Nutritional Herbs for Post-Menopausal Colon Cancer Therapy
NUTRITIONAL HERBS AS TESTABLE THERAPEUTIC ALTERNATIVES FOR POST-MENOPAUSAL COLON CANCER
Nitin T. Telang
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Telang, N.T., 2026. NUTRITIONAL HERBS AS TESTABLE THERAPEUTIC ALTERNATIVES FOR POST- MENOPAUSAL COLON CANCER. Medical Research Archives, [online]14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution license, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
Abstract
Background: Chemotherapeutic options for management of post-menopausal colon cancer are associated with systemic toxicity, therapy resistance and survival of cancer stem cells. These aspects promote progression of metastatic disease. In post-menopausal colon cancer estrogens are hypothesized to play a protective role, and phytoestrogenic nutritional herbs may provide valuable mechanistic leads for growth inhibition of cancer. Investigations using cellular models relevant to this cancer subtype provide mechanistic evidence for carcinogenesis, role of estrogens in post-menopausal colon cancer and identify susceptible molecular targets.
Objectives: This mini-review provides a systematic discussion of published evidence on i) experimental systems developed from colon carcinoma and colonic epithelium that facilitate identification of mechanistic link between Wnt/β-catenin signaling and estrogen receptor signaling, ii) inhibitory efficacy of natural products such as dietary phytochemicals and Chinese nutritional herbs and iii) Conceptual and technical limitations that define rationale for future research.
Conclusions: Molecular characterization and mechanistic leads have validated the experimental systems to investigate growth inhibitory efficacy of natural products. Major limitations provide rationale for future research that facilitate clinical relevance and translatability of the data.
Keywords: Colon cancer, Wnt/β-catenin pathway, Estrogen receptor-β, nutritional herbs
Introduction
Progression to advanced stage metastatic colon cancer remains a major cause of mortality. Although the incidence of colon cancer in female population is projected to be lower than that in the male population. In the female population the American Cancer Society has projected the incidence of newly diagnosed colorectal cancer has been projected as 71,810 and colorectal cancer related deaths has been projected as 24,000 in 2026. Epidemiological evidence from oral contraception usage and hormone replacement therapy have been considered as possible protective factors in post-menopausal colon cancer.
17β-estradiol (E2) mediated estrogen receptor (ER) signaling via ER-α and ER-β isoforms are responsible for genomic effects in receptor positive target cells. These isoforms exhibit functional antagonism via positive and negative growth regulation, respectively. Binding of estradiol to ER-α activates anti-apoptotic pathways, while that to ER-β activates pro-apoptotic caspase-3 and induction of cellular apoptosis. Compared to normal colonic epithelium decreased expression of ER-β has been reported in sporadic colon cancer, as well as in familial adenomatous polyps. Thus, status of ER-β expression represents prognostic marker of colorectal cancer progression. Protective role of E2 functioning as ER-β ligand, has been hypothesized in post-menopausal colon cancer.
The Wnt/β-catenin signaling pathway, via expression of adenomatous polyposis coli (Apc) tumor suppressor gene, plays an essential role in normal cellular homeostasis in colonic crypts by regulating proliferation, differentiation and apoptosis. Germline or somatic mutation in Apc produces truncated gene product that lacks β-catenin, axin and microtubule binding sites. This structural alteration deregulates the signaling pathway, facilitates nuclear translocation of β-catenin and upregulates the expression of Apc target genes cylin D1 and c-Myc. Loss-of-function germline mutation of Apc in familial adenomatous polyposis (FAP) patients represents a primary genetic defect facilitating initiation of colon cancer development. Somatic mutation in Apc has been detected in majority of sporadic colon cancers. The Wnt/β-catenin signaling pathway in general, and Apc expression in particular represent potential therapeutic targets for colorectal carcinogenesis including chemo-resistant cancer initiating colorectal cancer stem cells.
The role of ER isoforms α and β in Apc-dependent colon carcinogenesis has been investigated in Min/+ mice crossed with ER-α[+/-] and ER-β[+/-] mice. These mice exhibited increased incidence of colon tumors and micro-adenomas. The ER-α[+/-] Min/+ mice exhibited invasive carcinomas. The ER isomers α and β exhibit a distinct opposing gradient of expression along the colonic crypt-villus axis. This distribution is suggestive of functional antagonism between the two ER receptors. This preclinical evidence supports the clinical concept that the loss of estradiol and ER signaling in post-menopausal women may contribute to colorectal cancer development. Collectively, published evidence on the role of Wnt/β-catenin signaling and estrogen receptor signaling pathways in postmenopausal colon carcinogenesis provides a strong mechanistic support to clinical and preclinical data.
Conventional chemotherapy for colorectal cancer includes the use of the DNA synthesis inhibitor 5-Fluoro-uracil, DNA strand intercalating agent oxaliplatin and DNA topoisomerase inhibitor irinotecan. Targeted therapy includes the use of non-steroidal anti-inflammatory drugs and selective cyclooxyganase-2 (COX-2) inhibitors. The preclinical and clinical inhibitory efficacy of anti-inflammatory drugs has been documented. COX-2 represents a rate limiting enzyme for prostaglandin biosynthesis via conversion of polyunsaturated fatty acid arachidonic acid to prostaglandin E2 (PGE2). Upregulated expression of COX-2 is documented during colon carcinogenesis, and COX-2 inhibition represents a major molecular target for anti-inflammatory drugs.
The use of pharmacological agents for preventive/therapeutic effects on colon cancer is associated with systemic toxicity, therapy resistance and emergence of chemo-resistant cancer initiating stem cell population. Collectively, these limitations promote cancer progression and compromise patient compliance. Natural products such as Chinese nutritional herbs and dietary phytochemicals have documented human consumption and exhibit preclinical efficacy. Relatively nontoxic natural bioactive agents including polyphenols, flavonoids, and terpernoids, or constitutive bioactive agents from nutritional herbs may represent testable therapeutic alternatives for colon cancer. Present mini-review provides a systematic discussion of published evidence relevant to i) cellular systems developed from human colon carcinoma models and from genetically predisposed mouse models, providing a mechanistic link for the role of estrogen receptor signaling and Wnt/β-catenin signaling in colon cancer, ii) growth inhibitory efficacy of dietary phytochemicals and Chinese nutritional herbs and iii) conceptual and technical limitations in experimental strategies that define rationale for future investigations.
Cellular models
HUMAN COLON CARCINOMA-DERIVED CELL LINES:
These cellular models are developed from sporadic colon adenocarcinoma detected in male patients. Several publications have used these models to investigate the effect of E2 on status of ER-β expression. Mechanistic leads for growth inhibitory effects include inhibition of cell proliferation, inhibition of G1 to S phase transition, downregulation of cyclin E, and c-Myc and upregulation of CDKI p21cip1/waf1 and p27Kip1. Genotypic characterization and mechanism of action relevant to the role of ER-β is summarized in Table 1.
| Cell Line | Gene Expression | Mechanism of Action |
|---|---|---|
| T84 | APC MT, KRAS, PI3KCA TP 53 MT | ER-β growth inhibition. APC target gene expression. RAS signaling. TP53 signaling. |
| SW1116 | APC MT, KRAS, TP53 MT | ER-β growth inhibition. G1 to S inhibition. COX-2, AKT, ERK1/2 expression. APC target gene expression. KRAS signaling. TP53 signaling. |
| LoVo | APC, KRAS, MSH2 | ER-β. Growth inhibition. KRAS signaling. DNA mismatch repair. |
| SW480 | APC MT, β-catenin WT | APC target gene c-MYC inhibition. CDKI upregulation. |
| SW640 | APC MT, β-catenin WT | ER-β growth inhibition. Cyclin D1, c-MYC inhibition. CDKI upregulation. |
| HCT116 | APC WT, β-catenin MT | ER-β growth inhibition. β-catenin expression. |
COLONIC EPITHELIAL CELL LINES FOR GENETICALLY PREDISPOSED COLON CANCER:
Mono-allelic mutation In the Apc gene is responsible for primary genetic defect in FAP syndrome, functioning as a high risk precursor to colon cancer. Apc mutation deregulates Wnt/β-catenin signaling pathway and upregulates the expression of Apc target genes cyclin D1 and c-Myc in hyper-proliferative cancer phenotype. The cell lines for FAP include 1638N COL and 850MIN COL. These cell lines exhibit hyper-proliferation, downregulated cellular apoptosis, anchorage independent growth in vitro and tumor formation in vivo.
Germline mutations in Mlh1 gene and Apc 1638 N gene provide a mouse model for HNPCC. The cell line Mlh1/1638N COL developed from the animal model for HNPCC exhibits defective expression of Apc and DNA mismatch repair gene Mlh1, hyper-proliferation, downregulated cellular apoptosis, anchorage independent growth in vitro and tumor formation in vivo. The expression status of DNA mismatch repair gene MSH2 in human colon carcinoma derived LoVo cells, and of Mlh1 gene in Mlh1/1638N COL cells, provides a rationale to investigate the role of DNA mismatch repair in post-menopausal colon cancer. The cellular and molecular characteristics of mouse colonic epithelial cell lines are summarized in Table 2.
| Cell Line | Genotype | Origin | Mechanism of Action |
|---|---|---|---|
| C57 COL | Apc+/+ | Mouse colon. Non-tumorigenic | Colonic epithelial cellular homeostasis |
| 850MIN COL | Apc +/- | Apc+/- mouse colon. Tumorigenic FAP model | Apc target gene expression. |
| 1638N COL | Apc +/- | Apc+/- mouse colon. Tumorigenic. FAP model. | Apc target gene expression |
| Mlh1/1668N COL | Apc +/- | Mlh1/1638N mouse colon. Tumorigenic. HNPCC model. | Apc target gene expression. DNA mismatch repair inhibition. |
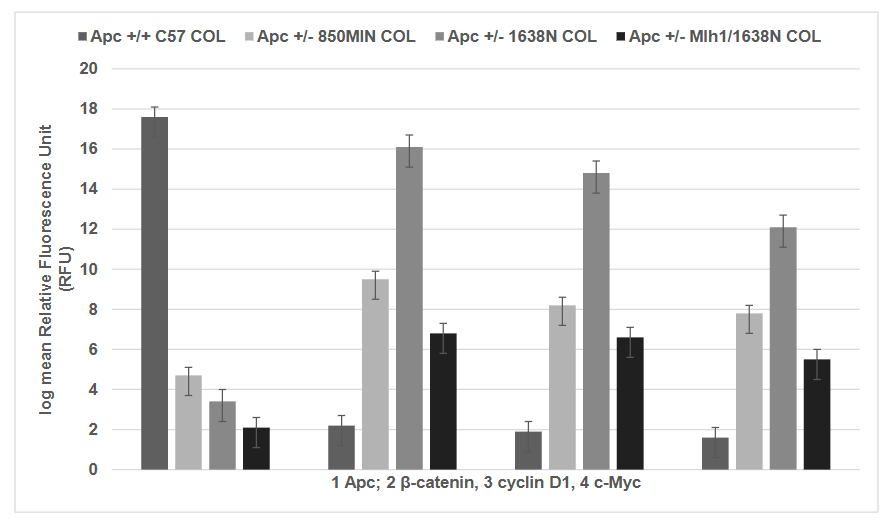
It is notable that compared to the Apc +/+ C57COL cells all the Apc +/- cell lines, 850MIN COL, 1638 N COL and Mlh1/1638N COL cell lines exhibit decreased expression of Apc and increased expression of Apc target genes β-catenin, cyclin D1 and c-Myc. Increased expression of these genes is predominantly due to the loss-of function mutation in Apc producing truncated gene product that exhibits loss of β-catenin binding sites, axin binding sites and microtubule binding sites. The altered gene product compromises the normal regulatory function of Apc. The expression of Apc and its target gene products (protein) are examined using a quantitative immuno-fluorescence assay. This assay monitors the cellular uptake of fluorescently labelled antibodies. The data are quantified by fluorescent units relative to the isotype control. The expression of Apc in 850 MIN COL, 1638N COL and Mlh1/1638N COL cells exhibited a reduction by about 88 to 89 %, relative to C57 COL cells. In contrast, the expression of β-catenin in the three Apc mutant cell lines was increased by about 40% to 80%, cyclin D1 was increased by about 3.0X to 4.3X, and c-Myc was increased by about 1.5x to 2.5x, respectively, relative to the Apc wild type C57 COL cells.
GROWTH INHIBITORY EFFICACY OF NATURAL PRODUCTS:
Human colon carcinoma derived cellular models have been utilized to investigate the role of ER-β functioning as a potent negative growth regulator. At the mechanistic levels, ER-β inhibits G1-S phase transition, downregulates expression of cyclin E, and c-Myc and upregulated the expression of cyclin dependent kinase inhibitors p21CIP1/waf1, p27Kip1. Predominantly documented function of Chinese nutritional herbs includes anti-inflammatory activity and phytoestrogenic activity. Selection of appropriate nutritional herbs, source of extract for investigation and mechanism of action are summarized in Table 3.
| Herb | Source of Extract | Mechanism of Action |
|---|---|---|
| Coptis sinensis (CS) | Root | Anti-inflammatory |
| Magnolia officinalis (MO) | Root | Anti-inflammatory. Anti-proliferative. Pro-apoptitic |
| Panax ginseng (PG) | Root | Anti-inflammatory. Anti-proliferative. Phyto-estrogenic |
| Radix paeoniae (RP) | Root | Anti-inflammatory. Anti-proliferative. Pro-apoptotic. Phyto-estrgenic. |
| Radix scutellariae (RS) | Bark | Anti-proliferative. Pro-apoptotic |
| Radix salvia miltorrhizae (RSM) | Root | Anti-proliferative. Pro-apoptotic. Phyto-estrogenic. |
In this context, It also notable that synthetic industrial chemicals and naturally occurring phytoestrogens compete with 17β-estradiol (E2) for binding to ER-β, and estradiol inhibits PGE2-induced COX-2 expression via suppression of the AKT and ERK ½ signaling pathways. Collectively, these lines of evidence provide a rationale to investigate agonistic effects of nutritional herbs or constituent bioactive agents on ER-β in cellular models FAP. Preliminary evidence has demonstrated anti-proliferative, cell cycle inhibitory and pro-apoptotic effects of nutritional herbs. Preliminary evidence has demonstrated that in the 850MIN COL model selective COX-2 inhibitor CLX inhibits COX-2, and reduces the expression of select Apc target genes. This evidence suggests that CLX may represent a valuable positive control for investigations with anti-inflammatory nutritional herbs or constituent bioactive agents.
STEM CELL MODELS:
Resistance to conventional/targeted chemotherapy presents a formidable challenge and emphasizes investigations on molecular mechanisms for drug resistance and on targeting of chemo-resistant cancer initiating stem cell population. Phenotypic resistance to pharmacological chemotherapeutic agents selects putative stem cells. These cellular models are characterized for the expression of stem cell specific markers including expression of CD44, CD133 and c-Myc. These drug resistant stem cell models may facilitate investigations on stem cell targeting efficacy of natural products.
Conclusion
Systematic discussion of published evidence, validation of cellular models for genetically predisposed FAP and HNPCC syndromes and mechanism-based growth inhibitory efficacy of natural products provide scientifically robust rationale for future research. Published evidence also identifies several unmet needs and unanswered questions. Major limitations include lack of clinically relevant models from post-menopausal patients and mechanistic leads for the role of ER signaling in growth inhibitory efficacy of nutritional herbs in post-menopausal colon cancer. Intrinsic limitations of cell line derived preclinical data include extrapolation for clinical relevance. Patient derived tumor explants and organoids from female patients reduce the need for extrapolation by facilitating clinically translatable mechanistic data. Additionally, efficacious bioactive agents from nutritional herbs may identify new drug candidates for post-menopausal colon cancer.
Conflict of interest statement:
The author has no conflicts of interest to declare.
Funding statement:
The current study has not been funded by extramural grants.
Acknowledgements:
The author has conceptualized the subject matter, reviewed published data and has approved the final version of the manuscript.
References:
- American Cancer Society Facts and Figures 2025. American Cancer Society. Atlanta, GA.
- Campbell-Thompson M, Lynch IJ, Bharadwaj B: Expression of estrogen receptor (ER) subtypes and ER-β isoforms in colon cancer. Cancer Res. 2001, 61: 632-640. PMID: 11212261.
- Ditonno I, Losurdo G, Rendina M, Pricci M, Girardi B, Lerardi E, Di Leo A. Estrogen receptors in colorectal cancer: Facts, Novelties and perspectives. Curr. Oncol. 2021, 28: 4256-4263. DOI: 10.3390/curoncol28060361.
- Filho PRS, Junior SA, Begnami MD, et al: Oestrogen receptor beta isoform expression in sporadic colorectal cancer, familial adenomatous polyposis and progressive stages of colorectal cancer. BMC Cancer 17: 754. DOI: 10.1186/s12885-017-3688-4.
- Filho PRS, Junior SA, Begnami MD, et al: Estrogen receptor β as a prognostic marker of tumor progression in colorectal cancer with familial adenomatous polyposis and sporadic polyps. Pathol. Oncol. Res. 2018, 24: 533-540. DOI: 10.1007/s12253-017-0268-5.
- Ditonno I, Novielli D, Celiberto F, et al: Molecular pathways of carcinogenesis in familial adenomatous polyposis. Int. J. Mol. Sci. 2023, 24: 5687. DOI: 10.3390/ijms24065687.
- Fearon ER, Vogelstein B: A genetic model for colorectal tumorigenesis. Cell 1990, 61: 759-76 DOI: 10.1016/0092-8674(90)90186-i.
- Fodde R, Smits R, Clevers H: APC, signal transduction and genetic instability in colon cancer. Nat. Rev. Cancer 2001, 1: 55-67. DOI: 10.1038/35094067.
- Anastas JN, Moon RT: Wnt signaling pathways as therapeutic targets in cancer. Nat. Rev. Cancer 2013, 13: 11-26. DOI: 10.1038/nrc3419.
- Bian J, Dannappel M, Wan C, et al: Transcriptional regulation of Wnt/β-catenin pathway in colorectal cancer. Cells 2020, 9: 2125. DOI: 10.3390/cells9092125.
- Shi G, Li Y, Shen H, et al: Intestinal stem cells in intestinal homeostasis and colorectal tumorigenesis. Life Med. 2024, 3: Inae042. DOI:10.1093/lifemedi/Inae042.
- Cho NL, Javid SH, Carothers AM, et al: Estrogen receptors alpha and beta are inhibitory modifiers of Apc-dependent tumorigenesis in the proximal colon of Min/+ mice. Cancer Res. 2007, 67: 2366-2372. DOI: 10.1158/0008-5472.CAN-06-3026.
- National Comprehensive Cancer network. Clinical practice guidelines in oncology: Colon cancer. 2025. www.http://nccn.org.
- Beazer-Barkley Y, Levy DB, Moser AR, et al: Sulindac suppresses tumorigenesis in the min mouse. Carcinogenesis 1996, 17: 1757-1760. DOI: 10.1093/carcin/17.8.1757.
- Boolbol SK, Dannenberg AJ, Chadburn A, et al: Cyclooxygenase-2 overexpression and tumor formation are blocked by sulindac in a murine model of familial adenomatous polyposis. Cancer Res. 1996, 56: 2556-2560. PMID: 8653697.
- Jacoby RF, Seibert K, Cole CE, et al: The cyclooxygenase-2 inhibitor celecoxib is a potent preventive and therapeutic agent in the min mouse model of adenomatous polyposis. Cancer Res. 60: 5040-5044. PMID: 11016626.
- Giardiello FM, Hamilton SR, Krush AL, et al: Treatment of colonic and rectal adenomas with sulindac in familial adenomatous polyposis. N. Engl. J. Med. 1993, 328: 1313-1316. DOI: 10.1056/NEJM199305063281805.
- Steinbach G, Jynch PM, Phillips RK, et al: The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N. Engl. J. Med. 2000, 342: 1946-1952. DOI: 10.1056/NEJM20000629.
- Hong M, Tan HY, Li S, et al: Cancer stem cells: the potential targets of Chinese medicines and their active compounds. Int. J. Mol. Sci. 2016, 17: 893.
- Aiello P, Sharghi M, Mansourkhani SM, et al: Medicinal plants in the prevention and treatment of colon cancer. Oxidat. Med. Cell Longev. 2019, 2019, 2075614. DOI: 10.1155/2019/2075614.
- Manogaran P, Umapathy D, Karthikeyan M, et al: Dietary phytochemicals as a potential source for targeting cancer stem cells. Cancer Investig. 2021, 39: 349-368. DOI: 10.1080/07357907.2021.1894569.
- Naujokat C, Mc Kee DL: The big five phytochemicals targeting cancer stem cells: Curcumin, EGCG, sulforaphane, resveratrol and genistein. Cur. Med. Chem. 2021, 28: 4321-4342. DOI: 10.2174/092986732766620028110738.
- Kuiper GG, Lemmen JG, Carisson B, et al: Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998, 139: 4252-4263, DOI: 10.1210/endo.139.10.6216.
- Lai T-Y, Chen L-M, Lin J-Y et al: 17beta-estradiol inhibits prostaglandin E2-induced COX-2 expression and cell migration by suppressing Akt and ERK1/2 signaling pathways in human LoVo colon cancer cells. Mol Cell Biochem. 2010, 342: 63-70. DOI: 10.1007/11010-010-0469-7.
- Martineti V, Picariello L, Tognarini I, et al: ER beta is a potent inhibitor of cell proliferation in the HCT8 human colon cancer cell line through regulation of cell cycle components. Endocr. Relat. Cancer 2005, 12: 455-469. DOI:10.1677/erc.1.00861.
- Hartman J, Edvardsson K, Lindberg K, et al: Tumor repressive functions of estrogen receptor beta in SW480 colon cancer cells. Cancer res. 2009, 69: 6100-6106. DOI: 10.1158/0008-5472.CAN-09-0506.
- Su LK, Kinzler KW, Vogelstein B, et al: Multiple intestinal neoplasia caused by a mutation in the murine homolog of the APC gene. Science 1992, 256 (5057): 668-670. DOI: 10.1126/science.1350108.
- Fodde R, Edelmann W, Yang K, et al: A targeted chain termination mutation in the mouse Apc gene results in multiple intestinal tumors. Proc. Natl. Acad. Sci. USA 1994, 91: 8969-8973. DOI: 10.1073/pnas.91.19.8969.
- Telang N, Katdare M: Novel cell culture model for prevention of carcinogenic risk in familial adenomatous polyposis syndrome. Oncol. Rep. 2009, 21: 1017-1021. DOI: 10.3892/or_00000318.
- Telang N: Anti-inflammatory drug resistance selects putative cancer stem cells in a cellular model for genetically predisposed colon cancer. Oncol. Letts. 2018, 15: 542-648. DOI: 10.3892/ol.2017.7147.
- Telang N: Drug resistant stem cells: Novel approach for colon cancer therapy. Int. J. Mol. Sci. 2022, 23: 2519. DOI: 10.3390/ijms23052519.
- Edelmann W, Yang K, Karaguchi M, et al: Tumorigenesis in Mlh1/1638N mutant mice. Cancer Res. 1999, 59: 1301-1307. PMID: 10096563.
- Telang N: Isolation and characterization of chemo-resistant stem cells from a mouse model for hereditary non-polyposis colon cancer. Stem Cells and Cloning: Advances and Applications. 2021, 14: 19-25. DOI: 10.2147/SCCAA.S312929.
- Fiorelli G, Picanello L, Martineti V, et al: Functional estrogen receptor beta in colon cancer cells. Biochem. Biophys. Res. Commun. 1999, 261: 521-527. DOI: 10.1006/bbrc.1999.1062.

