Next-Gen COVID-19 Vaccine for Immunocompromised Patients
GEO-CM04S1: A Dual-Antigen COVID-19 Vaccine for Immunocompromised Patients
Kelly T McKee, Jr. MD, MPH¹; Mark J Newman, PhD²
- Chief Medical Officer, GeoVax Inc, USA
- Chief Scientific Officer, GeoVax, Inc, USA
OPEN ACCESS
PUBLISHED 28 February 2026
CITATION McKee, KT., and Newman, MJ., 2026. GEO-CM04S1: A Dual-Antigen COVID-19 Vaccine for Immunocompromised Patients. Medical Research Archives, [online] 14(2).
COPYRIGHT © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
The multiantigen design and viral vector delivery platform of GEO-CM04S1 position this vaccine candidate as a next generation COVID-19 vaccine. Data generated to date indicate that the vaccine induces a durable, broadly specific and broadly functional humoral and cellular immune response to SARS-CoV-2 in both healthy adults and those with compromised immunity. The safety profile, as well as the breadth and magnitude of vaccine-induced T-cell responses supports its use in immunocompromised patients for prevention of severe COVID-19, addressing a critical unmet need. Targeting sequence-conserved structural proteins of this continually evolving pathogen should reduce the burdensome requirement for frequent updating and booster strategies designed to “chase the variant” to maintain efficacy.
Keywords
COVID-19, vaccine, immunocompromised, SARS-CoV-2, T-cell response
Introduction
The novel betacoronavirus that emerged in Wuhan, China, during late 2019, and that was subsequently identified as SARS-CoV-2, proved to be the causative agent for a highly contagious and lethal respiratory infection that spread rapidly throughout China, elsewhere in Asia, and ultimately the world. By the end of January 2020, the World Health Organization (WHO) had declared what became known as COVID-19 a public health emergency of international concern (PHEIC)1, and by March of that same year, had recognized it as a global pandemic. Unprecedented efforts to develop and put in place countermeasures to control and stem the relentless spread of this virus resulted in the successful development, testing, and emergency use authorization of efficacious vaccines in approximately 1 year. Despite these record-breaking accomplishments, and the declaration in 2023 of the end to the COVID-19 PHEIC2, by late 2024, COVID-19 had claimed the lives of more than 7 million persons worldwide3.
An often-overlooked patient group are those with medical conditions that impact vaccine effectiveness, such as individuals that are partially immunocompromised by disease and/or medication use. This includes patients suffering from and/or being treated for numerous types of malignancies, autoimmune disorders, transplant patients, dialysis patients, and potentially, even the aged population. The proportion of individuals that can be categorized as immunocompromised varies depending on which patients are included, but the most recent population estimate for the US is 6.6%4. This is a significant increase from the commonly reported rate of 3%, calculated in 2013, and is likely representative of other high-income countries with similar healthcare infrastructure.
A recent meta-analysis of case control and cohort studies reported effectiveness against hospitalization for multiple XBB.1.5 COVID-19 vaccines of 46-50%, with lower effectiveness (14-54%) during periods when the JN.1 variant dominated5. A single case-control study with a KP.2 mRNA COVID-19 vaccine reported a vaccine effectiveness of 68%6. Notably, vaccine effectiveness against hospitalization reported among immunocompromised individuals in 4 case-control studies ranged from 29-44% during this same survey interval5.
For individuals with functioning immunity receiving these continually updated, single-antigen SARS-CoV-2 vaccines, exposure to newer viral variants may result in infection, but these infections tend to result in relatively short-lived COVID-19 syndromes of modest severity. Not so for individuals with dysfunction of the innate, humoral, and/or cellular immune system(s), alone or in combination, who are at increased risk for poor control of SARS-CoV-2 infections and becoming more seriously ill (Table 1).
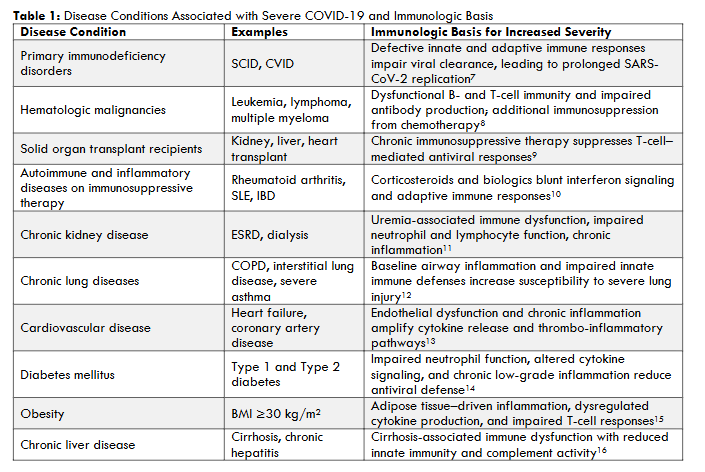
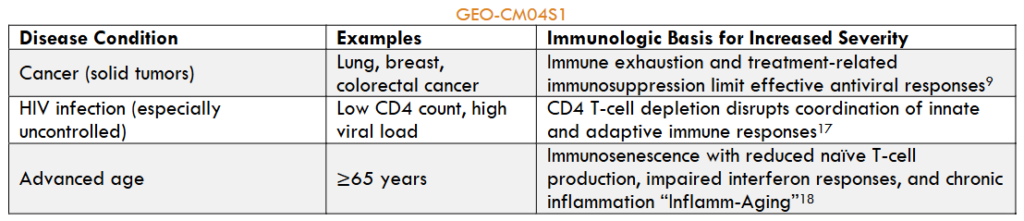
For many of these individuals, the ability to generate protective immune responses that contain SARS-CoV-2 infections and modulate disease outcomes through receipt of currently approved vaccines is far less certain, and in the case of some conditions, frankly inadequate. Thus, for individuals with underlying conditions recognized as being associated with poor control of SARS-CoV-2 infections (Table 1), COVID-19 remains at the present time the serious, and potentially lethal, health threat first encountered in 2019.
Importance of T-cells in COVID-19 Defense and Vaccine Response
Effective control and elimination of SARS-CoV-2 infection depends on a coordinated response of innate and adaptive immunity19. Impaired Type 1 interferon (IFN) response has been described in patients with severe COVID-19, but while important to effective clearance of infection, innate immunity will not be discussed further here.
The adaptive immune system is essential to control of virus infections through the function of three major effector cell components: (1) B cells, which produce antibodies; (2) CD4+T-cells, which provide a range of helper and effector functions; and (3) CD8+T-cells, which kill virus-infected cells20. The speed and efficiency with which each of these components responds to infection with most viruses, including SARS-CoV-2, determines the effectiveness of the host response and therefore the clinical outcome.
A prompt (within 7 days) T-cell response to SARS-CoV-2 infection that is followed by coordinated antibody production appears necessary for successful immune control21. Studies of patients early in the pandemic demonstrated clearly the importance of both CD4+ and CD8+ T-cells in this response. Early and coordinated virus-specific CD4+ and CD8+ T-cell responses were found to be correlated with lower SARS-CoV-2 viral loads and milder disease22,23, while virus-specific bystander CD8+T-cell responses, without systemic inflammation, were found to be characteristic of asymptomatic infection or mild disease. In contrast, SARS-CoV-2 infection induced inflammation with delayed CD8+ responses and was associated with severe disease and hospitalization24.
Pre-existing CD8+ T-cells specific for epitopes conserved among seasonal coronaviruses that were able to respond to SARS-CoV-2 epitopes, were found to be more abundant among patients with mild COVID-19 than those with more severe disease25,26. Similar observations were noted for healthcare workers that were previously exposed to coronaviruses27-32. Additional studies found correlations between the magnitude of the T-cell response33,34, as well as the magnitude and breadth of CD4+ responses34, and mild disease. A more recent study confirmed that virus-specific, polyfunctional CD4+ and CD8+ T-cells could be detected within days of symptom onset in the majority of adults with outpatient COVID-19, and that these early T-cell responses were associated with reduced upper respiratory tract SARS-CoV-2 virus RNA levels independent of neutralizing antibody36,37. In summary, the extent to which patients with COVID-19 are able mount early and robust T cell responses against SARS-CoV-2 to efficiently control and clear the virus, correlates with reduced progression to severe or fatal disease20, 23, 38-40.
COVID-19 vaccine efficacy in healthy individuals
There is no question that the widespread deployment of COVID-19 vaccines authorized for use since the 2020 has served an important role in blunting overall mortality from SARS-CoV-2 infections. All have been effective in preventing severe disease and death, with efficacy premised on the assumption that the induction of robust adaptive immune responses specific to the immunodominant viral S protein is the key. Studies of protection against SARS-CoV-2 infection offered by mRNA COVID-19 vaccines are characterized by high rates of protection from severe disease (>70%) and symptomatic infection (>60%) over the short term, up to 6 months. However, there is a dramatic fall-off in the rate of protection, generally to less than 35%, against symptomatic infection after 6 months41-45.
A recent matched case-control study provided a direct clinical correlation for the role of vaccine-induced T-cell responses in protecting against severe COVID-1946. Blood samples collected in PAXgene RNA tubes from adults receiving the Ad26/COV.2.S adenovirus vectored, S-based SARS-CoV-2 vaccine were used to evaluate the relation between T-cell response, assessed using T-cell receptor (TCR) β-chain sequencing, and clinical breakthrough infections. Through sequencing and computational analyses, levels of S-antigen specific TCRs were demonstrated to be significantly correlated with a lower risk of severe COVID-19. This study directly links the contribution of T-cell responses, first defined using blood samples obtained from naturally infected individuals, to control of SARS-CoV-2 infection in vaccinees.
Yet, despite these successes, limitations associated with first-generation vaccine products are recognized, and include:
- concerns over rare but serious adverse events associated with certain vaccine platforms 39,40
- diminished efficacy over time as new viral variants emerge, requiring constant updating and routine administration of boosters
- limited duration of antibody levels associated with protection following vaccination, and
- other factors41
These limitations support the need for next-generation COVID-19 vaccines offering safer, broader levels of efficacy across multiple variants and more durable protective immune responses.
COVID-19 vaccine efficacy in immunocompromised individuals
Studies of COVID-19 vaccine efficacy have been conducted primarily in healthy adult populations, excluding individuals with moderate-to-severe levels of compromised immunity47. It is well recognized that these immunocompromised individuals are at increased risk of poor control of SARS-CoV-2 infection and consequent progression to severe COVID-19, hospitalization, and death48. A meta-analysis of prospective, observational studies in immunocompromised patients performed in late 2021 reported much lower seroconversion rates among those with hematologic malignancies, solid tumors, immune-mediated inflammatory disorders, and transplant recipients after a single vaccine dose compared with rates in immunocompetent individuals. A second vaccine dose improved response rates among most of these, with the exception of transplant patients, while a third dose was associated with seroconversion in most non-responders49. In contrast, a population cohort study in patients with compromised immunity due to drug therapies or transplant procedures studied between December 2020 and April 2022, in which high vaccine uptake was seen (94% receiving at least one vaccine dose and 84% receiving 3 or more doses), found high vaccine effectiveness between 14 and 41 days following a second vaccine dose (78%), and up to 90% following a third dose50. However, effectiveness decreased relatively quickly over time, to 60% after 6 months post-second dose, and to <80% post-third dose after 3 months. Approximately 3% of these immunocompromised patients had a COVID-19-related hospitalization50.
Additional studies have shown diminished humoral immune response to COVID-19 vaccines for solid organ transplant recipients51, patients with hematologic malignancies52, patients treated with B-cell depleting agents53, patients with hematologic malignancies receiving cell transplants54,55, as well as for immunosuppressed patients with other disorders56. Patients with malignant solid tumors, hematologic malignancies (especially those with chronic lymphocytic leukemia), and those receiving hematopoietic stem cell transplants (HSCT) and chimeric antigen receptor T-cell (CAR-T) therapies, represent important patient populations characterized by inferior humoral immune responses to current COVID-19 vaccines. There is, therefore, a need for a safe SARS-CoV-2 vaccine that can offer broader and more durable protection against COVID-19 than that currently available to these and other individuals with profound immune dysfunction.
GEO-CM04S1
GEO-CM04S1 (originally termed COH-CM04S1) is a COVID-19 vaccine developed by scientists at the City of Hope Medical Center (Duarte, California, USA) to provide protection against severe COVID-19 for immunocompromised patients with hematologic malignancies. The vaccine is based on an attenuated Modified Vaccinia Ankara (MVA) vector platform expressing both spike (S) and nucleocapsid (N) antigens of ancestral (Wuhan) SARS-CoV-257.
Through expression of full-length S and N antigens, the latter of which is highly conserved across the multiple variants and subvariants of SARS-CoV-2, GEO-CM04S1 stimulates a robust T-cell response, and is less susceptible to the immune pressures that have plagued currently available vaccines and immunotherapies. In addition, the GEO-CM04S1 vaccine MVA backbone may also be more effective at inducing long-term immunity since MVA is known to elicit durable antibody and T-cell responses. These aspects of the multivalent vaccine design position this vaccine candidate as a logical option as a next-generation COVID-19 vaccine.
To address the continued evolution of SARS-CoV-2, the GEO-CM04S1 (Wuhan) vaccine was recently updated to produce a version wherein the encoded S gene is based on the sequence of Omicron-KP.2 linage, which matches the currently approved (2024/2025 COVID season) vaccines. Specifically, the original S gene based on the Wuhan sequence has been replaced with a S gene based on the SARS-CoV-2 KP.2 variant sequence using an established heterologous recombination method to preserve integrity of GEO-CM04S1. This updated construct is termed GEO-CM04S1-KP2.
Pre-clinical Studies
Animal model testing was completed to assess CM04S1 vaccine immunogenicity, efficacy and general safety prior to clinical (Phase 1) evaluation. Testing was extensive and included the use of the transgenic hACE2-mouse model (K18), the Syrian hamster model and African Green monkeys, all of which documented efficacy of vaccine-induced immune responses57.
Continued testing using experimental MVA-vectored vaccines encoding the S or N alone in the transgenic mouse model with challenge infections based on Omicron variants generated direct evidence for the importance of T-cell responses in protection from lethal SARS-CoV-2 infection. Critically, vaccine efficacy was observed in the absence of neutralizing antibodies58. T-cell contributions to vaccine efficacy against Omicron variants was largely anticipated because 85-95% of algorithm-predicted epitopes are highly conserved between ancestral and Omicron variants. However, the experiments also demonstrated that breadth of T-cell response was important because the combination of S and N in the GEO-CM04S1 vaccine induced optimal protection against lethal infection. Immune system mediated protection against severe disease was characterized by reduced levels of virus replication in the lungs, viral clearance and recovery with lower levels of lung inflammation and damage.
The ability to manipulate the mouse model using antibody-mediated in vivo deletion of CD4+ or CD8+ T-cells or B-lymphocytes documented the critical requirement for CD4+ T-cell functions. This observation is consistent with that reported for humans following both symptomatic and asymptomatic SARS-CoV-2 infections where effector CD4+ T-cells are the dominant component of T-cell responses.
While GEO-CM04S1 is the most advanced multi-antigen next generation SARS-CoV-2 construct, similar preclinical data has been generated independently. Specifically, an experimental mRNA vaccine based on the Wuhan sequences of S and N protected hACE2-transgenic mice against both Wuhan and Omicron virus challenges59. Separately, an MVA-vectored vaccine encoding Wuhan-sequence derived S and N genes protected rhesus macaques from severe disease pathogenesis following infection with Wuhan and Delta SARS-CoV-2 variants and hamsters from Omicron variants60,61, all in the presence of diminished neutralizing antibodies. Thus, the value of inducing T-cell responses cannot be disputed for next generation vaccine designs.
Clinical Studies
Testing of GEO-CMO4S1 was completed in Phase 1 and Phase 2 clinical trials in healthy adults and is currently undergoing evaluation in two Phase 2 trials in severely immunocompromised patients.
Healthy Adult Trials (NCT04639466)
Findings from the Phase 1 portion of this Phase 1/2 trial, the first-in-human study with GEO-CM04S1, were published in March 202262. A total of 56 participants were enrolled before the trial was closed to accrual on 21 May 2021. In brief, on Day 0 and 28, study participants received 1.0 mL intramuscular injections of GEO-CM04S1: 17 received 1.0 x 107 plaque forming units (PFU), 8 received 1.0 X 108 PFU, 9 received 2.5 X 108 PFU, 5 received placebo, and 13 received 1.0 x 107 PFU followed by placebo. Grade 3 fever was observed in 1 participant who received low-dose GEO-CM04S1 and placebo, and Grade 2 anxiety or fatigue was seen in 1 participant who received medium-dose GEO-CM04S1. No severe adverse events (SAEs) were reported. Seroconversion to the S protein by enzyme immunoassay was observed in all 34 participants after a single dose of vaccine and at all dose levels, while 32 (94%) responded similarly for N protein (p<0·0001 versus placebo for each comparison). Four times or more increase in SARS-CoV-2 neutralizing antibodies within 56 days was measured in nine of 17 participants in the low-dose GEO-CM04S1 group, all 8 participants in the medium-dose GEO-CM04S1 group, and 8 of 9 participants in the high-dose GEO-CM04S1 group (p=0.0035 combined dose levels vs placebo).
Post-prime and post-boost 4-times increase in S-specific or N-specific T-cells secreting IFNγ was measured in 48 (98%; 95% CI 89–100) of 49 participants who received at least 1 dose of GEO-CM04S1 and provided a sample for immunological analysis. Similar levels of T-cell responses were induced by all vaccine dose levels and responses appeared to approach the assay maximum after a single vaccine dose, with only marginal boosting. These data provided the necessary demonstration that GEO-CM04S1 was sufficiently safe, potent, and effective at inducing both antibody and T-cell responses in healthy adults to enable advancement to Phase 2 testing.
The Phase 2 portion of the trial was completed in September 2024. A total of 63 participants who had previously received one or more injections of an authorized COVID-19 (mRNA or Ad-vectored) were randomized to receive a single 1 mL booster injection of 1.0 X 107 PFU or 1.0 X 108 PFU of GEO-CM04S1 intramuscularly. Subjects were followed for 1 year and monitored periodically for safety and immune response; 56 subjects completed the trial. The primary immune endpoint was a 5-fold or greater anti-S IgG antibody response at day 28 following boosting as determined using a commercially available (Ortho-VITROS) ELISA assay. The safety profile was benign, and comparable in both dose groups. The primary immune endpoint was not met for either dose group. However, as compared with Ph 1 subjects, baseline antibody titers were high in all participants in this trial, indicating prior vaccination and/or natural COVID-19 infection. Spike IgG binding antibody levels were similar in both dose groups, while anti-receptor binding domain and anti-nucleocapsid antibodies also rose above baselines. There were increases in neutralizing antibody levels in both dose groups against alpha (D614G) and omicron (BA.1 and XBB.1.5) SARS-CoV-2 virus variants. Despite relatively high baseline levels of S and N-specific T-cell responses across both dose cohorts, there were significantly higher neutralizing antibody responses at the higher (1 X 108 PFU) dose level.
Immunocompromised Patient Trials
Patients with blood cancers who have undergone therapeutic cell transplants (HSCT or CAR-T), and patients with chronic lymphocytic leukemia are among those at greatest risk for severe consequences from SARS-CoV-2 infection. For these individuals, efficacy from currently available COVID-19 vaccines is unpredictable, and often suboptimal, highlighting a significant gap in protection for these highly vulnerable patients. Two clinical trials in these severely immunocompromised patient groups are currently ongoing to address this need:
- Phase 2 randomized, multi-center study of GEO-CMO4S1 (SARS-CoV-2 vaccine) versus mRNA SARS-CoV-2 vaccine in patients post cellular therapy for hematological malignancies (NCT04977024) Beginning in September 2021, patients with blood cancers ≥3 months after receiving autologous hematopoietic stem cell transplants (HSCT), allogeneic HSCT, or chimeric antigen T-cell (CAR-T) therapy were enrolled into open-label lead-in cohorts (up to 6 patients/cell therapy) to receive 2-4 injections (spaced 1 month apart) of 2.5X108PFU of GEO-CM04S1 intramuscularly. Subsequent patients were then randomized 1:1 to receive 2 injections of GEO-CM04S1 or a standard-of-care mRNA COVID-19 vaccine, then actively followed for 1 year for safety and immune assessments. The open-label safety lead-in portion of the study has been completed for 16 patients in this study who have received 2-4 injections of GEO-CM04S1, while 47 patients have been randomized to date. Six of the patients in the safety-lead-in portion of the trial are allogeneic HSCT recipients, six are autologous HSCT recipients, and two patients have received CAR-T therapy. The randomized portion has enrolled a total of 29 allogeneic HSCT, 16 autologous HSCT, and 2 CAR-T recipients.
- No significant safety events attributable to receipt of GEO-CM04S1 have been observed among study subjects over a follow-up period of 6-12 months. Treatment-emergent adverse events (TEAE) related to vaccination were either Grade 1 or 2, with no Grade 3 or higher recorded. There have been 8 SAEs (5 among GEO-CM04S1 recipients, 3 among those receiving an mRNA COVID-19 vaccine); none were attributed to receipt of vaccine. Breakthrough COVID-19 infections have been reported in 8 patients (5 among GEO-CM04S1 recipients, 3 among those receiving an mRNA COVID-19 vaccine); all cases were mild or moderate in severity. No cases of myocarditis or pericarditis have been observed.
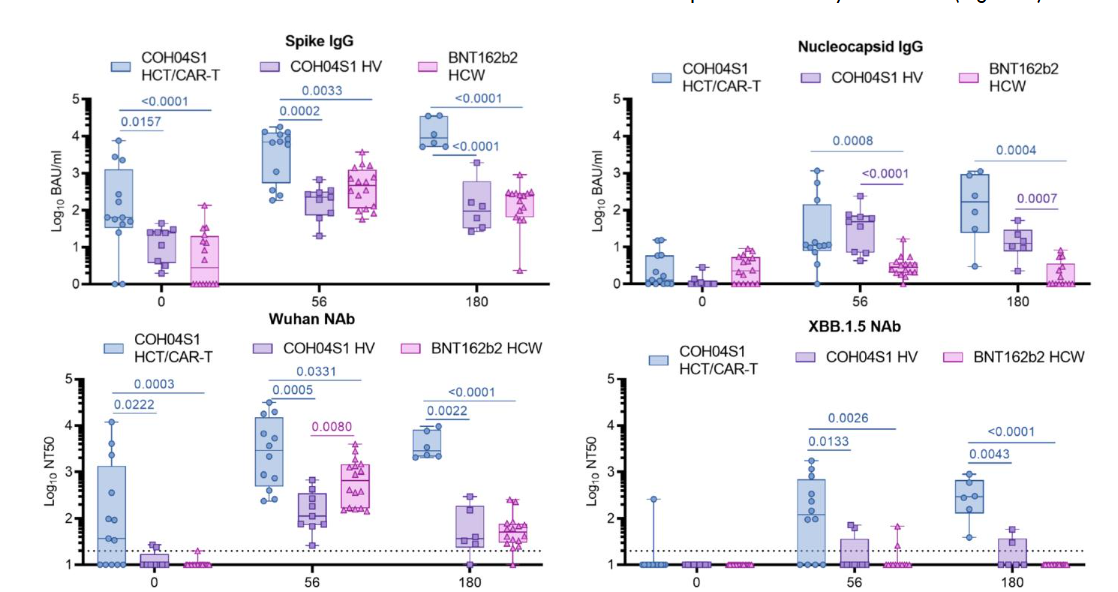
Analyses of antibody and T-cell responses in 13 of the patients enrolled in the open-label safety-lead-in portion, all of whom received GEO-CM04S1 (2.5 X 108 PFU/dose), showed robust binding and neutralizing antibody, as well as T-cell responses. Immune responses were functional for 6 months or longer against the omicron XBB.1.5 variant, despite the vaccine having been designed using ancestral Wuhan SARS-CoV-2 sequences. Antibody responses were comparable or superior in magnitude and duration to GEO-CM04S1 and mRNA vaccine responses in healthy individuals (Figure 2).
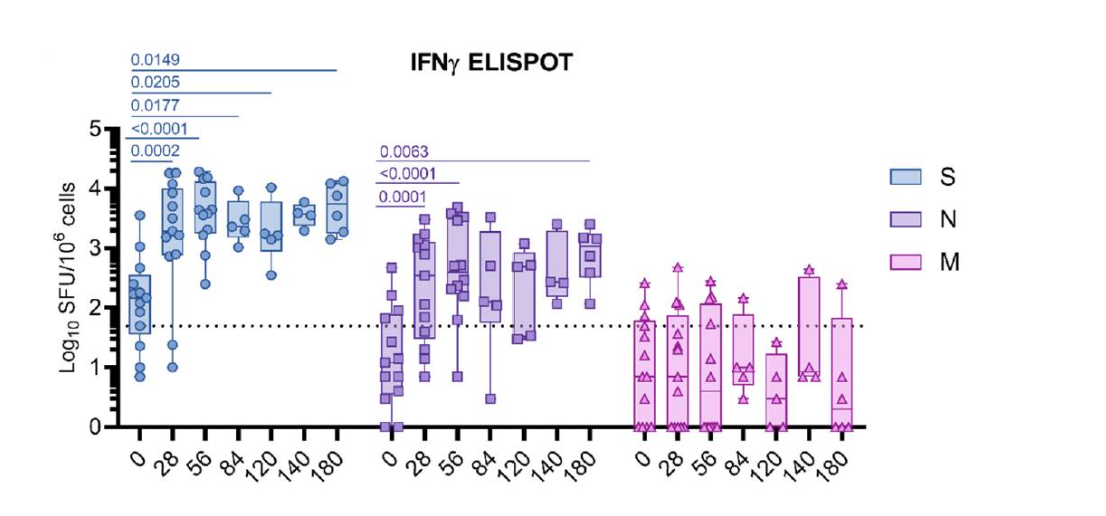
Activation of T-cells, measured using the Activation-Induced Marker (AIM) assay, was assessed using flow cytometry in a subset of samples. This assay demonstrated significant increases in both S- and N-specific CD4+ T cells following one or two vaccine injections. S- and N-specific CD8+ T cell responses were also measured, though at lower levels (Figure 4).
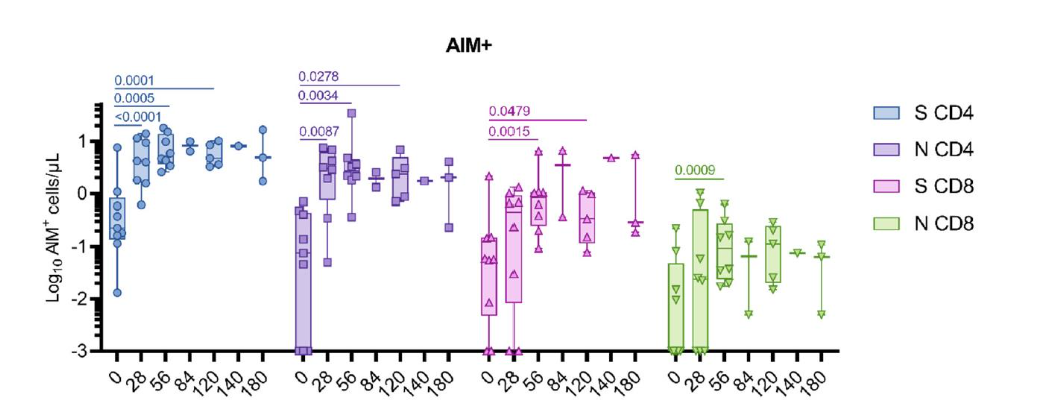
Immune responses of patients enrolled into the randomized portion of the trial remain blinded at the time of writing.
Randomized observer-blinded phase 2 trial of COVID-19 booster with GEO-CM04S1 or mRNA vaccine in patients with chronic lymphocytic leukemia (NCT05672355)
In this investigator-initiated study that began in July 2023, patients with chronic lymphocytic leukemia (CLL) ≥3 months after receiving 2 or more mRNA COVID-19 vaccinations were randomized under a Simon 2-stage design to receive 2 doses of 2.5 X 108 PFU of GEO-CM04S1 or a standard-of-care mRNA COVID-19 vaccine (BNT 162b2) spaced 3 months apart. After fourteen patients in each arm completed Stage 1, an interim analysis was planned. Patients were to be actively followed for 1 year for safety and immune assessments.
A total of 31 patients have been enrolled in this trial to date. At interim analysis, 6 of 15 patients (40%) in the GEO-CM04S1 arm showed S- or N-specific IFN-γ T-cell responses ≥3-fold over baseline at day 56 (the primary immune assessment [PIA] endpoint), compared with only 2 of 14 (14%) participants receiving the mRNA COVID-19 vaccine64. Thus, the proportion of study subjects receiving GEO-CM04S1 that achieved the study’s PIA met the statistical requirement to continue enrollment, while those in the mRNA COVID-19 vaccine arm did not (Figure 5).
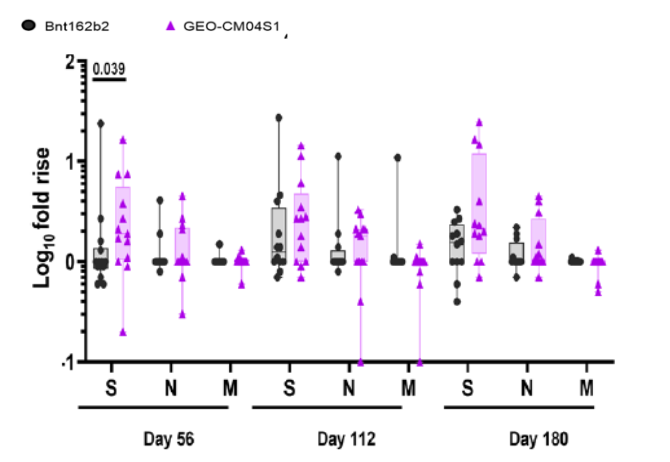
CLL patients receiving GEO-CM04S1 who had no prior CLL therapy showed significantly higher geometric mean fold rises in S-specific IFN-γ secretion than those observed in the mRNA COVID-19 arm on days 56 and 84 after their first dose. Among patients who had received CLL treatments, such differences were not observed, though S-specific IFN-γ secretion in mRNA COVID-19 vaccinees trended higher than that in those randomized to the GEO-CM04S1 arm64. While the sample size studied to date is relatively small, these findings indicate that the cellular immune response to both S- and N- antigens of SARS-CoV-2 induced by GEO-CM04S1 CLL patients is stronger than that induced by a standard-of-care mRNA COVID-19 vaccine. Enrollment of additional patients in the GEO-CM04S1-only arm of this trial is ongoing.
Future Directions
The GEO-CM04S1 vaccine represents a first step in the development of next generation vaccine products designed to induce or expand T-cell immunity and protect against severe COVID-19. Vaccines with additional viral proteins could also prove beneficial, but what viral proteins are the best vaccine immunogens?
Characterization of T-cell responses using samples from convalescent blood donors have documented CD4+ and CD8+ T-cell effector functions specific for all of the major viral structural proteins, S, N and Membrane, as well multiple nonstructural proteins (NSP)26, indicating a broad range of T-cell responses in natural infection and recovery. The observations that early or pre-existing T-cell responses, likely induced by infections involving seasonal coronavirus or subclinical infections, were significantly associated with lower levels of disease pathogenesis are of particular importance and supports the targeting of genetically conserved coronavirus proteins27-30.
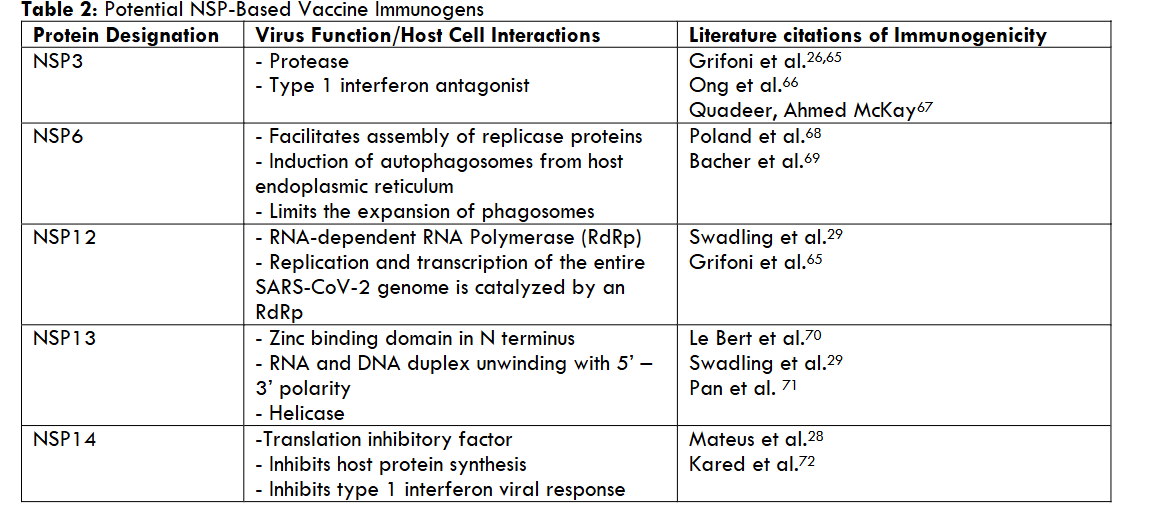
We believe T-cell responses to NSPs could be highly effective for limiting viral replication and severe disease because effector activity could impact the earliest stage of viral replication. Based on high levels of genetic conservation and documented immunogenicity in natural infection studies, we have identified several NSPs, notably NSP3, NSP6, NSP12, NSP13 and NSP14, as potential targets for use as vaccine immunogens. The properties associated with these NSPs are summarized in Table 2.
However, complex the desired immune response, vaccine design needs to result in a relatively simple product, with respect to format and administration and, we are focused on the MVA vaccine vector:
- MVA has a large and available genetic coding capacity allowing for the insertion of SARS-CoV-2 genes into different sites, supporting the simultaneous expression of multiple immunogenic proteins.
- MVA preferentially targets antigen presenting cells in vivo, in particular cells of the dendritic cell lineage73-75. This is of particular importance for the induction of CD8+ T-cell responses through the proteosome antigen-processing pathway.
- MVA also presents antigens through the cross-presentation pathway which is highly effective for the induction of antibody and CD4+ T-cell responses76.77.
- Pre-existing immunity, generated as a function of prior immunization with vaccinia virus during the global smallpox eradication campaign, does not limit the utility of MVA as a vaccine vector in older adults62.
- MVA can be safely and effectively used in people of all ages, including immunocompromised individuals78; MVA is the basis for the licensed MPOX and SPOX vaccine for use in immunocompromised patients79.
The technical processes for constructing the MVA-vectored vaccines are well established and building on GEO-CM04S1 is feasible. Experimental evaluation using mouse models and infectious challenge using a variety of SARS-CoV-2 variants and other coronaviruses can likely address most issues related to efficacy and potential deleterious immunopathology associated with function of the NSP and the T-cell responses.
Conclusions
It is difficult to overstate the importance of the current generation of SARS-CoV-2 vaccines, early versions of which were developed and tested in record time through the unprecedented collaboration of academia, governments, and industry, to the control of the global COVID-19 pandemic. The positive impact that these vaccines and their successors have had on blunting the ongoing morbidity and mortality caused by this virus is, likewise, undeniable. However, while the current generation of widely deployed single antigen S-based vaccines have proved moderately effective at protecting individuals with sound immune defenses against hospitalization at levels similar to those seen with influenza vaccines, there remains a need for improvement. The limited durability of protection afforded by these vaccines dictates a need for timely and regular updating in order to maintain protective levels of immunity against this constantly evolving pathogen. Moreover, for a substantial and growing segment of the population with dysfunctional innate and/or adaptive immunity, and who are therefore vulnerable to poor control of infection with SARS-CoV-2, the current generation of COVID-19 vaccines often falls short of providing a protective immune barrier to keep them safe.
Observational and experimental studies have repeatedly found that an efficient and effectively functioning cellular immune system, specifically T-cells, is critical to control of SARS-CoV-2 once infection has occurred. Currently available COVID-19 vaccines, based on only the S antigen, while effective in stimulating neutralizing antibodies, stimulate T-cells that are restricted in specificity and less likely to effectively mediate viral replication and clearance. There are, as well, concerns around the safety of these vaccines due to infrequent but significant toxicities that have come to light through their extensive use over time. There is, therefore, a need for a safe, next-generation vaccine that offers more durable immunity and that is less vulnerable to becoming “outdated” and ineffective because of immune escape by viral variants.
GEO-CM04S1 was developed specifically to address these deficiencies. With a well-documented safety profile, the vaccine vector, MVA, is widely recognized for its ability to induce durable humoral and cellular immune responses that provide long-term protection against a number of infectious diseases. The vaccine construct contains genes for full-length S and N proteins, thereby offering a broader range and number of epitope targets for stimulating B-cell, and especially T-cell, responses.
Importantly, the N protein, which is highly conserved across the coronaviruses that are known to infect humans, is less susceptible to the immune pressures that have plagued currently available vaccines and immunotherapies, and vesting in GEO-CM04S1 the potential to obviate the need to “chase the variants” of this continually evolving and mutating pathogen. These aspects of the multivalent vaccine design position GEO-CM04S1 (as well as its successor, GEO-CM-4S1-KP2), as a logical option as a next generation COVID-19 vaccine.
Conflicts of Interest
The authors are employees of, and hold equity interest in, GeoVax, Inc.
Funding
This work was funded in full by GeoVax Inc and performed in conjunction with employment activities of the authors.
References
- World Health Organization. (2020, January 30). Statement on the second meeting of the International Health Regulations (2005) Emergency Committee regarding the outbreak of novel coronavirus (2019-nCoV). WHO. Link
- World Health Organization. (2023, May 5). Statement on the fifteenth meeting of the International Health Regulations (2005) Emergency Committee regarding the coronavirus disease (COVID-19) pandemic. WHO. Link
- World Health Organization. (2024). COVID-19 epidemiological update, edition 174, 24 December 2024. World Health Organization. Link
- Martinson ML, Lapham J, Prevalence of immunosuppression among US adults. JAMA. 2024;331(10):880-882. doi:10.1001/jama.2023.28019.
- Scott J, Abers MS, Marwah HK, et al. Updated evidence for Covid-19, RSV, and influenza vaccines for 2025-2026. N Engl J Med. 2025;393(22):2221-2242. doi:10.1056/NEJMsa2514268.
- Appaneal HJ, Lopes VV, Puzniak L, et al. Early effectiveness of the BNT162b2 KP.2 vaccine against COVID-19 in the US Veterans Affairs Healthcare System. Nat Commun. 2025;16(4033):1-6. doi:10.1038/s41467-025-59344-7.
- Esenboga, S., Ocak, M., Akarsu, A. et al. COVID-19 in patients with primary immunodeficiency. J Clin Immunol. 2021;41:1515–1522. doi.org/10.1007/s10875-021-01065-9
- Passamonti F, Cattaneo C, Arcaini L, et al. Clinical characteristics and risk factors associated with COVID-19 severity in patients with haematological malignancies in Italy: a retrospective, multicentre, cohort study. Lancet Haematol. 2020;10:e737-e745. doi:10.1016/S2352-3026(20)30251-9.
- Pereira MR, Mohan S, Cohen DJ, et al. COVID-19 in solid organ transplant recipients: Initial report from the US epicenter. Am J Transplant. 2020;20(7):1800-1808. doi:10.1111/ajt.15941.
- Fajgenbaum DC, June CH. Cytokine storm. N Engl J Med. 2020;3;383(23):2255-2273. doi:10.1056/NEJMra2026131.
- Betjes MG. Immune cell dysfunction and inflammation in end-stage renal disease. Nat Rev Nephrol. 2013;9(5):255-265. doi:10.1038/nrneph.2013.44.
- Halpin DMG, Faner R, Sibila O, Badia JR, Agusti A. Do chronic respiratory diseases or their treatment affect the risk of SARS-CoV-2 infection? Lancet Respir Med. 2020;8(5):436-438. doi:10.1016/S2213-2600(20)30167-3.
- Libby P, Lüscher T. COVID-19 is, in the end, an endothelial disease. Eur Heart J. 2020;41(32):3038-3044. doi:10.1093/eurheartj/ehaa623.
- Berbudi A, Rahmadika N, Tjahjadi AI, Ruslami R. Type 2 diabetes and its impact on the immune system. Curr Diabetes Rev. 2020;16(5):442-449. doi:10.2174/1573399815666191024085838.
- Green WD, Beck MA. Obesity impairs the adaptive immune response to influenza virus. Ann Am Thorac Soc. 2017;14(Supplement_5):S406-S409. doi:10.1513/AnnalsATS.201706-447AW.
- Albillos A, Lario M, Álvarez-Mon M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance. J Hepatol. 2014;61(6):1385-1396. doi:10.1016/j.jhep.2014.08.010.
- Hoffmann C, Casado JL, Härter G, et al. Immune deficiency is a risk factor for severe COVID-19 in people living with HIV. HIV Med. 2021;22(5):372-378. doi:10.1111/hiv.13037.
- Fulop T, Larbi A, Dupuis G, et al. Immunosenescence and Inflamm-Aging as two sides of the same coin: Friends or foes? Front Immunol. 2018;10:8:1960. doi:10.3389/fimmu.2017.01960.
- Sette A, Crotty S. Adaptive immunity to SARS-CoV-2 and COVID-19. Cell. 2021;184(4):861-880. doi:10.1016/j.cell.2021.01.007.
- Almendro-Vázquez P, Laguna-Goya R, Paz-Artal E. Defending against SARS-CoV-2: The T cell perspective. Front Immunol. 2023;14:1107803. doi:10.3389/fimmu.2023.1107803.
- Zhao Z, Kumanovics A, Love T, et al. T Cell responses correlate with self-reported disease severity and neutralizing antibody responses predict protection against SARS-CoV-2 breakthrough infection. Viruses. 2023 Mar 9;15(3):709. doi: 10.3390/v15030709.
- Rydyznski Moderbacher C, Ramirez SI, Dan JM, et al. Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell. 2020;183(4):996-1012.e19. doi:10.1016/j.cell.2020.09.038.

