transit. 2012;14(7):1-151.
- Darciuc R, Boiciuc I, Ivanov D, Diker E. Cardiac resynchronization therapy in the Republic of Moldova: The beginning of the journey. Heart Rhythm O2. 2022;3(6Part B):728-730. Published 2022 Dec 16. Doi:10.1016/j.hroo.2022.09.019
- Keilegavlen H, Schuster P, Hovstad T, Faerestrand S. Clinical outcome of cardiac resynchronization therapy in patients randomized to an active fixation bipolar left ventricular lead versus a passive quadripolar lead. Scand Cardiovasc J. 2021;55(3):153-159. Doi:10.1080/14017431.2020.1869299
- Hakemi EU, Doukky R, Parzynski CS, Curtis JP, Madias C. Quadripolar versus bipolar leads in cardiac resynchronization therapy: An analysis of the National Cardiovascular Data Registry. Heart Rhythm. 2020;17(1):81-89. Doi:10.1016/j.hrthm.2019.07.028
- Maurhofer J, Asatryan B, Haeberlin A, et al. Acute and long-term outcomes of quadripolar IS-4 versus bipolar IS-1 left ventricular leads in cardiac resynchronization therapy: A retrospective registry study. Pacing Clin Electrophysiol. 2023;46(5):365-375. Doi:10.1111/pace.14686
- Vanderheyden M, De Backer T, Rivero-Ayerza M, et al. Tailored echocardiographic interventricular delay programming further optimizes left ventricular performance after cardiac resynchronization therapy. Heart Rhythm. 2005;2(10):1066-1072. Doi:10.1016/j.hrthm.2005.07.016
- Darciuc R, Boiciuc I, Diker E. Cryoballoon ablation of atrial fibrillation in Republic of Moldova. Three years of experience. One Health & Risk Management, Supplement. 2021;2(4):41.
- Heeger CH, Tscholl V, Salloum O, et al. What is the real recurrence rate after cryoballoon-based pulmonary vein isolation? Lessons from rhythm follow-up based on implanted cardiac devices with continuous atrial monitoring. Heart Rhythm. 2018;15(12):1844-1850. Doi:10.1016/j.hrthm.2018.07.016
- Choudhury R, Coutino HE, Darciuc R, et al. Continuous monitoring after second-generation cryoballoon ablation for paroxysmal atrial fibrillation in patients with cardiac implantable electronic devices. Heart Rhythm. 2019;16(2):187-196. Doi:10.1016/j.hrthm.2018.08.015
- IsHak WW, Hamilton MA, Korouri S, et al. Comparative Effectiveness of Psychotherapy vs Antidepressants for Depression in Heart Failure: A Randomized Clinical Trial. JAMA Netw Open. 2024;7(1):e2352094. Published 2024 Jan 2. Doi:10.1001/jamanetworkopen.2023.52094

