Obstructive Sleep Apnea in Children with Down Syndrome
Obstructive Sleep Apnea in Children and Adolescents with Down Syndrome: A Clinical Study
Lorena Luana Batista Favaro1, Marllon Marques Ferreira da Cunha2, Pedro Nassar dos Santos Costa1, Lídia Raquel de Carvalho2, Débora Avellaneda Penatti1, Silke Anna Theresa Weber3, Cátia Regina Branco da Fonseca1
- São Paulo State University, Botucatu Medical School, Department of Pediatrics, Botucatu, São Paulo State, Brazil
- São Paulo State University, Biosciences Institute, Botucatu, São Paulo State, Brazil
- São Paulo State University, Botucatu Medical School, Otolaryngology, Botucatu, São Paulo State, Brazil
OPEN ACCESS
PUBLISHED 28 February 2025
CITATION Batista Favaro, LL., da Cunha, MMF., et al., 2025. Obstructive Sleep Apnea in Children and Adolescents with Down Syndrome: A Clinical Study. Medical Research Archives, [online] 13(2). https://doi.org/10.18103/mra.v13i2.6336
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6336
ISSN 2375-1924
Abstract
Objectives: To assess the prevalence of obstructive sleep apnea in children and adolescents with Down syndrome followed up in an outpatient clinic in a university hospital and to identify which factors are related to the disease and its severity.
Methods: A total of 35 patients with Down syndrome were included in this cross-sectional study after being invited to undergo polysomnography and applying structured and standardized questionnaires, between March 2021 and June 2022, with data description by mean ± standard deviation for continuous variables and frequency and percentage in categorical, being considered significant p ≤ 0.05.
Results: The selected sample consists of 57.1% girls, with mean age and body mass index z-score, respectively, 87.5 months (± 45.5) and 0.6 (± 1.3). Obstructive sleep apnea was diagnosed in 96.3% of cases, with a mean apnea and hypopnea rate of 11.3 events per hour (± 12.5) and moderate or severe disease in more than half of the population (59.2%). The severity of the condition was significantly associated with parental report of habitual snoring (p 0.002) and excessive daytime sleepiness according to the modified Epworth scale (p 0.001). Patients diagnosed with obstructive sleep apnea were referred for otorhinolaryngological follow-up.
Conclusion: The results show the high prevalence of obstructive sleep apnea in this population, as well as the association between report of snoring and daytime sleepiness with the severity of the disease, which demonstrates the applicability of screening tools and reinforces the importance diagnosis to improve the follow-up of these patients.
Keywords
Down syndrome; sleep disorders; obstructive sleep apnea; pediatrics.
1. Introduction
Down syndrome (DS) is the most common chromosomal abnormality in the population with an estimated incidence of 1 in 700 live births. Children who have the condition are predisposed to have several comorbidities that interfere with their growth and development. The set of orofacial alterations presented by this population is associated with a high risk for the development of obstructive sleep apnea (OSA), a pathology characterized by partial or complete obstruction of the upper airways, with potentially deleterious effects on the health of these patients.
The Down syndrome is a genetic condition characterized by an excess of chromosome 21 in the individual’s genetic information, so instead of having 46 chromosomes per cell, there are 47 chromosomes. This additional chromosome determines changes in the development in addition to physical and mental alterations.
There are three types of DS differentiated through karyotype examination, being: 1) free trisomy of chromosome 21 (occurs in 92% to 95% of cases where there is an extra chromosome 21, resulting from an abnormal division of germ cells); 2) translocation (occurs in 3% to 5% of cases). It is a rearrangement in which a fragment of chromosome 21 is translocated to other acrocentric chromosomes, the most common being between chromosomes 14 and 21 (it can also occur between 15 and 21, and 22 and 21). In this case, there is a structural disorder or partial trisomy, in which the individual has 46 chromosomes, one of which is normal chromosome 14 and another structurally abnormal one with the translocated addition of the long arm of chromosome 21; 3) chromosomal mosaicism (occurs in 1 to 2% of cases), presenting some normal cells and others with trisomy 21, resulting from the abnormal cell division after fertilization. These individuals usually have fewer physical anomalies and higher intellectual capacity than those with free trisomy of chromosome 21, possibly due to the presence of some normal cells.
Children with DS are commonly diagnosed with OSA, with an estimated prevalence of 30 to 60% compared to 2% in the general pediatric population. Recent studies show that more than half of the children and adolescents with this chromosomal alteration have OSA documented by full-night polysomnographic examination. However, despite the high prevalence of OSA in patients with DS being well established in the literature, this association still remains underreported. Studies show that parents and caregivers underestimate the severity of sleep disorders in children and adolescents with DS.
Obstructive sleep apnea is a complex condition associated with multiple risk factors for intermittent upper airway obstruction in those with DS. Well-defined predisposing conditions are mandibular and midface hypoplasia, narrow nasopharynx, relative macroglossia, adenotonsillar hypertrophy, and high-arched palate, in addition to generalized hypotonia and obesity. These patients also have neurobehavioral complications, aggravation of cardiovascular comorbidities with high rates of heart disease and systemic arterial hypertension, and an increase in the incidence of respiratory tract infections resulting from intermittent upper airway obstruction. It is therefore essential to study the treatable health issues associated with OSA, what can optimize the quality of life for both DS patients and their caregivers. The therapeutic approach has demonstrated positive effects on neurocognitive functions, behavioral symptoms, and the individual’s overall well-being. The institution of a protocol becomes fundamental for a highly accurate screening, accelerating the objective diagnosis of this condition and the establishment of therapeutic indications.
The American Academy of Pediatrics recommends that all children with DS should have an overnight polysomnographic study at the age of 4 years, as significant upper airway obstruction may be present despite a lack of indications identified in the clinical history and physical examination. However, polysomnography is an exam that is difficult to access, and its performance can be critical in young children. Therefore, it is necessary to develop an alternative screening tool for the therapeutic definition and follow-up of this population.
Despite the resources currently available for studies of OSA in DS, few have evaluated diagnostic criteria and the impact of associated comorbidities and treatment strategies. Early recognition and the establishment of effective therapeutic measures are essential to improve the living conditions of this population. Thus, the purpose of the present study is to determine the prevalence of OSA in a sample of children and adolescents with DS as an outpatient in a University hospital. The association of the demographic factors and symptoms presented by the patients and the OSA severity were analyzed, in addition to the impact on the quality of life of the children, aiming at the construction of a local institutional protocol for diagnosis, management, and follow-up.
2. Material And Methods
In this single-center cross-sectional study, we invited all 87 patients, aged between 2 to 15 years old, at follow-up at the Pediatric Genetics and Adolescent Medicine outpatient clinics, and who had a confirmed diagnosis of DS by karyotype examination. A sample of 35 children agreed to participate, 34 of which were due to free trisomy of chromosome 21 and one due to mosaicism. As the clinical manifestations of the DS were similar, they were all analyzed as a single group.
The study protocol was applied to all children, independent of respiratory complaints. Patients were summoned by phone or at outpatient clinics to undergo full-night polysomnography (PSG) at the Sleep Laboratory. between March 2021 and June 2022. Standardized questionnaires were applied to identify signs and symptoms associated with obstructive respiratory disorders, as well as to investigate associated conditions on the day of the PSG.
The diagnosis of OSA was established by the apnea and hypopnea index (AHI) with the number of apneas and hypopneas > 1 event per hour (e/h) and classified as mild – AHI 1 to 5 e/h, moderate – AHI 5.1 at 10 e/h and severe – AHI > 10 e/h. Apnea is defined as obstructive (OA) when no airflow is observed with the maintenance of chest wall movements at the PSG for a period of at least 2 breaths duration, central (CA) when no nasal flow and chest movements are identified for more than 20 seconds, or more than a 2-breath-period if accompanied by desaturation or awakening; and mixed (MA) when characterized by obstructive and central components. Hypopneas correspond to a reduction in airflow by at least 50% during the period of at least two breaths with a corresponding drop in oxygen saturation by 3% and/or awakening. Scoring of the events, and definitions of diagnosis and severity are in agreement with the standard classification.
There was no exclusion criterion, but polysomnographic examinations were prioritized. The 35 participants included were divided into 3 groups defined by age to reduce sample heterogeneity: group 1 – 1 to 4 years old, group 2 – 5 to 11 years old, and group 3 – ≥ 12 years old. For the final analysis, all children undergoing polysomnography and data collection were included in this study, regardless of their history of upper airway surgery or the use of any drug therapy.
2.1. DATA COLLECTION
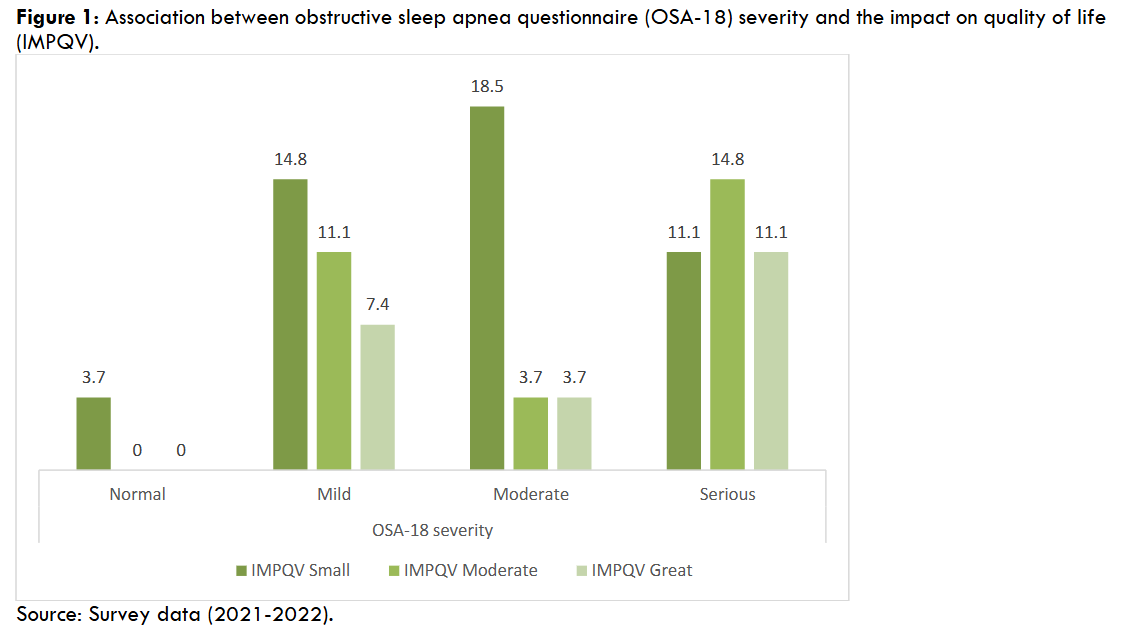
The research was carried out through the application of standardized questionnaires: 1.modified Epworth scale, composed of 8 questions that indicate excessive daytime sleepiness when scored > 10 points; 2. Pediatric Sleep Questionnaire (PSQ), consisting of 22 questions suggesting the presence of sleep breathing disorder when scored > 7; 3. the Obstructive Sleep Apnea questionnaire in children (OSA-18), consisting of 18 questions that assess the impact of OSA on the quality of life of the patients and their caregivers stratified into small (< 60), moderate (60 – 80) and large impact (> 80) (Supplementary files A.1, A.2 and A.3), and a structured qualitative interview (Appendix B). Demographic and clinical data, sociodemographic characteristics, and anthropometric measures were obtained from electronic medical charts. The questionnaires were applied by four previously trained participants, to standardize the instruments, and to reduce divergences and failures in the acquisition of information.
Tonsillar hypertrophy was considered through clinical analysis using the Brodsky scale to assess the size of the palatine tonsil; patients who presented more than 50% airway obstruction by the palatine tonsil (grades III and IV) were considered to have tonsillar hypertrophy. Hypertrophy of the adenoids were assessed by the otolaryngologist by nasofibroscopy or X-ray at a later moment. All data collection was performed by the pediatric team, except the polysomnographic exam. All children with a positive PSG were sent to the otolaryngologic evaluation at a later moment.
2.2. STATISTICAL ANALYSIS
Data were analyzed using SAS statistical software version 9.2 and statistical analyzes performed with chi-square test, analysis of variance, Tukey’s test and Fisher’s Exact test. The significance level of 0.05 was considered for all analyses. Continuous variables were expressed as mean ± standard deviation (SD) and categorical variables were described as frequency and percentage. Correlations were made between disease severity and gender, ethnicity, age, habitual (> 3 times per week) or sporadic (≤ 3 times per week) reported snoring, comorbidities, excessive daytime sleepiness, presence of respiratory disorder, and impact on quality of life.
2.3. ETHICAL CONSIDERATIONS
Following ethical principles in research, this project was submitted and approved by the Research Ethics Committee – CAAE: 35462720.8.0000.5411, according to Resolution n.466-CNS-MS. The inclusion of patients took place after the consent of the person responsible, with the signature of the Free and Informed Consent Term and, when possible, of the Term of Assent for minors aged between 12 and 17 years / 11 months and 29 days.
3. Results
3.1. CHARACTERISTICS OF THE POPULATION
In our study, 35 SD patients accepted the invitation, being 57.1% girls and 42.9% boys. Obesity was observed in 11.4%, prematurity was reported in 42.9%. The main comorbidities were wheezing/asthma (22.9%), recurrent otitis (11.5%), enuresis (14.2%), allergic rhinitis (5.7%) and hypothyroidism (2.8%), present only in group 2. Tonsillar hypertrophy was diagnosed by the pediatrician in 42.9% of the patients, and behavioral changes were observed by the parents in 51.4% (Table 1).
| Variables | Categories | Group 1 | Group 2 | Group 3 | Total |
|---|---|---|---|---|---|
| Sex | Female | 4 (44.4) | 13 (61.9) | 3 (60.0) | 20 (57.1) |
| Male | 5 (55.6) | 8 (38.1) | 2 (40.0) | 15 (42.9) | |
| Snoring | Yes | 8 (88.9) | 16 (76.2) | 3 (60.0) | 27 (77.2) |
| No | 1 (11.1) | 5 (23.8) | 2 (40.0) | 8 (22.8) | |
| Prematurity | Yes | 6 (66.7) | 6 (28.6) | 3 (60.0) | 15 (42.9) |
| No | 3 (33.3) | 15 (71.4) | 2 (40.0) | 20 (57.1) | |
| Comorbidities | Wheezing/Asthma | 3 (33.3) | 4 (19.1) | 1 (20.0) | 8 (22.9) |
| Allergic rhinitis | – (0.0) | 2 (9.5) | – (0.0) | 2 (5.7) | |
| Otitis | 1 (11.1) | 2 (9.5) | 1 (20.0) | 4 (11.5) | |
| Enuresis | 3 (33.3) | 2 (9.5) | – (0.0) | 5 (14.2) | |
| Hypothyroidism | – (0.0) | 1 (4.8) | – (0.0) | 1 (2.8) | |
| Not mentioned | 2 (22.3) | 10 (47.6) | 3 (60.0) | 15 (42.9) |
*Analysis of categorical variables described by frequency and percentage. Groups defined by age: group 1 – 1 to 4 years old, group 2 – 5 to 11 years old and group 3 – ≥ 12 years old. The mean age and body mass index (BMI) z-score were, respectively, 87.5 months (± 45.5) and 0.6 (± 1.3). The PSG exam was finalized by 27 patients, three missed the date due to flu symptoms and five did not accept to perform the exam due to agitation. OSA was diagnosed in 96.3% of the cases (n=25) and 37% were diagnosed with severe disease (n=10), with a mean AHI of 11.3 e/h (± 12.5). The mean oxygen saturation was 95.8% (± 1.4) and the mean minimum oxygen saturation was 84.7% (± 6.0) (Table 2).
| Variables | Average | SD | Minimum | Maximum |
|---|---|---|---|---|
| Age (months) | 87.5 | 45.5 | 22.0 | 184.0 |
| BMI z-score | 0.6 | 1.3 | -2.0 | 3.0 |
| AHI (e/h) | 11.3 | 12.5 | 0.5 | 61.8 |
| OA (e/h) | 3.0 | 8.0 | 0.0 | 41.5 |
| CA (e/h) | 2.7 | 4.9 | 0.0 | 19.8 |
| MA (e/h) | 0.1 | 0.2 | 0.0 | 1.1 |
| Hypopnea | 5.5 | 5.5 | 0.2 | 17.6 |
| Average SatO2 | 95.8 | 1.4 | 93.0 | 98.0 |
| Minimum SatO2 | 84.7 | 6.0 | 71.0 | 93.0 |
Analysis of continuous variables described by average and SD = standard deviation; BMI = body mass index; AHI = apnea and hypopnea index; OA = obstructive apnea; CA = central apnea; MA = mixed apnea; SatO2 = oxygen saturation; kg/m² = kilogram per square meter; e/h = events per hour.
The distribution of the patients according to the OSA severity showed moderate or severe disease in more than half of the population (59.2%), mainly in groups 2 and 3. Snoring was reported by parents in 79.7% of the cases, with the complaint being > 3 times a week in 47.0% of severe OSA, 42.9% of moderate and 32.4% of mild one.
3.2. OBSTRUCTIVE SLEEP APNEA SEVERITY
The parental report of snoring showed a significant correlation with the OSA severity in the study population (p 0.002). No relation was found regarding the characterization of snoring > 3 times a week, and the presence of comorbidities (p 0.32 and 0.43, respectively) (Table 3).
| Variables | Severe OSA | Moderate OSA | Mild OSA | Normal | P | |
|---|---|---|---|---|---|---|
| Snoring* | Yes | 7 (25.9) | 8 (29.6) | 12 (44.5) | – (0.0) | 0.0002 |
| No | – (0.0) | – (0.0) | – (0.0) | 3 (100.0) | ||
| Usual | 5 (31.2) | 6 (37.5) | 5 (31.3) | – (0.0) | 0.32 | |
| Sporadic | 2 (18.2) | 2 (18.2) | 7 (63.6) | – (0.0) | ||
| Comorbidities | Wheezing/Asthma | 3 (42.9) | 3(42.9) | 1 (14.2) | – (0.0) | 0.43 |
| Allergic rhinitis | – (0.0) | 2 (100.0) | – (0.0) | – (0.0) | ||
| Otitis | 1 (25.0) | 1 (25.0) | 2 (50.0) | – (0.0) | ||
| Enuresis | – (0.0) | 1 (33.3) | 2 (66.7) | – (0.0) | ||
| Hypothyroidism | – (0.0) | – (0.0) | – (0.0) | 1 (100.0) | ||
| Not mentioned | 3 (30.0) | 5(50.0) | 2 (20.0) | – (0.0) | ||
| Tonsillar hypertrophy | Yes | 3 (21.4) | 4 (28.6) | 6 (42.9) | 1 (7.1) | 0.78 |
| No | 4 (25.0) | 4 (25.0) | 6 (37.5) | 2 (12.5) | ||
| Not mentioned | – (0.0) | 3(75.0) | 1 (25.0) | – (0.0) | ||
| Obesity | Yes | 1 (25.0) | 1 (25.0) | 2 (50.0) | – (0.0) | 0.57 |
| No | 2 (8.3) | 8 (33.3) | 11 (45.9) | 3 (12.5) | ||
| Not mentioned | – (0.0) | 2 (100.0) | – (0.0) | – (0.0) |
Analysis of categorical variables described by frequency and percentage with Fisher’s Exact-test and significance level of 0.05. Percentage calculated on each line. * Not mentioned = 20.6%
Variables Categories Group 1 Group 2 Group 3 Total
Tonsillar hypertrophy
Yes 5 (55.6) 8 (38.2) 2 (40.0) 15 (42.9)
No 4 (44.4) 13 (61.8) 3 (60.0) 20 (57.1)
Obesity Yes 1 (25.0) 3 (14.3) – (0.0) 4 (11.4)
No 8 (88.9) 18 (85.7) 5 (100.0) 31 (88.6)
Behavior change Yes 6 (66.7) 12 (57.1) – (0.0) 18 (51.4)
No 3 (33.3) 5 (23.8) 5 (100.0) 13 (37.2)
Not mentioned 0 (0.00) 4 (19.1) 0 (0.0) 4 (11.4)
The Epworth scale was significantly associated with the severity of the disease (p 0.001), with higher scores in the severe OSA group (Table 4a). Regarding the comparisons of the variables according between the age groups, mean AHI showed (p 0.03), with a significant difference between group 1 and group 3, whereas the applied questionnaires and the BMI z-score showed no associated with the age groups. The PSQ instrument was not related to the severity of OSA (Table 4b).
| Variables | Measure | Severe OSA | Moderate OSA | Mild OSA | p |
|---|---|---|---|---|---|
| Epworth | Average | 10.2a | 5.0b | 3.2b | 0.001 |
| SD | 3.4 | 2.0 | 4.3 | ||
| PSQ | Average | 11.3 | 7.0 | 8.5 | 0.2 |
| SD | 6.4 | 2.6 | 4.4 | ||
| OSA-18 | Average | 67.1 | 54.4 | 62.3 | 0.6 |
| SD | 23.6 | 23.3 | 24.4 | ||
| BMI z-score | Average | 0.9 | 0.4 | 0.3 | 0.5 |
| SD | 1.3 | 0.9 | 1.6 |
Analysis of variance and Tukey’s test with significance level p ≤ 0.05. Means followed by at least 1 common letter do not differ statistically. SD = standard deviation; PSQ = pediatric sleep questionnaire; BMI = body mass index.
| Variables | Measure | Group 1 | Group 2 | Group 3 | p |
|---|---|---|---|---|---|
| AHI | Average | 4.7b | 9.8ab | 22.5a | 0.03 |
| SD | 3.5 | 7.6 | 20.5 | ||
| Epworth | Average | 5.9 | 6.3 | 6.0 | 0.9 |
| SD | 3.5 | 4.3 | 4.8 | ||
| PSQ | Average | 7.3 | 10.4 | 6.7 | 0.12 |
| SD | 4.7 | 4.8 | 2.7 | ||
| OSA-18 | Average | 52.4 | 69.4 | 49.3 | 0.06 |
| SD | 20.9 | 22.6 | 12.0 | ||
| BMI z-score | Average | 0.3 | 0.7 | 0.4 | 0.8 |
| SD | 1.7 | 1.3 | 0.9 |
Analysis of variance and Tukey’s test with significance level p ≤ 0.05. Means followed by at least 1 common letter do not differ statistically. AHI = apnea and hypopnea index; SD = standard deviation; PSQ = pediatric sleep questionnaire; BMI = body mass index.
4. Discussion
In our study population, the DS patients had a high prevalence of OSA, and more than one-third were diagnosed with severe disease. The predominance of the condition in the present sample was documented in the age group between 5 and 11 years old, as also observed in previous studies enrolling DS children. Abijai et al observed a similar prevalence of severe OSA in 60 out of 81 (74%) patients in his sample. The association between the report of snoring by the parents and the severity of OSA found in our study group was also reported in other studies, enhancing the importance of the investigation of respiratory symptoms in the anamnesis, mostly in the high-risk age group. However, we consider that the high prevalence of OSA in our population might be overestimated, as during the pandemic of covid-19, lasting during the whole study period, only the caregivers of more symptomatic children agreed to participate. At a time when hospitals were focused on combating the pandemic (National Contingency Plan (PCN), participation in the study and the performing of an objective diagnosis for OSA was essential for the referral to the otolaryngologist and a possible surgical treatment.
The small sample size is also related to the study period during the pandemic. DS patients are more vulnerable to the severe form of covid-19, and parents and caregivers did not feel safe to bring their children for a PSG exam. Besides this, the risk of transmission made the use of personal protection equipment mandatory, those being more difficult for DS children to wear.
The various craniofacial characteristics, as midface and mandibular hypoplasia, small oropharynx, narrow nasopharynx, relative macroglossia and glossoptosis, adenotonsillar hypertrophy, besides the generalized hypotonia, and obesity are found in all three types of chromosomal alterations associated with DS. These anatomic and functional alterations of the upper airway lead to intermittent airway obstruction with accumulation of secretion and impairment of adequate ventilation, increasing the predisposition to recurrent respiratory infections, obesity, and OSA. In addition, hypoxemia and hypercapnia resulting from the collapse of the upper airways during sleep trigger alternating events of abnormal breathing and awakening, stimulating the sympathetic nervous system with systemic vasoconstriction and arterial hypertension, as well as altering perfusion, with the production of inflammatory mediators that contribute significantly to cardiovascular outcomes and progression to pulmonary hypertension due to prolonged hypoxia. Thus, the outcomes of untreated OSA can result in severe problems, including developmental delays, behavioral changes and impacts on cognitive and social functions. In our study group, we found a high prevalence of severe OSA, tonsillar hypertrophy in almost half of the children, although not associated with the severity of OSA, and important hypoxia, with a mean minimum oxygen saturation of 85%, but drifting as low as 71%. This means that all of them were at high risk for any of the systemic repercussions cited above, and all would benefit from an earlier diagnosis and treatment planning.
In our analysis, the higher prevalence of severe OSA in older age groups has caveats due to the studied population. First, the reduced number of participants in group 3 should be highlighted, which may have compromised the association with disease severity. In addition, group 2 (5 to 11 years old) is known to cover the age group with the highest prevalence of adenotonsillar hypertrophy, which overlaps with the anatomical and functional alterations of DS. The serious consequences associated with the high prevalence of OSA in children and adolescents with DS make it imperative to perform screening in this population. Regardless of age or parental report, polysomnography is recommended as the gold standard for diagnosis. However, pediatric sleep centers are in limited numbers, mostly in the public health system there are only few sleep centers performing sleep studies in children. Given this reality, the proposal to use tools such as the modified Epworth scale becomes attractive. The results of this study are original with the Epworth scale significantly associated with OSA severity, and corroborate the current literature that emphasizes the relationship between parents’ reports of habitual snoring and the presence of obstructive sleep-disordered breathing.
It is noteworthy that in this sample, the Epworth scale was associated with the severity of OSA, with more severe cases being diagnosed in older patients, who are also more likely to score due to the better perception of daytime sleepiness by them and their parents. Children younger than 5 years old still have daytime sleep habits that may impair the recognition of excessive daytime sleepiness. In addition, it is emphasized that the use of the PSQ instrument was satisfactory as a screening tool to identify sleep-breathing disorders in the studied population but did not present a statistically significant association with the severity of OSA.
The strengths of this study include the analysis of variables that predict the severity of OSA in DS through the application of questionnaires that are easy to understand and to be performed. The full-night polysomnography of each of the participants enhances our representative results and allowed the authors to develop a protocol for the screening and follow-up of these patients in hospitals with low resources. Despite the highlighted aspects, the limitations are recognized: this study was carried out in a single center of a university hospital, limiting the sample size. As a public hospital, access to polysomnographic studies needed to be aligned with the long waiting lists, in addition to the impacts of the covid-19 pandemic, impeding an increase in the sample size.
The proposal to develop a clinical protocol has the clear aim of helping general pediatricians to suspect OSA in patients with DS. The Epworth scale was chosen as a screening tool since it represents an instrument easy to apply and which was significantly associated with severe health impairment in the present study population. Early diagnosis through an interdisciplinary approach is of fundamental importance in preventing cardiovascular and neurocognitive worsening, besides changes in the growth and facial development of these patients. We reinforce that a better diagnostic accuracy will be achieved with the early involvement of the otorhinolaryngologist, and other specialized examinations that are necessary. Multidisciplinary planning of the treatment is mandatory, including the postoperative period when surgery was indicated. Thus, through the flowchart, the pediatrician is able to identify the risk factors for OSA and coordinate the reference to the multidisciplinary team, participating in the decision of the indication of polysomnography in children with DS even before the age of four years, if necessary.
5. Conclusion
The high prevalence of OSA in the studied sample reinforces the importance of a multidisciplinary team for early diagnosis and treatment design in children and adolescents with DS. The results of this study demonstrate that the parental reports of snoring and the identification of excessive daytime sleepiness according to the Epworth scale and disease severity are good screening tools. The entirety of the study enabled the construction of an institutional protocol to improve the diagnostic steps of these patients.
References
- Kaditis AG, Alonso Alvarez ML, Boudewyns A, Alexopoulos EI, Ersu R, Joosten K, et al. Obstructive sleep disordered breathing in 2- to 18-year-old children: diagnosis and management. Eur Respir J. 2015 Nov 5;47(1):69-94.
- Austeng ME, Overland B, Kværner KJ, Andersson E-M, Axelsson S, Abdelnoor M, et al. Obstructive sleep apnea in younger school children with Down syndrome. Int J Pediatr Otorhinolaryngol. 2014 Jul;78(7):1026-9.
- Waters KA, Castro C, Chawla J. The spectrum of obstructive sleep apnea in infants and children with Down syndrome. Int J Pediatr Otorhinolaryngol. 2020 Feb;129:109763.
- Centers for Disease Control and Prevention. Facts about Down syndrome. Accessed September 24th, 2023. https://www.globaldownsyndrome.org/about-down-syndrome/facts-about-down-syndrome/
- Shin M, Siffel C, Correa A. Survival of children with mosaic Down syndrome. Am J Med Genet. Part A. 2010 Mar;152A:800-1.
- Cielo CM, Konstantinopoulou S, Hoque R. OSAS in specific pediatric populations. Curre Probl Pediat Adolesc Health Care. 2016 Jan;46(1):11-8.
- Knollman PD, Heubi CH, Meinzen-Derr J, Smith DF, Shott SR, Wiley S, et al. Adherence to guidelines for screening polysomnography in children with Down syndrome. Otolaryngol Head Neck Surg. 2019 Mar 26;161(1):157-63.
- Thottam PJ, Trivedi S, Siegel B, Williams K, Mehta D. Comparative outcomes of severe obstructive sleep apnea in pediatric patients with trisomy 21. Int J Pediat Otorhinolaryngol. 2015 Jul;79(7):1013-6.
- Brooks LJ, Olsen MN, Bacevice AM, Beebe A, Konstantinopoulou S, Taylor HG. Relationship between sleep, sleep apnea, and neuropsychological function in children with Down syndrome. Sleep Breath. 2014 May 7;19(1):197-204.
- Konstantinopoulou S, Tapia IE, Kim JY, Xanthopoulos MS, Radcliffe J, Cohen MS, et al. Relationship between obstructive sleep apnea cardiac complications and sleepiness in children with Down syndrome. Sleep Med. 2016 Jan;17:18-24.
- Lal C, White DR, Joseph JE, Van Bakergem K, LaRosa A. Sleep-disordered breathing in Down syndrome. Chest. 2015 Feb;147(2):570-9.
- Trucco F, Chatwin M, Semple T, Rosenthal M, Bush A, Tan H-L. Sleep disordered breathing and ventilatory support in children with Down syndrome. Pediatr Pulmonol. 2018 Jul 10;53(10):1414-21.
- Skotko BG, Macklin EA, Muselli M, Voelz L, McDonough ME, Davidson E, et al. A predictive model for obstructive sleep apnea and Down syndrome. Am J Med Genet. Part A. 2017 Jan 26;173(4):889-96.
- Friedman NR, Ruiz AG, Gao D, Ingram DG. Accuracy of parental perception of nighttime breathing in children with Down syndrome. Otolaryngol Head Neck Surg. 2017 Sep 5;158(2):364-7.
- Nehme J, LaBerge R, Pothos M, Barrowman N, Hoey L, Monsour A, et al. Predicting the presence of sleep-disordered breathing in children with Down syndrome. Sleep Med. 2017 Aug;36:104-8.
- Nehme J, LaBerge R, Pothos M, Barrowman N, Hoey L, Kukko M, et al. Treatment and persistence/recurrence of sleep disordered breathing in children with Down syndrome. Pediatr Pulmonol. 2019 Jun 6;54:1291-6.
- Bertolazi AN, Fagondes SC, Hoff LS, Pedro VD, Menna Barreto SS, Johns MW. Portuguese-language version of the Epworth sleepiness scale: validation for use in Brazil. J Bras Pneumol. 2009 Sep;35(9):877-883.
- Martins CAN, Deus MM, Abile IC, Garcia DM, Anselmo-Lima WT, Miura CS, et al. Translation and cross-cultural adaptation of the pediatric sleep questionnaire (PSQ*) into Brazilian Portuguese. Braz J Otorhinolaryngol. 2021 Apr.
- Santos Fernandes FM, da Cruz Vieira Veloso Teles R. Application of the Portuguese version of the Obstructive Sleep Apnea-18 survey to children. Braz J Otorhinolaryngol. 2013 Nov;79(6):720-726.
- Brodsky L. Modern assessment of tonsils and adenoids. Pediatr Clin North Am. 1989;36(6):1551-69. PMID: 2685730.
- Ministry of Health. Brazil. Guidelines and regulatory standards for research involving human. Resolution no. 466. Brasília: National Health Council. 2012.
- Chamseddin BH, Johnson RF, Mitchell RB. Obstructive sleep apnea in children with Down syndrome: demographic, clinical, and polysomnographic features. Otolaryngol Head Neck Surg. 2018 Aug 28;160(1):150-7.
- Abijay CA, Tomkies A, Rayasam S, Johnson RF, Mitchell RB. Children With Down Syndrome and Obstructive Sleep Apnea: Outcomes After Tonsillectomy. Otolaryngol Head Neck Surg. 2022 Mar;166(3):557-564. doi: 10.1177/01945998211023102. Epub 2021 Jul 13. PMID: 34253070.
- Bigoni A, Malik AM, Tasca R, Carrera MBM, Schiesari LMC, Gambardella DD, Massuda A. Brazil’s health system functionality amidst of the COVID-19 pandemic: An analysis of resilience. Lancet Reg Health Am. 2022, 10.
- Santos TBS, Andrade LRD, Vieira SL, Duarte JA, Martins JS, Rosado LB, Pinto I CM. Hospital contingency in coping with COVID-19 in Brazil: governmental problems and alternatives. Cienc Saude Colet.2021: 26, 1407-1418.
- Khan IA. Role of adenotonsillectomy and tonsillectomy in children with Down syndrome who develop obstructive sleep apnea by obesity as a risk factor. Int J Pediatrics. 2022 May 6;2022:1-8.
- Maris M, Verhulst S, Wojciechowski M, Van de Heyning P, Boudewyns A. Sleep problems and obstructive sleep apnea in children with Down syndrome, an overview. Int J Pediatric Otorhinolaryngol. 2016 Mar;82:12-5.
- Horne RSC, Wijayaratne P, Nixon GM, Walter LM. Sleep and sleep disordered breathing in children with Down syndrome: effects on behaviour, neurocognition and the cardiovascular system. Sleep Med Rev. 2019 Apr 1;44:1-11.
- Lin SC, Davey MJ, Horne RSC, Nixon GM. Screening for obstructive sleep apnea in children with Down syndrome. J Pediatr. 2014 Jul;165(1):117-22.
- Brockmann PE, Damiani F, Nuñez F, Moya A, Pincheira E, Paul MA, et al. Sleep-disordered breathing in children with Down syndrome: usefulness of home polysomnography. Int J Pediatr Otorhinolaryngol. 2016 Apr;83:47-50.
- Hsieh A, Gilad A, Wong K, Cohen M, Levi J. Obstructive sleep apnea in children with Down syndrome: Screening and Effect of Guidelines. Clin Pediatr. 2019 Apr 29;58(9):993-9.
Supplementary Files
A.1: MODIFIED EPWORTH SCALE
What is your child’s chance of dozing off or falling asleep in the following situations?
- 0 = no chance
- 1 = little chance
- 2 = moderate chance
- 3 = high chance
- Sitting and reading.
- Watching TV.
- Sitting inactive in a public place (cinema, meeting).
- As a passenger in a car for a period of 1 hour without breaks.
- Lying down to rest in the afternoon when possible.
- Sitting and talking to someone.
- Sitting quietly after lunch.
- In the car, while standing in traffic for a few minutes.
A.2: PEDIATRIC SLEEP QUESTIONNAIRE (PSQ)
PSQ Yes No Don’t know
During sleep, your child:
- snores more than half the time
- always snores
- snores loudly
- has deep or noisy breathing
- Do you have difficulty breathing or struggle to breathe?
- Have you ever seen your son (or daughter) stop breathing in their sleep?
Your son (or daughter):
- tends to breathe with an open mouth during the day
- wake up with a dry mouth
- Pee in bed from time to time
Your son (or daughter):
- Do you wake up tired in the morning?
- has a problem with daytime drowsiness?
- Has a teacher or other person ever commented that your child seems sleepy during the day?
- Is it difficult to wake up your child in the morning?
- Does your child wake up with a headache in the morning?
- Has your child stopped growing normally at some point since birth?
- Is your child overweight?
Does your child often:
- Doesn’t seem to hear when you talk to him directly
- Has difficulty organizing tasks and activities
- Is easily distracted by extraneous stimuli
- Has restless hands or feet or is restless when sitting
A.3: OBSTRUCTIVE SLEEP APNEA QUESTIONNAIRE (OSA-18)
In the past 4 weeks, how often has your child had…
Never Almost never Few times Some times Many times Often Always
- …snoring loudly?
- …breathing stops at night?
- …gagging or wheezing while sleeping?
- …restless sleep or frequent awakening?
Physical symptoms
- …mouth breathing due to nasal obstruction?
- …colds or upper respiratory infections?
- …nasal discharge and congestion?
- …difficulty swallowing food?
Emotional problems
- …mood swings or tantrums?
- …aggressive or hyperactive behavior?
- …disciplinary problems?
Everyday problems
- …excessive daytime sleepiness?
- …episodes of lack of attention or concentration?
- …difficulty getting out of bed in the morning?
Informant opinion
- How often do the above problems…
- …caused concern?
- …worried that your child could not get enough air?
- …interfered with daily activities?
- …left you frustrated?
B: INSTRUMENT FOR DATA COLLECTION
Date: Age: Weight:
Sex: ( ) Female ( ) Male Ethnicity (self-reported):
- Does your child experience snoring while sleeping, witnessed apnea, restless sleep or mouth breathing? ( ) Yes ( ) No
- If your child only snores, how often? ( ) ≤ 3 times a week ( ) > 3 times a week
- Does your child have one or more of the following conditions? ( ) History of prematurity ( ) Parents/siblings diagnosed with OSA / adenotonsillar hypertrophy / history of childhood adenotonsillectomy ( ) Enuresis ( ) Growth delay / failure ( ) Systemic arterial hypertension ( ) Obesity ( ) Metabolic syndrome ( ) History of recurrent otitis media / placement of a tympanostomy tube ( ) Recurrent wheezing or asthma ( ) Excessive daytime sleepiness ( ) Academic difficulties ( ) Behavioral changes




