NT-proBNP and Insulin Resistance in Metabolic Syndrome
N-terminal pro B-type natriuretic peptide and insulin resistance in patients with metabolic syndrome
Dr Ipsita Dash 2, Dr Pratima Kumari sahu 3, Dr Rasmita Kumari Padhy 4, Dr om Padarabinda Dash 1
- Professor Department of Biochemistry SCB MCH Cuttack Odisha
- Professor Department of Biochemistry Saheed rendo majhi medical college and hospital kalahandi
- Assistant professor, Pulmonary medicine (Saheed rendo majhi medical college and hospital kalahandi), Odisha
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Author1, X., Author2, Z., et al., 2024. N-terminal pro B-type natriuretic peptide and insulin resistance in patients with metabolic syndrome. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5811
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5811
ISSN 2375-1924
ABSTRACT
Background: B-type natriuretic peptide (BNP) and N-terminal pro-atrial natriuretic peptide (NT-proANP) plasma levels are linked to metabolic risk. Metabolic syndrome has been associated with lower levels of N-terminal pro B-type natriuretic peptide (NT-proBNP) and insulin resistance.
Aim: This study aims to evaluate the association between NT-proBNP and insulin resistance, diabetes and cardiovascular (CV) risk factors.
Methods: A total of 183 subjects, aged 25-70 years were included in the study based on the National Cholesterol Education Program (NCEP) Adult Treatment Panel III (ATP-III) guidelines for metabolic syndrome. Anthropometric parameters, subclinical measurements and biochemical markers were measured. Gender-specific comparisons were conducted using unpaired t-tests. Correlation between NT-proBNP and insulin resistance in metabolic syndrome was assessed using regression analysis. A p-value of less than 0.05 was considered statistically significant.
Results: Of the 183 subjects, 58% were male and 42% were female. Key clinical parameters – such as blood pressure, body mass index (35.57±6.02 kg/m²) and waist circumference (95.50±9.75 cm) were measured. Other biochemical parameters, including elevated triglycerides, Low-Density Lipoprotein (LDL) cholesterol and blood sugar levels, as well as reduced insulin and NT-proBNP levels were observed. The study found an inverse relationship between NT-proBNP and insulin levels.
Conclusion: Females exhibited higher levels of NT-proBNP and insulin. This study identified an inverse relationship between NT-proBNP and insulin resistance.
Keywords: Metabolic Syndrome; Insulin Resistance; NT pro BNP; Dyslipidemia; cardiovascular risk.
INTRODUCTION
A decade ago, metabolic syndrome was first introduced as ‘Syndrome X’. Over time, different researchers have referred to it by various names – such as the ‘deadly quartet’ and the ‘silent killer’. Later, it was defined as insulin resistance syndrome and ultimately as metabolic syndrome, characterized by an aggregation of metabolic abnormalities. These abnormalities include insulin resistance, glucose intolerance, central obesity, dyslipidemia and increased blood pressure. The striking rise in the incidence of metabolic syndrome can be attributed to the growing global burden of diabetes and obesity. According to a World Health Organization report, approximately 20–25% of the global population suffers from metabolic syndrome.
In India, the prevalence of metabolic syndrome is increasing due to urbanization, lack of physical activity and high-calorie diets. Numerous studies have documented the prevalence of metabolic syndrome among the Indian population, which ranges between 11% and 41%. Sinha SK et al. reported that the prevalence in India ranges from 22.1% to 41%. A higher prevalence of metabolic syndrome among female patients has been documented globally and our study also had a greater number of female subjects.
Insulin resistance contributes to increased fatty acid synthesis and the production of apolipoprotein B-containing triglyceride-rich very low-density lipoprotein (LDL) in the liver. Visceral, metabolically active tissue synthesizes bioactive secretory molecules that promote a prothrombotic state, smooth muscle proliferation and vascular remodelling. A combination of these mechanisms is believed to play a key role in the pathophysiology of metabolic syndrome.
Metabolic syndrome is strongly linked to an increased risk of developing atherosclerotic cardiovascular disease (CVD). Its pathogenesis involves both genetic and acquired factors that contribute to the final inflammatory pathway leading to CVD. The exponential increase in obesity has made metabolic syndrome increasingly relevant in recent years. Insulin resistance, neurohormonal activation and chronic inflammation are the primary drivers in the initiation, progression and transition of metabolic syndrome to CVD. The activation of pro-atherogenic pathways in metabolic syndrome leads to a final common inflammatory pathway, which eventually results in clinical manifestations of the condition. Alongside lifestyle modifications, addressing risk factors is essential for preventing complications associated with metabolic syndrome. This study was planned to evaluate the relationship between NT-proBNP and insulin resistance in metabolic syndrome patients.
MATERIALS & METHODS
An analytical study was conducted in the Department of Biochemistry in collaboration with the Department of Medicine at Saheed Laxman Nayak Medical College and Hospital, Koraput, Odisha, India. After obtaining written informed consent – a total of 183 subjects, aged 25-70 years and of both sexes were enrolled in the study. These subjects met at least three criteria of the National Cholesterol Education Program (NCEP) Adult Treatment Panel III (ATP III) definition for metabolic syndrome over a study period of 24 months. Subjects with a history of diabetes, hypertension, obesity, or hypothyroidism were excluded. Additionally, individuals diagnosed with liver diseases, hepatitis, or other vulnerable conditions such as HIV or sexually transmitted diseases (STDs) were also excluded. The study received approval from the institutional ethics committee (IEC Approval No………).
General and clinical Characteristics: Anthropometric parameters, including height, weight, waist circumference (WC) and body mass index (BMI) were measured using standard methods followed at Saheed Hospital. Blood pressure, both systolic and diastolic, was measured for each participant in accordance with the 2017 American College of Cardiology and American Heart Association Hypertension Guidelines, using a sphygmomanometer.
Sample collection and storage: After an overnight fast – a total of 8 ml of intravenous blood was collected from each patient using aseptic precautions. The blood samples were collected in Plain, Lavender and Fluoride vacutainers. The collected samples were centrifuged at 4000 rpm for 8-10 minutes to separate serum and plasma. The Lavender vacutainer, containing 3 ml of blood, was used to measure glycosylated haemoglobin. A 2 ml plasma sample was used to analyze fasting and postprandial blood sugar, 1 ml of serum was used for lipid profile analysis and 2 ml of serum was stored at -20°C for other biochemical analyses.
Laboratory Investigations: After blood sample collection and proper distribution, blood sugar was analyzed using enzymatic methods (GOD-POD method). Lipid parameters – such as total cholesterol and triglycerides were measured using the cholesterol oxidase and glycerol kinase methods, respectively. HDL cholesterol was analyzed using immunoprecipitation methods and LDL cholesterol was calculated using the Friedewald formula. Human insulin and NT-proBNP levels were analyzed using an ELISA kit. The Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) was calculated by multiplying fasting glucose by fasting insulin levels and dividing by 22.5.
Statistical analysis: Study tools: Data were collected using case record forms (CRF), which included details regarding clinical history, medication history, general characteristics, anthropometric data and biochemical marker values. The data were then entered into Microsoft Excel for further organization and analysis. MedCalc statistical software for Windows was used for statistical analysis. Descriptive statistics were reported as mean, standard deviation and standard error of the mean. The normal distribution of data was assessed using the Shapiro-Wilk test with an alpha value of less than 0.05 – represented in the form of a histogram. Since the data did not follow a normal distribution, non-parametric tests were used to calculate the p-value for variables between males and females. A correlation coefficient scatter plot was employed to determine the relationship between NT-proBNP and metabolic syndrome. A p-value of less than 0.05 was considered statistically significant.
RESULTS
A total of 183 subjects who met the NCEP-ATP-III criteria for metabolic syndrome were studied retrospectively. Demographic data were analyzed based on age and gender. Of the study population, 58% were male and 42% were female. The mean age of the subjects was 55.29±9.99 years. A descriptive analysis of the medical conditions showed that 14% had type-II diabetes (T2DM), 20% had hypertension (HTN), 27% had both T2DM and HTN, 8% were obese, 7% had T2DM with obesity, 5% had T2DM with both HTN and obesity and 16% had T2DM with HTN and were on medication.
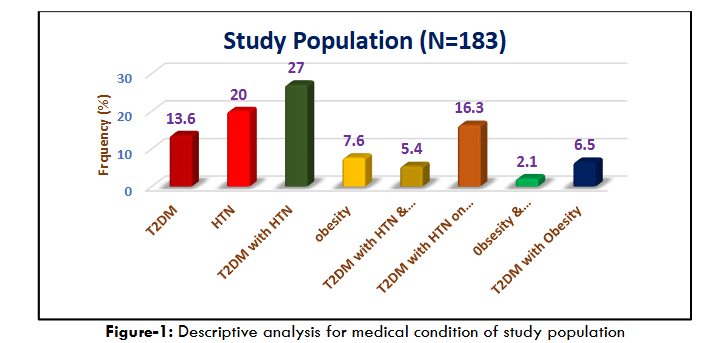
Basic clinical measurements such as blood pressure, body mass index (BMI: 35.57±6.02 kg/m²) and waist circumference (95.50±9.75 cm) were recorded. Additionally, biochemical parameters, including lipid profile, blood sugar, insulin and NT-proBNP levels were evaluated to assess the relationship between insulin resistance and NT-proBNP. A correlation analysis revealed a nonlinear, significant correlation between NT-proBNP and both BMI and waist circumference. However, there was a significant linear inverse correlation between NT-proBNP and insulin resistance and diabetes (P<0.05).
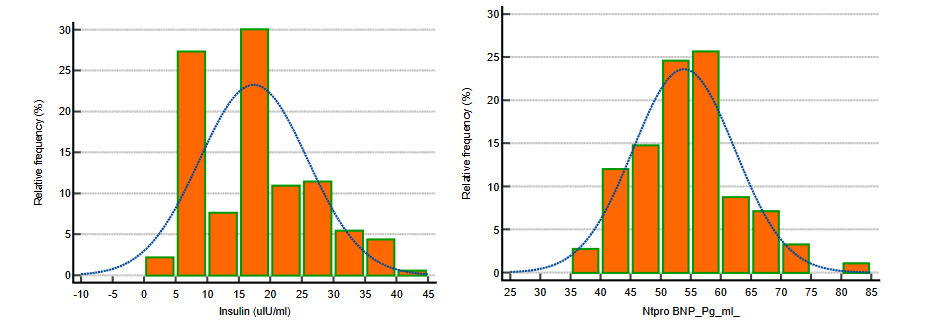
To assess the distribution of NT-proBNP and insulin levels in the study population, we performed a Shapiro-Wilk test. The test was applied to check the normality of the data distribution for both NT-proBNP and insulin. Below are the histograms representing the relative frequency of NT-proBNP and insulin levels in the study population. The fitted curve illustrates the normal distribution model applied to the data.
The histogram in Figure 2(a) shows the distribution of NT-proBNP levels in the population. The data exhibit a near-normal distribution, as indicated by the bell-shaped curve. However, there are some deviations in the tails of the distribution, which may suggest slight skewness. In Figure 2(b), the histogram for insulin levels shows a skewed distribution. The data are not normally distributed with a right-skewed pattern indicating a higher concentration of participants with lower insulin levels and fewer individuals with elevated insulin levels. This skewness reinforces the decision to use non-parametric tests for further statistical analyses. In summary, the Shapiro-Wilk test confirmed that the insulin data did not follow a normal distribution, while NT-proBNP data were closer to normality, although with slight deviations. Thus, non-parametric methods were employed to analyze relationships between NT-proBNP, insulin resistance and other metabolic parameters.
Clinical and Biochemical Characteristics of the Study Group
| Parameters | Study variables (N=183) | Maximum | Minimum | Mean | SD | SEM |
|---|---|---|---|---|---|---|
| Age (years) | 77 | 21 | 55.29 | 9.99 | 0.74 | |
| Body Mass Index (Kg/m²) | 59.9 | 17.9 | 35.57 | 6.02 | 0.45 | |
| Waist circumference (Cm) | 123 | 36 | 95.50 | 9.75 | 0.72 | |
| Systolic Blood Pressure (mm/Hg) | 190 | 110 | 146.85 | 17.73 | 1.31 | |
| Diastolic Blood Pressure (mm/Hg) | 116 | 70 | 89.66 | 6.29 | 0.47 | |
| FBS (mg/dl) | 324 | 81 | 163.31 | 52.34 | 3.87 | |
| PP2BS (mg/dl) | 431 | 81 | 163.31 | 52.34 | 4.48 | |
| Total cholesterol (mg/dl) | 560 | 100 | 278.82 | 90.58 | 6.70 | |
| Triacylglyceride (mg/dl) | 630 | 113 | 130.48 | 90.58 | 9.93 | |
| HDL (mg/dl) | 68 | 17 | 31.08 | 9.12 | 0.67 | |
| LDL (mg/dl) | 173 | 58 | 124.95 | 14.42 | 1.07 | |
| HbA1C (gm%) | 15.4 | 5.8 | 7.56 | 1.98 | 0.15 | |
| Insulin (uIU/ml) | 40 | 0.8 | 17.39 | 8.58 | 0.63 | |
| IR HOMA | 25.9 | 0.26 | 7 | 4.36 | 0.32 | |
| NT pro BNP (pg/ml) | 82 | 36 | 53.92 | 8.45 | 0.62 |
Interpretation of Results: The mean age of the study participants was 55.29 years with a BMI of 35.57 kg/m² and a waist circumference of 95.50 cm, suggesting that the majority of participants fell into the category of central obesity. The mean systolic blood pressure was 146.85 mmHg and indicates that a substantial proportion of participants had elevated blood pressure – a key component of metabolic syndrome. Lipid profile analysis revealed elevated total cholesterol (mean: 278.82 mg/dl), triglycerides (mean: 130.48 mg/dl) and LDL cholesterol (mean: 124.95 mg/dl), all of which are associated with increased cardiovascular risk. Notably, the mean HDL cholesterol was lower (31.08 mg/dl), further contributing to the overall atherogenic risk in this population. In terms of glucose metabolism, the mean fasting blood sugar (FBS) and postprandial blood sugar (PP2BS) were both elevated, reinforcing the presence of insulin resistance among the subjects. Additionally, the mean HOMA-IR value of 7, combined with an average insulin level of 17.39 μIU/ml, suggests a significant level of insulin resistance within the group. The NT-proBNP levels averaged 53.92 pg/ml, which is relevant for exploring its association with metabolic parameters and insulin resistance in subsequent analyses.
Correlation Analysis Between NT-proBNP, Insulin, HOMA-IR and HbA1C
To explore the relationship between NT-proBNP and metabolic indicators such as insulin, HOMA-IR and HbA1C, we performed a Spearman correlation analysis. The scatter plots below show the correlation of NT-proBNP with insulin, HOMA-IR and HbA1C. A significant inverse relationship was observed and indicates that lower NT-proBNP levels are associated with higher insulin resistance and elevated HbA1C levels, both of which are critical markers for metabolic syndrome.
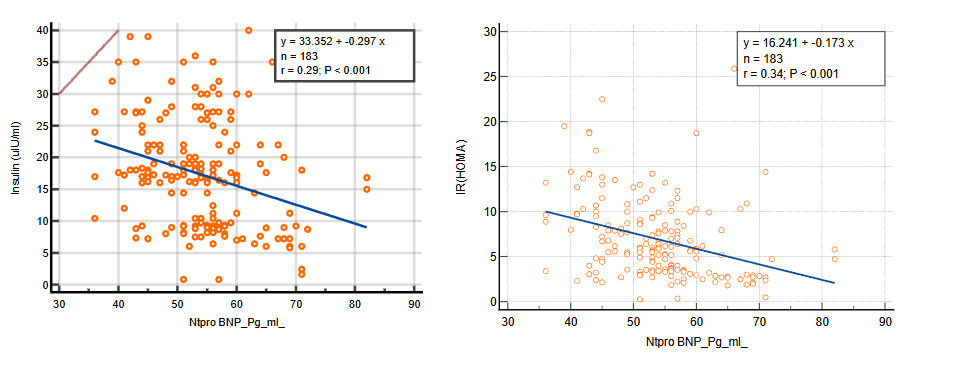
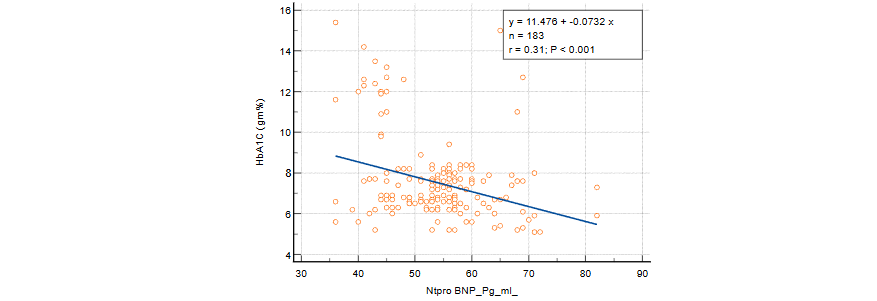
Figure 3: Correlation (Spearman scatter plot) of NT-proBNP with (a) Insulin, (b) HOMA IR and (c) HbA1C.
Interpretation of Results: In Figure 3(a), the scatter plot illustrates the correlation between NT-proBNP and insulin levels. The negative slope of the regression line (r = -0.29, P < 0.001) suggests a significant inverse relationship between NT-proBNP and insulin, meaning that higher insulin levels are associated with lower NT-proBNP values. This relationship reinforces the role of NT-proBNP in metabolic regulation. Figure 3(b) shows the correlation between NT-proBNP and HOMA-IR with a similarly strong inverse relationship (r = -0.34, P < 0.001). This finding indicates that as NT-proBNP levels decrease, insulin resistance (as measured by HOMA-IR) increases. This supports the hypothesis that NT-proBNP could be a marker for insulin resistance in individuals with metabolic syndrome. In Figure 3(c), we see the correlation between NT-proBNP and HbA1C levels. The inverse association (r = -0.31, P < 0.001) further strengthens the evidence that lower NT-proBNP levels are linked to poor glycemic control over time, as indicated by elevated HbA1C levels. This finding is particularly relevant for understanding the long-term glycemic burden in patients with metabolic syndrome.
Comparison of Clinical and Biochemical Characteristics Between Male and Female Participants
| Parameters | Male (N=106) | Female (N=77) | P value | df | Mean± SD | SEM | Mean± SD | SEM |
|---|---|---|---|---|---|---|---|---|
| Age (years) | 56.21±10.21 | 0.97 | 54.18±9.91 | 1.13 | 0.52 | 0.64 | ||
| Body Mass Index (Kg/m²) | 35.98±9.19 | 0.89 | 35.95±5.64 | 0.64 | 0.97 | 0.02 | ||
| Waist circumference (Cm) | 97.23±8.14 | 0.79 | 93.53±11.32 | 1.29 | 0.01* | 2.57 | ||
| SBP (mm/Hg) | 146.78±17.00 | 1.65 | 146.86±18.80 | 2.14 | 0.97 | 0.03 | ||
| DBP (mm/Hg) | 96.46±67.59 | 6.57 | 89.36±6.43 | 0.73 | 0.35 | 0.91 | ||
| FBS (mg/dl) | 156.50±47.08 | 4.57 | 171.66±58.00 | 6.61 | 0.05* | 1.94 | ||
| PP2BS (mg/dl) | 180.77±51.05 | 4.96 | 201.30±70.31 | 8.01 | 0.02* | 2.28 | ||
| Total cholesterol (mg/dl) | 266.90±86.19 | 8.37 | 293.56±94.69 | 10.79 | 0.04* | 1.98 | ||
| Triacylglyceride (mg/dl) | 296.05±126.52 | 12.29 | 328.75±143.08 | 16.31 | 0.10 | 1.63 | ||
| HDL (mg/dl) | 31.53±10.24 | 0.99 | 30.17±7.29 | 0.83 | 0.32 | 0.99 | ||
| LDL (mg/dl) | 123.23±15.21 | 1.48 | 127.55±12.96 | 1.48 | 0.04* | 2.06 | ||
| HbA1C (gm%) | 7.51±1.91 | 0.19 | 7.57±2.10 | 0.24 | 0.84 | 0.20 | ||
| Insulin (uIU/ml) | 16.93±8.24 | 0.80 | 17.94±9.05 | 1.03 | 0.43 | 0.78 | ||
| IR HOMA | 6.49±4.09 | 0.40 | 7.54±4.65 | 0.53 | 0.10 | 0.61 | ||
| NT pro BNP (pg/ml) | 53.56±8.33 | 0.81 | 54.30±8.65 | 0.99 | 0.56 | 0.58 |
INTERPRETATION OF RESULTS: The comparison of clinical and biochemical characteristics between males and females reveals some key differences. Notably, females had a significantly lower waist circumference compared to males (P = 0.01), although both groups exhibited elevated BMI values with no significant difference (P = 0.97). Fasting blood sugar (FBS) and postprandial blood sugar (PP2BS) were significantly higher in females compared to males (P = 0.05 and P = 0.02, respectively) and indicates that females in this study group had poorer glycemic control. In terms of lipid profiles, females had significantly higher total cholesterol (P = 0.04) and LDL cholesterol levels (P = 0.04) compared to males. However, HDL levels were comparable between the two groups (P = 0.32). Interestingly, although triglyceride levels were higher in females, this difference was not statistically significant (P = 0.10).
There were no significant differences observed in insulin levels (P = 0.43) or HOMA-IR (P = 0.10) between males and females, suggesting similar levels of insulin resistance in both groups. NT-proBNP levels – a key marker of cardiovascular risk were also similar between males and females (P = 0.56) and indicates no significant sex-based variation in this biomarker within the study group. These findings highlight gender-based differences in metabolic syndrome components with females showing greater risk in terms of lipid profile and blood glucose levels – which may have implications for cardiovascular risk and metabolic health in this population.
DISCUSSION
Over the past decade – there has been a significant rise in the prevalence of insulin resistance, affecting approximately 20 to 25% of the general population. Insulin resistance is recognized as one of the central contributors to metabolic syndrome, driving the growing epidemics of diabetes and cardiovascular abnormalities. Abnormal fat distribution in the visceral region plays a key role in the pathophysiology of metabolic syndrome. Numerous studies have demonstrated a strong association between visceral adiposity and insulin resistance. Visceral fat is considered a strong determinant of beta-cell function and insulin sensitivity. Waist circumference is a well-established measure of visceral fat or obesity and is preferred over body mass index (BMI) for quantifying central obesity in routine clinical practice. In this study, anthropometric analysis revealed an increased BMI and waist circumference (P<0.05), both of which are prominent risk factors for obesity and associated with an elevated risk of diabetes and cardiovascular disease.
A study conducted by Singh O et al. (2015) found that the mean waist circumference was elevated (98.61 ± 7.64 cm), highlighting its role as an indicator of visceral adipose tissue, which serves as a source of free fatty acids that are converted into triglycerides (TG) by the liver. Hypertension is another major burden in the general population and is strongly associated with metabolic syndrome. The exact mechanisms underlying hypertension in metabolic syndrome are multifactorial. Insulin-related alterations in metabolic syndrome provoke sodium reabsorption, sympathetic hyperactivation, endothelin-1–mediated vasoconstriction and increased blood pressure, all of which contribute to elevated inflammatory mediators. Globally, the prevalence of hypertension—defined as systolic blood pressure (SBP) ≥140 mmHg or diastolic blood pressure (DBP) ≥90 mmHg—is 2 to 3 times higher in patients with diabetes and obesity compared to those without these conditions with over 60% of type 2 diabetes patients affected.
In our study, there was no significant influence on systolic and diastolic values and a decrease in diastolic blood pressure was observed. Abnormal lipid levels in the bloodstream are a clustering cardiovascular risk associated with insulin resistance in metabolic syndrome. Elevated triglycerides, apolipoprotein B and small-dense low-density lipoprotein (LDL) particles, along with reduced high-density lipoprotein (HDL) cholesterol, are key factors in the development of atherosclerosis in metabolic syndrome. These factors are closely linked to hepatic and peripheral insulin resistance.
We examined the relationship between lipid profiles and insulin resistance in metabolic syndrome and found that high levels of TG and low levels of HDL-C in patients with metabolic syndrome result from decreased clearance of lipoproteins from circulation. Lipoprotein lipase (LPL) is the major enzyme responsible for clearing TG-containing lipoproteins from circulation and insulin resistance is associated with impaired LPL activity. A low level of HDL-C is an important risk factor for cardiovascular disease (CVD). A similar observation was made in a study conducted on the North Indian population with metabolic syndrome (MetS), where triglycerides (TG) were identified as the most prevalent lipid abnormality. In another study on the Indian population with type 2 diabetes mellitus (T2DM), hypertriglyceridemia and high serum LDL-C levels (≥100 mg/dL) were found to be major components of dyslipidemia with most patients showing mixed dyslipidemia. These findings are consistent with the results of our study.
Beta cells in the pancreas secrete insulin – a signalling molecule that initiates the absorption of glucose from the bloodstream into liver and muscle cells. A reduction in this signalling triggers insulin resistance, which disrupts metabolic actions. This defective metabolic response leads to hyperinsulinemia, contributing to the development of conditions such as obesity, hypertension, diabetes and cardiovascular abnormalities. Insulin resistance (IR) is defined by a reduced ability of insulin to stimulate glucose utilization. The Homeostasis Model Assessment-Insulin Resistance (HOMA-IR) index is used to estimate IR based on glycemia and basal insulin levels. Numerous cross-sectional and longitudinal studies have demonstrated significant changes in insulin sensitivity with aging. HOMA-IR is considered a surrogate marker for estimating insulin resistance based on fasting plasma insulin and glucose levels. The formula used to calculate HOMA-IR is insulin (mUI/L) x blood glucose (mmol/L) / 22.5. In this study, HOMA-IR values greater than 4.65 were used for participants with a body mass index (BMI) above 28.9 kg/m² and values greater than 3.60 for those with a BMI above 27.5 kg/m². Lower HOMA-IR values indicate insulin sensitivity, while higher levels suggest insulin resistance or an increased likelihood of developing diabetes. Esteghamati et al. (2008) found that hypertensive patients without diabetes had significantly higher fasting plasma insulin levels (9.24 vs. 7.86 mIU/L) and higher HOMA-IR (2.16 vs. 1.75) compared to normotensive, non-diabetic patients. In our study, we reported a higher insulin level (7 mIU/L) among participants with metabolic syndrome. The elevated HOMA-IR in our study aligns with the findings of Akande et al., who observed that a HOMA-IR cut-off value greater than 3.8 is indicative of insulin resistance.
Energy balance and glucose metabolism are regulated differently in males and females with gender and biological sex influencing the pathogenesis of insulin resistance and metabolic syndrome. Several researchers have reported that insulin resistance is more prevalent in men than in women, particularly in middle-aged populations. However, in our study, we found slightly higher levels of insulin, HOMA-IR, diabetes screening and NT-proBNP in females compared to males. These higher levels may be attributed to minor errors encountered during sample handling and participant enrolment – which included a wide age range. In addition – our study found significantly higher postprandial blood glucose levels and atherogenic lipid parameters in females than in males (P<0.05). This finding is directly associated with increased cardiovascular risk, oxidative stress and diabetes in individuals with metabolic syndrome.
Our study assessed the cross-sectional relationship between NT-proBNP and conditions related to insulin resistance – such as obesity, diabetes, hypertension and metabolic syndrome. We did not find any significant correlation between NT-proBNP and blood pressure or anthropometric indices – a finding consistent with the studies by Chang et al. and Baldassarre et al., which also lacked relevant associations. However, after stratifying subjects based on symptoms, we performed Pearson correlation analysis and observed an inverse association between NT-proBNP, insulin resistance and glycated haemoglobin (P<0.05). Similarly, Paweł Krzesinski et al. also reported an inverse relationship between NT-proBNP and insulin resistance.
HbA1c and fasting glucose levels measure different aspects of glycemic burden. HbA1c levels change gradually and reflect average glucose levels over 2-3 months, offering a chronic glycemic profile, whereas fasting glucose levels can be affected by acute changes. In our study, we found a significant inverse relationship between NT-proBNP and glycemic burden (P<0.05). In the absence of a diabetes diagnosis, several studies from around the world have reported that HbA1c offers better diagnostic accuracy for metabolic syndrome than fasting glucose levels.
CONCLUSION
The relationship between lower levels of NT-proBNP and metabolic components has significant clinical and pathophysiological implications. Increased waist circumference and body mass index (BMI) appear to be linked to a reduction in NT-proBNP and insulin levels, which may serve as potential predictive markers for metabolic syndrome. Additionally – a more androgenic hormone profile in women could be associated with a greater increase in NT-proBNP levels compared to men and possibly reflects a higher risk of cardiovascular disease (CVD) after menopause. Overall plasma NT-proBNP levels are inversely associated with metabolic syndrome and emphasize their potential role in assessing metabolic risk.
Conflict of Interest: The author has not declared any potential conflict of interest.
Funding Details: None
Acknowledgement: The author gratefully acknowledges the efforts of Saheed Laxman Nayak Medical College and Hospital, Koraput, Odisha, for their support in gathering patient data and details for this study.
REFERENCES
- Rochlani Y, Pothineni NV, Kovelamudi S, Mehta JL. Metabolic syndrome: pathophysiology, management and modulation by natural compounds. Ther Adv Cardiovasc Dis 2017;11(8):215–225.
- Beltran-Sanchez H, Harhay MO, Harhay MM, McElligott S. Prevalence and trends of metabolic syndrome in the adult US population, 1999–2010. J Am Coll Cardiol 2013;62(8):697–703.
- Ruderman NB, Shulman GL. Metabolic syndrome. In: Jameson JL, Groot JD, editors. Endocrinology: adult and pediatric. Elsevier; 2015. p. 752–764.
- Sinha SK, Goel A, Madaan A, Thakur R, Krishna V, Singh K, et al. Prevalence of metabolic syndrome and its clinical and angiographic profile in patients with naive acute coronary syndrome in North Indian population. J Clin Med Res. 2016; 8:667–73.
- Lewis GF, Steiner G. Acute effects of insulin in the control of VLDL production in humans. Implications for the insulin-resistant state. Diabetes Care 1996; 19(4): 390–393.
- Rochlani Y, Pothineni NV, Kovelamudi S, Mehta JL. Metabolic syndrome: pathophysiology, management and modulation by natural compounds. Ther Adv Cardiovasc Dis. 2017 Aug;11(8):215-225.
- Boden G, Shulman GI. Free fatty acids in obesity and type 2 diabetes: defining their role in the development of insulin resistance and beta-cell dysfunction. Eur J Clin Invest 2002; 32(Suppl. 3): 14–23.
- National Cholesterol Education Program (NCEP): Expert Panel on Detection and Treatment of High Blood Cholesterol in Adults (2002). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 106, 3143–342.
- Baldassarre S, Fragapani S, Panero A, Fedele D, Pinach S, Lucchiari M, Vitale AR, Mengozzi G, Gruden G, Bruno G. NTproBNP in insulin-resistance mediated conditions: overweight/obesity, metabolic syndrome and diabetes. The population-based Casale Monferrato Study. Cardiovasc Diabetol. 2017 Sep 25;16(1):119.
- Alberti KGMM, Zimmet P, Shaw J: Metabolic syndrome: A new world-wide definition. A consensus statement from the International Diabetes Federation. Diabet Med 2006; 23:469–80. Wallace TM, Matthews DR: The assessment of insulin resistance in man. Diabet Med 2002; 19:527–34.
- Singh O, Gupta M, Khajuria V. Lipid profile and its relationship with blood glucose levels in Metabolic Syndrome. Natl J Physiol Pharm Pharmacol 2015;5:134-137.
- Lent-Schochet D., McLaughlin M., Ramakrishnan N., Jialal I. Exploratory Metabolomics of Metabolic Syndrome: A Status Report. World J. Diabetes. 2019; 10:23–36.
- Scott CL: Diagnosis, prevention and intervention for the metabolic syndrome. Am J Cardiol 2003; 92:35–42.
- Hall J.E., Mouton A.J., da Silva A.A., Wang Z., Li X., do Carmo J.M. Obesity, Kidney Dysfunction and Inflammation: Interactions in Hypertension. Cardiovasc. Res. 2021; 117:1859–1876.
- Avramoglu RK, Basciano H, Adeli K: Lipid and lipoprotein dysregulation in insulin resistant states. Clinica Chimica Acta 2006; 368:1–19.
- Deedwania P, Barter P, Carmena R, Fruchart JC, Grundy SM, Haffner S, Kastelein JJ, LaRosa JC, Schachner H, Shepherd J, Waters DD, Treating to New Targets Investigators: Reduction of low-density lipoprotein cholesterol in patients with coronary heart disease and metabolic syndrome: Analysis of the Treating to New Targets study. Lancet 2006; 368:919–28.
- Maheux P, Azhar S, Kern PA, Chen YD, Reuven GM. Relationship between insulin-mediated glucose disposal and regulation of plasma and adipose tissue lipoprotein lipase. Diabetologia. 1997; 40:850–8.
- Assmann G, Gotto AM Jr. HDL Cholesterol and protective factors in atherosclerosis. Circulation. 2004;109: III-8–III-14.
- Pandya H, Lakhani JD, Dadhania J, Trivedi A. The prevalence and pattern of dyslipidemia among type 2 diabetic patients at rural based hospital in Gujarat, India. Indian J Clin Pract. 2012;22(12):36–44.
- Wilcox G. Insulin and insulin resistance. Clin Biochem Rev. 2005;26(2):19.
- Vasques AC, Rosado LH, Alfenas RC, Geleneze B. Critical analysis on the use of the homeostasis model assessment (HOMA) indexes in the evaluation of the insulin resistance and the pancreatic beta cells functional capacity. Arq Bras Endocrinol Metab. 2008;52(1):32-9.
- George L, Bacha F, Lee S, Tfayli H, Andreatta E, Arslanian S. Surrogate estimates of insulin sensitivity in obese youth along the spectrum of glucose tolerance from normal to prediabetes to diabetes. J Clin Endocrinol Metab. 2011;96:2136–2145.
- Stern SE, Williams K, Ferrannini E, DeFronzo RA, Bogardus C, Stern MP. Identification of individuals with insulin resistance using routine clinical measurements. Diabetes. 2005;54(2):333-9. PMID: 15677489.
- Esteghamati A, Khalilzadeh O, Abbasi M, Nakhjavani M, Novin L, Esteghamati AR. HOMA-estimated insulin resistance is associated with hypertension in Iranian diabetic and non-diabetic subjects. Clin Exp Hypertens. 2008; 30:297–307.
- Akande TO, Adeleye JO, Kadiri S. Insulin resistance in Nigerians with essential hypertension. Afr Health Sci. 2013;13:655–60.
- Lentner M, Harreiter J. Sex differences in type 2 diabetes. Diabetologia. 2023;66:986–1002.
- S. Baldassarre, S. Fragapani, A. Panero et al., “NTproBNP in insulin-resistance mediated conditions: overweight/obesity, metabolic syndrome and diabetes. The population-based Casale Monferrato Study,” Cardiovascular Diabetology, vol. 16, no. 1, p. 119, 2017.
- H.-R. Chang, J.-C. Hsieh, M. Y.-C. Chen et al., “N-terminal pro- B-type natriuretic peptide is inversely associated with metabolic syndrome in hypertensive patients,” The American Journal of the Medical Sciences, vol. 348, no. 3, pp. 210–214, 2014.
- Paweł Krzesinski, Wiesław Piechota, Katarzyna Piotrowicz, Grzegorz Gielerak, Agnieszka Wozniak-Kosek. Association of Estimated Insulin Resistance with N-Terminal B-Type Natriuretic Peptide Concentration in Men with Metabolic Syndrome. Cardiology Research and Practice. Volume 2019, Article ID 8571795, 6 pages.
- Bonora, E.; Tuomilehto, J. The pros and cons of diagnosing diabetes with A1C. Diabetes Care 2011, 34 (Suppl. 2), S184–S190.

