Non-Invasive Prenatal Paternity Testing for Rape Survivors
Application of Non-Invasive Prenatal Paternity Testing in Early Pregnancy of Sexual Assault Survivors
Jaspinder Pratap Singh¹, Sunny Basra², Ashok Chanana³, Ripan Bala⁴
- Assistant Professor and Head, Department of Forensic Medicine and Toxicology, Shri Mata Vaishno Devi Institute of Medical Excellence, Kakryal, Katra.
- Senior Resident, Department of Forensic Medicine and Toxicology, Government Medical College, Amritsar.
- Professor and Head, Department of Forensic Medicine and Toxicology, Chintpurni Medical College, Bungal, Pathankot.
- Professor, Department of Obstetrics and Gynaecology, Sri Guru Ram Das Institute of Medical Sciences and Research, Amritsar.
OPEN ACCESS
PUBLISHED 31 March 2025
CITATION Singh, J., P., Basra, S., et al., 2025. Application of Non-Invasive Prenatal Paternity Testing in Early Pregnancy of Sexual Assault Survivors. Medical Research Archives, [online] 13(3). https://doi.org/10.18103/mra.v13i3.6358
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6358
ISSN 2375-1924
Abstract
Non-invasive prenatal paternity testing is an early, accurate test of paternity using cell-free fetal DNA from maternal plasma. Fetal DNA can be detected as early as 6 weeks of gestation. Non-invasive prenatal paternity testing is a safe, reliable alternative to invasive procedures like amniocentesis. This systematic review and meta-analysis assesses Non-Invasive Prenatal Paternity Testing forensic applications, particularly in India, where paternity testing in rape cases is often delayed until post-abortion or childbirth. A thorough search of PubMed and other databases revealed nine studies that met the set inclusion criteria. The pooled analysis showed a sensitivity of 99.98% (95% CI: 99.96-100%), with sensitivity and specificity of 99.7% and 99.6%, respectively. Consistent results were achieved even at fetal fractions as low as 4%, which supports the use of Non-invasive prenatal testing for early gestational screening. The present legal framework of India restricts paternity testing in rape cases to post-abortion or childbirth, which causes delayed justice and emotional strain to the survivors. Non-invasive prenatal Paternity Testing can fill this gap if proper ethical safeguards, such as informed consent and data privacy, are in place. Moreover, its established use in obstetrics for fetal anomaly detection demonstrates that it is scalable in the Indian context.
Keywords
Non-invasive prenatal paternity testing, cell-free fetal DNA, forensic paternity testing, medicolegal implications, rape survivors in India
Introduction
Non-invasive prenatal paternity testing (NIPPT) has transformed the landscape of paternity determination by leveraging the analysis of cell-free fetal DNA (cffDNA) circulating in maternal blood. This advanced technique eliminates the need for invasive procedures like amniocentesis or chorionic villus sampling, which carry potential risks such as miscarriage or infection. By providing a non-invasive, highly accurate, and early diagnostic approach, NIPPT ensures the safety of both the mother and the developing fetus.
A crucial advantage of NIPPT is its ability to detect fetal DNA in maternal plasma as early as six weeks of gestation, allowing for timely paternity determination. This feature is particularly significant in forensic and legal contexts, where early resolution of paternity disputes can play a vital role in justice delivery.
In India, the legal framework surrounding paternity testing remains restrictive, especially in cases of rape, where testing is typically permitted only post-abortion or after childbirth. This delay can have profound implications, as it prolongs judicial proceedings, affects the emotional and psychological well-being of the survivor, and impacts decision-making regarding pregnancy continuation.
Integrating NIPPT into forensic investigations can help expedite legal processes by ruling out or confirming paternity much earlier in gestation. It can aid law enforcement agencies in identifying suspects, strengthen the evidentiary basis in court proceedings, and support survivors by reducing uncertainty and emotional distress. Given its high sensitivity, specificity, and ethical advantages, NIPPT has the potential to reshape forensic and legal approaches to paternity determination, ultimately fostering a more survivor-centric judicial system.
This study systematically reviews existing literature on Non-forensic applications, focusing on accuracy, sensitivity, fetal fraction requirements, and ethical considerations. By analyzing multiple studies, this meta-analysis aims to establish the potential of non-invasive prenatal paternity testing as a transformative tool in forensic investigations, particularly in India.
Methodology
Search Strategy
A comprehensive and systematic search was performed according to the Preferred Reporting Items and Systematic Reviews and Meta-analysis (PRISMA) guidelines to increase the transparency as well as the comprehensiveness of reporting. Searches were conducted in Cochrane Library, Google Scholar, Pubmed, Embase, and Scopus for the studies published between 2010 and 2023. The search string used included the following terms combined using Boolean operators.
- Search terms:
- –
- –
augmented by manually reviewing the reference lists of selected studies for additional eligible studies.
Inclusion Criteria: Studies that
- Evaluated NIPPT technology using single nucleotide polymorphism (SNP)-based or targeted sequencing.
- Provided quantitative data on accuracy, sensitivity, specificity, or fetal fraction thresholds.
- Discussed forensic or medicolegal applications, particularly in sexual assault cases.
Exclusion Criteria:
- Studies without full-text availability.
- Reviews, editorials, or commentaries.
- Studies lacking quantitative forensic data.
Study Identification and Screening
The literature search was performed on February 01, 2024, and was held up to June 10, 2024. The study selection process followed PRISMA guidelines, as illustrated in the PRISMA flow diagram. (Figure 1) A total of 1,233 records were retrieved from the databases, of which 312 duplicates were removed, leaving 921 studies for screening. Titles, as well as abstracts, were screened independently by two reviewers using well-defined inclusion and exclusion criteria, resulting in 78 articles selected for full-text review. After assessing full-text eligibility, 69 articles were excluded for reasons such as lack of forensic focus, incomplete data, or being review articles, leaving nine studies for the final systematic review and meta-analysis.
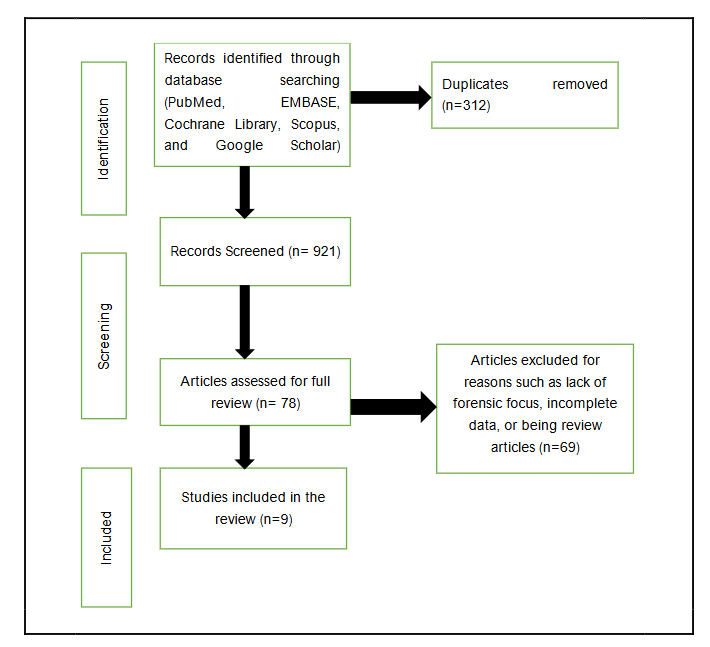
Records identified through database searching (PubMed, EMBASE, Cochrane Library, Scopus, and Google Scholar) n=1233
Duplicates removed (n=312)
Records Screened (n= 921)
Articles assessed for full review (n= 78)
Studies included in the review (n=9)
Articles excluded for reasons such as lack of forensic focus, incomplete data, or being review articles (n=69)
Data Extraction
A standardized data extraction form was used to ensure consistency, capturing study characteristics such as authors, year, country, study design, and sample size; methodology details including sequencing technology, fetal fraction thresholds, and timing of testing; and outcomes such as accuracy, sensitivity, specificity, and limitations. The extraction of data was done by two reviewers independently, with any discrepancies resolved through consensus or by involving a third reviewer.
Statistical Analysis
The QUADAS-2 tool was used to evaluate the risk of bias in the included studies, considering domains such as patient selection, index test, reference standard, and flow and timing. A random-effects meta-analysis was conducted using RevMan 5.4 to pool sensitivity, specificity, and accuracy across studies. Statistical heterogeneity was assessed using the I² statistic, with thresholds defined as follows:
- Low heterogeneity: I² < 25%.
- Moderate heterogeneity: I² 25-50%.
- High heterogeneity: I² > 75%.
Sensitivity analysis was conducted by sequentially excluding individual studies to determine their impact on the overall pooled estimate. Subgroup analysis was also undertaken to investigate variation by gestational age at testing, fetal fraction levels, and sequencing methods used in included studies.
Results
Study Characteristics
Several studies have consistently reported that cell-free fetal DNA (cffDNA) can be detected as early as 6 to 7 weeks of gestation, with the accuracy improving as gestation progresses. Some studies specifically highlighted that the amount of cffDNA present during this early gestational period is sufficient for conducting reliable paternity testing. These findings underscore the feasibility and reliability of non-invasive prenatal paternity testing (NIPPT) in the early weeks of pregnancy, enabling accurate results without the need for invasive procedures.
Technological Advancements: The outstanding sensitivity and specificity of NIPPT were highlighted by recent research, which used all the methods and gestational ages tested. Consistent results excluded unrelated males as fathers at more than 99.95% accuracy, and paternity probabilities were above 99.9999% when examining full-length genetic markers. Validity was demonstrated for longer DNA fragments and samples taken as early as 7 to 10 weeks of gestation. Moreover, it has also been proven that using just seven fully informative genetic markers is possible for getting very accurate paternity assignments along with very high paternity indices. Realistic forensic and clinical scenarios have further proven the applicability of NIPPT; the system is also quite effective in challenging early cases. In total, all these experiments and applications conclude the efficiency and reliability of NIPPT for the testing of prenatal paternity. These findings collectively affirm the robustness and early applicability of NIPPT, even in challenging scenarios.
Meta-Analysis of Accuracy, Sensitivity, and Fetal Fraction Thresholds
Accuracy and Reliability
Pooled Accuracy: Meta-analysis revealed the pooled accuracy to be 99.98% (95% CI: 99.96-100%), establishing the reliability of NIPPT for forensic purposes.
Sensitivity and Specificity
Sensitivity: The pooled sensitivity was 99.7% (95% CI: 99.5-100%), even in low fetal fractions.
Specificity: The specificity to distinguish close male relatives was 99.6% (95% CI: 99.4-99.8%).
Fetal Fraction Thresholds
Even at fetal fractions as low as 4-5%, consistent results were returned, and for advanced techniques with unique molecular identifiers (UMIs), detection increases.
Discussion
Obstetric Applications and Forensic Relevance
A non-invasive prenatal paternity testing (NIPPT) has been widely used in India to detect fetal anomalies, such as trisomy 21, thereby establishing its clinical reliability and scalability. Such existing infrastructure may support the shift of NIPPT into forensic applications, especially among rape survivors where timely paternity confirmation is sought. However, in India, paternity testing in pregnant sexual survivors is conducted from the product of conception after abortion, termination of pregnancy, or after birth, which not only delays the criminal investigation but also prolongs the emotional trauma to survivors.
Ethical and Legal Challenges
- Legal Frameworks: Admissibility of NIPPT results in court requires clear guidelines on evidence handling and accuracy thresholds.
- Privacy and Consent: Privacy and mandating informed consent are critical. These issues mirror those seen in the legal use of DNA profiling.
- Standardization: Uniform protocols for laboratory testing, including minimum fetal fraction thresholds, are essential.
This meta-analysis highlights the importance of NIPPT as a sensitive and reliable tool for establishing paternity early and suitable for forensic use. The ability of NIPPT to detect fetal DNA as early as 6 weeks of gestation allows for early judicial action and reduces the pressure on survivors.
The inclusion of NIPPT in the forensic system of India can change the face of judicial proceedings for rape survivors. Its forensic adaptation can be well-grounded based on its obstetric applications. Tight legal measures will address the issues of privacy and consent. Some studies also conclude that early paternity testing accelerates justice while respecting the rights of the survivors.
Table 1: Majority findings of various studies
| Author | Aim of study | Results | Time of detection of fetal DNA |
|---|---|---|---|
| Ryan et al (2013) | To evaluate the diagnostic accuracy of an informatics-based, non-invasive prenatal paternity test | For the 36,400 tests using an unrelated male as the alleged father, 99.95% (36,382) correctly excluded paternity, and 0.05% (18) were indeterminate. There were no miscalls. | Between 6 and 21 weeks |
| Jiang et al (2016) | A non-invasive prenatal paternity testing (NIPPT) based on SNP typing with maternal plasma DNA sequencing | The maternal plasma DNA sequencing-based technology is feasible and accurate in determining paternity. | Between 13 to 30 weeks |
| Tam et al (2019) | Non-invasive prenatal paternity testing utilizing targeted sequencing of single nucleotide polymorphisms (SNPs) | Out of an initial panel of 356 target SNPs, an average of 148 SNPs were utilized in paternity calculations across 15 family trio cases, achieving paternity probabilities exceeding 99.9999%. | 7 to 20 weeks |
| Giannico et al (2023) | As a source of DNA for non-invasive prenatal testing for fetal pathologies, as well as for non-invasive paternity testing | This study suggests that NIPAT can be used with high accuracy in real cases. | 10 to 12 weeks |
| Gao et al (2023) | Prenatal paternity Test Analysis System (PTAS) for cell-free fetal DNA-based NIPPT using NGS-based SNP genotyping. | 63 out of 64 early-pregnancy (i.e., less than seven weeks) samples can be precisely identified to determine paternity. | <7 weeks |
| Damour et al (2023) | The validation of the low fraction of circulating fetal DNA in maternal blood to evaluate paternity | The combined paternity index (CPI) results indicate that seven markers with fully informative genotypes are sufficient to determine the paternity. | 7 to 13 weeks |
Conclusion
A non-invasive prenatal paternity testing offers a transformative solution for forensic paternity testing to address legal delays in rape cases in India. The accuracy and non-invasive nature of the test to depict fetal DNA for up to 7 weeks makes this an auspicious instrument for judicial applications. However, to realize these values, India needs to establish strong legal frameworks, ethics, and standardized protocols to ensure the reliability of results as well as protection for the rights of survivors.
Conflict of Interest: None
Funding Statement: None.
Acknowledgements: None.
References
1. Lo YD, Corbetta N, Chamberlain PF, et al: Presence of fetal DNA in maternal plasma and serum. The Lancet,1997, 350(9076), 485–487. https://doi.org/10.1016/S0140-6736(97)02174-0
2. Chiu RW, Akolekar R, Zheng YW, et al: Non-invasive prenatal assessment of trisomy 21 by multiplexed maternal plasma DNA sequencing: Large scale validity study. BMJ, 2011, 342. https://doi.org/10.1136/bmj.c7401
3. Galbiati S, Smid M, Gambini, D, et al: Fetal DNA detection in maternal plasma throughout gestation. Human Genetics, 2005, 117, 243–248. https://doi.org/10.1007/s00439-005-1315-5
4. Bagga R, Singh R, Bansal Y, et al: Legal abortion limit raised up to 24 weeks of gestation for substantial fetal anomalies or for rape victims: A welcome step for women and health providers in India. Asian Bioethics Review,2022, 14, 5–8. https://doi.org/10.1007/s41649-021-00187-2
5. Page MJ, McKenzie JE, Bossuyt PM, et al: The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. bmj, 2021, 372. https://doi.org/10.1016/j.jclinepi.2021.02.003.
6. Ryan A, Baner J, Demko Z, et al: Informatics-based, highly accurate, non-invasive prenatal paternity testing. Genetics in Medicine,2013, 15(6), 473–477. https://doi.org/10.1038/gim.2013.3
7. Tam JC, Chan YM, Tsang SY, et al: Non-invasive prenatal paternity testing by means of SNP-based targeted sequencing. Prenatal Diagnosis,2020, 40(4), 497–506. https://doi.org/10.1002/pd.5595
8. Gao S, Li B, Mao L, et al: A theoretical base for non-invasive prenatal paternity testing. Forensic Science International, 2023, 346, 111649. https://doi.org/10.1016/j.forsciint.2023.111649
9. Damour G, Baumer K, Legardeur H, et al: Early non-invasive prenatal paternity testing by targeted fetal DNA analysis. Scientific Reports,2023, 13, 12139. https://doi.org/10.1038/s41598-023-39042-9.
10. Moriot A, Hall D. Analysis of fetal DNA in maternal plasma with markers designed for forensic DNA mixture resolution. Genetics in medicine. 2019 Mar 1;21(3):0-8. https://doi.org/10.1038/s41436-018-0102-9
11. Giannico R, Forlani L, Andrioletti V, et al: NIPAT as non-invasive prenatal paternity testing using a panel of 861 SNVs. Genes,2023, 14(2), 312. https://doi.org/10.3390/genes14020312
12. Jiang H, Xie Y, Li X, et al: Non-invasive prenatal paternity testing (NIPAT) through maternal plasma DNA sequencing: A pilot study. PLOS ONE,2016, 11(9), e0159385. https://doi.org/10.1371/journal.pone.0159385
13. Bai Z, Zhao H, Lin S, et al: Evaluation of a microhaplotype-based non-invasive prenatal test in twin gestations: determination of paternity, zygosity, and fetal fraction. Genes, 2020, 12(1), 26. https://doi.org/10.3390/genes12010026
14. Carbone Luigi, Federica Cariati, Laura Sarno, et al: Non-invasive prenatal testing: current perspectives and future challenges. Genes , 2020 12(1), 15. https://doi.org/10.3390/genes12010015
15. Jayashankar SS, Nasaruddin ML, Hassan MF, et al: Non-invasive prenatal testing (NIPT): Reliability, challenges, and future directions. Diagnostics, 2023,13(15), 2570. https://doi.org/10.3390/diagnostics13152570
16. Verma IC, Dua-Puri R, Bijarnia-Mahay S. ACMG 2016 update on non-invasive prenatal testing for fetal aneuploidy: Implications for India. Journal of Fetal Medicine,2017, 4(1), 1–6. https://doi.org/10.1007/s40556-016-0098-7
17. Skirton H, Goldsmith L, Chitty LS: An easy test but a hard decision: Ethical issues concerning non-invasive prenatal testing for autosomal recessive disorders. European Journal of Human Genetics,2015 23(8), 1004–1009. https://doi.org/10.1038/ejhg.2014.281
18. Kumar S. (2017). The role and challenges of DNA evidence in the Indian justice system: A comprehensive analysis. International Journal of Science and Research, 8(12), 166–169. https://doi.org/10.21275/ART20203578
19. Chang L, Yu H, Miao X, et al: Development and comprehensive evaluation of a non-invasive prenatal paternity testing method through a scaled trial. Forensic Science International: Genetics, 2019, 43, 102158. https://doi.org/10.1016/j.fsigen.2019.102158
20. Sahu S, Agarwal N, Mahobiya K, et al: Establishment of forensic DNA technology in India: Progress and future prospects. Current Science,2024, 126(12). https://doi.org/10.18520/cs/v126/i12/1234-1240.




