NLR Trajectories in ICU Patients: Impact on Outcomes
Dynamic Clustering Analysis of Neutrophil to Lymphocyte Ratio Evolution Overtime in Intensive Care Unit Patients
Alejandra Restrepo 1*†, Muzhda Haem Rahimi 1,2*, Maxime Bodinier 2,3, Morgane Gossez 1,4, Anne-Claire Lukaszewicz 2,5, Thomas Rimmelé 2,5, Fabienne Venet 1,4, Guillaume Monneret 1,2
1. Hospices Civils de Lyon, Immunology Laboratory, Hôpital E. Herriot, Lyon France
2. Université de Lyon, EA 7426 “Pathophysiology of Injury-Induced Immunosuppression”, Université Claude Bernard Lyon_1, Lyon, France
3. Open Innovation & Partnerships, bioMérieux S.A., Marcy l’Etoile, France
4. CIRI, Centre International de Recherche en Infectiologie, Univ Lyon, Inserm U1111, Université Claude Bernard-Lyon 1, CNRS, UMR5308, ENS de Lyon, F-69007, Lyon, France
5. Hospices Civils de Lyon, Anesthesiology and Critical Care Medicine department, Hôpital E. Herriot, Lyon, France
* AR and MHR contributed equally to the work
‡ Current address: Internal Medicine, Hospital Ramón y Cajal, Madrid, España
OPEN ACCESS
PUBLISHED: 28 February 2025
CITATION: Restrepo, A., Rahimi, MH., et al., 2025. Dynamic Clustering Analysis of Neutrophil to Lymphocyte Ratio Evolution Overtime in Intensive Care Unit Patients. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6315
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6315
ISSN 2375-1924
ABSTRACT
Elevated neutrophil to lymphocyte ratio (NLR) upon ICU admission has been reported to be associated with disease progression, severity, or mortality in critically ill patients. However, the overtime trajectories of NLR after admission and their association with other markers of intensive care units patients’ immuno-inflammatory status have not be evaluated so far. In a cohort of 353 critically ill patients (sepsis, trauma, major surgery), we evaluated the association between NLR trajectories and patients’ deterioration (mortality or occurrence of nosocomial infection) both by a conventional analysis at each time-point and through K-means clustering analysis. Additionally, these trajectories were delineated alongside established markers of hyper-inflammation (e.g., IL-6) or immunosuppression (including HLA-DR expression on monocytes, proportion of immature neutrophils, and plasma IL-10 levels). The results showed that persistently elevated NLR values were associated with an increased risk of patient deterioration, as demonstrated in a multivariate analysis that included usual clinical confounders. Patients belonging to the persistently elevated NLR endotype simultaneously exhibited heightened inflammation and pronounced immunosuppression. In conclusion, by providing integrated information on both hyper-inflammation and immunosuppression, NLR measurement holds significant promise for monitoring the immune status of ICU patients. This straightforward and standardized marker could serve to assess immune organ failure in ICU and assist in guiding potential immunomodulation strategies.
Keywords: Neutrophil, lymphocyte, NLR, sepsis, mHLA-DR, immunosuppression
INTRODUCTION
Serious injuries such as sepsis, severe trauma, major surgery, and pancreatitis often lead to patients’ admission to intensive care units (ICU), as they provoke a complex host immune response marked by simultaneous pro-inflammatory and anti-inflammatory/immunosuppressive elements. These responses disrupt the body’s mechanisms for maintaining homeostasis. While some understanding of the underlying mechanisms driving excessive inflammation and profound immunosuppression has emerged, a significant knowledge gap remains regarding the link between these two facets of the same disease, both in terms of mechanistic and temporal aspects. An integrated perspective is urgently required to address the complexity and heterogeneity of these conditions, as the absence of such framework impedes progress away from the ineffective “one size fits all” approach in their management. A shift towards personalized precision medicine in sepsis is imperative.
Many markers, ranging from soluble proteins to cell surface markers and mRNA signatures, have been reported to delineate the immuno-inflammatory response of ICU patients and its association with initial severity, organ failures, and mortality. Among these markers, the neutrophil to lymphocyte ratio (NLR), easily obtained from a routine white blood cell count, holds the advantage of encapsulating both the inflammatory and innate immunity aspects, signified by elevated neutrophil counts, and the immunosuppressive and adaptive immunity facets, indicated by the extent of lymphopenia. Initially introduced by Zahorec in surgical patients, NLR has since been evaluated across various clinical conditions—from cancer to systemic inflammation—and has demonstrated associations with disease progression, severity, or mortality.
In the context of ICU patients, most articles focused on a single NLR measurement upon admission. Only a few studies have presented serial measurements over time and most of them focused on the very first hours and days after ICU admission. In addition, none of them reported on the concurrently evolution of additional markers of inflammation/immunosuppression. The aim of this study was to investigate the overtime evolution of NLR in a large cohort of ICU patients with diverse etiologies (including sepsis, major surgery, or severe trauma) during the initial week after their ICU admission and its association with patients’ deterioration. We established NLR trajectories both by conventional analysis at each time-point and through K-means clustering analysis. We correlated these results with established markers of inflammation (such as plasma IL-6 concentration) or immunosuppression (such as HLA-DR expression on monocytes, percentage of immature neutrophils, plasma IL-10 concentration).
MATERIALS AND METHODS
STUDY POPULATION
The current study constitutes an ancillary analysis of the REALISM protocol (REAnimation Low Immune Status Marker), a prospective longitudinal observational study conducted at Anesthesia and Critical Care Medicine Department in E. Herriot Hospital (Hospices Civils de Lyon, France). The primary aim of the REALISM study was to assess immune parameters in a critically ill patient cohort following injury, with the intent of refining the understanding of host response in ICU patients. The comprehensive study protocol has been previously published, along with primary results. The study received approval from the IRB: Comité de Protection des Personnes Sud-Est II (first posted on 23/12/2015) under the registration number: #2015-42-2) and was registered on clinicaltrials.gov under NCT02638779. The REALISM protocol complied with the Declaration of Helsinki, principles of Good Clinical Practice and the French personal data protection act. Written informed consent was obtained from each patient. The cohort included injured patients across various etiologies such as sepsis, trauma, and surgery. Detailed inclusion criteria are described in the primary results. Blood samples were collected at three specific time points within the initial week post admission to the ICU: on day 1 or 2 (D1–2), day 3 or 4 (D3–4), and day 5, 6, or 7 (D5–7). The REALISM report provides detailed information on the methods and protocols used for assessing immunological parameters and conducting complete blood counts. In the present study, the primary endpoint was the occurrence of patients’ deterioration, defined as the occurrence of death or of a nosocomial infection within the 28 days following ICU admission as previously proposed. A blinded adjudication committee, comprising 2 ICU physicians and 1 infectious disease specialists not involved in patients’ recruitment, was responsible for establishing the diagnosis of nosocomial infection. Their criteria for diagnosis were consistent with the definitions established by the European Centre for Disease Prevention and Control. NLR results were censored from the moment nosocomial infection was diagnosed. All other immunological parameters were generated as part of the REALISM study.
STATISTICAL ANALYSIS
Qualitative variables are presented as numbers and percentages, and quantitative variables as medians and 25th/75th percentiles. In alignment with the findings of the initial REALSIM study, wherein we demonstrated that a consistent host response occurred regardless of etiology following adjustment for initial severity, we analyzed the present NLR values in the entire cohort. Chi-square or Fisher’s exact test were used for qualitative variables assessment. Quantitative variables were compared with the Mann–Whitney U test. In order to investigate the relationship between parameters and adverse outcome, Kaplan-Meier analyses were conducted at multiple time points, and the log-rank test was employed to compare differences between groups categorized according to median values or 4th quartile (above and below). In multivariate analysis, the Cox proportional hazards model was performed to examine the independency of NLR in associating with adverse outcome when including usual ICU confounders (age, SOFA score and exposure to invasive devices: intubation, urinary catheter and central venous line). Regarding clustering, in order to identify patients’ group with a common NLR dynamic over time (trajectories endotypes) we used “kmL” – K-means for longitudinal data. The number of endotypes (n = 2) was defined a priori. All statistical analyses were conducted using R software version 4.3.2 (R Studio, Boston, MA).
RESULTS
The cohort comprised 353 patients including 107 individuals diagnosed with sepsis (30%), 137 individuals with trauma (39%), and 109 individuals who had undergone major surgery (31%). Patients’ deterioration was observed in 25% of patients. The main clinical characteristics of patients based on this adverse outcome are depicted in Table 1. Patients with adverse outcome tended to be older, had more severe disease (higher SOFA and SAPSII scores), and had a higher exposure to invasive devices compared to patients with favorable outcomes. Further details on clinical characteristics can be found elsewhere.
| Whole Cohort (n= 353) | Adverse outcome (n= 90) | Favorable outcome (n= 263) | p-value | |
|---|---|---|---|---|
| Gender male (yes_n_%) | 65.4 (%) | 57 (63.3) | 174 (66.2) | 0.72 |
| Age (Years) | 60 (47 – 71) | 67.5 (55.25 – 75) | 57 (44 – 70) | < 0.0001 |
| SAPS II score | 29 (20 – 43) | 35 (26.25 – 49.75) | 26 (18 – 40) | < 0.0001 |
| SOFA score | 4 (1 – 8) | 7 (2 – 9.75) | 4 (1 – 8) | < 0.0001 |
| Exposure to invasive devices (yes n_%) | 315 (89%) | 88 (97.7%) | 227 (84.4%) | 0.004 |
Compared to the control values proposed by Zahorec (i.e., NLR < 2), we noted that NLR values were markedly elevated on days 1-2 in patients and gradually decreased throughout the first week without returning to normal ranges (Figure 1). Details regarding NLR values by quartiles and for each etiological group are provided in Table 2. NLR levels were higher in patients experiencing adverse outcomes, with significant statistical differences observed at time points D3-4 and D5-7 (p=0.006 and p<0.0001, respectively).
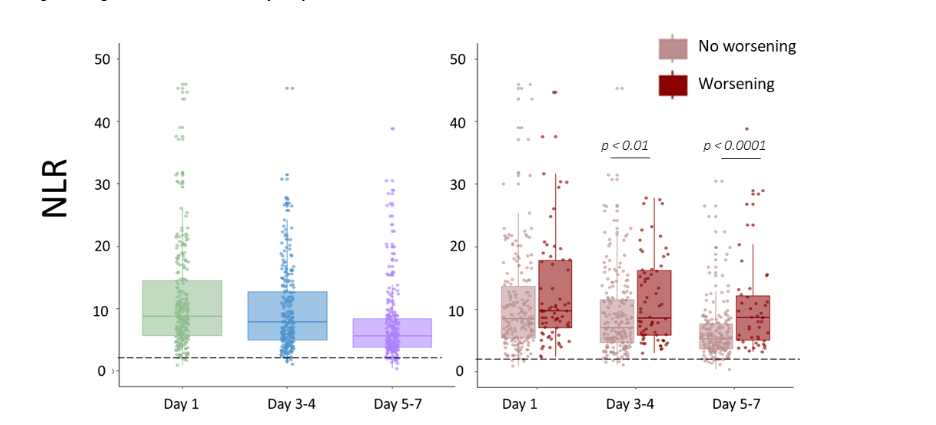
| Day 1-2 | Day 3-4 | Day 5-7 |
|---|---|---|
| Whole cohort (n = 353) Median 8.8 IQR 5.7-14.5 |
Median 7.8 IQR 4.9-12.6 |
Median 5.6 IQR 3.8-8.7 |
| Sepsis (n = 107) Median 11.3 IQR 7.9-21 |
Median 10.4 IQR 6.8-16.3 |
Median 6.9 IQR 4.1-10.2 |
| Trauma (n = 137) Median 7.9 IQR 4.7-13.6 |
Median 5.4 IQR 3.9-8.9 |
Median 4.6 IQR 3.4-6.3 |
| Surgery (n = 109) Median 8.3 IQR 5.5-10.5 |
Median 7.8 IQR 5.1-12.2 |
Median 5.7 IQR 4.3-8.9 |
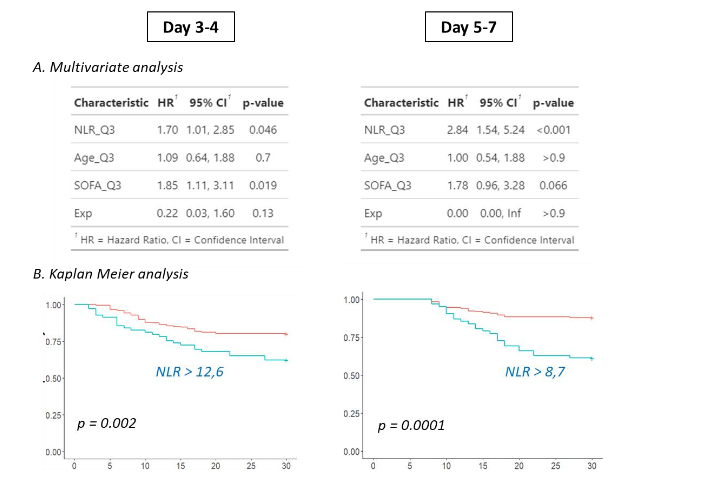
At D3-4 and D5-7, we subsequently conducted multivariate analysis by including usual clinical confounders (age, SOFA score, exposure to invasive devices) for predicting patients’ deterioration. When utilizing NLR medians to dichotomize patients, NLR was not significantly associated with adverse outcome after multivariate analysis (data not shown). However, considering previous studies which demonstrated that the 4th NLR quartile was notably linked to higher mortality, we directed our attention to this specific quartile at day 3-4 and day 5-7 (i.e., NLR = 12.6 and 8.7 respectively). By employing these cutoffs to classify patients, NLR was independently associated with deterioration at day 3-4 (hazard ratio = 1.7, p = 0.046) and day 5-7 (hazard ratio = 2.8, p < 0.0001). At the latter time point, NLR was the only parameter independently associated with deterioration (Figure 2). Importantly, we also conducted multivariate analysis separately for lymphocytes and neutrophils by categorizing patients into the worst quartile (the lowest for lymphocytes, the highest for neutrophils). Lymphocyte values were never significantly associated with deterioration, whereas neutrophils were significant only at day 5-7 (p = 0.01). This underscores the benefit of calculating a ratio based on these parameters.
Next, we conducted a K-means clustering analysis, arbitrarily setting the number of clusters to 2, in line with the previous multivariate analyses. We identified 2 clusters characterized by different NLR trajectories which tended to be parallel and did not converge.
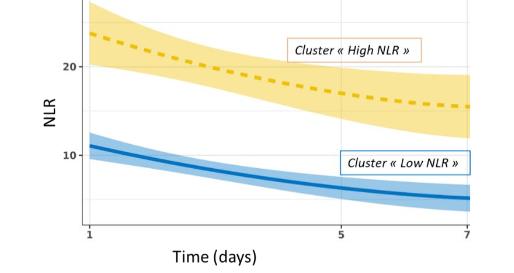
Cluster “low NLR” (n = 260, 83 % of patients) presented with moderately elevated NLR values at day 1-2 which tended to return toward normal at day 5-7, whereas cluster “high NLR” (n = 51, 17 % of patients) exhibited extremely and persistently high NLR values during the monitoring (ranging from 18 to 13 over the week). Cluster “high NLR” was characterized by higher clinical scores upon admission (age, Charlson, SAPS II, SOFA scores) and increased deterioration compared to “low NLR” cluster. Importantly, at each time point of the monitoring, cluster “high NLR” was characterized by elevated IL-6 values associated with markers of immunosuppression: lower mHLA-DR and CD4+ counts, increased IL-10, and percentage of immature neutrophils.
| Variable | Cluster « low NLR » | Cluster « high NLR » | p.value |
|---|---|---|---|
| n | 260 | 51 | |
| Admission Age | 58 [46 -70] | 68 [62-77.5] | <0.001 |
| Charlson Score | 1 [0-2] | 2 [0-3] | 0.015 |
| SAPSII score | 27 [19-40] | 43 [26.5-55] | <0.001 |
| SOFA score | 4 [1-8] | 7 [3.5-10.5] | <0.001 |
| Outcome Occurrence of nosocomial infection (D28) | 52 (20.0%) | 20 (39.2%) | 0.005 |
| Mortality (D28) | 5 (1.9%) | 6 (11.8%) | 0.004 |
| Mortality (D90) | 17 (6.5%) | 9 (17.6%) | 0.022 |
| Immune Parameters – D1-D2 Immature neutrophils (% of total neutrophils) | 45.3 [21.6-73.4] | 73.5[47.8-91.4] | <0.001 |
| HLA-DR per Monocyte (AB/C) | 6990 [4436-9544] | 4668 [3779-7166] | 0.038 |
| IL6 (pg/ml) | 105 [48-329] | 302 [93-667] | 0.001 |
| IL10 (pg/ml) | 14 [8.6-27] | 38 [23-57] | <0.001 |
| NLR | 8 [5-12] | 18 [11-30] | <0.001 |
| Neutrophils (G/L) | 10.2 [7.5-12.9] | 13.9 [10.5-20.9] | <0.001 |
| Lymphocytes (G/L) | 1.3 [0.9-1.8] | 0.8 [0.6-1.1] | <0.001 |
| CD4+ T lymphocytes (cells/µL) | 490 [329-685] | 288 [184-485] | <0.001 |
| Immune Parameters – D3-4 Immature neutrophils (% of total neutrophils) | 16 [6.5-44] | 37.6 [19-68] | <0.001 |
| HLA-DR per Monocyte | 7480 [5170-12340] | 5355 [3343-8214] | 0.001 |
| IL6 (pg/ml) | 45 [20-105] | 76 [40-170] | 0.003 |
| IL10 (pg/ml) | 9 [6.5-15] | 16.5 [12-29] | <0.001 |
| NLR | 6 [5-9] | 19 [15-23] | <0.001 |
| Neutrophils (G/L) | 7.2 [5.4-10.3] | 12.6 [9.6-16.9] | <0.001 |
| Lymphocytes (G/L) | 1.2 [0.9-1.5] | 0.7 [0.5-0.8] | <0.001 |
| CD4+ T lymphocytes (cells/µL) | 465 [322-670] | 293 [194-370] | <0.001 |
| Immune Parameters – D5-7 Immature neutrophils (% of total neutrophils) | 12.42 [5.74-26.28] | 27.62 [10.73-37.10] | 0.006 |
| mHLA-DR (AB/C) | 10062 [6482-13904] | 6461 [4727-9275] | <0.001 |
| IL6 (pg/ml) | 31 [16-58] | 49 [30-87] | <0.001 |
| IL10 (pg/ml) | 7 [5-12] | 13 [9-17] | <0.001 |
| NLR | 5 [4-8] | 13 [10-21] | <0.001 |
| Neutrophils (G/L) | 6.8 [5-9] | 11.6 [7-14] | <0.001 |
| Lymphocytes (G/L) | 1.3 [1.1-1.7] | 0.7 [0.5-0.8] | <0.001 |
| CD4+ T lymphocytes (cells/µL) | 558 [405-779] | 363 [235-526] | <0.001 |
DISCUSSION
In the quest to discover new parameters aiding in the identification of critically ill patients at higher risk of adverse events, the NLR has consistently been highlighted as an informative biomarker in various clinical contexts such as cancer, geriatrics, and systemic inflammation. Additionally, it is cost-effective and readily available everywhere since it derives from routine blood tests, making it applicable in various settings to provide an initial insight into immune system balance. The present results overall align with previous reports while providing important complementary data insights.
Firstly, in this cohort of patients, it is the persistence of elevated NLR that is associated with the occurrence of adverse events while values measured upon admission appeared less informative. So far, in ICU patients, most studies have reported on the association between early NLR values and prognosis. For example, in their meta-analysis, including 14 studies and >11,500 patients, Huang et al. found in patients with sepsis that higher NLR at admission was associated with mortality (hazard ratio = 1.75, p < 0.01). In contrast, in the present study, we observed a delayed significant and independent association (based on multivariate analysis) between persistently higher NLR and patients’ deterioration. This was especially true at later time points (hazard ratio = 2.8). This aligns with results from Dilektasli et al. who also find more elevated hazard ratio at day 5 (HR = 3.8) vs day 2 (HR = 1.6) in critically ill trauma patients.
Secondly, at a given time point, our results underline the importance of considering the highest NLR value. Specifically, when categorizing patients according to their belonging to the 4th quartile (n = 62) of NLR values compared to the sum of patients from the 3 lower quartiles, the results were highly significant at day 5-7 (hazard ratio = 2.84, p < 0.001). Accordingly, the subgroup with high NLR values identified by K-means clustering included 51 patients. This aspect, namely the consideration of the highest values, should be kept in mind in guiding any putative therapeutic options based on NLR values. Clinically pertinent thresholds need to be defined in further studies.
Thirdly, the present results offer a trajectory perspective of NRL evolution overtime after ICU admission that has, to our knowledge, never been reported. By defining 2 trajectory clusters, the results reinforce the idea that it is the persistence over time of high values which is associated with poor prognosis. This is consistent with two previous studies that examined NLR according to time evolution by calculating the NLR ratio between two time points, between day 1 and day 2, or between day 2 and day 5. In both studies, a non-decreasing NLR was associated with a poorer outcome.
Fourthly, and most importantly, we show for the first time that patients with persistently elevated NLR (“high NLR” cluster) presented with typical features of persistently deregulated immune response including hyper-inflammation and marked immunosuppression. For instance, at day 5-7, these patients presented with high percentages of immature neutrophils, which are likely myeloid derived suppressor cells, although not demonstrated in the present work. Additionally, they exhibited elevated IL-10 plasma concentration and very low mHLA-DR values. Accordingly, “high NLR” patients presented with significantly more nosocomial infections (39%) than those of “low” NLR cluster (20%), which is likely a consequence of underlying decreased immune defenses. Furthermore, this “high NLR” cluster exhibited significantly increased mortality at both 28 and 90 days.
Overall, the current findings underscore novel aspects regarding NLR monitoring in ICU patients, revealing distinct trajectories during the first week after admission and highlighting the association between persistently elevated NLR and immunosuppression. NLR captures dual information: an increase in neutrophils (as a surrogate marker of inflammation) and lymphopenia (as a surrogate marker of immunosuppression). As such, this simple biomarker presents a potentially major interest in monitoring ICU patients, and upon further evaluation, it may help in better defining immunoadjuvant therapeutic strategy to be administered in patients. Indeed, the paradigm of an exclusively hyperinflammatory component during severe host aggression has evolved towards a more balanced understanding of the condition. The immune response to sepsis is actually a complex phenomenon involving both an inflammatory and an immunosuppressive response aimed at modulating the initial inflammatory reaction. The intensity of these two aspects of the response varies from one patient to another and, more importantly, evolves over time in one patient. As a result, the immuno-inflammatory trajectories of patients are heterogeneous, and any intervention aimed at modulating these trajectories in ICU patients (whether pro- or anti-inflammatory) should be considered from the perspective of precision medicine. Considering its simplicity and informative value, NLR could be incorporated into upcoming studies, whether observational or interventional. Furthermore, integrating this immune parameter into an updated version of the SOFA score could help evaluate immune failure alongside other organ failures.
AUTHOR CONTRIBUTION
AR, MHR: Statistical analysis, Writing / MG: Validation, review & editing / MB: Statistical analysis (K-means) / TR: patients’ inclusion, Project administration, review & editing / ACL: Validation, review & editing / FV, GM: Conceptualization, Methodology, Supervision, Validation, Writing, review & editing. All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
FUNDING
This work was supported by intramural Program of the EA7426 (Hospices Civils de Lyon, University Lyon-I & bioMérieux). The REALISM study received funding from the Agence Nationale de la Recherche through a grant awarded to BIOASTER (Grant number #ANR-10-AIRT-03) and from bioMérieux, Sanofi and GSK.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The REALISM study is registered at ClinicalTrials.gov (NCT02638779) and have been approved by the Institutional Study Board (2015-42-2). The patients/participants provided their written informed consent to participate in this study.
DECLARATION OF COMPETING INTEREST
MB is employee of bioMérieux. The other authors declare that they have no competing financial interests or personal relationships that could have influenced the work reported in this paper.
ACKNOWLEDGEMENTS
This work is as ancillary study of the REALISM study. The authors thank all members of REALISM study group.
REFERENCES
- Medzhitov R, Schneider DS, Soares MP. Disease tolerance as a defense strategy. Science. Feb 24 2012;335(6071):936-941.
- Rubio I, Osuchowski MF, Shankar-Hari M, et al. Current gaps in sepsis immunology: new opportunities for translational research. Lancet Infect Dis. 2019;In press.
- van der Poll T, Shankar-Hari M, Wiersinga WJ. The immunology of sepsis. Immunity. Nov 9 2021;54(11):2450-2464.
- Hotchkiss RS, Monneret G, Payen D. Sepsis-induced immunosuppression: from cellular dysfunctions to immunotherapy. Nat Rev Immunol. Dec 2013;13(12):862-874.
- Vincent JL, van der Poll T, Marshall JC. The End of “One Size Fits All” Sepsis Therapies: Toward an Individualized Approach. Biomedicines. Sep 12 2022;10(9).
- Stanski NL, Wong HR. Prognostic and predictive enrichment in sepsis. Nat Rev Nephrol. Jan 2020;16(1):20-31.
- Sinha P, Meyer NJ, Calfee CS. Biological Phenotyping in Sepsis and Acute Respiratory Distress Syndrome. Annual review of medicine. Jan 27 2023;74:457-471.
- Cajander S, Kox M, Scicluna BP, et al. Profiling the dysregulated immune response in sepsis: overcoming challenges to achieve the goal of precision medicine. Lancet Respir Med. Apr 2024;12(4):305-322.
- Pierrakos C, Vincent JL. Sepsis biomarkers: a review. Crit Care. 2010;14(1):R15.
- Demaret J, Venet F, Friggeri A, et al. Marked alterations of neutrophil functions during sepsis-induced immunosuppression. J Leukoc Biol. Dec 2015;98(6):1081-1090.
- Finfer S, Venkatesh B, Hotchkiss RS, Sasson SC. Lymphopenia in sepsis-an acquired immunodeficiency? Immunol Cell Biol. Dec 5 2022.
- Zahorec R. Ratio of neutrophil to lymphocyte counts–rapid and simple parameter of systemic inflammation and stress in critically ill. Bratislavske lekarske listy. 2001;102(1):5-14.
- Zahorec R. Neutrophil-to-lymphocyte ratio, past, present and future perspectives. Bratislavske lekarske listy. 2021;122(7):474-488.
- Firment J, Hulin I. Zahorec index or Neutrophil-to-lymphocyte ratio, valid biomarker of inflammation and immune response to infection, cancer and surgery. Bratislavske lekarske listy. 2024;125(2):75-83.
- Huang Z, Fu Z, Huang W, Huang K. Prognostic value of neutrophil-to-lymphocyte ratio in sepsis: A meta-analysis. Am J Emerg Med. Mar 2020;38(3):641-647.
- Dilektasli E, Inaba K, Haltmeier T, et al. The prognostic value of neutrophil-to-lymphocyte ratio on mortality in critically ill trauma patients. J Trauma Acute Care Surg. Nov 2016;81(5):882-888.
- Younan D, Richman J, Zaky A, Pittet JF. An Increasing Neutrophil-to-Lymphocyte Ratio Trajectory Predicts Organ Failure in Critically-Ill Male Trauma Patients. An Exploratory Study. Healthcare (Basel). Mar 14 2019;7(1).
- Torres LK, Pickkers P, van der Poll T. Sepsis-Induced Immunosuppression. Annu Rev Physiol. Oct 27 2021.
- Rol ML, Venet F, Rimmele T, et al. The REAnimation Low Immune Status Markers (REALISM) project: a protocol for broad characterisation and follow-up of injury-induced immunosuppression in intensive care unit (ICU) critically ill patients. BMJ Open. Jun 21 2017;7(6):e015734.
- Venet F, Textoris J, Blein S, et al. Immune Profiling Demonstrates a Common Immune Signature of Delayed Acquired Immunodeficiency in Patients With Various Etiologies of Severe Injury. Crit Care Med. Apr 1 2022;50(4):565-575.
- Bidar F, Bodinier M, Venet F, et al. Concomitant Assessment of Monocyte HLA-DR Expression and Ex Vivo TNF-alpha Release as Markers of Adverse Outcome after Various Injuries-Insights from the REALISM Study. J Clin Med. Dec 24 2021;11(1).
- Bodinier M, Peronnet E, Brengel-Pesce K, et al. Monocyte Trajectories Endotypes Are Associated With Worsening in Septic Patients. Front Immunol. 2021;12:795052.
- Salciccioli JD, Marshall DC, Pimentel MA, et al. The association between the neutrophil-to-lymphocyte ratio and mortality in critical illness: an observational cohort study. Crit Care. 2015;19:13.
- Roldgaard M, Benfield T, Tingsgard S. Blood neutrophil to lymphocyte ratio is associated with 90-day mortality and 60-day readmission in Gram negative bacteremia: a multi-center cohort study. BMC Infect Dis. Feb 23 2024;24(1):255.
- Hwang SY, Shin TG, Jo IJ, et al. Neutrophil-to-lymphocyte ratio as a prognostic marker in critically-ill septic patients. Am J Emerg Med. Feb 2017;35(2):234-239.
- Di Rosa M, Sabbatinelli J, Soraci L, et al. Neutrophil-to-lymphocyte ratio (NLR) predicts mortality in hospitalized geriatric patients independent of the admission diagnosis: a multicenter prospective cohort study. J Transl Med. Nov 21 2023;21(1):835.
- Coudereau R, Bodinier M, Lukaszewicz AC, et al. Persistent NLRP3 inflammasome activation is associated with delayed immunosuppression in septic patients. J Leukoc Biol. Mar 29 2024;115(4):706-713.
- Hotchkiss RS, Monneret G, Payen D. Immunosuppression in sepsis: a novel understanding of the disorder and a new therapeutic approach. Lancet Infect Dis. Mar 2013;13(3):260-268.

