Multimodal Imaging of Orbital Necrobiotic Xantogranuloma
Multimodality Imaging Features of Orbital Necrobiotic Xantogranuloma: A Case Report and Literature Review
Ghazal Shadmani, MD¹; Athar Shadmani, MD²
- Mallinckrodt Institute of Radiology, Washington University School of Medicine, St. Louis, MO, USA
- Bascom Palmer Eye Institute, University of Miami health system, FL, USA
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: Shadmani, G., and Shadmani, A., 2024. Multimodality Imaging Features of Orbital Necrobiotic Xantogranuloma: A Case Report and Literature Review. Medical Research Archives, [online] 12(11). https://doi.org/10.18103/mra.v12i11.6014
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i11.6014
ISSN 2375-1924
Abstract
Background: Necrobiotic xanthogranuloma (NXG) is a rare non-Langerhans histiocytosis that primarily manifests as skin lesions and is often associated with hematologic disorders, particularly monoclonal gammopathy. The orbit is the most common site of extracutaneous involvement in NXG. Since its first description in 1980, the molecular pathology and dermatologic characteristics of NXG have been extensively studied. However, there is no comprehensive multimodality description of the imaging features of orbital NXG in the literature. This case report aimed to describe the imaging features of NXG and provide a brief overview of the imaging differentials.
Patient presentation: A 16-year-old white young man was referred to our center for evaluation of a 6-year history of a slowly growing right orbital mass, which resulted in vision loss in his right eye. His medical history was notable for systemic juvenile rheumatoid arthritis (sJRA) and a gain-of-function (GOF) mutation in the IKAROS gene, which predisposed him to immune dysregulation. To further characterize the orbital lesion, orbital computed tomography (CT), magnetic Resonance Imaging (MRI), and Fluorodeoxyglucose Positron Emission Tomography/Magnetic Resonance Imaging (FDG PET/MRI) were performed. A biopsy of the most hypermetabolic component of the infiltrative retro-orbital mass confirmed the diagnosis of NXG. He received high-dose immunosuppressive and IVIG therapy and bone marrow stem cell transplantation, which resulted in a slight reduction in the right orbital mass.
Conclusion: This paper presents the first multimodality imaging features of orbital necrobiotic xanthogranuloma in an adolescent patient.
Keywords
Necrobiotic xanthogranuloma, imaging features, orbital mass, histiocytosis, case report
Introduction:
Necrobiotic xanthogranuloma (NXG), first described by Kossard and Winkelmann in 1980, is a rare multisystemic form of non-Langerhans cell histiocytosis. It is primarily characterized by chronic and progressively enlarging yellow‒orange skin papules and plaques, most frequently around the eyes. The mean age of onset is at the 6th decade of life, and there is no known sex predilection. Monoclonal proteins can be detected in up to 85% of cases (most notably, IgG kappa), and multiple myeloma (MM) or monoclonal gammopathy of undetermined significance (MGUS) is observed in 25% of cases. The specific cause of NXG is still unknown, but it is theorized that paraproteins might act as catalysts or play a supporting role in the development of granulomas. Furthermore, there is an association between NXG and immune system dysregulation. Treatment consists mainly of corticosteroids and IVIG. Orbital involvement is one of the most common extracutaneous manifestations in 27-50% of NXG patients and is associated with variable presentations of uveitis, scleritis, and proptosis. Despite extensive research on the molecular pathology and dermatologic features of NXG, the imaging characteristics of orbital NXG have not been fully elucidated in the literature. In general, the imaging differential diagnosis of orbital masses includes infectious, inflammatory, or tumoral processes. Tumoral involvement can be further categorized into benign and malignant types. Differentiating NXG from other inflammatory and infiltrative conditions can be challenging, particularly given the significant overlap with other subtypes of histiocytosis, such as Erdheim-Chester disease (ECD) and Rosai-Dorfman disease (RDD), as well as additional inflammatory orbitopathies like IgG4-related disease and nonspecific orbital inflammatory syndrome (NSOIS). A definitive diagnosis requires a combination of clinical assessment, imaging findings, and histopathological analysis. This paper aims to review the CT, MRI, and FDG PET characteristics of orbital NXG and compare these imaging features with the primary differential diagnoses. To the best of our knowledge, this is the first description of the multimodality imaging features of orbital NXG.
Case Report:
The patient’s father provided consent for the publication of the images in a medical journal.
A 16-year-old young white man presented to our institution with a 6-year history of a slowly enlarging right orbital mass, unresponsive to multiple immunosuppressive treatments and several surgical interventions. The mass caused vision loss in the right eye due to compressive optic neuropathy and corneal scar from exposure keratitis. Repeated biopsies showed chronic inflammation and negative microbial staining or malignancy. Other notable medical problems include systemic juvenile idiopathic arthritis (sJIA) at age 5, moderate seasonal allergies, polyarthralgia, and myalgia. The family history was notable for multiple female relatives with immune-related arthritis and pulmonary diseases, including the patient’s mother, who had been diagnosed with optic neuritis and autoimmune connective tissue disease.
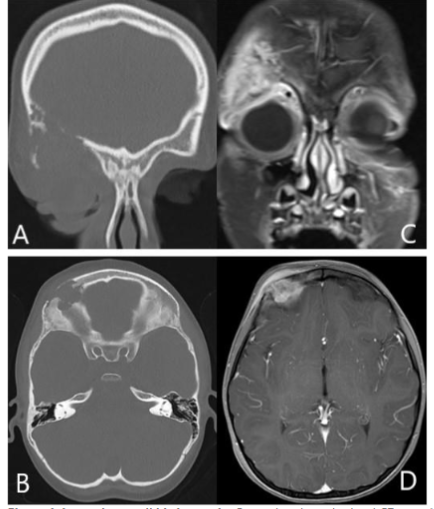
An orbital CT scan showed severe right orbital proptosis, right-sided pre- and post-septal soft tissue thickening with intraconal/retrobulbar space extension and diffuse thickening of the adjacent bone secondary to chronic inflammation without erosion. There was also diffuse paranasal sinus mucosal thickening.
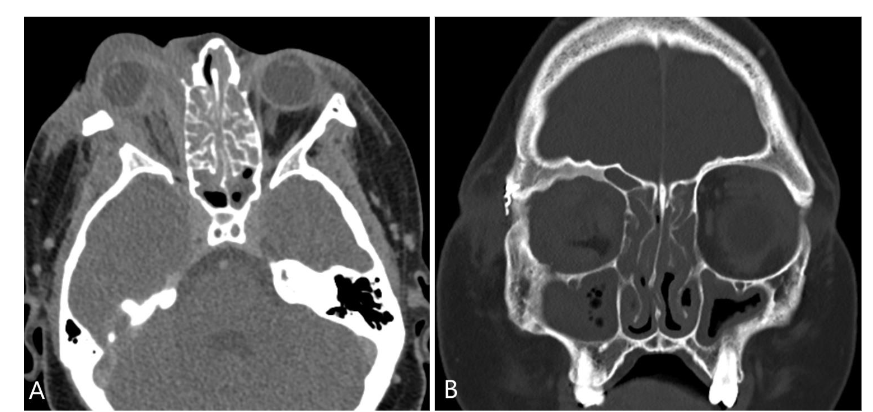
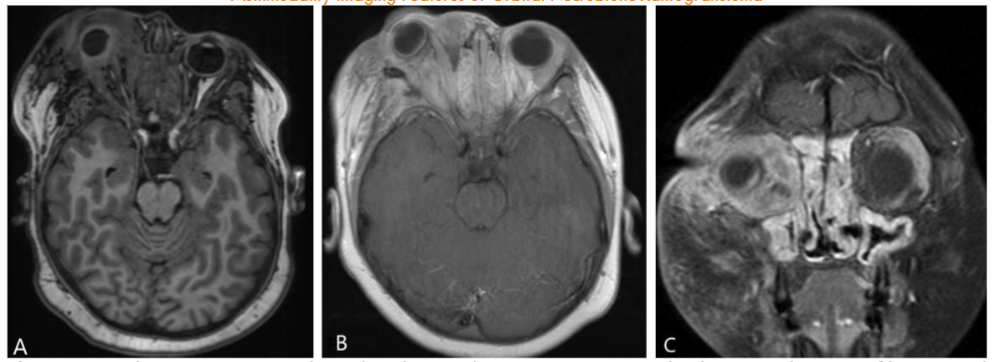
Orbital MRI provided a clearer characterization of diffuse retro-orbital soft tissue thickening, involving the fat, extraocular muscles, and lacrimal glands, leading to pronounced right orbital proptosis and globe eversion. The right globe was flattened with optic nerve stretching. The retrobulbar mass extended posteriorly into the orbital apex without cavernous sinus involvement, and there was also further extension into the lateral face, affecting the temporalis muscle.
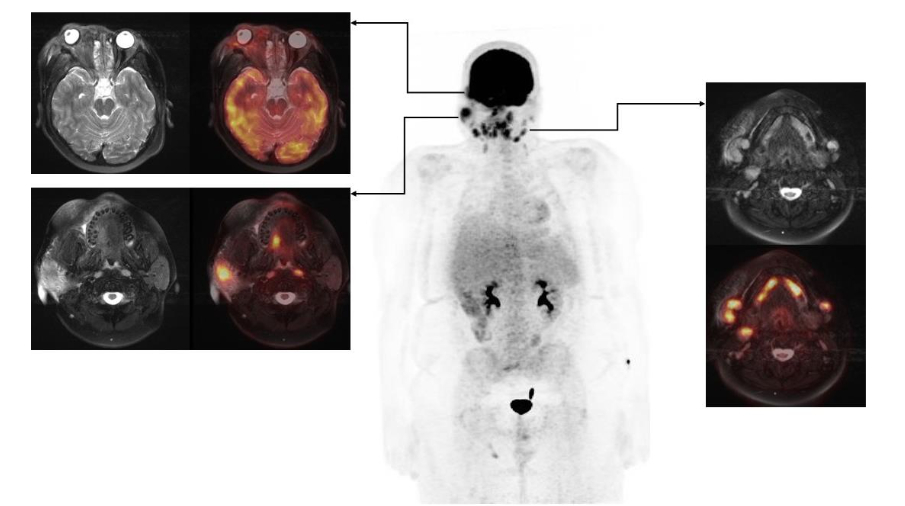
FDG PET/MRI was used to characterize the extent of the disease and determine the optimal site for tissue biopsy. FDG PET/MRI revealed multiple markedly FDG-avid cervical lymph nodes, focal intense uptake in the right parotid region, and focal uptake in the right orbital infiltrative mass with maximum Standardized Uptake Value (SUV) of 13. Guided by FDG PET/MRI, the patient underwent a biopsy of the most FDG-avid region of the right orbital mass, which was diagnosed as necrobiotic xanthogranuloma with increased IgG4 expression. The patient was treated with cyclophosphamide, corticosteroid, and high-dose IVIG followed by bone marrow stem cell transplantation and showed a good response in terms of both imaging and clinical evaluation. The genetic and histopathologic findings of this case are discussed in detail by Guess et al.
Discussion:
Histiocytosis are a group of rare inflammatory disorders marked by the buildup of macrophages, dendritic cells, or monocyte-derived cells in various tissues and organs that affect both children and adults. This group of disorders includes more than 100 different subtypes, which have been recently categorized into five main groups: (1) Langerhans-related histiocytosis, (2) Rosai-Dorfman histiocytosis, (3) cutaneous and mucocutaneous histiocytosis, (4) malignant histiocytosis, and (5) hemophagocytic lymphohistiocytosis and macrophage activation syndrome.
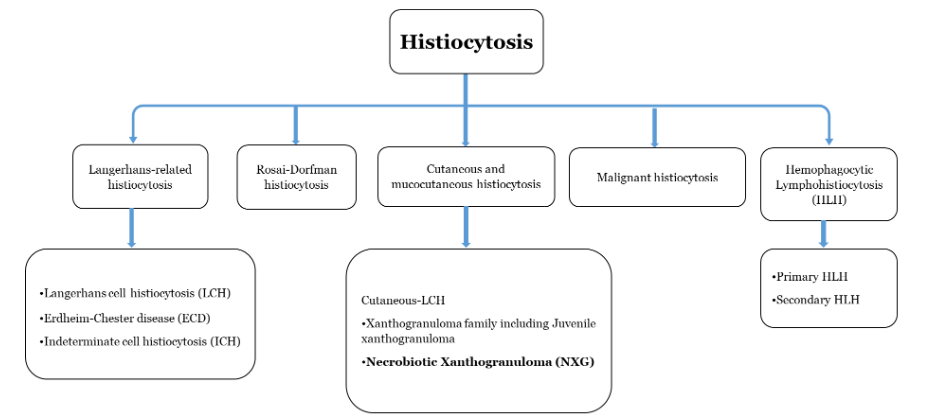
Langerhans cell histiocytosis (LCH), Erdheim-Chester disease (ECD), and Rosai-Dorfman disease (RDD) are different subtypes of histiocytosis that can potentially involve the orbit. Langerhans cell histiocytosis (LCH) is the most prevalent form of histiocytosis, impacting multiple organs and systems, with the lungs and bones being the most commonly affected, particularly in pediatric patients. Orbital involvement occurs in approximately 20% of LCH patients, often presenting as lesions in the bony orbit, most frequently in the upper outer region. These lesions can extend to nearby soft tissue, leading to symptoms such as proptosis, swelling, and restricted eye movement. Orbital LCH is more common in children than in adults, but it can also occur in adults. The presence of additional destructive skull base lesions can be a valuable clue in narrowing down the differential diagnosis. In this case, the absence of adjacent osseous erosions and other systemic indicators of LCH helped to lower LCH on the differential diagnosis list.
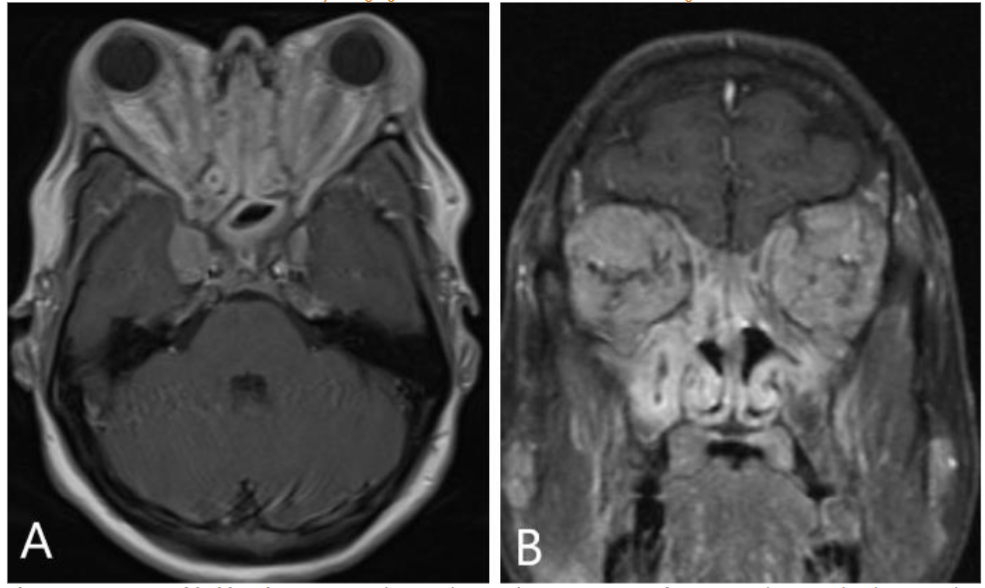
Erdheim-Chester disease (ECD) is a rare subtype of histiocytosis that predominantly affects the skeletal system in more than 95% of cases. Orbital involvement occurs in 25% of ECD patients, typically appearing on imaging as an infiltrative intraconal or retrobulbar mass, leading to exophthalmos.
Rosai-Dorfman disease (RDD), also known as sinus histiocytosis with massive lymphadenopathy, is a benign, self-limited condition that primarily affects lymph nodes. As its name suggests, the paranasal sinuses are the most common sites of extranodal involvement. Orbital involvement is seen in approximately 7% of patients, typically manifesting as unilateral, infiltrative, and enhancing retro-orbital soft tissue masses affecting the extraocular muscles, retro-orbital fat, and lacrimal gland. This results in proptosis, with contrast enhancement that may be either uniform or heterogeneous.
Both NXG and Juvenile xanthogranuloma (JXG) are under the subgroup of cutaneous and mucocutaneous histiocytosis. Although JXG can involve the orbit with well-defined lesion and bone erosions, it primarily affects the iris and is more common in children. The characteristic yellow-orange iris lesions and the potential for hyphemia are distinctive.
Two other chronic inflammatory conditions of the eye that can exhibit similar imaging features, though not classified under histiocytosis family, are IgG4-related disease (IgG4-RD) and nonspecific orbital inflammatory syndrome (NSOIS).
IgG4-RD is a systemic inflammatory condition of unclear origin marked by the infiltration of tissues by IgG4-positive plasma cells and the presence of sclerosing inflammation. Orbital involvement can be observed in up to 40% of cases, with proptosis and periorbital edema being the most common clinical presentations. Pain is reported in approximately 7% of these patients. Imaging of IgG4-related orbitopathy typically reveals an infiltrative retro-orbital mass, primarily involving the extraocular muscles, with the lateral rectus being most commonly affected. Notably, the anterior tendons are spared in the majority of cases. It is often accompanied by lacrimal gland involvement and paranasal mucosal thickening.
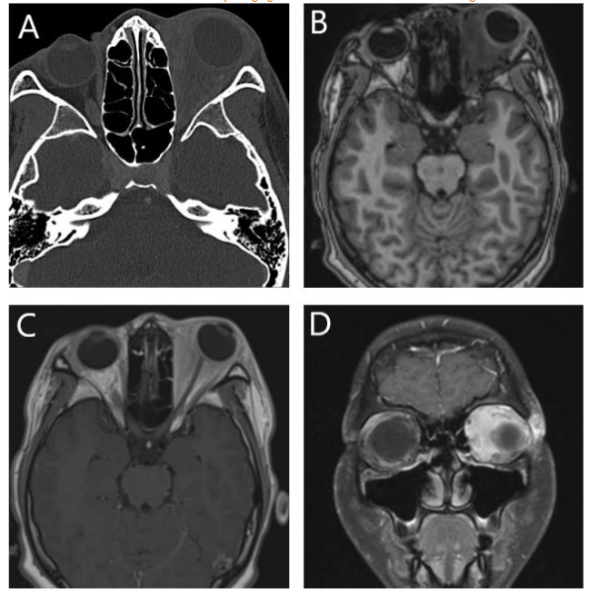
Nonspecific orbital inflammatory syndrome (NSOIS), formerly known as orbital pseudotumor, is another differential diagnosis to be considered in this patient, although it is uncommon in the pediatric population. NSOIS can affect various orbital structures, including the extraocular muscles, retrobulbar fat, optic nerves, and globe. The extraocular muscles are the most frequently involved muscles, with characteristic involvement of the musculotendinous insertions. The clinical presentations typically include bilateral proptosis, painful ophthalmoplegia, and diplopia. CT and MRI findings differ based on the disease’s location but typically reveal thickening of the extraocular muscles, including musculotendinous insertion, enlargement of the lacrimal gland with surrounding inflammatory changes, and a thickened, enhancing sclera.
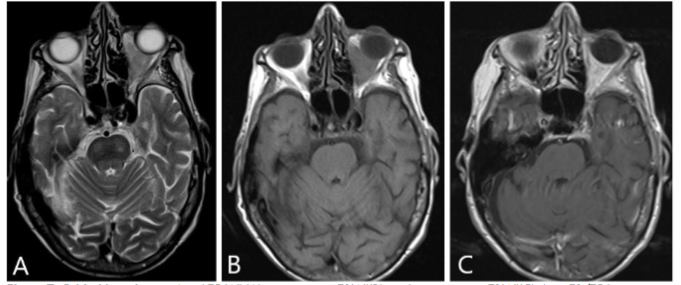
Differentiating between NXG and conditions such as IgG4-RD, RDD, ECD, and NSIOS is particularly challenging, especially in advanced cases such as this one, where accurate assessment of musculotendinous involvement of extraocular muscles is difficult. Orbital lymphoma, another imaging consideration in this case, typically presents as a well-defined soft tissue mass, most commonly located in the superolateral quadrant of the orbit. It usually has an extraconal component with intraconal extension, particularly in larger retrobulbar masses. Compared to orbital NXG, orbital lymphoma generally has a more well-defined appearance with less surrounding retrobulbar fat stranding. Furthermore, the absence of additional supporting evidence for lymphoma, combined with the presence of serum paraproteinemia in this case, argue against lymphoma. Leiomyosarcoma, a rapidly progressing orbital malignancy common in teenagers, was considered highly unlikely in this case due to the chronic nature of the symptoms and lack of aggressive osseous erosion.
Conclusion:
Herein, we present a multimodality imaging description of orbital necrobiotic xanthogranuloma and demonstrate the imaging features of most of its differential diagnoses. While certain imaging features can help distinguish orbital NXG from other orbital infiltrative and inflammatory conditions, a combination of clinical findings, imaging, and histopathological analysis is essential for accurate diagnosis and appropriate management.
References:
- Kossard steven, Winkelmann RK. NECROBIOTIC XANTHOGRANULOMA. Aust J Dermatol [Internet]. 1980 Aug 2 [cited 2024 Sep 3];(25):85–8. Available from: doi: 10.1111/j.1440-0960.1980.tb00148.x.
- Nelson CA, Zhong CS, Hashemi DA, Ashchyan HJ, Brown-Joel Z, Noe MH, et al. A Multicenter Cross-Sectional Study and Systematic Review of Necrobiotic Xanthogranuloma with Proposed Diagnostic Criteria. JAMA Dermatol. 2020 Mar 1;156(3):270–9.
- Hilal T, DiCaudo DJ, Connolly SM, Reeder CB. Necrobiotic xanthogranuloma: a 30-year single-center experience. Ann Hematol. 2018 Aug 1;97(8):1471–9.
- Steinhelfer L, Kühnel T, Jägle H, Mayer S, Karrer S, Haubner F, et al. Systemic therapy of necrobiotic xanthogranuloma: a systematic review. Vol. 17, Orphanet Journal of Rare Diseases. BioMed Central Ltd; 2022.
- Guess R, Harocopos G, Bednarski JJ, Hassmann LM, Bigley TM. Pediatric Necrobiotic Xanthogranuloma as a Novel Phenotype of IKAROS Gain of Function. Vol. 44, Journal of Clinical Immunology. Springer; 2024.
- Olson RM, Harrison AR, Maltry A, Mokhtarzadeh A. Periorbital necrobiotic xanthogranuloma successfully treated with intravenous immunoglobulin. Case Rep Ophthalmol. 2018 Jan 23;9(1):70–5.
- Huynh KN, Nguyen BD. Histiocytosis and neoplasms of macrophage-dendritic cell lineages: Multimodality imaging with emphasis on PET/CT. Radiographics. 2021;41(2):576–94.
- Duong Nguyen B. F-FDG PET/CT and MRI of Necrobiotic Xanthogranuloma [Internet]. 2013. Available from: www.nuclearmed.com
- Emile JF, Abla O, Fraitag S, Horne A, Haroche J, Donadieu J, et al. Revised classification of histiocytoses and neoplasms of the macrophage-dendritic cell lineages. 2016; Available from: http://ashpublications.org/blood/article-pdf/127/22/2672/1393781/2672.pdf
- Zaveri J, La Q, Yarmish G, Neuman J. More than just langerhans cell histiocytosis: A radiologic review of histiocytic disorders. Radiographics. 2014;34(7):2008–24.
- Das J, Soibam R, Tiwary B, Magdalene D, Paul S, Bhuyan C. Orbital manifestations of Langerhans Cell Histiocytosis: A report of three cases. Oman J Ophthalmol. 2009;2(3):137.
- Wu C, Li K, Hei Y, Lan P, Mu X. MR imaging features of orbital Langerhans cell Histiocytosis. BMC Ophthalmol. 2019 Dec 19;19(1).
- Pan Z, Kleinschmidt-DeMasters BK. CNS Erdheim-Chester disease: A challenge to diagnose. Vol. 76, Journal of Neuropathology and Experimental Neurology. Oxford University Press; 2017. p. 986–96.
- Civit JJR, Godoy D, Conde A, Arencibia J, Medel R, Limeres M, et al. Orbital histiocytosis with systemic involvement: A case with complex affiliations. Saudi Journal of Ophthalmology. 2020 Oct 1;34(4):319–23.
- Wang E, Anzai Y, Paulino A, Wong J. Rosai-Dorfman Disease Presenting with Isolated Bilateral Orbital Masses: Report of Two Cases. Vol. 22, AJNR Am J Neuroradiol. 2001.
- La Barge D V., Salzman KL, Harnsberger HR, Ginsberg LE, Hamilton BE, Wiggins RH, et al. Sinus histiocytosis with massive lymphadenopathy (Rosai-Dorfman disease): Imaging manifestations in the head and neck. Vol. 191, American Journal of Roentgenology. 2008.
- Yataco-Vicente JA, Araujo-Castillo R V, Fuentes MHL. Orbital presentation of Rosai-Dorfman disease Presentación orbitaria de enfermedad de Rosai-Dorfman [Internet]. 2018. Available from: http://www.revistasbolivianas.org.bo/scielo.php?script=sci
- Chalard F, Nguyen T, Morel B, Leiber LM, Roux CJ, Petit P, et al. Juvenile Xanthogranuloma of the Head and Neck: Imaging Findings in 11 Cases. J Pediatr Hematol Oncol. 2024 Jun 3;46:368–80.
- Samara WA, Khoo CTL, Say EAT, Saktanasate J, Eagle RC, Shields JA, et al. Juvenile xanthogranuloma involving the eye and ocular adnexa: Tumor control, visual outcomes, and globe salvage in 30 patients. Ophthalmology. 2015 Oct 1;122(10):2130–8.
- Tiegs-Heiden CA, Eckel LJ, Hunt CH, Diehn FE, Schwartz KM, Kallmes DF, et al. Immunoglobulin G4-related disease of the orbit: Imaging features in 27 patients. American Journal of Neuroradiology. 2014;35(7):1393–7.
- Ebbo M, Patient M, Grados A, Groh M, Desblaches J, Hachulla E, et al. Ophthalmic manifestations in IgG4-related disease. Medicine (United States). 2017 Mar 1;96(10).
- Paydar A, Jenner ZB, Simkins TJ, Chang YM, Hacein-Bey L, Ozturk A, et al. Autoimmune disease of head and neck, imaging, and clinical review. Vol. 35, Neuroradiology Journal. SAGE Publications Inc.; 2022. p. 545–62.
- Joseph AK, Guerin JB, Eckel LJ, Dalvin LA, Keating GF, Liebo GB, et al. Imaging Findings of Pediatric Orbital Masses and Tumor Mimics. Radiographics. 2022 May 1;42(3):880–97.
- Gerrie SK, Rajani H, Navarro OM, Lyons CJ, Marie E, Frayn CS, et al. Pediatric orbital lesions: non-neoplastic extraocular soft-tissue lesions. Pediatr Radiol. 2024 May 1;54(6):910–21.
- Priego G, Majos C, Climent F, Muntane A. Orbital lymphoma: Imaging features and differential diagnosis. Vol. 3, Insights into Imaging. 2012. p. 337–44.
- Huynh KN, Kong MJ, Nguyen BD. Anatomic and Functional Imaging of Immunoglobulin G4–related Disease and Its Mimics. Radiographics. 2023 Mar 1;43(3).
- Nagaraj UD, Koch BL. Imaging of orbital infectious and inflammatory disease in children. Pediatric Radiology. 2021 May 12;51:1149–61.

