Nodular Gastritis and Intestinal Metaplasia: A Review
Nodular gastritis and gastric intestinal metaplasia: Two bedfellows with the same dream who wake up in different worlds
Shiu Kum Lam, MD¹, George Ka Kit Lau, MD¹
- The Humanity & Health GI & Liver Centre, Suite 2101, 9 Queen’s Road Central, Hong Kong.
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Lam, SK., and Lau, GKK., 2024. Nodular gastritis and gastric intestinal metaplasia: Two bedfellows with the same dream who wake up in different worlds. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5796
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5796
ISSN 2375-1924
Abstract
Nodular gastritis (NG) and gastric intestinal metaplasia (GIM) are both the aftermath of H. pylori infection. Despite vigorous attempts to eradicate H. pylori in the past 4 decades, the infection has dropped, rather unimpressively, from 58.2% to 43.1%. Prevalence has been reported as 5% for NG and 25% for GIM. NG affects children and young adults especially females, and the symptoms are those of non-specific dyspepsia, whereas GIM is asymptomatic. NG is usually diagnosed at endoscopy with classical multitudinous nodules covering the antrum but also the body, and confirmed by the histological feature of lymphoid follicles, the basis of the nodules. GIM is detected by multiple biopsies (8 has been proposed, particularly for regions with high gastric cancer rates), and is graded histologically as mild, moderate, and severe. NG is treated by eradication of the H. pylori infection; if untreated, there is a 5% chance that extensive NG could develop into diffuse gastric cancer in 3 to 5 years. GIM does not improve despite successful eradication of H. pylori, nor does it respond to any other medical treatment. If untreated, there is a 5.8% chance for severe GIM to develop into gastric cancer in 5 years. Management of GIM has been by surveillance, with treatment instituted once early cancer appears. Management, however, has been revolutionized recently with the introduction of endoscopic mucosal resection using Lam’s technique. It is proposed that once detected, both NG and GIM should be treated vigorously until they are healed, to prevent possible malignant change.
Keywords
Nodular gastritis, gastric intestinal metaplasia, H. pylori, gastric cancer, endoscopic mucosal resection
Introduction
Gastric cancer is the 5th most common cancer in the world, and is the 4th leading cause of cancer deaths worldwide, following lung, colorectal and liver cancer in overall mortality; over a million new cases are diagnosed, worldwide, each year. The age-standardized rate (×1/105) of gastric cancer is 32.8 in East Asia (China, Japan, S Korea), 16.8 in European Union, and 4.2 in USA. Despite vigorous eradication of H. pylori infection in the past 4 decades, the decrease in world frequency of the infection has been recorded as from 58.2% to 43.1%, an unimpressive drop; 1.1 million new cases and 770,000 deaths of gastric cancer estimated in 2020 are expected to rise to 1.8 million and 1.3 million, respectively by 2040.
Nodular gastritis (NG) and gastric intestinal metaplasia (GIM) are precursors to gastric cancer. NG is responsible for the diffuse type of gastric cancer, and GIM for the intestinal type of gastric cancer. Both are the aftermath of H. pylori infection: they are bedfellows with the same dream and they wake up in different worlds.
The purpose of this review is to emphasize the same etiological agent, i.e. H. pylori, of these two conditions, and their totally diversified outcome. We began with a brief history of “gastritis”, went on to include the same etiology of NG and GIM, their different prevalence, pathology, classification, clinical picture, diagnosis, malignant potential, and treatment.
Methodology
We searched in the greatest detail possible the literature in relation to NG and GIM, including their etiology, prevalence, pathology including histology, classification, clinical picture, diagnosis, malignant potential and treatment.
History of gastritis
A right time to start the history of gastritis would be in 1939, when Abraham Friedberg and Louis Baron suspected that bacteria might be a cause of duodenal ulcer, gastric ulcer, and gastric cancer. They published two papers in the subsequent year that were mostly ignored.
Haot was the first to discover NG in 1988, when the group described lymphocyte gastritis, characterized by nodular and eroded lesions running along the gastric rugae in the corpus, although this most probably corresponded to the varioliform gastritis first proposed by Moutier and Martin in 1947. In 1998, Estham went on to describe the clinical picture in children.
Pelayo Correa first proposed in 1975 that under the effect of a mutagen (e.g. nitroso compound) the glandular gastric epithelium is gradually changed to intestinal-type epithelium, which he expanded in 1992 in the First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention into a sequential cascade: chronic gastritis; atrophy; intestinal metaplasia; and dysplasia.
Etiology
H. pylori infection causes of both nodular gastritis (NG) and gastric intestinal metaplasia (GIM), and both conditions have malignant potential of different types: two bedfellows with the same dream and wake up in different worlds.
H. pylori infection is a major cause of NG. NG has been considered a sine qua non condition of infection with H. pylori, with 100% of NG being caused by H. pylori. In another study involving 273 patients in Netherland, 87.5% had H. pylori infection. In one study of 97 adults with H. pylori infection and NG, regression of the nodules was observed in 63% after eradication of the infection. Regression of nodules had also been observed in another study.
Prevalence
NG
NG is a disease of children and of young adults particularly females, but no age is immune. NG is uncommon, with a reported incidence of 0.18–0.44% in endoscopy centres, although 1.9%, 5.1% and 7.2% have been reported. In one study of 114 patients with NG, 5% developed gastric cancer, all of which were of the diffuse type.
Interestingly, while NG has been well described in eastern nationalities like Koreans, Japanese, and Taiwanese, and while gastric cancer has been particularly common in China, the authors could locate, including searching in the Chinese language, only one report on NG from China, and this was a review article of subjects outside China, and in a recent study on national chronic gastritis, NG was not mentioned. In two studies in Japan, of 674 patients with H. pylori infections, NG was observed in 114 (17%) and in 40/265 (15.1%) in the other. It was more prevalent in women (69%) and young adults.
GIM
GIM is an adult disease and has no particular sex distribution. It has a worldwide prevalence of 25% in a meta-analysis of 107 studies involving 30,960 subjects, and its prevalence varies between western and eastern countries, being 7–25% in USA and Europe, and 24–84% in east Asia.
Pathology
Endoscopy
Both occur in the antrum, and in severe cases extend into the body of the stomach. GIM most frequently occurs in the prepyloric region, but is often multi-focal, spreading to involve the lower and upper antrum, incisura, and in extensive cases to the body of the stomach. While the nodular appearance of NG is readily discernible endoscopically, although it has been reported to be better visualized with high resolution endoscopy with NBI. GIM is invisible at routine endoscopy.
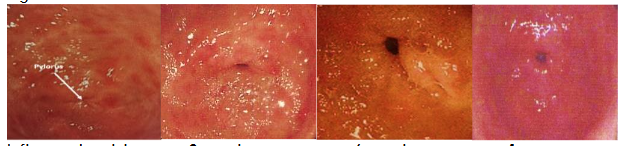
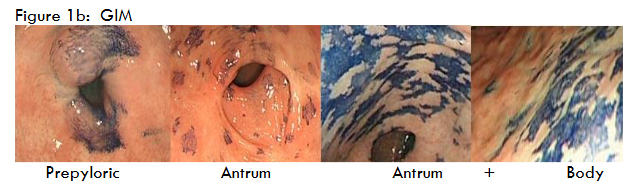
While there are features to identify GIM by white light endoscopy with the help of narrow-band imaging (NBI), GIM remains generally undetected at routine endoscopy; the correlation between endoscopy findings and histology is poor. Diagnosis is made after multiple biopsies of the antrum and body. Sydney system for grading of gastritis recommended five: two from antrum, two from corpus, and one from incisura. One study observed that the Sydney system might miss 50% of GIM and recommended 8 biopsies. The European MAPSII 2019 recommends 4 biopsies: two antral biopsies from the lesser and greater curvature, and two from the corpus, also from the lesser and greater curvature.
Histology
NG
Pangastritis is the commonest type of gastritis. Biopsy specimens of these lesions reveal lymphoid follicles with active and chronic inflammation. Intestinal metaplasia and atrophy are absent. The histopathology has been well described. Increase in intraepithelial lymphocytes is considered to have contributed to the macroscopic nodular appearance of this form of gastritis.
GIM
Histology remains the best way of recognizing its presence and assessing its severity. OLGIM (operative link on gastric intestinal metaplasia) is fast replacing OLGA (operative link on gastric atrophy). Classification of GIM.
Traditional
- Classification by mucin secretion
This early classification classifies GIM into three subtypes, based on morphology and mucin staining methods such as periodic acid-Schiff, Alcian blue, and high iron diamine: Type I contains absorptive cells, Paneth cells and goblet cells secreting sialomucin; type II has few absorptive cells, consists of columnar cells and goblet cells secreting predominantly sialomucin but some sulphomucin, and has presence of Paneth cells; and type III consists of columnar cells and goblet cells secreting predominantly sulphomucin, and absence of Paneth cells. In this subclassification, type I corresponds to complete GIM and types II and III to incomplete GIM. Because special stains are used — including high-iron diamine/Alcian blue stain— and because they carry toxicity, such staining is not routinely used.
- Complete and incomplete
This is the classical classification. The complete form has a complete set of enzymes, with well-defined brush border (microvilli) and well-formed goblet cells, and sometimes Paneth cells. The incomplete colonic form doesn’t have all these digestive enzymes and there is no brush border or Paneth cells. Simply stated, complete intestinal metaplasia closely resembles small intestinal epithelium and goblet cells. Incomplete intestinal metaplasia resembles the colonic epithelium.
Practical
- Classification by severity
The simplicity of this classification: mild, moderate, and severe, using hematoxylin and eosin routine staining, and its practical use in clinical medicine has made it an appeal to clinicians. The main purpose of classification is for deciding whether and when endoscopic surveillance should be done. This is because there has been no treatment for GIM until recently (wide infra) and gastric atrophy. For example, the use of Cox-2 inhibitors plus H. pylori eradication had been shown to be unsuccessful.
These conditions usually persist despite eradication of H. pylori. The situation has been described as “a point of no return.”
Clinical
NG occurs most commonly between 30 and 40 years of age. Epigastric pain, nausea, vomiting and bloating have been reported in NG, possibly by virtue of its intense inflammation, as revealed by routine endoscopy. GIM, on the other hand, is asymptomatic, and cannot be detected visually by routine endoscopy. Mean age has been reported as 62.3 ± SE 2.9 years for males and 58.6 ± SE 2.4 years for females, and there is no sex preference.
Diagnosis
NG
Because of symptoms, many patients with NG end up in an endoscopy examination, which itself, together with biopsies, will reveal the diagnosis. Others, because of positive 13C-breath test, would be revealed at subsequent endoscopy and biopsies.
IM
As the condition is asymptomatic, it is usually discovered on biopsy specimens following an endoscopy for non-specific reasons including a family history of stomach cancer, or when a 13C-breath test reveals H. pylori infection. Because routine endoscopy does not reveal IM, an adequate number of biopsies is important for the diagnosis. A gastroscopy with five biopsies is recommended by the updated Sydney classification of gastritis. El-Zimaity and Graham found that IM was missed in more than 50% of the biopsies from “Sydney” sites in patients with confirmed gastric IM on multiple site sampling. These authors concluded that the minimum number of biopsies needed to identify IM should probably be eight, and emphasized that current and future studies that use the Sydney system as a basis for detecting gastric IM are not likely to be reliable.
In ethnic groups and regions with increased prevalence for gastric cancer, however, we agree that a total of eight biopsies that cover prepyloric region, the broader antrum, incisura, body and fundus of the stomach is recommended.
Malignant potential
To classify GIM histologically, the extent, or number of sites involved in the stomach, and the grade, or severity of GIM involvement of the biopsied fragments, should both be measured. The subtype, an additional histologic classification for GIM, is based on distinct microscopic appearances. For the purpose of using intestinal metaplasia to identify high-risk individuals, the AGA guidelines recommend using the extent and subtype. The European guidelines classify GIM as limited if one site—antrum/incisura or corpus—is involved, and as extensive if both sites are involved.
Gastric cancer, diffuse type
NG has been regarded as premalignant, with increased presence of premalignant pathologies including gastric atrophy, GIM, and dysplasia. There is strong evidence that NG is associated with the diffuse type of gastric cancer, including two young females of 16 and 29 years.
Gastric cancer, intestinal type
GIM is a precursor to gastric cancer, with a pooled odds ratio (OR) of 3.6 in a meta-analysis of 21 studies comprising 402,636 participants, and an OR of 29.3 in rural China. Annual progression from GIM to cancer also differs, being 129 x 10-5 in Sweden and 600 x 10-5 in China. GIM is not reversible. It is often extensive, affecting major areas of the stomach, and extensiveness is associated with increased cancer risk. Individuals with widespread metaplasia affecting both gastric antrum and body were at higher risk, with an absolute risk of 5.8% at 5 years. Hence, the extent of the metaplasia determined with mapping biopsies, regardless of the subtype, should also be incorporated into the risk assessment of the patient. A rapid progression to adenocarcinoma in 20 months has been reported.
H. pylori is known to start a cascade of chronic gastritis, gastric atrophy, GIM, and gastric cancer. In a meta-analysis of 24 studies involving 48,064 individuals, eradication of H. pylori reduced the consequence of cancer by 54%; the failure in the rest was attributed to pre-existing GIM, as affirmed by a meta-analysis of 16 studies involving 52,363 subjects. Attempts to treat GIM with long-term supplements or selective COX-2 inhibitor had failed. There is no treatment for GIM.
The grading of OLGIM according to pathology: mild, moderate, and severe, and according to site: antrum, and body is fast replacing the grading into complete and incomplete. Patients with GIM in both gastric antrum and body were significantly more likely to progress to dysplasia than those with GIM in only one location.
Treatment
NG
In one study, 40 adults with NG (94% with H. pylori) and 30 controls without NG (30% with H. pylori), H. pylori therapy in patients with NG led to significant decrease in symptoms (epigastric pain, nausea, vomiting, bloating) and histologic abnormalities. Another study showed that after H. pylori eradication, two-thirds of the patients have their nodules regressed in 38 months.
GIM
Management of GIM, discovered usually from gastric biopsy specimens, had been expectant, as there was no treatment available, including drug treatment and endoscopy treatment. GIM is not reversible. Surveillance, followed by endoscopic resection when feasible or by open surgery, had been the approach.
| Mild | Moderate | Severe |
|---|---|---|
| 1 – 30% | 31 – 60% | >60% |
Timing of surveillance proposed based on recommendations in the literature
| OLGIM | Antrum only | Fundus only | Antrum + Fundus |
|---|---|---|---|
| Mild (1 – 30%) | Nil | Nil | Nil |
| Moderate (31 – 60%) | 3 years | 3 years | 1 – 2 years |
| Severe (>60%) | 3 years | 3 years | 1 – 2 years |
| Family history of gastric cancer | 3 years | 3 years | 1 – 2 years |
| Family history + moderate or severe GIM | 1 – 2 years | 1 – 2 years | 1 – 2 years |
| Autoimmune gastritis | 3 years | 3 years | 3 years |
A breakthrough treatment, however, has been discovered using an endoscopic approach. Lam’s endoscopy treatment uses two endoscopes. One endoscope was used to perform chromoendoscopy with methylene blue to disclose and mark the areas of GIM. Submucosal saline injections were used to lift the stained mucosa to form multiple safety cushions. Before the saline diffuses out of the cushions, a second endoscope, fitted with a cap for ligation of esophageal varices, was quickly inserted after withdrawal of the first endoscope. With the second endoscope, the cushions were transformed into artificial polyps by suction and ligation, using the cap for ligation of esophageal varices. The second endoscope was withdrawn and the first endoscope was reintroduced. EMRs were then achieved by snare polypectomy. By rotating two gastroscopes, one designated to perform lift and snare and the other to perform suction and ligation, cycles of lift–ligate–snare were carried out until all stained mucosa was removed.
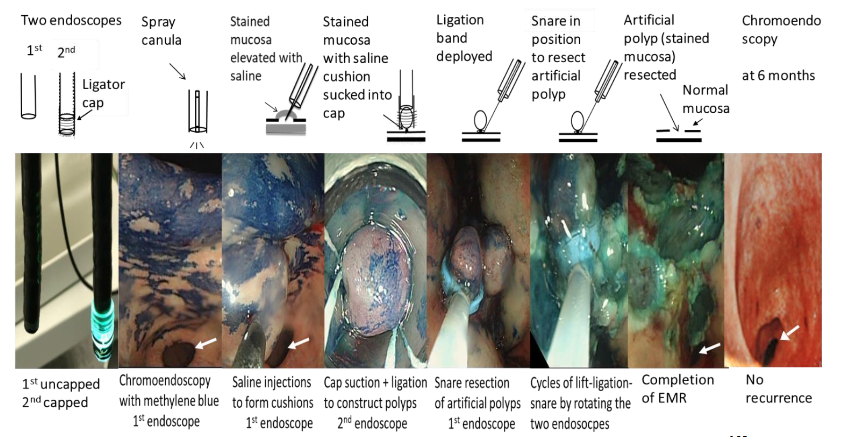
A major advantage of Lam’s technique of EMR is its ability to resect gastric lesions larger than 2 cm, a limit beyond which has not been endoscopically achieved in the past, and GIM is often larger than 2 cm.
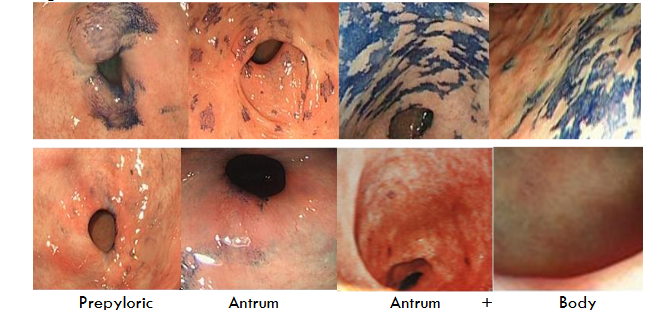
Discussion
| NG | GIM |
|---|---|
| Etiology: H. pylori | Etiology: H. pylori |
| Prevalence: 5% | Prevalence: 25% |
| Age and sex: Affects children & young adults (< 29 yr old), especially females | Age and sex: No age & sex preference |
| Symptoms: Non-specific abdominal discomfort | Symptoms: Silent |
| Endoscopy: Characteristic nodules | Endoscopy: Invisible. Shows up on chromoendoscopy |
| Histology: Lymphoid follicles | Histology: From simple brush-border & globet cells to colonic cells |
| Malignant potential: 5% develops diffuse cancer in 3.5 years for extensive NG | Malignant potential: 5.8% develops intestinal type cancer in 5 years for severe IM |
| Treatment: Eradication of H. pylori | Treatment: Lam’s endoscopic mucosal resection |
Both nodular gastritis (NG) and gastric intestinal metaplasia (GIM) are precursors to gastric cancer, the diffuse type for NG and the intestinal type of GIM, and both are the aftermath of H. pylori infection. While eradication of H. pylori helps to prevent NG from developing into cancer, this does not help GIM to develop into cancer; and in fact, no medication has been shown to help. This is not surprising since NG itself has been regarded as a sine qua non condition of infection with H. pylori, whereas in GIM mutation has taken place and the resultant morphology is likened to that of small or large intestinal cells that are irreversible, including eradication of H. pylori. It is heartening that GIM can now be treated without recurrence by endoscopic mucosal resection using Lam’s technique.
This has made the detection of NG and GIM an important tool. Detection of NG is simple, since NG is sine qua non with H. pylori infection, which can simply be diagnosed by 13C-breath test or at endoscopy; and NG itself is often symptomatic, leading to investigations that often reveal its presence. GIM detection, on the other hand, can be difficult, since the condition itself is clinically silent, and is often discovered incidentally when endoscopy with multiple biopsies is done.
Despite vigorous eradication of H. pylori infection in the past 4 decades, the decrease in world frequency of the infection has been recorded as from 58.2% to 43.1%, an unimpressive drop. This can be interpreted that there remains an abundance of nodular gastritis and intestinal metaplasia. Yet, 1.1 million new cases and 770,000 deaths of gastric cancer estimated in 2020 are expected to rise to 1.8 million and 1.3 million, respectively by 2040. While such results are open to interpretations including methods and vigor in diagnosis and treatment of the infection, and non-abate of food carcinogens, a more serious, collaborative world effort is needed to rid the H. pylori infection from this earth and educate the public in a concerted effort to reduce known carcinogens from our food, particularly since gastric cancer has become a largely preventable disease.
Conflicts of interests
The authors have no conflict of interest or financial relationship relevant to this article. The technique reported in this study was accepted by the Australia Patent Office, patent number 2017272212 Title: Two-Endoscope Technique of Endoscopic Mucosal Resection.
Funding
This study is not supported by any funding.
Acknowledgements
The authors gratefully acknowledged the support of the staffs at The Humanity & Health GI & Liver Centre, Hong Kong, particularly Ms. C Ho (endoscopy nurse), and Ms. A Wong, and were particularly grateful to the expertise of Dr. SC Yuen (pathologist).
Ethics
This study was approved by Hong Kong Clinical Research Ethics Committee.
References
- Sung, H, Ferlay, J, Siegel, RL, Laversanne, M, Soerjomataram, I, Jemal, A, Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021: 71: 209-249. https://doi.org/10.3322/caac.21660.
- Yan X, Lei L, Li H, Cao M, Yang F, He S, Zhang S, Teng Y, Li Q, Xia C, Chen W. Stomach cancer burden in China: Epidemiology and prevention. Chin J Cancer Res. 2023 Apr 30;35(2):81-91. doi: 10.21147/j.issn.10009604.2023.02.01. PMID: 37180831; PMCID: PMC10167608.
- Shao L, Li P, Ye J, Chen J, Han Y, Cai J, Lu X. Risk of gastric cancer among patients with gastric intestinal metaplasia. Int J Cancer 2018;143:1671–1677. PMID: 29707766, doi:10.1002/ijc.31571.
- Butt J, Epplein M. How do global trends in Helicobacter pylori prevalence inform prevention planning? Lancet 2023;8:498-499.
- Morgan E, Arnold M, Camargo MC, et al. The current and future incidence and mortality of gastric cancer in 185 countries, 2020-40: A population-based modelling study. EClinicalMedicine. 2022 Apr 21;47:101404. doi: 10.1016/j.eclinm.2022.101404. PMID: 35497064; PMCID: PMC9046108.
- Freedberg, A. Stone, “An Early Study of Human Stomach Bacteria” 1939, in Helicobacter Pioneers, pp. 25–28.
- Freedberg, A.S.; L.E. Baron (1940). “The presence of spirochaetes in human gastric mucosa”. American Journal of Digestive Diseases. 7 (10): 443–445. doi:10.1007/BF02997393.
- Haot J, Hamichi L, Wallez L, Mainguet P. Lymphocytic gastritis: a newly described entity: a retrospective endoscopic and histological study. Gut 1988;29:1258-64. https://doi.org/10.1136/gut.29.9.1258.
- Moutier F. Martin J. Deaux cas de gastrite varioliforme. Arch Mal App Dig 1947;36:155-161.
- Eastham EJ, Elliott TSJ, Berkeley D, Jones DM. Campylobacter pylori infection in children. J. Infect. 1988; 16: 77–9.
- Correa P, Haenszel W, Cuello C, Tannenbaum S, Archer M. A model for gastric cancer epidemiology. Lancet. 1975 Jul 12;2(7924):58-60. doi: 10.1016/s0140-6736(75)90498-5. PMID: 49653.
- Correa P. Human gastric carcinogenesis: a multistep and multifactorial process–First American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res. 1992 Dec 15;52(24):6735-40. PMID: 1458460.
- Miyamoto M, Haruma K, Yoshihara M, Hiyama T, Sumioka M, Nishisaka T, Tanaka S, Chayama K. Nodular gastritis in adults is caused by Helicobacter pylori infection. Dig Dis Sci. 2003 May;48(5):968-75. doi: 10.1023/a:1023016000096. PMID: 12772798.
- Sugano K, Tack J, Kuipers EJ, et al. Kyoto global consensus report on Helicobacter pylori gastritis. Gut 2015; 64: 1353-1367.
- Malfertheiner P, Megraud F, O’Morain CA, et al. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut 2017; 66: 6-30.
- Mahachai V, Vilaichone RK, Pittayanon R, et al. Thailand Consensus on Helicobacter pylori Treatment 2015.Asian Pac J Cancer Prev. 2016; 17: 2351-2360.
- Authors. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. The Eur Res J 2019;1-5. DOI: 10.18621/eurj.420164.
- Pennelli G, Grillo F, Galuppini F, Ingravallo G, Pilozzi E, Rugge M, Fiocca R, Fassan M, Mastracci L. Gastritis: update on etiological features and histological practical approach. Pathologica. 2020 Sep;112(3):153-165. doi: 10.32074/1591-951X-163. PMID: 33179619; PMCID: PMC7931571.
- Chen, Ming-Jen, & Wang, Tsang-En & Chang, Wen-Hsiung & Liao, Ta-Chuan & Lin, Ching-Chung & Shih, Shou-Chuan. (2007). Nodular Gastritis: An Endoscopic Indicator of Helicobacter Pylori Infection. Digestive diseases and sciences. 52. 2662-6. 10.1007/s10620-006-9281-3.
- Nakamura, S.; Mitsunaga, A.; Imai, R.; et al. Clinical Evaluation of Nodular Gastritis in Adults. Dig. Endosc. 2007, 19, 74–79.
- Nakashima R.; Nagata N.; Watanabe K.; et al. Histological Features of Nodular Gastritis and its Endoscopic Classification. J. Dig. Dis. 2011, 12, 436–442.
- Önal İK, Sökmensüer C, Demir Önal E, Ersoy O, Yeniova Ö, Aydınlı M, et al. Clinical and pathological features of nodular gastritis in adults. Turk J Med Sci 2009;39:719-23.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Netherlands J Med 2015;73:236-241.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Kim JW, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, Han HS. Nodule Regression in Adults With Nodular Gastritis. Gastroenterology Res. 2015;8:296–302. doi: 10.14740/gr692w.
- Bahú Mda G, da Silveira TR, Maguilnick I, Ulbrich-Kulczynski J. Endoscopic nodular gastritis: an endoscopic indicator of high-grade bacterial colonization and severe gastritis in children with Helicobacter pylori. J Pediatr Gastroenterol Nutr. 2003 Feb;36(2):217-22. doi: 10.1097/00005176-200302000-00011. PMID: 12548057.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med. 2015 Jun;73(5):236-41. PMID: 26087803.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Yalak S. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. Eur Res J 2019;5:822-826.
- Chen, Ming-Jen, & Wang, Tsang-En & Chang, Wen-Hsiung & Liao, Ta-Chuan & Lin, Ching-Chung & Shih, Shou-Chuan. (2007). Nodular Gastritis: An Endoscopic Indicator of Helicobacter Pylori Infection. Digestive diseases and sciences. 52. 2662-6. 10.1007/s10620-006-9281-3.
- Dwivedi, M., Misra, S.P. and Misra, V. (2008), Nodular gastritis in adults: Clinical features, endoscopic appearance, histopathological features, and response to therapy. Journal of Gastroenterology and Hepatology, 23: 943-947. https://doi.org/10.1111/j.1440-1746.2007.05044.x.
- Miyamoto, M.; Haruma, K.; Yoshihara, M.; et al. Nodular Gastritis in Adults is Caused by Helicobacter pylori Infection. Dig. Dis. Sci. 2003, 48, 968–975.
- Nakamura, S.; Mitsunaga, A.; Imai, R.; et al. Clinical Evaluation of Nodular Gastritis in Adults. Dig. Endosc. 2007, 19, 74–79.
- Nakashima, R.; Nagata, N.; Watanabe, K.; et al. Histological Features of Nodular Gastritis and its Endoscopic Classification. J Dig Dis 2011, 12, 436–442.
- Önal İK, Sökmensüer C, Demir Önal E, Ersoy O, Yeniova Ö, Aydınlı M, et al. Clinical and pathological features of nodular gastritis in adults. Turk J Med Sci 2009;39:719-23.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Netherlands J Med 2015;73:236-241.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Kim JW, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, Han HS. Nodule Regression in Adults With Nodular Gastritis. Gastroenterology Res. 2015;8:296–302. doi: 10.14740/gr692w.
- Bahú Mda G, da Silveira TR, Maguilnick I, Ulbrich-Kulczynski J. Endoscopic nodular gastritis: an endoscopic indicator of high-grade bacterial colonization and severe gastritis in children with Helicobacter pylori. J Pediatr Gastroenterol Nutr. 2003 Feb;36(2):217-22. doi: 10.1097/00005176-200302000-00011. PMID: 12548057.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med. 2015 Jun;73(5):236-41. PMID: 26087803.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Yalak S. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. Eur Res J 2019;5:822-826.
- Chen, Ming-Jen, & Wang, Tsang-En & Chang, Wen-Hsiung & Liao, Ta-Chuan & Lin, Ching-Chung & Shih, Shou-Chuan. (2007). Nodular Gastritis: An Endoscopic Indicator of Helicobacter Pylori Infection. Digestive diseases and sciences. 52. 2662-6. 10.1007/s10620-006-9281-3.
- Dwivedi, M., Misra, S.P. and Misra, V. (2008), Nodular gastritis in adults: Clinical features, endoscopic appearance, histopathological features, and response to therapy. Journal of Gastroenterology and Hepatology, 23: 943-947. https://doi.org/10.1111/j.1440-1746.2007.05044.x.
- Miyamoto, M.; Haruma, K.; Yoshihara, M.; et al. Nodular Gastritis in Adults is Caused by Helicobacter pylori Infection. Dig. Dis. Sci. 2003, 48, 968–975.
- Nakamura, S.; Mitsunaga, A.; Imai, R.; et al. Clinical Evaluation of Nodular Gastritis in Adults. Dig. Endosc. 2007, 19, 74–79.
- Nakashima, R.; Nagata, N.; Watanabe, K.; et al. Histological Features of Nodular Gastritis and its Endoscopic Classification. J Dig Dis 2011, 12, 436–442.
- Önal İK, Sökmensüer C, Demir Önal E, Ersoy O, Yeniova Ö, Aydınlı M, et al. Clinical and pathological features of nodular gastritis in adults. Turk J Med Sci 2009;39:719-23.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Netherlands J Med 2015;73:236-241.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Kim JW, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, Han HS. Nodule Regression in Adults With Nodular Gastritis. Gastroenterology Res. 2015;8:296–302. doi: 10.14740/gr692w.
- Bahú Mda G, da Silveira TR, Maguilnick I, Ulbrich-Kulczynski J. Endoscopic nodular gastritis: an endoscopic indicator of high-grade bacterial colonization and severe gastritis in children with Helicobacter pylori. J Pediatr Gastroenterol Nutr. 2003 Feb;36(2):217-22. doi: 10.1097/00005176-200302000-00011. PMID: 12548057.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med. 2015 Jun;73(5):236-41. PMID: 26087803.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Yalak S. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. Eur Res J 2019;5:822-826.
- Chen, Ming-Jen, & Wang, Tsang-En & Chang, Wen-Hsiung & Liao, Ta-Chuan & Lin, Ching-Chung & Shih, Shou-Chuan. (2007). Nodular Gastritis: An Endoscopic Indicator of Helicobacter Pylori Infection. Digestive diseases and sciences. 52. 2662-6. 10.1007/s10620-006-9281-3.
- Dwivedi, M., Misra, S.P. and Misra, V. (2008), Nodular gastritis in adults: Clinical features, endoscopic appearance, histopathological features, and response to therapy. Journal of Gastroenterology and Hepatology, 23: 943-947. https://doi.org/10.1111/j.1440-1746.2007.05044.x.
- Miyamoto, M.; Haruma, K.; Yoshihara, M.; et al. Nodular Gastritis in Adults is Caused by Helicobacter pylori Infection. Dig. Dis. Sci. 2003, 48, 968–975.
- Nakamura, S.; Mitsunaga, A.; Imai, R.; et al. Clinical Evaluation of Nodular Gastritis in Adults. Dig. Endosc. 2007, 19, 74–79.
- Nakashima, R.; Nagata, N.; Watanabe, K.; et al. Histological Features of Nodular Gastritis and its Endoscopic Classification. J Dig Dis 2011, 12, 436–442.
- Önal İK, Sökmensüer C, Demir Önal E, Ersoy O, Yeniova Ö, Aydınlı M, et al. Clinical and pathological features of nodular gastritis in adults. Turk J Med Sci 2009;39:719-23.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Netherlands J Med 2015;73:236-241.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Kim JW, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, Han HS. Nodule Regression in Adults With Nodular Gastritis. Gastroenterology Res. 2015;8:296–302. doi: 10.14740/gr692w.
- Bahú Mda G, da Silveira TR, Maguilnick I, Ulbrich-Kulczynski J. Endoscopic nodular gastritis: an endoscopic indicator of high-grade bacterial colonization and severe gastritis in children with Helicobacter pylori. J Pediatr Gastroenterol Nutr. 2003 Feb;36(2):217-22. doi: 10.1097/00005176-200302000-00011. PMID: 12548057.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med. 2015 Jun;73(5):236-41. PMID: 26087803.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Yalak S. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. Eur Res J 2019;5:822-826.
- Chen, Ming-Jen, & Wang, Tsang-En & Chang, Wen-Hsiung & Liao, Ta-Chuan & Lin, Ching-Chung & Shih, Shou-Chuan. (2007). Nodular Gastritis: An Endoscopic Indicator of Helicobacter Pylori Infection. Digestive diseases and sciences. 52. 2662-6. 10.1007/s10620-006-9281-3.
- Dwivedi, M., Misra, S.P. and Misra, V. (2008), Nodular gastritis in adults: Clinical features, endoscopic appearance, histopathological features, and response to therapy. Journal of Gastroenterology and Hepatology, 23: 943-947. https://doi.org/10.1111/j.1440-1746.2007.05044.x.
- Miyamoto, M.; Haruma, K.; Yoshihara, M.; et al. Nodular Gastritis in Adults is Caused by Helicobacter pylori Infection. Dig. Dis. Sci. 2003, 48, 968–975.
- Nakamura, S.; Mitsunaga, A.; Imai, R.; et al. Clinical Evaluation of Nodular Gastritis in Adults. Dig. Endosc. 2007, 19, 74–79.
- Nakashima, R.; Nagata, N.; Watanabe K.; et al. Histological Features of Nodular Gastritis and its Endoscopic Classification. J Dig Dis 2011, 12, 436–442.
- Önal İK, Sökmensüer C, Demir Önal E, Ersoy O, Yeniova Ö, Aydınlı M, et al. Clinical and pathological features of nodular gastritis in adults. Turk J Med Sci 2009;39:719-23.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Netherlands J Med 2015;73:236-241.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Kim JW, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, Han HS. Nodule Regression in Adults With Nodular Gastritis. Gastroenterology Res. 2015;8:296–302. doi: 10.14740/gr692w.
- Bahú Mda G, da Silveira TR, Maguilnick I, Ulbrich-Kulczynski J. Endoscopic nodular gastritis: an endoscopic indicator of high-grade bacterial colonization and severe gastritis in children with Helicobacter pylori. J Pediatr Gastroenterol Nutr. 2003 Feb;36(2):217-22. doi: 10.1097/00005176-200302000-00011. PMID: 12548057.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med. 2015 Jun;73(5):236-41. PMID: 26087803.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Yalak S. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. Eur Res J 2019;5:822-826.
- Chen, Ming-Jen, & Wang, Tsang-En & Chang, Wen-Hsiung & Liao, Ta-Chuan & Lin, Ching-Chung & Shih, Shou-Chuan. (2007). Nodular Gastritis: An Endoscopic Indicator of Helicobacter Pylori Infection. Digestive diseases and sciences. 52. 2662-6. 10.1007/s10620-006-9281-3.
- Dwivedi, M., Misra, S.P. and Misra, V. (2008), Nodular gastritis in adults: Clinical features, endoscopic appearance, histopathological features, and response to therapy. Journal of Gastroenterology and Hepatology, 23: 943-947. https://doi.org/10.1111/j.1440-1746.2007.05044.x.
- Miyamoto, M.; Haruma, K.; Yoshihara, M.; et al. Nodular Gastritis in Adults is Caused by Helicobacter pylori Infection. Dig. Dis. Sci. 2003, 48, 968–975.
- Nakamura, S.; Mitsunaga, A.; Imai, R.; et al. Clinical Evaluation of Nodular Gastritis in Adults. Dig. Endosc. 2007, 19, 74–79.
- Nakashima, R.; Nagata, N.; Watanabe K.; et al. Histological Features of Nodular Gastritis and its Endoscopic Classification. J Dig Dis 2011, 12, 436–442.
- Önal İK, Sökmensüer C, Demir Önal E, Ersoy O, Yeniova Ö, Aydınlı M, et al. Clinical and pathological features of nodular gastritis in adults. Turk J Med Sci 2009;39:719-23.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Netherlands J Med 2015;73:236-241.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Kim JW, Lee SY, Kim JH, Sung IK, Park HS, Shim CS, Han HS. Nodule Regression in Adults With Nodular Gastritis. Gastroenterology Res. 2015;8:296–302. doi: 10.14740/gr692w.
- Bahú Mda G, da Silveira TR, Maguilnick I, Ulbrich-Kulczynski J. Endoscopic nodular gastritis: an endoscopic indicator of high-grade bacterial colonization and severe gastritis in children with Helicobacter pylori. J Pediatr Gastroenterol Nutr. 2003 Feb;36(2):217-22. doi: 10.1097/00005176-200302000-00011. PMID: 12548057.
- Niknam R, Manafi A, Maghbool M, Kouhpayeh A, Mahmoudi L. Is endoscopic nodular gastritis associated with premalignant lesions? Neth J Med. 2015 Jun;73(5):236-41. PMID: 26087803.
- Kim YJ, Lee SY, Kim JH, Sung IK, Park HS. Nodular Gastritis as a Precursor Lesion of Atrophic and Metaplastic Gastritis. Korean J Gastroenterol 2019;73:332-340.
- Yalak S. The frequency of nodular gastritis in adult patients and the relation of this type of gastritis with Helicobacter pylori and histopathologic findings. Eur Res J 2019;5:822-826.

