Neurophysiological Monitoring in Laparoscopic Nerve Decompression
Multimodal neurophysiological monitoring during laparoscopic pelvic nerve decompression in deep endometriosis: results from a prospective single-center cohort
Sierra Brozon, A1*; Riveralainez Rios, X1; Ponce Flores, J1; Cabrera Carranco, R1, Kondo, W2; Rivas Cruz, M1; Obando Barrientos; A1; Chura Paco, J1; Pimentel Valladolid; A1; Diaz Roesch, F1.
- Minimal Invasive Surgery Department, Doyenne Institute, Angeles Hospital Network, Mexico City, Mexico.
- Minimal Invasive Surgery Department, Vita Batel Hospital, Curitiba, Paraná, Brazil.
OPEN ACCESS
PUBLISH: Jul 25, 2025
CITATION:SIERRA BROZON, Ana Gabriela et al. Multimodal neurophysiological monitoring during laparoscopic pelvic nerve decompression in deep endometriosis: results from a prospective single-center cohort. Medical Research Archives, [S.l.], v. 13, n. 7, july 2025.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ABSTRACT
Objective: To describe the technique and assess the clinical utility of intraoperative neurophysiological monitoring (IONM) during laparoscopic pelvic nerve decompression in women with deep endometriosis, with a focus on its integration into surgical workflow and its role in preserving pelvic neurological function.
Methods: A prospective descriptive case series was conducted at a Latin American tertiary center between January 2024 and May 2025. Fourteen women with MRI-confirmed deep endometriosis and pelvic nerve involvement underwent laparoscopic decompression assisted by a multimodal IONM protocol. Data on demographics, intraoperative findings, neurophysiological alerts, and clinical symptoms were recorded. Postoperative outcomes were assessed at 1, 3, and 6 months. Descriptive statistics were applied.
Results: A prospective follow-up was conducted in 14 patients, with clinical outcomes evaluated from a minimum of 1 month (92.9%) to up to 6 months (85.7%) postoperatively. All patients exhibited preoperative evidence of nerve compression, most commonly affecting the sciatic (57.1%) and pudendal (50%) nerves. Multimodal intraoperative neurophysiological monitoring (IONM) was successfully implemented in all cases (100%) without any major intraoperative complications. At the 1-month follow-up, 78.6% of patients reported symptom improvement, with sustained clinical benefit observed in 75% at 6 months. No permanent motor deficits were noted, and all transient postoperative symptoms resolved spontaneously. The most frequently improved symptoms included dyschezia, urinary dysfunction, and chronic pelvic pain.
Conclusion: Laparoscopic pelvic nerve decompression with real-time IONM is a safe and reproducible technique that may enhance surgical outcomes and neurological preservation in patients with deep endometriosis. The present study provides foundational evidence for its systematic incorporation in Latin American surgical practice, though larger and controlled studies are warranted to confirm these findings and evaluate long-term efficacy.
Keywords
neurophysiological monitoring, laparoscopic surgery, pelvic nerve decompression, deep endometriosis, intraoperative monitoring
Introduction
Deep endometriosis with neural involvement represents one of the most severe and functionally disabling variants of the disease. When this condition infiltrates the lumbosacral plexus, patients are often presented with chronic neuropathic pain, urinary or gastrointestinal disturbances, and sexual dysfunction symptoms that significantly deteriorate their quality of life and complicated treatment planning. Although laparoscopic surgery has become the preferred approach for managing deep pelvic endometriotic lesions, the anatomical complexity of the retroperitoneal space introduces a high risk of iatrogenic nerve injury during surgical dissection. Intraoperative neurophysiological monitoring (IONM) has emerged as a valuable adjunct in high-risk surgeries involving the nervous system. Its implementation in neurosurgical, orthopedic, and peripheral nerve procedures has demonstrated the potential to detect early signs of neural compromise, thereby enabling timely intraoperative adjustments to prevent long-term deficits. Specifically, modalities such as motor evoked potentials, somatosensory evoked potentials, and electromyography provide real-time insights into the functional integrity of critical neural pathways.
The evolving field of neuropelveology has brought renewed attention to intrapelvic nerve entrapment syndromes, which may arise from fibrosis, anomalous musculature, vascular abnormalities, or direct endometriotic infiltration. Clinical manifestations typically include sharp, radiating pain along dermatomal distributions, in addition to dyschezia, deep dyspareunia, or motor weakness depending on the nerves affected. Commonly implicated structures include the pudendal, obturator, and sciatic nerves, all of which traverse anatomically constrained regions that are vulnerable to pathological compression.
Despite the increasing recognition of nerve-sparing surgical principles and IONM as tools to enhance functional outcomes, standardized protocols remain scarce particularly in Latin America. Moreover, few publications detail the operative steps or neurophysiological workflows required for effective implementation of IONM in pelvic surgery. This prospective study not only evaluates the clinical outcomes of laparoscopic pelvic nerve decompression with multimodal IONM in patients with deep endometriosis but also aims to describe the intraoperative neuromonitoring protocol in a standardized and reproducible format. By documenting the technique in detail, we seek to promote its integration in other specialized centers and support the development of evidence-based guidelines in neuropelvic surgery.
Methods
STUDY DESIGN AND OBJECTIVES
This prospective, observational and descriptive case series was conducted between January 2024 and May 2025 at Instituto Doyenne (Mexico City), a referral center for endometriosis, minimally invasive gynecologic and neuropelvic surgery. The study enrolled all consecutive patients undergoing laparoscopic pelvic nerve decompression for deep endometriosis with documented use of intraoperative neurophysiological monitoring (IONM). No priori power calculation was performed, as the intent was descriptive rather than inferential. The primary aim was to describe the technical application and clinical relevance of multimodal IONM during laparoscopic pelvic nerve surgery. Secondary aims included the identification of the most involved pelvic nerves, the characterization of neurophysiological patterns observed during surgery, and the evaluation of postoperative symptom trajectories and safety outcomes.
Participants and Setting
The study included women with a confirmed diagnosis of deep endometriosis involving pelvic nerve structures, as documented by magnetic resonance imaging (MRI). All surgeries were performed between January 2024 and May 2025 at a specialized center for endometriosis, minimally invasive gynecologic and neuropelvic surgery. Only patients who underwent laparoscopic nerve decompression with full intraoperative neurophysiological monitoring and postoperative clinical follow-up of at least one month were eligible for inclusion.
Inclusion Criteria
- confirmed deep endometriosis involving pelvic nerves.
- Undergoing laparoscopic surgery with the intent of nerve decompression.
- Availability of complete IONM recordings and postoperative clinical follow-up of at least one month.
Exclusion Criteria
- Procedures without a decompressive component
- Lack of intraoperative neuromonitoring documentation.
- Patients with pre-existing non-endometriosis-related pelvic neuropathies.
All patients provided informed written consent for the surgical procedure, neuromonitoring, and academic use of anonymized clinical data.
SURGICAL TEAM AND SETTING
All procedures were performed by an interdisciplinary team consisting of an expert pelvic surgeon (specialized in endometriosis excision), a neuropelveologist, and a board-certified neurophysiologist who established and interpreted the IONM in real time. Under general anesthesia, patients were placed in lithotomy with a 25-30° Trendelenburg tilt. A four-port laparoscopic access was used to create pneumoperitoneum (14 mmHg), followed by retroperitoneal dissection using the standardized LANN/ISON approach. Depending on intraoperative findings, decompression was performed on one or more of the following structures: pudendal, sciatic nerves, and sacral roots L5-S4. Techniques were adapted to the anatomical segment, using vascular landmarks to identify nerves and ensure preservation of surrounding structures.
ANESTHETIC MANAGEMENT FOR NEUROMONITORING
Anesthesia consisted of total intravenous anesthesia (TIVA) with propofol and remifentanil continuous infusions; no volatile agents were used. After a single intubating dose of rocuronium, no additional neuromuscular blockers were given. Core temperature was maintained > 35.5°C; mean arterial pressure was kept > 80 mm Hg. End-tidal CO₂ was held at 35-40 mm Hg.
The intra-operative Neurophysiological Monitoring Protocol is described in table 1.
| Component | Specification |
|---|---|
| System | Cadwell ® Cascade Pro multimodality platform |
| Modalities | Train-of-four (TOF), trans-cranial motor evoked potentials (TcMEPs), somato-sensory evoked potentials (SSEPs), continuous EMG (cEMG), triggered EMG (tEMG) |
| Electrodes | Disposable sub-dermal needle pairs, 27 G × 10 mm; cork-screw scalp electrodes (10-20 system) at C3/C4 (stimulation) and Fz’, Cz’, C’3, C’4, C5 cervical (SSEP recording); adhesive surface electrodes for bilateral tibial-posterior and pudendal nerve stimulation; sub-dermal needles in: abductor pollicis (TOF), bulbocavernosus & transverse perineal (pudendal), adductor magnus (obturator), rectus femoris (femoral), biceps femoris (sciatic), medial gastrocnemius (S1), abductor hallucis (L5), and external anal sphincter (S2-S4) bilaterally. |
| Stimulation parameters | tEMG: monopolar probe 275 mm, 1-5 mA; TcMEP: 8-pulse trains, 450 V, ISI 2 ms; filters cEMG 10-3000 Hz, sweep 10 ms, sensitivity 100 µV/div. |
| Baseline tests | After induction and intubation, impedance check → TOF to confirm adequate neuromuscular recovery → baseline TcMEP |
| Alarms | TcMEP amplitude ↓ > 50 % from baseline; SSEP latency ↑ > 10 % or amplitude ↓ > 50 %; neurotonic discharges on cEMG |
| Corrective actions | Immediate surgical pause, irrigation, haemodynamic optimisation, and re-check of anaesthetic depth |
| Anaesthetic protocol | Total intravenous anaesthesia (propofol/remifentanil infusions); no volatile agents or bolus muscle relaxants; mean arterial pressure maintained > 80 mm Hg |
A full visualization of electrode positioning and neuromonitoring signals is available as supplementary material. (see Figure 2; https://www.youtube.com/watch?v=IBGasZb3Ehs)
DATA COLLECTION AND STUDY VARIABLES
Clinical and electrophysiological variables were recorded prospectively using standardized forms.
Primary qualitative variables:
- Nerves affected (nominal): pudendal, obturator, sciatic, S2-S4 roots.
- Preoperative symptoms
- Postoperative symptoms
- Intraoperative neurophysiological alerts:
- baseline.
- SSEP latency increase >10% or amplitude reduction >50%
- cEMG
Quantitative variables:
- Age (continuous)
- VAS pain scores (ordinal)
STATISTICAL ANALYSIS
A descriptive analysis was conducted using SPSS version 27. Categorical variables were expressed as frequencies and percentages. Continuous data were reported as mean ± standard deviation or median with interquartile range, based on Shapiro-Wilk normality testing. Due to the small sample size and exploration nature of the study, no inferential statistics or comparative analyses were applied.
ETHICAL CONSIDERATIONS
This study adhered to the ethical standards outlined in the Declaration of Helsinki. The institutional review board approved the protocol. All participants provided written informed consent covering the procedure, IONM, and publication of anonymized data.
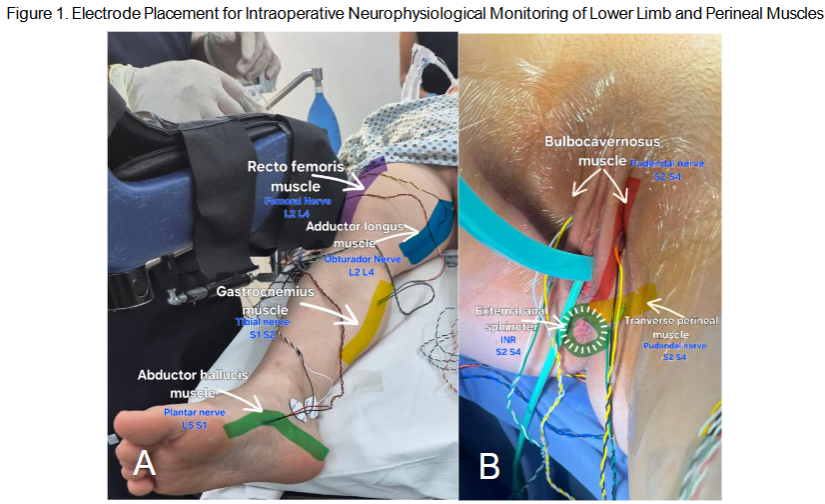
Figure 1. Intraoperative CMAP recordings using subdermal needle electrodes. A. Electrode placement on lower limb muscles: rectus femoris, adductor longus, gastrocnemius, and abductor hallucis. B. Perineal electrodes on bulbocavernosus, transverse perineal, and external anal sphincter muscles. All muscles are innervated by key pelvic and lumbosacral nerves (L2-S4) relevant for surgical neuromonitoring.
Figure 2. Bilateral Electrode Configuration for Intraoperative Neuromonitoring During Pelvic Nerve Decompression Figure 2. AD. Final setup of subdermal electrodes for intraoperative neurophysiological monitoring (IONM) prior to laparoscopic pelvic nerve surgery. The images illustrate the completed configuration of bilateral needle electrodes placed in lower limb and perineal muscles, following anatomical landmarks and prior to trocar placement and surgical draping.
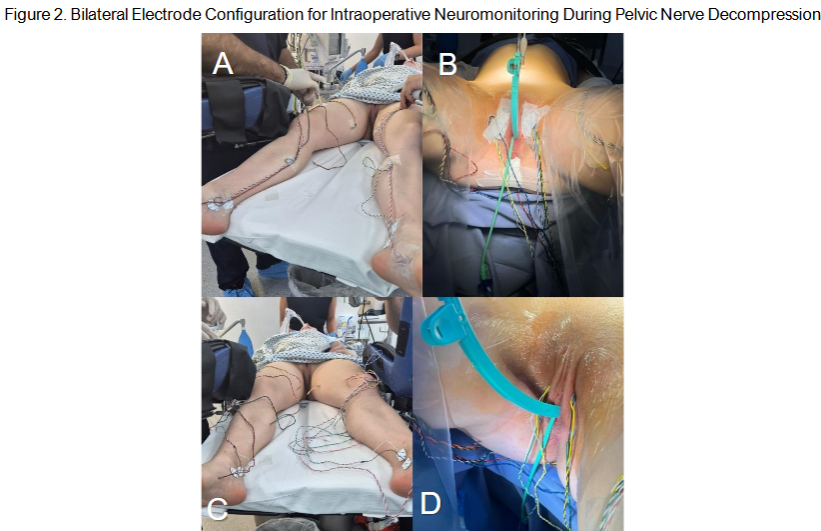
Figure 3. Intraoperative neurophysiological confirmation of pelvic nerve identification using triggered electromyography (tEMG). A 275 mm monopolar stimulation probe was employed to deliver pulses at intensities ranging from 1 to 5 mV. (A) Shows stimulation of the obturator nerve. (B) Stimulation of the sciatic nerve after complete exposure. These tests were performed to verify functional integrity of the nerves before proceeding with surgical dissectio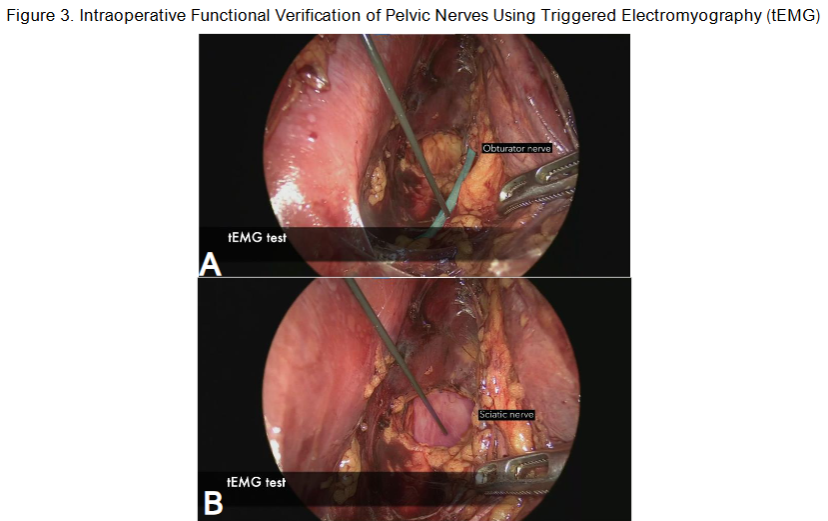
Surgical Decompression Approach and Anatomical Landmarks
After standard trocar placement and induction of baseline neuromonitoring (TcMEPs), the retroperitoneal space was accessed via a wide peritoneal incision. Dissection was carried medially between the iliopsoas muscle and the external iliac vessels to expose the obturator fossa and iliolumbar region, where vascular structures adjacent to the lumbosacral trunk were carefully preserved. Targeted exposure and decompression of key nerves were performed as follows:
- Obturator Nerve: Accessed in the lateral paravesical space, using the obturator artery as a landmark. Dissection followed the nerve superiorly to its exit through the obturator canal.
- Sacral Nerve Roots (S1-S4): Identified via blunt dissection of the pararectal space, medial to the ureter. The hypogastric fascia was opened to visualize the roots laterally.
- Sciatic Nerve and Plexus: Accessed via the lumbosacral space lateral to the external iliac vessels. Exposure extended to the suprapiriform area of the sciatic foramen and included selective vascular transections for visualization.
- Pudendal Nerve: Isolated by creating a surgical window medial to the obturator nerve, followed by dissection of the sacrospinous ligament to trace the nerve before its exit through the lesser sciatic foramen.
Results
A total of 14 women met the inclusion criteria and underwent laparoscopic pelvic nerve decompression with multimodal intraoperative neurophysiological monitoring (IONM). The patients ranged in age from 24 to 44 years, with a mean age of 34.0 ± 5.6 years. All participants (100%) reported at least one pelvic pain-related symptom prior to surgery, highlighting the significant clinical burden and functional impairment associated with deep endometriosis involving neural structures. The most frequently reported preoperative complaints included dyschezia in 10 patients (71.4%, 95% CI: 45.4-88.3%), urinary symptoms in 8 (57.1%, 95% CI: 32.6-78.6%), deep dyspareunia, chronic pelvic pain, and lower limb pain each in 6 patients (42.9%), superficial dyspareunia and sitting pain in 4 (28.6%), and dysmenorrhea in 8 patients 3 with a VAS score >6. Additional symptoms included gastrointestinal disturbances in 6 patients (42.9%), abnormal uterine bleeding in 2 (14.3%), and vulvodynia in 1 patient (7.1%). Intraoperative exploration confirmed compression of one or more pelvic nerves in all patients (100%). The most affected nerves were the sciatic nerve in 8 patients (57.1%), the pudendal nerve in 7 (50.0%), the sacral roots S2-S3 in 5 (35.7%), and the obturator nerve in 1 (7.1%). Combined nerve compressions were also observed, with simultaneous sciatic and pudendal nerve involvement in 4 patients (28.6%), pudendal and S2-S3 root compression in 1 (7.1%), and pudendal and obturator nerve compression in another 1 (7.1%). These findings are consistent with known anatomical pathways susceptible to endometriotic entrapment and reinforce previously described neuropelveological theories and anatomical studies.
Intraoperative Neurophysiological Monitoring and Safety
Multimodal IONM was successfully implemented in all 14 procedures (100%). No intraoperative technical failures or signal losses were reported. Neurophysiological alerts occurred in 3 patients (21.4%), prompting temporary surgical pausing and correction of intraoperative parameters. There were no major intraoperative complications or adverse events related to the monitoring setup.
Postoperative Outcomes
In the immediate postoperative period, three patients (21.4%) reported transient symptoms such as localized paresthesia and mild neuropathic discomfort, all of which resolved spontaneously within a few days. At the one-month follow-up (n = 13), 10 patients (76.9%) reported significant improvement or complete resolution of their pain-related symptoms. However, persistent symptoms were noted in three patients (21.4% of the total cohort), including chronic pelvic pain in two cases (14.3%) and lower limb pain in one case (7.1%). At the three-month follow-up (n = 12), symptom recurrence or persistence was documented in five patients (35.7%), with four experiencing chronic pelvic pain (28.6%) and one reporting rectal pain (7.1%). Additionally, a new case of transient lower limb weakness (7.1%) was identified, which resolved within weeks. By the six-month follow-up (n = 12), sustained clinical benefit was observed in most patients, with chronic pelvic pain persisting in only two (14.3%) and a single case of vulvodynia (7.1%) reported without any associated motor deficit. Overall, 75% of patients experienced meaningful relief of neuropathic and pelvic pain symptoms by six months, supporting the primary study hypothesis that IONM-assisted decompression is both safe and functionally effective.
| Total patients | 14 | 100% | IONM | 14 | 100% |
|---|---|---|---|---|---|
| Compressed Nerves | 14 | 100% | PO complications | 0 | 0% |
| Sciatic | 8 | 57.1% | Immediate PO symptoms | 3 | 21.4% |
| Obturator | 1 | 7.1% | 1-month PO follow-up | 13 | 92.9% |
| Pudendal | 7 | 50% | 1-month PO symptoms | 3 | 21.4% |
| S2-S3 | 5 | 35.7% | Chronic pelvic pain | 2 | 14.3% |
| Sciatic and Pudendal | 4 | 28.6% | Lower limb pain | 1 | 7.1% |
| Obturator and Pudendal | 1 | 7.1% | 3-month PO follow-up | 12 | 85.7% |
| Pudendal and S2-S3 | 1 | 7.1% | 3-month PO symptoms | 5 | 35.7% |
| Presurgical symptoms | 14 | 100% | Rectal pain | 1 | 7.1% |
| Vulvodynia | 1 | 7.1% | Chronic pelvic pain | 4 | 28.6% |
| Sitting pain | 4 | 28.6% | Dysmenorrhea (VAS> 6) | 3 | 21.4% |
| Dysmenorrhea (VAS< 6) | 5 | 35.7% | Superficial dyspareunia | 4 | 28.6% |
| Deep dyspareunia | 6 | 42.9% | Dyschezia | 10 | 71.4% |
| Urinary symptoms | 8 | 57.1% | Chronic pelvic pain | 6 | 42.9% |
| Gastrointestinal symptoms | 6 | 42.9% | Abnormal uterine bleeding | 2 | 14.3% |
| Lower limb pain | 6 | 42.9% |
Discussion
This prospective case series confirms the feasibility, safety, and potential clinical value of integrating intraoperative neurophysiological monitoring (IONM) during laparoscopic pelvic nerve decompression for deep endometriosis in a Latin American referral center. At 6 months, 75% of patients reported sustained relief from neuropathic pelvic pain without evidence of permanent motor deficits or intraoperative complications, an outcome that underscores the functional benefit of combining meticulous anatomical dissection with continuous electrophysiological surveillance.
Although pelvic nerve involvement in endometriosis remains underrecognized, its clinical burden is substantial, and current literature offers limited prospective guidance. Our findings enrich this landscape by providing structured, real-world evidence from a single-center cohort in a region where such protocols are seldom implemented or documented. The methodological standardization of both surgical and neurophysiological techniques represents a step toward closing the gap between high-resource and underrepresented healthcare settings.
The clinical improvement rates observed in our study (78.6% at 1 month, 75% at 6 months) are in line with previously reported series from specialized European centers. For instance, a 5-year follow-up on partial sciatic nerve resection in women with endometriosis showed a 78% rate of gait normalization and significant VAS pain reduction from 9.3 to 1.2 points highlighting the long-term effectiveness of targeted neurodecompression. Similarly, Lemos reported 78% of patients treated via the Laparoscopic Neuronavigation (LANN) technique at a mean follow-up of 3.2 years. Notably, our protocol differs in its systematic use of multimodal IONM including TcMEP, SSEP, free EMG, and tEMG which may have contributed to the absence of new or permanent neurological deficits, even in cases involving deep sacral root dissection.
Our results also resonate with the study by Gundogdu et al. (2023), who reported improved symptom control and no iatrogenic nerve injuries following the introduction of IONM into laparoscopic pelvic surgery. These findings support the hypothesis that continuous motor pathway monitoring facilitates immediate intraoperative corrective measures, particularly in TcMEP amplitude are detected. Moreover, our data complement the broader conclusions drawn by Allahqoli et al. (2024) in their systematic review and meta-analysis, which highlighted the efficacy of neuropelveological strategies including neurolysis, neuromodulation, and decompression in reducing pain recurrence by 50%-75%. However, as they noted, heterogeneity across studies and a lack of randomized controlled trials still limit the generalizability of these results. In this context, our series contributes prospective, protocol-driven evidence to support future comparative trials evaluating monitored versus unmonitored decompression strategies.
Anatomically, the predominance of sciatic (57.1%) and pudendal (50.0%) nerve involvement observed in our cohort is consistent with the neural pathways most affected by deep endometriotic. The frequent co-occurrence of dyschezia and urinary symptoms further reinforces the known overlap between pelvic visceral and somatic innervation territories and validates the clinical relevance of a neuropelveological surgical approach.
From a regional perspective, the systematic use of IONM remains largely absent in gynecologic surgical practice across Latin America. By demonstrating the applicability of an internationally benchmarked protocol within a Latin American healthcare context, our findings establish a foundation for the wider adoption of such techniques across similarly equipped centers. In addition, the interdisciplinary structure of our surgical and neurophysiology team reflects a model of care that can be scaled in tertiary referral environments with appropriate training.
Despite its strengths, this study has notable limitations. The small sample size limits statistical power and precludes inferential analysis. The absence of a control group without IONM prevents definitive attribution of outcomes solely to neuromonitoring. Additionally, while short- and medium-term outcomes were favorable, the six-month follow-up may be insufficient to capture long-term neural recovery or pain recurrence, especially considering that functional restitution after nerve decompression can extend beyond one year.
Future multicenter studies with larger sample sizes, control cohorts, and extended follow-up will be necessary to validate our findings and assess the cost-effectiveness and scalability of IONM in routine clinical practice. Moreover, quality-of-life outcomes, sexual function, and fertility metrics should be incorporated to provide a more comprehensive assessment of patient benefit.
Conclusions
In this prospective Latin American series, laparoscopic pelvic nerve decompression assisted by a standardized multimodal intraoperative neurophysiological monitoring (IONM) protocol proved to be both feasible and clinically effective. By six months postoperatively, three out of four patients experienced meaningful reductions in neuropathic pelvic pain without permanent neurological deficits or major complications. The integration of real-time IONM facilitated nerve identification and preservation, likely contributing to the absence of iatrogenic injury in anatomically complex cases. Furthermore, the protocol was successfully implemented in a specialized regional center, demonstrating its reproducibility in non-European healthcare settings. These findings support the progressive incorporation of neuromonitoring in the surgical management of deep endometriosis with pelvic nerve involvement. However, the lack of a control group, limited sample size, and short follow-up duration underscore the need for multicenter, randomized studies with longer-term endpoints to fully determine the added value and cost-effectiveness of IONM in gynecologic neuropelveology.
Conflict of Interest Statement:
None.
Funding Statement:
None.
Acknowledgements:
None.
References:
- Singh R, Husain AM. Neurophysiologic intraoperative monitoring of the glossopharyngeal and vagus nerves. Journal of Clinical Neurophysiology. 2011; 28(6):582-586. doi:10.1097/WNP.0B013E31823E9B00
- Gundogdu EC, Kale A, Mercan M, et al. Integration of Intraoperative Neurophysiological Monitoring into Laparoscopic Pelvic Nerve Decompression Surgery: A Novel Technique for Protecting Pelvic Nerves. Clin Exp Obstet Gynecol. 2023;50(9):198. doi:10.31083/J.CEOG5009198/2709-0094-50-9-198/FIG8.PNG
- Possover M. Neuropelveology: An Emerging Discipline for the Management of Pelvic Neuropathies and Bladder Dysfunctions through to Spinal Cord Injury, Anti-Ageing and the Mars Mission. Journal of Clinical Medicine 2020, Vol 9, Page 3285. 2020;9(10):3285. doi:10.3390/JCM9103285
- Possover M, Forman A. Pelvic Neuralgias by Neuro-Vascular Entrapment: Anatomical Findings in a Series of 97 Consecutive Patients Treated by Laparoscopic Nerve Decompression. Accessed May 10, 2025. www.painphysicianjournal.
- Kale A, Basol G, Topcu AC, Gundogdu EC, Usta T, Demirhan R. Intrapelvic Nerve Entrapment Syndrome Caused by a Variation of the Intrapelvic Piriformis Muscle and Abnormal Varicose Vessels: A Case Report. Int Neurourol J. 2021;25(2):177. doi:10.5213/INJ.2040232.116
- NUCELIO LEMOS KKCPSPPAG. Laparoscopic approach to intrapelvic nerve entrapments. Pelviperineology. doi:10.34057/PPJ
- Possover M, Chiantera V, Baekelandt J. Anatomy of the sacral roots and the pelvic splanchnic nerves in women using the LANN technique. Surg Laparosc Endosc Percutan Tech. 2007;17(6):508-510. doi:10.1097/SLE.0B013E31812F81E4
- Possover M, Baekelandt J, Flaskamp C, Li D, Chiantera V. Laparoscopic neurolysis of the sacral plexus and the sciatic nerve for extensive endometriosis of the pelvic wall. Minimally Invasive Neurosurgery. 2007;50(1):33-36. doi:10.1055/S-2007-970075
- Puntambekar S, Manchanda R. Surgical pelvic anatomy in gynecologic oncology. International Journal of Gynecology and Obstetrics. 2018;143:86-92. doi:10.1002/IJGO.12616
- Marc Possover NEUROPELVEOLOGY Latest Developments in Pelvic Neurofunctional Surgery PROGRESS IN PELVIC RESEARCH.
- Possover M. Five-Year Follow-Up After Laparoscopic Large Nerve Resection for Deep Infiltrating Sciatic Nerve Endometriosis. J Minim Invasive Gynecol. 2017;24(5):822-826. doi:10.1016/j.jmig.2017.02.027
- Lemos N, Sermer C, Fernandes G, et al. Laparoscopic approach to refractory extraspinal sciatica and pudendal pain caused by intrapelvic nerve entrapment. Sci Rep. 2021;11(1):1-7. doi:10.1038/S41598-021-90319-Y;SUBJMETA=308,375,617,692;KWRD=MEDICAL+RESEARCH,NEUROLOGICAL+DISORDERS
- Gundogdu EC, Kale A, Mercan M, et al. Integration of Intraoperative Neurophysiological Monitoring into Laparoscopic Pelvic Nerve Decompression Surgery: A Novel Technique for Protecting Pelvic Nerves. Clin Exp Obstet Gynecol. 2023;50(9):198. doi:10.31083/J.CEOG5009198/2709-0094-50-9-198/FIG8.PNG
- Allahqoli L, Hakimi S, Momenimovahed Z, et al. Neuropelveology for Endometriosis Management: A Systematic Review and Multilevel Meta-Analysis. J Clin Med. 2024;13(16). doi:10.3390/JCM13164676
- Kong X, Chai W, Chen J, Yan C, Shi L, Wang Y. Intraoperative monitoring of the femoral and sciatic nerves during total hip arthroplasty in high-riding developmental dysplasia of the hip. Bone Joint J. 2019;101-B(12):1438-1446. doi:10.1302/0301-620X.101B11.BJJ-2019-0341.R2
- Garry R. Laparoscopic surgery: best practice and research. Clin Obstet Gynecol. 2006;20(1):89-104. doi:10.1016/j.bpobgyn.2005.10.003
- Farrar JT, Young JP Jr, LaMoreaux L, Werth JL, Poole RM. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain. 2001;94(2):149-158. doi:10.1016/S0304-3959(01)00349-9
- Kneist W, Kauff DW, Lang H. Laparoscopic neuromapping in pelvic surgery: areas of application. Surg Innov. 2014;21(2):213-220. doi:10.1177/1553350613496907
- Zentner J. Noninvasive monitoring of motor evoked potentials during spinal cord neurosurgery. Neurosurgery. 1989;24(5):709-712. doi:10.1227/00006123-198905000-00008
- Tsutsui S, Yamada H. Basic principles and recent trends of transcranial motor evoked potentials in intraoperative neurophysiological monitoring. Neurol Med Chir (Tokyo). 2016;56(8):451-456. doi:10.2176/nmc.ra.2015-0307
- Molin CJ, Punga AR. Compound muscle action potential: an electrophysiological marker of muscle training. Clin Neurophysiol Pract. 2016;33(5):340-345. doi:10.1097/WNP.0000000000000252
- Possover M. Laparoscopic management of intrapelvic etiologies of pudendal pain in 134 consecutive patients. J Urol. 2009;181(4):1732-1736.




