Next-Gen Porcine Models in Cancer Research and Therapy
Next Generation Large Animal Oncology Preclinical Transitional Platform: Developing New Approach Methodologies and Emerging Regulatory Considerations
Kyle M. Schachtschneider¹, Stephen Baggott¹, F Edward Boas², Tanja Dominko³, Raimon Duran-Struuck⁴, Lobna Elkhadragy⁵, Ron C. Gaba⁵, Vera Mehta¹, Lars F. Mikkelsen⁶, Matthew Niemeyer³, Jessicca Rege¹, Lawrence B. Schoo¹˒⁷
- Sus Clinicals, Chicago.
- City of Hope, Los Angeles.
- University of Wisconsin, Madison.
- University of Washington, Seattle.
- University of Illinois at Chicago, Chicago.
- LarSolution, Copenhagen, Denmark.
- University of Illinois at Urbana-Champaign.
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Schachtschneider, K., M., Baggott S., et al. Next Generation Large Animal Oncology Preclinical Translational Platform: Developing New Approach Methodologies and Emerging Regulatory Considerations. [online] 14(1).
https://doi.org/10.18103/mra.v14i1.7194
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7194
ISSN 2375-1924
ABSTRACT
Preclinical cancer models are essential for assessing the safety and efficacy of new therapeutics. The goals of this review are to address the development of an in vivo cancer technology platform that provides a tool for addressing emerging preclinical regulatory processes required for advancing new therapeutics and devices for use into the clinic. Previous studies have established the utility of porcine models as an alternative large animal model for cancer research due to their similarity to human size, genetics, metabolism, and physiology. Additionally, tumorigenesis pathways are similar between human and pigs in that similar driver mutations are required for transformation. Due to their larger size porcine models can be harnessed for testing of new interventional devices and radiological/surgical approaches as well. Taken together, swine are a feasible option for preclinical therapeutic and device testing. This review provides insights into the value of in vivo porcine models to provide both toxicity and efficacy data to support pre-clinical trials. The article describes next generation large animal models for cancer research, focusing on how genetically engineered pigs particularly the Oncopig® and mini-Oncopig can bridge the gap between lab experiments and real-world human clinical trials. Evidence is provided to demonstrate how these models address major shortcomings of traditional methods and help accelerate safe and effective cancer treatment development. The inducible transgenic Oncopig and mini-Oncopig develop site and cell and driver mutation specific tumors for preclinical human cancer that supports preclinical evaluation of novel drugs, biologicals, devices and locoregional therapies. The Wisconsin Mini-swine-Oncopig is a minipig nutritionally inducible metabolic diseases (fatty liver disease and obesity) in a small animal applicable for pharmaceutical evaluation. A final and important goal of this review is to demonstrate that such cancer models are consistent with the 3-Rs and support emerging new approach methodologies evaluations.
Keywords
NAMs, AI, Oncopigs, mini-Oncopigs, safety and efficacy platforms, precision medicine
INTRODUCTION
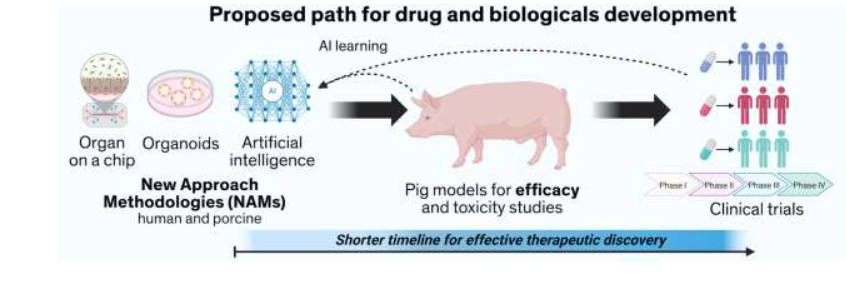
This review addresses current cancer research and its heavy reliance on mice and cell cultures, which often fail to predict how drugs will perform in humans. Presented here we advocate for and provide support to increase the use of pigs for therapeutic pre-clinical evaluation, based on their closer similarities with humans in size, organ function, genetics, metabolism, and immune responses. This makes them superior models for testing new cancer drugs, surgical technologies, interventional approaches, radiation therapy techniques, and diagnostic tools. Also, presented is the Oncopig Cancer Platform. The Oncopig is a genetically engineered pig that can develop precise, human-like tumors on demand using specific genetic “switches.” It carries human cancer-driving mutations (KRAS and TP53) that appear in roughly half of all human cancers. Because tumors can be induced in specific tissues and at defined times, scientists can study early cancer development, test treatments, and observe outcomes in a system that mirrors human disease. Traditional cancer research sees a 90% failure rate when moving from animal studies to human trials. This review argues that smarter, more humanlike preclinical testing could drastically improve success rates while also reducing costs and ethical issues. Furthermore, this review emphasizes how the Oncopig platform can be leveraged to generate New Approach Methodologies (NAMs) with the Oncopig Cancer tissue chips, and Artificial Intelligence (AI)-driven simulation models. Oncopigs support these models lack. The goal is a hybrid, tiered approach: screen drugs and devices through NAMs first, then validate promising ones in pigs before human trials. Such an integrated complementary approach supports real-world applications.
Table 1. Oncopig and Oncopig-based NAM Applications
| Applications |
|---|
| Testing new cancer drugs including various modalities such as monoclonal antibodies, antibody directed conjugates, oral targeting agents, and radiotherapies |
| Testing surgical and interventional technologies and devices using human size anatomy and physiology |
| Developing AI and robotic surgery training programs based on real tumor behavior |
| Exploring cancer with co-morbidities such as liver disease, obesity, and diabetes |
| Exploring the potential of tumor micro-environment effects to add in predictive or prognostic biomarkers for the clinic |
| Exploring monotherapy and combination standard of care approaches including dose optimization and kinetic and metabolic effects in actual diseased state |
| Harnessing in vivo genome editing of the Oncopig to enhance personalized medicine strategies by studying how specific genetic mutations influence treatment responses. |
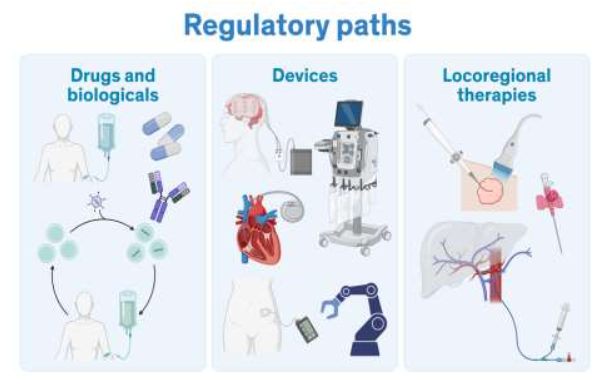
New drug and device regulatory review
Clinical testing for therapeutics (drug and biologicals), diagnostics and devices must first be assessed by federal agencies (Food and Drug Administration (FDA) in the US, European Medicines Agency in Europe) prior to clinical testing and/or marketing. Clinical trial success rates in oncology are low compared to other clinical fields. In a report that assessed clinical trials and FDA approval for therapeutic agents from 2009 to 2018 (for oncological therapeutics), only 3.4% of Phase 1 therapeutics made it to approval; this proportion was higher for therapeutics in Phase 2 (6.7% made it to approval) and Phase 3 (35.5% made it to approval). There are a variety of reasons for clinical trial failure including poor participant recruitment and compliance as well as lack of funds. However, the leading reasons are due to safety issues and failure to show efficacy in later phases. Currently, all therapies being considered by the FDA must have been assessed in animal models prior to entering a clinical trial. Taken together, there are opportunities to optimize preclinical assessment of new cancer therapeutics; particularly using in vivo platforms that demonstrate similar drug metabolism as humans, and organs with approximate size and anatomical structures to those found in humans, as well as the confounding co-morbidities.
Cancer is however not a single disease caused by identical driver mutations. The genetic mutations that contribute to the transformation of healthy cells into cancerous cells have been the subject of extensive research. Sinkala et. al. investigated the genomic sequences of 20,331 primary tumors representing 41 distinct human cancer types to identify and catalogue the driver mutations present in 727 known cancer genes. This revealed significant variations in the frequency of cancer gene mutations across different cancer types and highlight the frequent involvement of tumor suppressor genes (94%), oncogenes (93%), transcription factors (72%), kinases (64%), cell surface receptors (63%), and phosphatases (22%) in cancer. Additionally, their analysis revealed that cancer gene mutations are predominantly co-occurring rather than exclusive in all types of cancer. Notably, patients with tumors displaying different combinations of gene mutation patterns tend to exhibit variable survival outcomes. These findings provide new insights into the genetic landscape of cancer and bring us closer to a comprehensive understanding of the underlying mechanisms driving the development of various forms of cancer. Such findings further support the need for a pre-clinical in vivo platform in which any clinically relevant driver mutation can be introduced into a defined cell type within a defined microenvironment and at defined times in order establish time 0 to support early diagnosis and treatment responses.
As witnessed globally, the drug discovery and pre-clinical testing path prior to drug approval is a long, costly, and high-risk process that takes over 10 to 15 years with an average cost of over $1-2 billion for each new drug to be approved for clinical use. In addition, advancing a drug candidate to phase I clinical trials requires an extensive institutional infrastructure. Despite the creation of large comprehensive cancer centers, success has not been elevated with still nine out of ten drug candidates failing during phase I, II, or III clinical trials and at final drug approval. Suna et al. also note that the 90% failure rate is for the drug candidates that are already advanced to phase I clinical trial (no toxicological considerations), which does not include the drug candidates in the preclinical stages. Their analyses of clinical trial data from 2010 to 2017 identified four possible reasons attributed to the 90% clinical failures of drug development: 1) lack of clinical efficacy (40%-50%); 2) unmanageable toxicity (30%) in humans; 3) poor drug-like properties (10%-15%); and 4) lack of commercial needs and poor strategic planning (10%).
Evolving Pre-clinical Cancer Models
Over the past decade porcine models for pre-clinical cancer trials have emerged due to advances in genome editing capabilities and similarities between humans and pigs with respect to physiological, anatomical, and metabolic features. Cancer research progress has been markedly hampered by the lack of clinically relevant systems in which to study the effects of mutational interactions on cancer phenotypes. Relevant to cancer Joshi et. al. further demonstrated an increasingly vital role of pigs as translational biomedical models for studying human pathophysiology. They acknowledge the annotation of the pig genome supporting translatability of pigs as a biomedical model for various human diseases particularly due to their similarities with humans and pigs in terms of anatomy, physiology, genetics, and immunology. With a diverse range, from craniofacial and ophthalmology to reproduction, wound healing, musculoskeletal, and cancer, pigs have provided a seminal understanding of human pathophysiology. They further outline how the pig provides unique preclinical models for cancer research and highlight the strengths and opportunities for studying various human cancers.
Transition of Pre-clinical Models
A recent Economist editorial highlights the progress in fighting cancer that has been achieved over the past decades. Yet because cancer is not one illness, but a whole category, much of the progress has come not from big breakthroughs, but thousands of smaller advances in diagnostic screening, devices, surgical interventions and drugs and biologicals. Future gains will continue to come from such sources. And the last source of progress will be the clinical application of fresh science. This comes in two steps: 1. identifying who is most at risk of developing a cancer, and 2. finding ways to stop the disease in its tracks. The need to understanding the growing co-morbidities and emerging risk factors that increase specific cancer indices is also critical. Armed with new biomarkers in blood or breath and a deeper understanding of how combinations of genes and environmental exposure predispose people to develop cancers, physicians can target those who would benefit from treatment. That is important to prevent people undergoing needless surgery, chemo- and radiation therapy, at vast expense and with severe side-effects.
Emergence of New Alternative Models and Regulatory Considerations
Regulatory approval is required for drugs and biologicals with a different pathway for devices. Loco regional therapies represent the integrated mode of delivery of therapeutics (drugs, biologicals, and radioisotopes) with devices providing additional regulatory considerations. Finally, the early diagnosis of cancers is critical to providing timely interventions based on the size and location of the tumor; particularly in patients with metabolic considerations (diabetes, cardiovascular or obesity).
The proposed NAMs-based process, however, provides few available guidelines, and thus, significant efforts will be required to validate the platforms with respect to specificity and sensitivity. To ensure wider acceptance of NAMs in regulatory studies evaluating toxicology, it will be imperative to demonstrate that a given alternative method is not only robust and reproducible but also that it is biologically relevant. The market will soon be overwhelmed with cell lines, microchips and organoids representing all normal and diseased tissues. The need to ensure that they are robust and indicative of what is observed in health and sick individuals will require extensive efforts. Critical will be the further demonstration of in vivo (animals) studies with shared metabolic and mechanistic NAM-based predictions of what is observed in humans; especially with varying stages of disease(s) and co-morbidities. Workshops to test, train and knowledge transfer (intellectual property licenses) will be essential to ensure translational implementation from academic, governmental (both investigative and regulatory) and industry to support unmet clinical needs.
Technology-driven drug discovery and development
In response to ongoing debates regarding the value of animal experiments support regulatory clearance and clinical efficacy, et. al. performed a metanalysis to assess measures of translation across biomedical fields. They assessed the proportion of therapeutic interventions advancing to any human study, a randomized controlled trial (RCT), and regulatory approval. They determined that the median transition times from animal studies were 5, 7, and 10 years to reach any human study, an RCT, and regulatory approval, respectively. They concluded that, contrary to widespread assertions, the rate of successful animal-to-human translation may be higher than previously reported. However, the low rate of final approval indicates potential deficiencies in the design of both animal studies and early clinical trials. To ameliorate the efficacy of translating therapies from bench to bedside, they advocate for enhanced study design robustness and the reinforcement of generalizability.
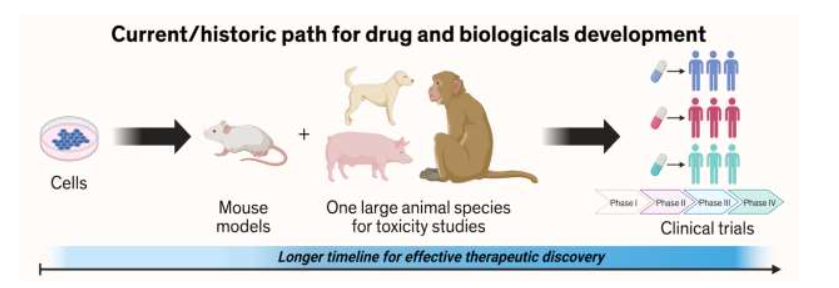
Recently, there has been a low success rate for the approval of new oncological therapeutics in clinical trials. The two leading reasons for these failures are either: (1) toxicity and safety issues or (2) efficacious responses. As all therapeutics must be tested within animal models prior to clinical testing, there are opportunities to expand the ability to assess efficacy and toxicity profiles within the preclinical testing phases of new therapeutics. Most preclinical in vivo testing is performed in mice, canines, and non-human primates. However, swine models are an alternative large animal model for cancer research with similarity to human size, genetics, and physiology. Additionally, tumorigenesis pathways are similar between human and pigs in that similar driver mutations are required for transformation. Due to their larger size, the development of orthotopic tumors is easier than in smaller rodent models; additionally, porcine models can be harnessed for testing of new interventional devices and radiological/surgical approaches as well. Taken together, pigs are a feasible option for preclinical therapeutic and device testing.
Transitional Platforms
Compared to humans, mice have 60 times faster drug diffusion, 7 times faster blood perfusion, and typically require 12 times the drug dosage (per kg). Therefore, we decided to evaluate drug delivery in pigs, which have similar physiology and drug dosing, compared to humans. Progress in creating the building blocks (genome sequence, genome editing and cloning) has clearly been achieved in the utility of the pig as a large animal biomedical model. The justification for using genetically modify organisms (GMO) to develop a disease on demand reflects the principles of the 3Rs (Replacement, Reduction and Refinement). The use of such a GMO animal replaces the need for intensive and painful chemical/radiation exposures to induce tumors; significantly reducing the need for large numbers of animals to ensure proper cohorts for pre-clinical trials; improves the time to treatment as some spontaneous models require constant monitoring over long periods of time, and refines pre-clinical modeling to an animal with inducible genome features that provides a single in vivo tool in which to conduct safety, risk, and efficacy studies. Such an in vivo technology platform supports NAMs needs for defining in vitro and ex vivo tools with the ability to then perform in vivo studies from those tools for defined driver mutation tissue tumors as a single pre-clinical platform.
Oncopig Cancer Platform
The Oncopig is a genetically engineered porcine model designed to recapitulate human cancer. Oncopigs harbor a transgenic cassette that expresses oncogenic mutant KRAS and TP53 under control of a Cre-Lox system, allowing for temporal and spatial control of tumor induction. Its versatility has enabled the development of diverse cancer models including liver, pancreatic, lung, and bladder cancer. Serving as a clinically relevant model for human cancer, the Oncopig addresses unmet clinical needs and holds immense promise for advancing preclinical cancer research and therapeutic development. In addition, using gene editing techniques like clustered regularly interspaced short palindromic repeats (CRISPR) editing, genetically tailored tumors can be developed in the Oncopig by somatic gene editing. This approach allows the introduction of specific gene mutations in Oncopig tumors, thereby broadening the scope of modeling human tumor mutational profiles that can be modeled and enable preclinical evaluation of precision medicine strategies. By leveraging somatic gene editing, Oncopigs can serve as a versatile platform for the development of genetically defined tumors, circumventing the need for the laborious and time-consuming process of developing additional genetically engineered pig models harboring distinct gene mutations.
The Oncopig harbors mutations found in >50% of human cancers- KRASG12D and TP53R167H and results in tumors that recapitulate the phenotype and physiology of human cancers. As in human disease, TERT is solely expressed in Oncopig cancer cells, and innate Oncopig KRASG12D and TP53R167H driver mutations are heterozygous in nature. The pig size supports utilization of human clinical instruments (radiology, imaging, and robotic surgery) which are unique for an in vivo platform for pre-clinical testing. Moreover, the pig has been shown to metabolize drugs similarly to human by the same p450 pathways (xenosensor pregnane X receptor) responsible for over 50% of existing prescription drugs.
Therapeutics (drugs and biologicals), Devices and Locoregional Therapies
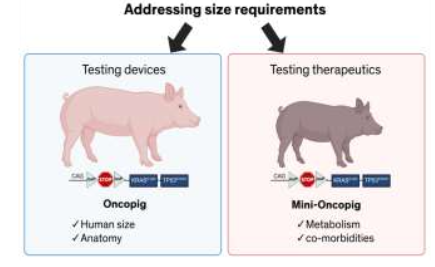
The development of tumors in a clinically relevant microenvironment is a crucial aspect of preclinical animal models. Cancer comorbidities have been modeled in Oncopigs, including alcohol-induced fibrosis, a precursor to liver cancer development. Excessive alcohol intake is a major etiological factor for liver cancer, often leading to liver cirrhosis. The development of liver cancer models within a cirrhotic background holds immense importance for preclinical studies, given the profound effect of cirrhosis on tumor progression and treatment responses. Although the size and anatomical resemblances between pigs and humans are crucial aspects of a porcine cancer model, equally significant are the genomic, epigenetic, physiological, metabolic, and immunological characteristics of pigs that mirror human biology. These similarities confer clinical relevance to porcine models, enabling the testing of drug efficacy, drug delivery, and immunotherapy.
The in vivo platforms that support dosing (mg/kg) of drugs require a smaller animal to support costs of therapeutics as well as size of housing pens as well as the longer duration of pre-clinical trials. Such therapeutics also need to address microenvironments and co-morbidities which can be modeled in the recently developed mini-Oncopig (Wisconsin Mini-swine (WMS) background). Whereas for device and locoregional therapies an in vivo platform that is similar in size is more appropriate.
The ability to induce tumors in Oncopigs with fully competent immune systems, along with the T cell mediated antitumor immune response observed in Oncopig models of liver, pancreatic, and lung cancers highlights the potential utility of Oncopigs for studying antitumor immune responses and preclinical testing of immunotherapies. Gaba et. al. recently underscored the clinical relevance of the Oncopig for testing the efficacy of clinically relevant drugs. Their study demonstrated similar expression of key genes involved in drug metabolism and transport between Oncopig and human HCC. Furthermore, Oncopig HCC cells accurately predicted chemotherapeutic susceptibility of human HCC cells in vitro and outperformed murine HCC cells in predictive accuracy. Notably, six Oncopig HCC cell lines exhibited consistent susceptibility to the chemotherapy drugs sorafenib and doxorubicin, commonly used for systemic and locoregional HCC therapy, respectively.
More recently, Segatto et. al. developed bladder cancer cell lines from Oncopigs and demonstrated consistent treatment responses between Oncopig and human cells to cisplatin, doxorubicin, and gemcitabine chemotherapeutics in vitro. This platform provides the opportunity to create pre-clinicals trials for any tumor, at any location and at any stage of development to support diagnostic, device, and drug development. Also, the platform can induce clinically relevant microenvironments and systemic co-morbidities. Such a platform supports the development of NAMs relevant to diverse human populations and health conditions; especially in using such a platform for training AI programs. Specifically, the Oncopig Cancer Platform addresses: 1. Disease and comorbidity models that account for complex systemic effects of cancer in relation to systemic disease on efficacy and toxicity; 2. provide anatomically scaled in vivo models for device-based (including robotic) and image-guided therapeutics; 3. controlled time of induction in vivo models for establishing early detection biomarkers, imaging-based diagnostics, and efficacy of treatment based on tumor stage; and 4. complimentary cooperation between NAMs and clinically relevant in vivo models for therapy screening and identifying and confirming/validating results.
Table 2. In vivo Large Animal Platform Applications
| Tumor Types | Applications | References |
|---|---|---|
| Hepatocellular Carcinoma | CRISPR genome editing | Elkhadragy 2025, 2022 |
| trans-arterial embolization | Nurili 2021 | |
| Intra-arterial pressure-enabled drug delivery | Jaroch 2024 | |
| ethanol-induced hepatic fibrosis co-morbidity | Magnetic Resonance Elastography | |
| drug eluding beads for embolization | Namur 2010; Isfort 2019 | |
| Yttrium-90 radioembolization | Schachtschneider 2024 | |
| locoregional therapies | Llovet 2021 | |
| micro-environment signaling | Patel 2021 | |
| staging fibrosis by PET | Pirasteh 2022 | |
| mets and robotic surgery | ||
| Pulmonary | in vivo induction | Ghosn 2023 |
| bronchoscope-induced to assess translatability | Joshi 2024 | |
| therapeutic biodistribution | Niemeyer 2025 | |
| bronchial Artery 90Y radioembolization | Wehrenberg-Klee 2025 | |
| microwave ablation | Carberry 2017 | |
| Colorectal | reverse electroporation assessment | Rugivarodom 2024 |
| Renal | microwave ablation | Sommer 2012 |
| theranostic treatment | Rice 2024 | |
| Pancreatic | safety and efficacy in vivo of a yttrium-90 resin microspheres glue formulation | Govindarajan 2025 |
| induction and characterization | Boas 2020, Mondal 2023 | |
| Induction of pancreatic ductal adenocarcinoma | Principe 2018 | |
| Bladder | recapitulate human bladder cancer treatment responses in vitro | Segatto 2024 |
| development of epithelial and intramuscular humanized pig models | Aulitzky 2023 | |
| Glioblastoma | imaging and characterization | Selek 2014 |
| Implantation and intratumoral isotope delivery | Khoshnevis 2017, 2020 | |
| high-grade spinal cord | Tora 2020 | |
| Sarcomas | ablation of IM and retroperitoneal sarcomas | Rund 2018 |
| Immune responses | Overgaard 2018 | |
| Melanoma | Conditional genetic engineering | Oh 2025 |
Disease and comorbidity models account for complex effects of cancer in relation to systemic disease on efficacy and toxicity. A major obstacle to advancing promising interventional oncology approaches is the lack of suitable tumor-bearing large animal models for preclinical evaluation of therapeutic dosing, delivery, safety, and efficacy. While many tumor-bearing small animal models benefit from availability of species-specific reagents, their small size limits applicability for interventional oncology studies. Large animal models, such as rabbits and pigs are better suited for interventional oncology research due to their size and anatomical similarities to humans. A commonly used model for interventional oncology research is the rabbit VX2 tumor implantation model, utilized to test locoregional therapeutic approaches for tumors in various organs, including liver, kidney, breast, head and neck, uterus, lung, bowel, bone, and pancreas. However, limitations including its small size, squamous cell carcinoma tumor biology, varying tumor kinetics, and spontaneous necrosis reduce the translational relevance and highlights the need for more representative preclinical models to advance interventional oncology research.
Lung cancer is the leading cause of cancer-related mortality worldwide. First line therapies for early-stage lung adenocarcinoma include surgical resection and stereotactic body radiation therapy (SBRT); however, only 25% of patients are treated surgically and toxicity limits SBRT for central/ultracentral tumors and stage III NSCLC. These limitations have sparked interest in locoregional therapies, including transarterial radioembolization (TARE) with 90Yttrium microspheres. Niemeyer et. al. demonstrated feasibility of intra-tracheal AdCre infusion for lung tumor induction in Oncopigs, and then used the platform to assess biodistribution of 99mTechnetium macroaggregated albumin (MAA) as a surrogate for 90Y to investigate the optimal infusion route for safe and predicable pulmonary TARE. First, three Oncopigs underwent intratracheal infusion of 1×1010 PFU of AdCre activated in 5 ml of 0.01M CaCl2 in PBS. This resulted in tumors visible on CT within 2-weeks post inoculation (mean size 2.9 x 2.2 cm, range 0.5 – 4 cm). Histological analysis confirmed presence of undifferentiated neoplasms without typical histological pattern of adenocarcinoma and KRASG12D expression indicating successful transgene activation. After histologic and radiographic tumor characterization, four additional Oncopigs underwent nonspecific lung tumor induction via intratracheal infusion of AdCre. After tumor formation, angiography and selective 99mTc-MAA infusions were performed from pulmonary and bronchial arteries, attempting to target the same tumor, with procedures separated by 7 days. Digital subtraction angiography (DSA) and cone beam computed tomography (CBCT) confirmed appropriate tumor perfusion from the selected pulmonary or bronchial artery. 5 mCi of 99mTc-MAA was infused from these locations, and SPECT/CT was performed. The mean tumor-to-normal ratio (TNR) after bronchial arterial infusion of 99mTc-MAA was significantly higher than after pulmonary arterial infusion of 99mTc-MAA (8.10±4.30 vs. 2.40±2.15, p=0.032). Off target esophageal (0.79±0.80 vs. 0.20±0.16, p=0.20), pericardial (8.83±5.34 vs. 5.87±5.15, p=0.43), spinal cord (0.08±0.09 vs. 0.08±0.08, p=1.0), renal (1.21±0.11 vs 2.07±0.36, p=0.04), and brain (0.01±0.01 vs. 0.00±0.01, p=1.0) biodistribution was low in both pulmonary and bronchial infusions.
Considerable investigations have confirmed the translational relevance of these Oncopig hepatocellular cancer (HCC) cells. Like human HCC, the Oncopig HCCs express the HCC diagnostic marker arginase-1, have similar cell cycle lengths and migration rates. Additionally, Oncopig HCC cells secrete alpha fetoprotein (AFP) consistent with most human HCCs. Furthermore, the Oncopig HCC cells were used to perform chemotherapeutic susceptibility assays and the results compared between Oncopig, human, and mouse HCC lines. This study compared 5 Oncopig and 5 different human HCC lines. Significant was that the Oncopig HCC responses were more like human responses for all 5 comparisons whereas the murine results varied. These findings further support the translational relevance of the model. Another example is a study by Chen (personal communication) of an arterially directed therapeutic study in the liver model. Their study focused on the local delivery of an NK cell-based immunotherapy for treatment of liver cancer. Using the Oncopig they were able to confirm significantly increased tumor regression in treated tumors compared to untreated.
To address how the Oncopig could be complementary to NAM efforts, we introduced PTEN, AXIN1, and ARID1A knockout (KO) mutations into Oncopig HCC cells. We demonstrated that PTEN and AXIN1 KOs significantly increased cell proliferation, while PTEN KO also results in increased cell migration. These mutated cells also were evaluated by therapeutic screening assays and confirmed that the PTEN KO mutations increase susceptibility to PI3K inhibitors consistent with human HCC. Such an approach further illustrates the power of the Oncopig Cancer Platform to provide a transitional model for in vitro (cell lines) to ex vivo (organoids) to in vivo (Oncopigs) to validate assays and prediction of in vivo outcomes. Both the Bay and GSK Pi3K inhibitors tested, demonstrated significantly more cell death in PTEN KO compared to AXIN1, ARID1A, and unedited cell lines. Importantly, both inhibitors have been shown to be effective against PTEN null human cells. However, increased susceptibility was not observed for the BKM inhibitor tested, which is also consistent with results from humans where this inhibitor has been shown to be ineffective for PTEN null cells.
Focus: Drugs and Biologicals
Sus Clinicals is also developing and validating new mini-pig Oncopig models. One is using the Wisconsin Miniature Swine as a background. The WMS was chosen based on its reduced size and predisposition to clinically relevant comorbidities including obesity, cardiovascular disease, and other metabolic syndromes. The inducible Oncopig construct was used to create a cloned mini-Oncopig line. The WMS was selected as the background genome since this line provides not only a lower body mass essential for drug and biological in vivo analyses but also has a genetic predisposition to obesity and metabolic disorders (e.g., fatty liver disease). These animals range from 32 kg (70 lbs.) at 6 months and 60 kg (130 lbs.) at 12 months of age and thus conducive to drug and biological studies.
Emerging challenges and opportunities
Historically, oncology drug development prioritized dosing to the maximum tolerated dose (MTD), often resulting in toxicity outweighing efficacy because of the urgent need to treat life-threatening cancers. With the emergence of novel and combinatorial therapies and interventions, it is now essential to adopt a more strategic approach defining the optimal dose, schedule, and treatment window preclinically to ensure safer and more effective translation into clinical trials including combination therapies (multiple drugs, beads with isotopes, and antibody targeted conjugates). MTD has been historically linked to murine toxicity studies rather than defining the Most Efficacious Dose (MED). Important to note is that mouse tumor cell lines only require a single driver mutation whereas human and pigs require two driver mutations. Thus, as stated earlier, human tumors from the same tissue can have different mutations and thus require new transitional testing models. Also, as presented the Oncopig Cancer Platform provides such a transitional platform that could support MED studies.
Locoregional therapy (LRT) continues to be a clinical intersection between Oncologists and Interventional Radiologists which provides targeted delivery to tumors increasing treatment efficacy as well as decreasing systemic side effects. Metastasis (Mets) is another opportunity for a new dimension of the Oncopig Cancer Platform. Through the ability to perform in vitro genome editing and to introduction of oncogenic cells through intervascular delivery allows seeding of organs with disparate tumorigenic cells and support efficacy studies of novel therapies in animals exhibiting metastatic disease.
Robotic surgery with respect to cancer will require pigs serve as a promising preclinical model for surgical applications given their similar physiology, size, genetics, immunity, and metabolism compared to humans. While pigs are frequently utilized in surgical studies, the inclusion of tumor-bearing pigs would enhance the translatability and clinical significance of these investigations. Here, we propose the Oncopig Cancer Platform as a valuable option for preclinical assessment and training on innovative AI and robotic surgery approaches. Oncopig tumor formation is successfully achieved across all models within two weeks, demonstrating the efficacy of the established protocols. Tumor sizes vary by organ type, with notable dimensions as follows: pancreatic tumors range from 0.5 to 5.1 cm, liver tumors from 0.5 to 4 cm, and lung tumors from 0.5 to 4 cm. Following tumor induction, Oncopigs can be utilized to test novel oncolytic robotic surgery devices and techniques in the context of real-world surgical conditions, including movement, bleeding, tissue responses, and defined tumor targets. In addition, reproducible induction of tumors in defined locations enables use for AI model training through generation of varied and realistic datasets. Finally, Oncopigs can be utilized to train surgeons on advanced AI and robotic surgery techniques in a controlled setting, allowing for development of skills and confidence prior to use in clinical practice. Use of organ-specific Oncopig tumor models represents an advancement in the preclinical evaluation landscape for AI and robotic surgical oncology. By mimicking human anatomy and physiology, these models provide a robust platform for assessing novel surgical techniques and training AI models and surgeons prior to translation into clinical practice.
CONCLUSION
Pigs offer an essential bridge between lab experiments and human testing allowing cancer therapies to be developed and approved faster, safer, and more responsibly. As presented here, we advocate a cooperative framework, blending human-relevant non-animal methods with high-fidelity animal models like the Oncopig platform, to advance oncology research and regulatory science in the next decade. The current reliance on traditional animal models, especially rodents, in cancer research poses significant hurdles to successful translation of preclinical findings to human treatments. The vast biological differences, methodological limitations, and systemic biases contribute to high failure rates in clinical trials and an overestimation of treatment efficacy. While animal models remain valuable for basic research, a paradigm shift towards more human-relevant models is critical. Pigs, particularly the Oncopig and mini-Oncopigs (WMS-Oncopig and Specipig-Oncopig), models offer a promising pre-clinical bridge due to their closer biological similarities to humans. These models provide more accurate insights into drug metabolism, tumor progression, and therapeutic responses. Complementary human-centric approaches such as 3D organoids, micro-physiological systems, and advanced computational models are essential for overcoming the current translational gap and accelerating the development of effective cancer therapies.
This review was designed to demonstrate that the obtainment of safe and efficacious therapies requires the integration of NAMs with refined animal technology platforms that address the need to reduce, replace and refine animal models. Simplistically it is not an either-or but the insertion of these approaches that will reduce risk of therapeutic discovery and regulatory trials while increasing the speed at which new solutions can be provided to the clinic; including rare and orphan therapies. The proposed NAMs-based process needs to ensure that they are robust and indicative of what is observed in healthy and sick individuals will require extensive efforts. Critical will be further demonstration in vivo (animals) studies of shared metabolic and mechanistic NAM-based predictions of what is observed in humans; especially with varying stages of disease(s) and co-morbidities. Workshops to test, train, and knowledge transfer (intellectual property licenses) will be essential to ensure translational implementation from academic, governmental (both investigative and regulatory) and industry to support unmet clinical needs.
Reardon provides strong arguments for the value of NAMs based on their relevance to human medicine, addressing mounting ethical concerns and development of technology platforms. As also stated, we have provided further support that additional standardization and validation of such platforms with respect to their robustness with regards to sensitivity and specificity. Reardon NAMs provide human-relevant, ethical, and innovative models but face challenges in biological completeness, validation, predictive scope, data availability, and regulatory. As stated by the German Toxicology Congress, the use of animal testing remains the international standard and requires its translation into NAMs to support AI applications and will require strong validation of NAMs. The growing consensus is that a hybrid complimentary model is essential. Where NAMs are used to screen drug and biological candidates and that the in vivo platform ensures full safety and efficacy prior to initiating clinical trials. Finally, Reardon provides a paradigm shift in biomedical research and regulatory science and further motivates the community to transition away from animal testing. Successful transition is dependent upon international validation and alignment of NAMs outputs that supports the 3Rs while not putting humans at risk.
CONFLICT OF INTEREST
KMS, JS, VM and LBS declare their employment and equity in Sus Clinicals. RCG declares Sus Clinicals, Fluidx Medical Technology, and ClearDynamic. FEB received research grants from City of Hope, Guerbet, Stephenson fund, COH-Caltech Biomedical Research Initiative (Merkin Institute), Society of Interventional Radiology, Society of Interventional Oncology, NIH/NCI, Department of Defense, Thompson family foundation, and Brockman Medical Research Foundation. He received research supplies (investigator-initiated) from Boston Scientific, and attended research consensus panels with the Society of Interventional Oncology. He is a co-founder of Claripacs, LLC, and an investor in Labdoor, Qventus, CloudMedx, Xgenomes, and Solugen. He has patent applications covering intratumoral drug delivery devices, immunoembolization, Y90 dosimetry, endoluminal ablation, CT metal artifact reduction, and other inventions.
Funding Statement
This manuscript was supported by funds received from the NIH (5R01CA283548).
Acknowledgements
Figures were created using BioRender.
References
- Bryda EC. The Mighty Mouse: the impact of rodents on advances in biomedical research. Mo Med. May-Jun 2013;110(3):207-11.
- Onaciu A, Munteanu R, Munteanu VC, et al. Spontaneous and Induced Animal Models for Cancer Research. Diagnostics (Basel). Aug 31 2020;10(9)doi:10.3390/diagnostics10090660
- Sajjad H, Imtiaz S, Noor T, Siddiqui YH, Sajjad A, Zia M. Cancer models in preclinical research: A chronicle review of advancement in effective cancer research. Animal Model Exp Med. Jun 2021;4(2):87-103. doi:10.1002/ame2.12165
- de Jong M, Maina T. Of mice and humans: are they the same?–Implications in cancer translational research. J Nucl Med. Apr 2010;51(4):501-4. doi:10.2967/jnumed.109.065706
- Hou N, Du X, Wu S. Advances in pig models of human diseases. Animal Model Exp Med. Apr 2022;5(2):141-152. doi:10.1002/ame2.12223
- Hicks WH, Bird CE, Pernik MN, et al. Large Animal Models of Glioma: Current Status and Future Prospects. Anticancer Res. Nov 2021;41(11):5343-5353. doi:10.21873/anticanres.15347
- Aravalli RN, Golzarian J, Cressman EN. Animal models of cancer in interventional radiology. Eur Radiol. May 2009;19(5):1049-53. doi:10.1007/s00330-008-1263-8
- Meyerholz DK, Burrough ER, Kirchhof N, Anderson DJ, Helke KL. Swine models in translational research and medicine. Vet Pathol. Jul 2024;61(4):512-523. doi:10.1177/03009858231222235
- Schachtschneider KM, Mehta V, Rege J, Schook LB. Oncopigs: an inducible transgenic large animal cancer model to address pre-clinical assessments. Medical Research Archives. 2025-03-29 2025;13(3)doi:10.18103/mra.v13i3.6392
- Schachtschneider KM, Jungersen G, Schook LB, Shanmuganayagam D. Editorial: “Humanized” Large Animal Cancer Models: Accelerating Time and Effectiveness of Clinical Trials. Front Oncol. 2019;9:793. doi:10.3389/fonc.2019.00793
- Schachtschneider KM, Redlon LN, Lokken RP, et al. Epigenetic regulation of individual components of combined hepatocellular-cholangiocarcinoma. PLoS One. 2025;20(5):e0324145. doi:10.1371/journal.pone.0324145
- Schachtschneider KM, Schwind RM, Darfour-Oduro KA, et al. A validated, transitional and translational porcine model of hepatocellular carcinoma. Oncotarget. Sep 8 2017;8(38):63620-63634. doi:10.18632/oncotarget.18872
- Schachtschneider KM, Schwind RM, Newson J, et al. The Oncopig Cancer Model: An Innovative Large Animal Translational Oncology Platform. Front Oncol. 2017;7:190. doi:10.3389/fonc.2017.00190
- Schook LB, Collares TV, Darfour-Oduro KA, et al. Unraveling the swine genome: implications for human health. Annu Rev Anim Biosci. 2015;3:219-44. doi:10.1146/annurev-animal-022114-110815
- Joshi K, Telugu BP, Prather RS, et al. Benefits and opportunities of the transgenic Oncopig cancer model. Trends Cancer. Mar 2024;10(3):182-184. doi:10.1016/j.trecan.2024.01.005
- Xu C, Wu S, Schook LB, Schachtschneider KM. Translating Human Cancer Sequences Into Personalized Porcine Cancer Models. Front Oncol. 2019;9:105. doi:10.3389/fonc.2019.00105
- Gaba RC, Elkhadragy L, Boas FE, et al. Development and comprehensive characterization of porcine hepatocellular carcinoma for translational liver cancer investigation. Oncotarget. Jul 14 2020;11(28):2686-2701. doi:10.18632/oncotarget.27647
- Lobianco F, Giurini E, Neto M, et al. 03:27 PM Abstract No. 221 Porcine and human hepatocellular carcinoma cell lines present similar drug-metabolizing enzyme expression levels and comparable sorafenib and doxorubicin cytotoxic responses. Journal of Vascular and Interventional Radiology. 2019;30(3):S99-S100. doi:10.1016/j.jvir.2018.12.279
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. May 2021;71(3):209-249. doi:10.3322/caac.21660
- Wong CH, Siah KW, Lo AW. Estimation of clinical trial success rates and related parameters. Biostatistics. Apr 1 2019;20(2):273-286. doi:10.1093/biostatistics/kxx069
- Fogel DB. Factors associated with clinical trials that fail and opportunities for improving the likelihood of success: A review. Contemp Clin Trials Commun. Sep 2018;11:156-164. doi:10.1016/j.conctc.2018.08.001
- Siegel RL, Kratzer TB, Giaquinto AN, Sung H, Jemal A. Cancer statistics, 2025. CA: A Cancer Journal for Clinicians. 2025;75(1):10-45. doi:https://doi.org/10.3322/caac.21871
- Sinkala M. Mutational landscape of cancer-driver genes across human cancers. Sci Rep. Aug 7 2023;13(1):12742. doi:10.1038/s41598-023-39608-2
- Sun D, Gao W, Hu H, Zhou S. Why 90% of clinical drug development fails and how to improve it? Acta Pharm Sin B. Jul 2022;12(7):3049-3062. doi:10.1016/j.apsb.2022.02.002
- Boettcher AN, Schachtschneider KM, Schook LB, Tuggle CK. Swine models for translational oncological research: an evolving landscape and regulatory considerations. Mamm Genome. Mar 2022;33(1):230-240. doi:10.1007/s00335-021-09907-y
- Pollock CB, Rogatcheva MB, Schook LB. Comparative genomics of xenobiotic metabolism: a porcine-human PXR gene comparison. Mamm Genome. Mar 2007;18(3):210-9. doi:10.1007/s00335-007-9007-7
- Elkhadragy L, Gaba RC, Niemeyer MM, Schook LB, Schachtschneider KM. Translational Relevance and Future Integration of the Oncopig Cancer Model in Preclinical Applications. Annu Rev Anim Biosci. Oct 17 2024;doi:10.1146/annurev-animal-111523-102125
- Honkala A, Malhotra SV, Kummar S, Junttila MR. Harnessing the predictive power of preclinical models for oncology drug development. Nature Reviews Drug Discovery. 2022/02/01 2022;21(2):99-114. doi:10.1038/s41573-021-00301-6
- Reardon S. US science academies take on human-genome editing. Nature. 2015/05/18 2015;doi:10.1038/nature.2015.17581
- Reardon S. Beyond lab animals. Science. Aug 14 2025;389(6761):676-679. doi:10.1126/science.aeb3933
- Ineichen BV, Furrer E, Grüninger SL, Zürrer WE, Macleod MR. Analysis of animal-to-human translation shows that only 5% of animal-tested therapeutic interventions obtain regulatory approval for human applications. PLOS Biology. 2024;22(6):e3002667. doi:10.1371/journal.pbio.3002667
- West GB, Brown JH, Enquist BJ. A general model for the origin of allometric scaling laws in biology. Science. Apr 4 1997;276(5309):122-6. doi:10.1126/science.276.5309.122
- Schmidt-Nielsen K. Scaling, why is animal size so important? Cambridge University Press; 1984.
- Nair AB, Jacob S. A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm. Mar 2016;7(2):27-31. doi:10.4103/0976-0105.177703
- Elkhadragy L, Carlino MJ, Jordan LR, et al. Development of a genetically tailored implantation hepatocellular carcinoma model in Oncopigs by somatic cell CRISPR editing. Disease Models & Mechanisms. 2025;18(1)doi:10.1242/dmm.052079
- Khalafalla M, Viswakarma N, Elkhadragy L, et al. Abstract 5169: Validation of genetically defined Oncopig hepatocellular carcinoma cell lines for in vitro evaluation of targeted therapeutic efficacy. Cancer Research. 2025;85(8_Supplement_1):5169-5169. doi:10.1158/1538-7445.Am2025-5169
- Schachtschneider KM, Liu Y, Mäkeläinen S, et al. Oncopig Soft-Tissue Sarcomas Recapitulate Key Transcriptional Features of Human Sarcomas. Scientific Reports. 2017/06/01 2017;7(1):2624. doi:10.1038/s41598-017-02912-9
- Robertson N, Schook LB, Schachtschneider KM. Porcine cancer models: potential tools to enhance cancer drug trials. Expert Opin Drug Discov. Aug 2020;15(8):893-902. doi:10.1080/17460441.2020.1757644
- Hieromnimon M, Regan DP, Lokken RP, Schook LB, Gaba RC, Schachtschneider KM. Single and multi-omic characterization of a porcine model of ethanol-induced hepatic fibrosis. Epigenetics. Dec 2025;20(1):2471127. doi:10.1080/15592294.2025.2471127
- Lee J, Boas FE, Duran-Struuck R, et al. Pigs as Clinically Relevant Models for Synergizing Interventional Oncology and Immunotherapy. J Vasc Interv Radiol. Jun 2024;35(6):809-817.e1. doi:10.1016/j.jvir.2024.01.005
- Rund LA, Schachtschneider KM, Gaba RC, et al. Abstract 3105: Oncopig carcinoma cell lines: A foundation for co-clinical trials. Cancer Research. 2018;78(13 Supplement):3105-3105. doi:10.1158/1538-7445.Am2018-3105
- Segatto NV, Simões LD, Bender CB, et al. Oncopig bladder cancer cells recapitulate human bladder cancer treatment responses in vitro. Original Research. Frontiers in Oncology. 2024-February-26 2024;Volume 14 – 2024doi:10.3389/fonc.2024.1323422
- Nurili F, Monette S, Michel AO, et al. Transarterial Embolization of Liver Cancer in a Transgenic Pig Model. Journal of Vascular and Interventional Radiology. 2021/04/01/ 2021;32(4):510-517.e3. doi:https://doi.org/10.1016/j.jvir.2020.09.011
- Jaroch DB, Liu Y, Kim AY, Katz SC, Cox BF, Hullinger TG. Intra-arterial Pressure-Enabled Drug Delivery Significantly Increases Penetration of Glass Microspheres in a Porcine Liver Tumor Model. J Vasc Interv Radiol. Oct 2024;35(10):1525-1533.e4. doi:10.1016/j.jvir.2024.06.030
- Gaba RC, Elkhadragy L, Pennix T, et al. Magnetic Resonance Elastography for Staging Liver Fibrosis in the Oncopig. Diagnostics (Basel). Aug 28 2024;14(17)doi:10.3390/diagnostics14171880
- Yasmin A, Regan DP, Schook LB, Gaba RC, Schachtschneider KM. Transcriptional regulation of alcohol induced liver fibrosis in a translational porcine hepatocellular carcinoma model. Biochimie. Mar 2021;182:73-84. doi:10.1016/j.biochi.2020.12.022
- Namur J, Wassef M, Millot JM, Lewis AL, Manfait M, Laurent A. Drug-eluting beads for liver embolization: concentration of doxorubicin in tissue and in beads in a pig model. J Vasc Interv Radiol. Feb 2010;21(2):259-67. doi:10.1016/j.jvir.2009.10.026
- Isfort P, Rauen P, Na HS, et al. Does Drug-Eluting Bead TACE Enhance the Local Effect of IRE? Imaging and Histopathological Evaluation in a Porcine Model. Cardiovasc Intervent Radiol. Jun 2019;42(6):880-885. doi:10.1007/s00270-019-02181-1
- Schachtschneider K, Arepally A, Blanco D, et al. Abstract No. LBA2 Preclinical Evaluation of Yttrium-90 Radioembolization in the Oncopig Liver Cancer Model. Journal of Vascular and Interventional Radiology. 2024;35(3):S226. doi:10.1016/j.jvir.2024.01.016
- Llovet JM, De Baere T, Kulik L, et al. Locoregional therapies in the era of molecular and immune treatments for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. May 2021;18(5):293-313. doi:10.1038/s41575-020-00395-0
- Patel SS, Sandur A, El-Kebir M, Gaba RC, Schook LB, Schachtschneider KM. Transcriptional Profiling of Porcine HCC Xenografts Provides Insights Into Tumor Cell Microenvironment Signaling. Front Genet. 2021;12:657330. doi:10.3389/fgene.2021.657330
- Pirasteh A, Periyasamy S, Meudt JJ, et al. Staging Liver Fibrosis by Fibroblast Activation Protein Inhibitor PET in a Human-Sized Swine Model. J Nucl Med. Dec 2022;63(12):1956-1961. doi:10.2967/jnumed.121.263736
- O’Connell RM, Horne S, O’Keeffe DA, et al. A novel low-cost high-fidelity porcine model of liver metastases for simulation training in robotic parenchyma-preserving liver resection. J Robot Surg. Nov 5 2024;18(1):394. doi:10.1007/s11701-024-02151-x
- Ghosn M, Elsakka AS, Petre EN, et al. Induction and preliminary characterization of neoplastic pulmonary nodules in a transgenic pig model. Lung Cancer. Apr 2023;178:157-165. doi:10.1016/j.lungcan.2023.02.013
- Joshi K, Suvilesh KN, Natesh NS, et al. Characterization of A Bronchoscopically Induced Transgenic Lung Cancer Pig Model for Human Translatability. bioRxiv. Nov 6 2024;doi:10.1101/2024.11.04.621940
- Niemeyer MM, Wang Y, Carlino MJ, et al. Biodistribution of Macroaggregated Albumin after Tumor Model Development and Characterization in a Porcine Lung Cancer Model. J Vasc Interv Radiol. Nov 2025;36(11):1735-1745.e3. doi:10.1016/j.jvir.2025.08.007
- Wehrenberg-Klee E, An T, Heidari P, et al. SPECT/CT Dosimetry of Bronchial Artery (99m)Tc Macroaggregated Albumin Injection in Pulmonary Malignancies: Feasibility Evaluation of Bronchial Artery (90)Y Radioembolization. Radiology. Feb 2025;314(2):e240331. doi:10.1148/radiol.240331
- Carberry GA, Nocerino E, Cristescu MM, Smolock AR, Lee FT, Jr., Brace CL. Microwave Ablation of the Lung in a Porcine Model: Vessel Diameter Predicts Pulmonary Artery Occlusion. Cardiovasc Intervent Radiol. Oct 2017;40(10):1609-1616. doi:10.1007/s00270-017-1689-y
- Rugivarodom M, Sonani H, Kamba S, Kee Song LW, Graham R, Rajan E. CREATING A PORCINE MODEL FOR COLORECTAL CANCER AND ASSESSING THE EFFICACY AND SAFETY OF ELECTROPORATION TREATMENT. Gastrointestinal Endoscopy. 2024;99(6):AB452-AB453. doi:10.1016/j.gie.2024.04.1426
- Sommer CM, Arnegger F, Koch V, et al. Microwave ablation of porcine kidneys in vivo: effect of two different ablation modes (“temperature control” and “power control”) on procedural outcome. Cardiovasc Intervent Radiol. Jun 2012;35(3):653-60. doi:10.1007/s00270-011-0171-5
- Rice SL, Muñoz FG, Benjamin J, et al. Transcatheter pseudo-vascular isolation for localization and concentration of a large molecule theranostic probe into a transgenic OncoPIG kidney tumor. Nucl Med Biol. Sep-Oct 2024;136-137:108939. doi:10.1016/j.nucmedbio.2024.108939
- Govindarajan N, Geschwind J, Bealo F, Janssen P, Bello PB, Danieli GD. Preclinical study of safety and efficacy in vivo of a yttrium-90 resin microspheres glue formulation in a large animal model of pancreatic cancer. Journal of Clinical Oncology. 2025;43(16_suppl):e16438-e16438. doi:10.1200/JCO.2025.43.16_suppl.e16438
- Boas FE, Nurili F, Bendet A, et al. Induction and characterization of pancreatic cancer in a transgenic pig model. PLOS ONE. 2020;15(9):e0239391. doi:10.1371/journal.pone.0239391
- Mondal P, Patel NS, Bailey K, et al. Induction of pancreatic neoplasia in the KRAS/TP53 Oncopig. Dis Model Mech. Jan 1 2023;16(1)doi:10.1242/dmm.049699
- Principe DR, Overgaard NH, Park AJ, et al. KRAS(G12D) and TP53(R167H) Cooperate to Induce Pancreatic Ductal Adenocarcinoma in Sus scrofa Pigs. Sci Rep. Aug 22 2018;8(1):12548. doi:10.1038/s41598-018-30916-6
- Aulitzky A VBR, Kim K, Al-Ahmadie H, Monette S, Coleman JA. MP14-02 DEVELOPMENT OF A PORCINE MODEL OF BLADDER CANCER USING THE ONCOPIG. Journal of Urology. 2023;209(Supplement 4):e182. doi:10.1097/JU.0000000000003234.02
- Segatto NV, Bender CB, Seixas FK, et al. Perspective: Humanized Pig Models of Bladder Cancer. Front Mol Biosci. 2021;8:681044. doi:10.3389/fmolb.2021.681044
- Selek L, Seigneuret E, Nugue G, et al. Imaging and histological characterization of a human brain xenograft in pig: the first induced glioma model in a large animal. J Neurosci Methods. Jan 15 2014;221:159-65. doi:10.1016/j.jneumeth.2013.10.002
- Khoshnevis M, Carozzo C, Bonnefont-Rebeix C, et al. Development of induced glioblastoma by implantation of a human xenograft in Yucatan minipig as a large animal model. J Neurosci Methods. Apr 15 2017;282:61-68. doi:10.1016/j.jneumeth.2017.03.007
- Khoshnevis M, Carozzo C, Brown R, et al. Feasibility of intratumoral 165Holmium siloxane delivery to induced U87 glioblastoma in a large animal model, the Yucatan minipig. PLoS One. 2020;15(6):e0234772. doi:10.1371/journal.pone.0234772
- Tora MS, Texakalidis P, Neill S, et al. Lentiviral Vector Induced Modeling of High-Grade Spinal Cord Glioma in Minipigs. Sci Rep. Mar 24 2020;10(1):5291. doi:10.1038/s41598-020-62167-9
- Rund LA, Ghoshal G, Williams E, et al. Abstract 2148: Image-guided catheter-based ultrasound thermal ablation of intramuscular and retroperitoneal sarcomas in the transgenic Oncopig cancer model. Cancer Research. 2018;78(13_Supplement):2148-2148. doi:10.1158/1538-7445.Am2018-2148
- Overgaard NH, Principe DR, Schachtschneider KM, et al. Genetically Induced Tumors in the Oncopig Model Invoke an Antitumor Immune Response Dominated by Cytotoxic CD8β(+) T Cells and Differentiated γδ T Cells Alongside a Regulatory Response Mediated by FOXP3(+) T Cells and Immunoregulatory Molecules. Front Immunol. 2018;9:1301. doi:10.3389/fimmu.2018.01301
- Overgaard NH, Fan TM, Schachtschneider KM, Principe DR, Schook LB, Jungersen G. Of Mice, Dogs, Pigs, and Men: Choosing the Appropriate Model for Immuno-Oncology Research. Ilar j. Dec 31 2018;59(3):247-262. doi:10.1093/ilar/ily014
- Oh D, Hong N, Eun K, et al. Generation of a genetically engineered porcine melanoma model featuring oncogenic control through conditional Cre recombination. Sci Rep. Jan 10 2025;15(1):1616. doi:10.1038/s41598-024-82554-w
- Lu T, Yang X, Huang Y, et al. Trends in the incidence, treatment, and survival of patients with lung cancer in the last four decades. Cancer Manag Res. 2019;11:943-953. doi:10.2147/cmar.S187317
- Timmerman R, McGarry R, Yiannoutsos C, et al. Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J Clin Oncol. Oct 20 2006;24(30):4833-9. doi:10.1200/jco.2006.07.5937
- Lindberg K, Grozman V, Karlsson K, et al. The HILUS-Trial-a Prospective Nordic Multicenter Phase 2 Study of Ultracentral Lung Tumors Treated With Stereotactic Body Radiotherapy. J Thorac Oncol. Jul 2021;16(7):1200-1210. doi:10.1016/j.jtho.2021.03.019
- Wang K, Eblan MJ, Deal AM, et al. Cardiac Toxicity After Radiotherapy for Stage III Non-Small-Cell Lung Cancer: Pooled Analysis of Dose-Escalation Trials Delivering 70 to 90 Gy. J Clin Oncol. May 1 2017;35(13):1387-1394. doi:10.1200/jco.2016.70.0229
- Ricke J, Großer O, Amthauer H. Y90-radioembolization of lung metastases via the bronchial artery: a report of 2 cases. Cardiovasc Intervent Radiol. Dec 2013;36(6):1664-1669. doi:10.1007/s00270-013-0690-3
- Bekiares NA, Chen AS, Shanmuganayagam D, et al. Effect of Caloric Restriction on Metabolic Dysfunction of Young Rapacz Familial Hypercholesterolemic Swine (Sus scrofa). Comp Med. Dec 1 2017;67(6):508-517.
- Schmeisser S, Miccoli A, von Bergen M, et al. New approach methodologies in human regulatory toxicology Not if, but how and when! Environment International. 2023/08/01/ 2023;178:108082. doi:https://doi.org/10.1016/j.envint.2023.108082

