Optimizing Heart Failure Management: Pre- and Postdischarge
Predischarge and Postdischarge Heart Failure Management: Treatment Optimisation, Adherence, and Multidisciplinary Care
Marija Polovina 1,2, Ovidiu Chioncel 3, Gianluigi Savarese 4, Magdy Abdelhamid 5, Gordana Krljanac 1,2, Carsten Tschöpe 6,7, Petar M. Seferović 2,7
- Marija Polovina Departmet of Cardiology, University Clinical Centre of Serbia, Belgrade, Serbia.; Faculty of Medicine, University of Belgrade, Belgrade, Serbia.
- Ovidiu Chioncel Emergency Institute for Cardiovascular Diseases “Prof. Dr. C.C. Iliescu” Bucharest; University for Medicine and Pharmacy “Carol Davila” Bucharest, Bucharest, Romania.
- Gianluigi Savarese Division of Cardiology, Department of Medicine, and Karolinska Institutet, Stockholm, Sweden.
- Magdy Abdelhamid Faculty of Medicine, Kasr Al Ainy, Cardiology Department, Cairo University, Cairo, Egypt.
- Gordana Krljanac Department of Cardiology, University Clinical Centre of Serbia, Belgrade, Serbia.; Faculty of Medicine, University of Belgrade, Belgrade, Serbia.
- Carsten Tschöpe Department of Cardiology, Angiology and Intensive Care, Campus Virchow German Heart Center at Charité, Berlin, Germany.; Berlin Institute of Health at Charite Center for Regenerative Therapies, Berlin, Germany
- Petar M. Seferović Faculty of Medicine, University of Belgrade, Belgrade, Serbia.; Serbian Academy of Sciences and Arts, Belgrade, Serbia.
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION:POLOVINA, Marija et al. Predischarge and Postdischarge Heart Failure Management: Treatment Optimisation, Adherence, and Multidisciplinary Care. Medical Research Archives, [S.l.], v. 13, n. 5, may 2025. ISSN 2375-1924. Available at: <https://esmed.org/MRA/mra/article/view/6550>
DOI: https://doi.org/10.18103/mra.v13i5.6550
ISSN 2375-1924
Abstract
Hospitalisation for heart failure presents a critical event associated with significant risk of readmission and mortality. It also offers a window of opportunity to optimise patient management with a goal to improve clinical outcomes, functional status, and quality of life. This narrative review summarises contemporary, evidence-based strategies for optimising heart failure management before and after hospital discharge. Firstly, comprehensive assessment of congestion status is necessary before discharge because residual congestion is a major contributor to poor outcomes. In addition, robust evidence supports the early initiation and rapid up-titration of core guideline-directed medical therapies in all patients without known contraindications, irrespective of left ventricular ejection fraction. The core guideline-directed medical therapies classes include renin-angiotensin system inhibitors, sacubitril/valsartan, beta-blockers, mineralocorticoid receptor antagonists, and sodium-glucose cotransporter-2 inhibitors. Intensive strategy to optimisation of renin-angiotensin system inhibitors, beta-blockers, and mineralocorticoid receptor antagonists has been shown to reduce the risk of death or readmission by 34% at six months compared to standard care. Likewise, initiating sodium-glucose cotransporter-2 inhibitors during hospitalisation has demonstrated favourable effects on clinical outcomes, including lower risk of all-cause mortality and readmission. Furthermore, multidisciplinary care and early and sustained postdischarge follow-up are essential to address comorbidities, ensure continuity of care and allow further optimisation of medical therapy. They also enable timely management of potential issues concerning drug intolerance, side effects, nonadherence, or changes in clinical status. Successful long-term management and adherence to treatment recommendations also requires structured patient education and empowerment for self-care.
Keywords: heart failure, management, guideline-directed medical therapy, congestion, adherence, multidisciplinary care, education
Introduction
Heart failure (HF) is a global health challenge, affecting an estimated 64 million people worldwide, and associated with significantly impaired survival and frequent hospitalisations 1. An estimated 2.1 million hospitalisations for HF take place in Europe annually 2. Hospitalisation for HF is a critical event, since it is linked to a higher mortality, greater risk of complications, and a vulnerable postdischarge period marked by a significant risk of readmission3. Hospitalisation for HF also offers a valuable opportunity to optimise patient management and improve clinical outcomes. Current European Society of Cardiology (ESC) Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure highlighted several key strategies in pre- and postdischarge management of patients with HF 4,5. These include complete decongestion, or minimal residual congestion, rapid initiation and up-titration of guideline directed medical therapies (GDMT) following patient stabilisation, structured postdischarge follow-up, and engaging patients in multidisciplinary care that addresses care transition, comorbidities, and patient education. These recommendations are based on recent clinical trial evidence demonstrating that such strategies can improve clinical outcomes, functional capacity, and quality of life following hospitalisation for HF 6-8. However, real-world data reveal that a significant proportion of patients are discharged with residual congestion, whilst multiple challenges and gaps persist in implementing Guideline-recommended management strategies for HF, significantly affecting patient outcomes and prognosis 9-11.
This narrative review is intended for physicians across all levels of care, aiming to highlight key elements in the pre- and postdischarge management of patients with HF. It addresses effective management of congestion, timely GDMT initiation and up-titration, and the importance of structured follow-up. Emphasis is placed on adopting a multidisciplinary approach, ensuring a successful transition of care, and promoting patient education – all essential for improving GDMT implementation and adherence, and enhancing quality of care and patient outcomes.
Predischarge management of congestion: implications for the implementation of medical therapies for heart failure
Congestion is defined as fluid accumulation in the intravascular compartment and the interstitial space, resulting from increased cardiac filling pressures and/or increased vascular permeability because of inflammation12. Congestion in HF may progress chronically due to maladaptive renal sodium and water retention, or acutely from fluid mobilization from the splanchnic reservoir 13. It is the most frequent cause of hospitalisation for HF, present in ~90% of admitted patients according to the ESC and Heart Failure Association (HFA) HF long-term registry 9. Residual congestion persists in 30.9% of patients at discharge and is associated with worse prognosis in terms of mortality and readmission rates 9,14. Residual congestion often indicates a more advanced disease state in HF, as it is more frequently seen in older patients, those with worse symptoms and multiple comorbidities, and may be associated with diuretic resistance and/or worsening renal function or acute kidney injury 15-17.
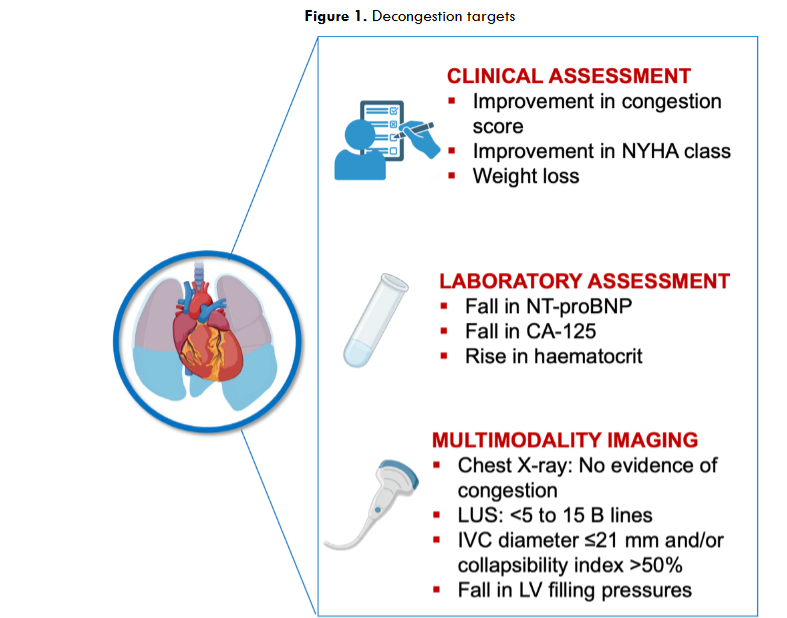
Given its detrimental effects, predischarge assessment of congestion by multiple targets has important clinical implications 4. Current evidence supports an integrative approach that combines clinical evaluation, laboratory markers and multimodality imaging to assess changes in congestion from baseline to discharge (Figure 1) 18. This is important because clinical improvement may occur whilst tissue congestion persists and using multiple tools can increase the sensitivity of detecting residual congestion. A reduction of >30% in N-terminal pro-B-type natriuretic peptide (NT-proBNP) from baseline before discharge, or a predischarge NT-proBNP level <1500 pg/mL, is associated with a significantly lower risk of postdischarge adverse events 19. Additional biomarkers, such as carbohydrate antigen 125, ghrelin and galectin 3, can complement natriuretic peptides in evaluating congestion 20-22. Several imaging tools can be used, including chest X-ray, echocardiographic evaluation of left ventricular (LV) filling pressures, imaging of inferior vena cava (IVC) size and collapsibility, and detection of B-lines with lung ultrasound (LUS) (Figure 1). Chest X-ray has the benefit of being affordable and accessible but suffers from low sensitivity (i.e. one in five patients with congestion may have normal radiogram) 23, and the requirement for repeat radiation exposure. Despite these limitations, high radiographic congestion scores at discharge predict greater risk of rehospitalisation 24. Non-invasive assessment of predischarge LV filling pressures with echocardiography was shown to be superior to clinical evaluation alone in the prediction of readmission 25,26. It is considered appropriate to obtain a fall in E/e’ ratio <10-15 18. IVC imaging and LUS provide a reliable estimation of right atrial pressure and lung congestion, respectively, and rapidly reflect changes in volume status in response to treatment. Persistently dilated IVC with low collapsibility index before discharge predicts greater risk of readmission 27. Likewise, the number of B-lines indicates the severity of pulmonary congestion, with <5 B-lines indicating no congestion and >30 B-lines indicating severe congestion28. Furthermore, residual pulmonary congestion as assessed with LUS at discharge is strongly associated with adverse outcomes 29,30.
Intravenous loop diuretics remain the cornerstone therapy for relieving fluid overload. To enhance diuretic response and achieve more complete decongestion, multiple strategies have been investigated. Among them, combining loop diuretics with “non-loop” agents that act at different sites along the nephron to induce sequential nephron blockade which leads to increased sodium and water excretion has been evaluated in several clinical trials. Addition of intravenous acetazolamide (i.e. inhibits proximal tubule sodium reabsorption) to furosemide, compared to furosemide alone, in the ADVOR trial, has provided more successful decongestion and shorter hospital stay, without significantly increasing the rate of adverse events (i.e. worsening kidney function, hypokalaemia, hypotension) 31. Similarly, a combination of intravenous furosemide with an oral thiazide-like diuretic, compared to furosemide alone in the CLOROTIC trial, enhanced diuretic response, but did not lead to improved symptom relief and was associated with a higher risk of worsening renal function 32. Furthermore, a decongestive strategy guided by urinary sodium excretion has demonstrated improved natriuresis and diuresis; however, it did not impact 180-day HF rehospitalisation or all-cause mortality, which was the co-primary endpoint of this trial 33. Of note, these and other strategies aimed to improve diuretic response failed to show improvements in clinical outcomes, including mortality and HF readmission rates (Table 1).
| Strategy |
|---|
| Shorter door-to-diuretic time (i.v. furosemide <60 min from admission) |
| Furosemide intravenous bolus every 12 h vs continuous furosemide infusion |
| Furosemide combined with “non-loop” diuretics: |
| ▪ Acetazolamide |
| ▪ Hydrochlorothiazide |
| ▪ Metolazone |
| ▪ Spironolactone |
| Natriuresis-guided diuretic therapy |
| Vasopresin-2 receptor agonists (tolvaptan) |
| Rollofyline |
| Ultrafiltration/renal replacement therapy |
Although successful decongestion (i.e. attaining euvolemia) is a desirable therapeutic goal in patients hospitalised for HF, it is noteworthy to consider that overzealous diuretic use can have adverse effects, including worsening renal function/acute kidney injury, dehydration, hypotension and electrolyte disturbances that may delay or preclude institution of GDMT 34. Current evidence suggests that euvolemia is not a prerequisite for predischarge initiation and optimisation of GDMT. Patients involved in the recent STRONG-HF trial, that assessed efficacy and safety of rapid GDMT implementation in hospitalised HF patients, regardless of left ventricular ejection fraction (LVEF), were required to be haemodynamically stable (i.e. systolic blood pressure ≥100 mmHg and heart rate >60 bpm within prior 24 h) and without significant renal dysfunction or electrolyte abnormalities (i.e. estimated glomerular filtration rate, eGFR ≥30 mL/min/1.73 m2 and K+ ≤5.0 mmol/L) before randomisation 35. Persistent congestion, evidenced by a screening NT-proBNP >2500 pg/mL and >10% decrease between screening and before randomization (but still >1500 pg/mL), was required for inclusion 35. The patients were randomized to either an intensive strategy involving initiation of the three classes of neurohormonal inhibitors – angiotensin-converting enzyme inhibitor (ACEI), angiotensin receptor blocker (ARB) or angiotensin receptor–neprilysin inhibitor (ARNI); beta-blocker (BB); and mineralocorticoid receptor antagonist (MRA) – two days before discharge, followed by rapid up-titration to target doses within two weeks postdischarge, or to standard of care 35 6. Compared to standard care, the intensive treatment strategy resulted in a 34% risk reduction in HF rehospitalisation or all-cause mortality at 6 months (hazard ratio, HR 0.66; 95% confidence interval, 0.50–0.86) 6. Further analysis demonstrated that timely initiation and rapid up-titration of HF medications (ACEI/ARB/ARNI, BB and MRA) in haemodynamically stable, but still modestly congested patients, is associated with attaining more complete and sustained decongestion over time, ultimately translating into improved clinical outcomes and quality of life 6,36,37. Re-assessment of natriuretic peptide levels after discharge can help guide clinical decisions, such as increasing the diuretic dose or slowing GDMT up-titration if a rise in NT-proBNP is observed 38. These observations are further supported by the EMPULSE trial, which assessed the efficacy and safety of a sodium-glucose-contransporter-2 (SGLT2) inhibitor, empagliflozin (on top of usual care) vs placebo in patients hospitalised for HF, regardless of LVEF 7. In addition to showing a significant improvement in clinical outcomes with empagliflozin vs. placebo, this trial also demonstrated more effective and sustained decongestion with early empagliflozin initiation (median three days after admission) 39. Moreover, contemporary GDMT can help reduce loop diuretic doses needed to prevent subsequent development of fluid overload in the long run 40.
Initiation and optimisation of medical therapies in patients hospitalised for heart failure
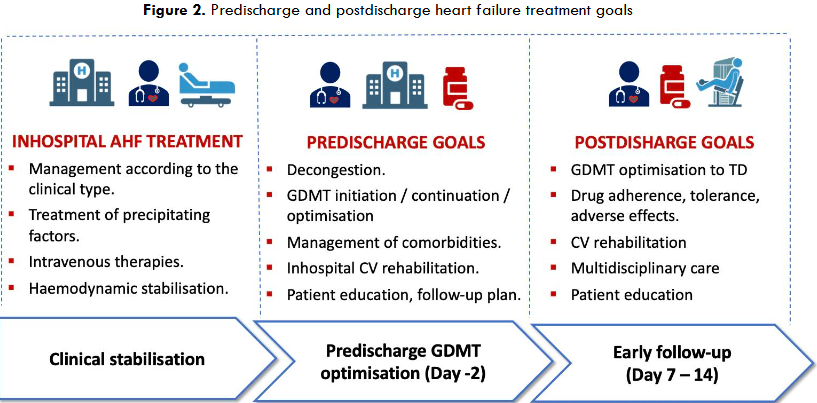
The current evidence-based approach to reducing the risk of rehospitalisation and mortality in patients admitted for HF involves provision and optimisation of the four fundamental classes of GDMT upon clinical stabilisation and before discharge (Figure 2) 5. This includes the use of neurohormonal inhibitors (i.e. ACEI/ARB/ARNI, BB, and MRA) along with an SGLT2 inhibitor (i.e. dapagliflozin or empagliflozin) in all patients without contraindications, regardless of LVEF, based on the STRONG-HF results 5,6. Neurohormonal inhibitors should be up-titrated to 50% target doses before discharge and then up-titrated to the effective target (100%) doses (or at least maximum tolerated doses) during the careful follow-up visits within 2 – 6 weeks postdischarge 6. Since SGLT2 inhibitors come in a single dose and have a negligible or no effect on blood pressure, heart rate or electrolyte levels, they can be easily implemented along other HF medications. For each patient, an optimal timing should be identified before discharge to provide all four GDMT classes, due to their synergistic and rapid beneficial effects. This is emphasised by the recent FINEARTS-HF trial assessing the efficacy of a nonsteroidal MRA, finerenone vs. placebo in patients with HF and mildly reduced (HFmrEF) or preserved ejection fraction (HFpEF) for the risk reduction in total HF events and CV death 41. The trial included more than 50% of patients with a recent HF event (i.e. hospitalisation) before randomisation (~20%, <7 days before randomisation) and demonstrated a 26% lower rate of the composite primary outcome (rate ratio, 0.84; 95% CI, 0.74 – 0.95) with finerenone vs. placebo 41. Of note, a prespecified analysis of this trial demonstrated a statistically significant improvement in outcomes with finerenone after only 28 days from the treatment institution, and sustained thereafter 42, supporting similar earlier observations of rapid treatment benefits with contemporary medications 43,44.
However, decisions on how and when to provide GDMT in individual patients may be influenced by haemodynamic factors (i.e. blood pressure, heart rate), renal function, electrolyte levels (i.e. hyperkalaemia), comorbidities, frailty and concerns about intolerance and adverse effects. These concerns may be even more challenging in therapy-naïve patients. Whereas the STRONG-HF trial demonstrated feasibility, safety, and beneficial effects of continuing and rapidly up-titrating GDMT in patients who were already on at least some (but not yet optimized) HF drugs prior to randomisation, evidence supporting GDMT initiation in treatment-naïve patients is less robust. Available data, derived mostly from smaller-scale clinical trials and observational studies, support benefits of GDMT initiation in therapy naïve haemodynamically stable individuals, mostly with HF and reduced ejection fraction (HFrEF). For example, the IMPACT-HF trial demonstrated that predischarge initiation of carvedilol in stabilised BB naïve patients with LVEF <40% was feasible, and not associated with more adverse effects or prolonged hospital stay 45. Improved outcomes with ACEI/ARB initiation have been reported in HFrEF patients without a history of prior ACEI/ARB use 46. Furthermore, PIONEER-HF and TRANSITION trials demonstrated that ARNI initiation during hospitalisation is safe and well tolerated, even in patients without prior exposure to ACEI/ARB 47,48. In the PIONEER-HF trial, the benefits of ARNI over enalapril were consistent among HFrEF patients who were ACEI/ARB-naïve 49. Finally, the EMPULSE trial confirms that initiating empagliflozin in stabilised HF patients is both effective and safe when started after a median of three days following hospital admission 7. Of note, in clinical practice most patients admitted for newly diagnosed acute HF have often been prescribed one or more GDMT drug classes for the treatment of comorbidities and are therefore not truly therapy naïve.
Another challenge in optimising GDMT during hospitalisation arises when it is necessary to reduce the dose or temporary discontinue drug therapy, most frequently due to hypotension, bradycardia, worsening renal function/acute kidney injury, and/or significant electrolyte imbalances 46. Whilst multiple studies have suggested improved outcomes with in-hospital continuation of medical therapies, medication withdrawal or even dose reduction has been associated with higher rates of mortality and readmission and lower likelihood of subsequent drug re-initiation at follow-up 46,50,51. Despite challenges, therefore, GDMT optimisation should be prioritised in high-risk patients.
Follow-up strategies to improve implementation of medical therapies for heart failure
Postdischarge continuity of care is crucial for HF patients since it facilitates multiple goals and ultimately improves quality of life and reduces the risk of readmission or mortality (Figure 2). The ESC Guidelines for HF management recommend first follow-up visit at 1 – 2 weeks after discharge 4. This is supported by extensive data suggesting that an early follow-up visit, within 7 – 14 days after discharge is associated with lower risk of death or rehospitalisation within 30 days 52,53. Even a telephone call within this period has been linked to better outcomes 52, whereas delays or inability to secure follow-up visits have been associated with poorer outcomes 53. Follow-up visits can be performed by HF specialists, cardiologists, or even general practitioners and trained nurses, and follow-up with a familiar physician yields better outcomes than with an unfamiliar one, highlighting the importance of continuity of care 53,54. Early optimisation of GDMT to target doses should be prioritised, as supported by the STRONG-HF results. This trial has shown that an intensive treatment strategy with an initial follow-up visit within 7 days after discharge, followed by subsequent visits up to 6 weeks can provide more successful GDMT optimisation (i.e. greater proportion of patients achieving target doses of ACEI/ARB/ARNI, BB, and MRA) compared to usual care, eventually translating into improved outcomes 6.
Rapid treatment optimisation, rather than follow-up frequency or settings, seems to be crucial for clinical outcomes after discharge. A randomised trial (ECAD-HF) assessed a strategy of optimized care and patient education by a HF specialist over the 2 – 3 weeks, compared to standard care in high-risk HFrEF patients characterised by recent HF hospitalisation, low blood pressure, renal dysfunction and/or increased levels of natriuretic peptides 55. The trial failed to show a reduction in all-cause mortality or HF hospitalisation at 6 months with intensified management compared to usual care (HR 0.97; 95% CI 0.74 – 1.26) 55. Importantly, there was no significant difference in treatment optimisation between the intensive and usual care groups in this trial, which might have influenced clinical outcomes. However, reasons for the sub-optimal treatment optimisation remain unclear and may reflect greater intolerance/side effects, characteristic of the high-risk patients involved in the ECAD-HF trial 55.
Despite its significance, real-world data continue to reveal delayed and/or suboptimal GDMT implementation and up-titration. A multinational cohort study using routine-care data demonstrated a delay in the postdischarge initiation of “novel” GDMT (i.e. an SGLT2 inhibitor and ARNI) compared with “older” HF medications (i.e. ACEI/ARB, BB, MRA) 56. Up-titration to target doses remained low, while drug discontinuation rates were high 56. A registry-based data from the Netherlands, suggested that, among chronic and worsening HF patients, 44% of those with HFrEF and 35% of those with HFmrEF were prescribed the quadruple GDMT, but only 1% achieved target doses for all drug classes 10. Similar results were reported earlier in HFrEF outpatients in the United States 11. An Italian registry involving individuals with severe HFrEF demonstrated that BB, ACEI/ARB/ARNI and MRA were provided in 82%, 55% and 60% of patients, respectively, whilst ≥50% of target doses were reached in 41%, 22%, and 56% of those taking BB, ACEI/ARB/ARNI and MRA, respectively 57.
The reasons for suboptimal GDMT implementation are complex and multifactorial 58. Patient-related factors involve concerns about intolerance or side effects due to chronic kidney disease, hypotension, bradycardia and/or hyperkalaemia 57. Furthermore, older age, female sex and frailty are often associated with under-prescription 59, despite proven GDMT benefits among vulnerable patient categories 60. Physicians’ lack of confidence and clinical inertia, polypharmacy, and factors related to drug availability, affordability and access to care also have a significant impact on GDMT implementation 58,61. However, reasons for drug non-prescription remain unknown in up to 50% of patients 57. Another major limitation in providing rapid treatment optimisation through multiple patient visits is the lack of sufficient workforce to support seamless integration between inpatient and outpatient care 62. Various digital health solutions (teleconsultations, non-invasive and invasive telemonitoring, electronic clinical decision support systems etc.) have been explored to support care transition and GDMT uptake (Table 2), and their applicability across different healthcare systems deserves further assessment.
| Study | Intervention | Outcome |
|---|---|---|
| IMPLEMENT-HF 87 | Pharmacist-physician GDMT Team provided suggestions to treating teams based on an evidence-based algorithm for GDMT optimisation in patients with HFrEF. | Improved HF GDMT uptake and optimisation |
| Rao et al. 88 | Randomised clinical trial of multidisciplinary virtual HF consultation vs usual care for GDMT optimisation in patients with HFrEF. | Improved HF GDMT implementation and dose titration |
| TIM-HF89 | Randomised trial of a secure web-based system of remote patient management plus usual care vs usual care alone. | Reduction in the percentage of days lost due to unplanned CV hospitalisation and all-cause mortality (RR, 0.80, 95% CI 0.65-1.00; p=0.0460) |
| DAVID-HF 90 | Home monitoring system consisting of wearable armband monitors paired with the smartphone application in HFrEF patients | Improved HF GDMT up-titration |
| MONITOR-HF 91 | Randomised clinical trial of invasive monitoring of pulmonary artery pressure (CardioMEMS-HF system, Abbott Laboratories, Abbott Park, IL, USA) vs standard care in patients with HF, regardless of LVEF | Improvement in quality of life and reduction in HF hospitalisations |
| PROMPT-HF 92 | Randomised trial of tailored electronic health record alerts vs usual care for GDMT optimisation in HFrEF patients | Significantly higher rates of the four classes of GDMT provision at 30 days. |
| EPIC-HF93 | Randomised trial of patient activation tools designed to encouraged patients to work collaboratively with their clinicians to “make one positive change” vs. standard care | Improved GDMT initiation and dose intensification |
Addressing patient adherence and persistence to medical therapies for heart failure
In addition to suboptimal GDMT implementation by physicians, non-adherence (i.e. not taking medications as prescribed) or lack of persistence (i.e. drug discontinuation) among HF patients impose a significant impediment to successful treatment outcomes. Accordingly, medication non-adherence was identified in 21% – 64% of patients preceding an episode of worsening HF in different studies 63. Multiple factors can impact medication adherence and persistence, as summarised in Table 3.
| Patient-related factors |
|---|
| Socioeconomic deprivation |
| Low health literacy |
| Patients’ perception of drug ineffectiveness |
| Living alone |
| Frailty |
| Cognitive impairment |
| Depression |
| Multimorbidity |
| Treatment-related |
| Polypharmacy |
| Multiple healthcare providers |
| Healthcare system-related |
| Availability |
| Affordability/Reimbursement |
| Access to care |
Given the complexity of factors influencing adherence and persistence, interventions aimed to improve these aspects of care likely need to be multifactorial, patient-centred and tailored to specific healthcare circumstances. Appropriate transition and coordination of postdischarge care involving scheduled follow-up visits, long-term follow-up commitment, patient education and empowerment for self-management, as well as the amount of time spent with patients by healthcare professionals have shown to provide some improvements in adherence 64,65. Persistence in patient education and motivation to accept medical treatment as a necessary part of living with HF, and empowerment to understand the benefits of treatment and how to deal with potential mild and transient symptoms of intolerance/side effects can favourably impact patients’ perceptions and adherence. A multidisciplinary approach including pharmacist-led education and follow-up, assisted with modern technologies, compared to standard care has also shown modest improvement in adherence 66. Medication reminders utilising electronic pillboxes or health-apps have been well-accepted by the patients and associated with improved adherence 67.
Management of patients with HF is almost inevitably linked to polypharmacy (i.e. use of ≥5 medications), due to the GDMT complexity and frequent need to treat concomitant comorbidities. Treatment simplification by carefully deprescribing unnecessary medications (taking into consideration patient preferences and risk of post-withdrawal symptom exacerbations), treating comorbidities with appropriate HF drugs (e.g. arterial hypertension, angina, diabetes, chronic kidney disease etc.), and even development of a polypill containing several of the disease-modifying HF medications have been proposed to improve adherence/persistence 68,69. Currently, a polypill-based strategy containing low fixed doses of spironolactone, empagliflozin, and a titratable dose of metoprolol-succinate, added to a titratable dose of renin-angiotensin system blocker (not included in the polypill) vs standard care is being assessed to evaluate the impact on left ventricular function in HFrEF patients (NCT04633005) 69. Changes in health policies aimed to improve access to care and drug reimbursement may also foster treatment adherence. Notably, successful interventions that enhance patient adherence in HF have been shown to reduce the risk of death by 11% (HR, 0.89; 95% CI, 0.81 – 0.99), and decrease the odds for hospital readmission by 21% (odds ratio, 0.79; 95% CI, 0.71 – 0.89) 70.
Multidisciplinary care, cardiovascular rehabilitation, and patient education
Multimorbidity and frailty are prevalent in patients with HF and are associated with adverse outcomes, greater utilisation of healthcare resources and more complex management that can impede GDMT implementation and adherence 71,72. To address the need for the multifaceted and coordinated care by multiple specialists, the Heart Failure Association (HFA) of the ESC proposed a concept of the Quality-of-Care Centres for HF management 73. These centres are intended to provide an integrated multidisciplinary approach and smooth transition of care between different specialists and across different levels of care 73. This approach is essential to manage both CV (e.g. atrial fibrillation, valvular heart disease, ischaemic heart disease etc.) and non-CV comorbidities (e.g. diabetes, chronic lung disease, chronic kidney disease, anaemia, electrolyte disturbances) ensuring compliance with different guideline recommendations and avoidance of therapeutic competition. It offers an opportunity to initiate cardiovascular rehabilitation and promote interventions such as smoking cessation, nutritional improvements, and other lifestyle modifications that can impact patient outcomes 74. It is also essential for ensuring optimal care of patients with advanced HF. A meta-analysis of 74 trials has shown that multidisciplinary HF management can reduce HF admissions (relative risk, RR 0.87, 95% CI 0.79 – 0.95) and all-cause mortality (RR 0.70, 95% CI 0.61 to 0.81), supporting the integration of multidisciplinary care into existing healthcare systems 73,75. However, many European countries either lack dedicated HF centres or have less than one such centre per million inhabitants, indicating the need for further reorganisation and development of healthcare infrastructure that can deal with a growing burden of HF 2.
Pre- and postdischarge cardiovascular rehabilitation, including structured exercise training and risk factor modification can help improve health status and quality of life and reduce the risk of hospitalisation and mortality in a broad spectrum of HF patients, including the elderly and frail individuals 76-78. Despite proven benefits, cardiac rehabilitation remains underutilised, and is further challenged by poor adherence, with dropout rates reaching 63% 79. Improving implementation requires raising awareness among physicians and patients, alongside health policies that ensure affordable or reimbursed multidisciplinary rehabilitation programmes.
Patient education about HF symptoms, treatment significance, and necessary lifestyle modifications is another vital component of HF management. Education should be extended to family members and caregivers with an emphasis on disease management and psychological and social support. Effective self-care among HF patients in the community, including adherence to medications and life-style recommendations, appropriate symptom response, and consulting behaviours (i.e. contacting a healthcare provider when appropriate), has been demonstrated to significantly reduce the risk of clinical outcomes (HR, 0.66, 95%CI 0.46 – 0.96) compared to poorer self-care 80. According to the HFA/ESC recommendations, patient education for effective self-care should encompass several domains as presented in Table 4 81. Multidisciplinary care offers the most effective environment for engaging patients in education and promoting self-empowerment 73,81. The multidisciplinary team should incorporate various specialists and trained nurses to provide educational sessions, informative materials, telemonitoring, and e-health support, tailored to individual patient characteristics, disease severity, health literacy, values, and preferences 81. However, a unified approach to patient education and self-care has not yet been adopted across the heterogenous healthcare systems in Europe 2. Therefore, healthcare professionals engaged in the management of HF patients should dedicate time and effort in patient education adapted to individual clinical needs and health literacy levels.
| Self-care maintenance |
|---|
| Adherence to medications |
| Healthy diet and nutritional supplementation |
| Physical activity |
| Smoking cessation |
| Moderate alcohol consumption |
| Immunisation |
| Self-care monitoring |
| Early recognition of worsening HF signs and symptoms |
| Self-care management |
| Adjustments in diuretic doses in response to signs of fluid overload |
| Adjustments in other medication doses in response to symptoms |
| Adjustment in physical activity |
| Seeking medical assistance when necessary |
Conclusions
Over the recent years pre- and postdischarge management of patients admitted for HF has received considerable scientific interest and advanced significantly. It now offers evidence-based options to improve patients’ well-being and reduce the risk of hospitalisation and mortality. These options entail appropriate management of congestion, continuation or initiation and optimisation of GDMT before discharge, and postdischarge efforts to secure early and sustained follow-up, rapid GDMT up-titration to target doses, and access to multidisciplinary care addressing comorbidities, cardiac rehabilitation, and patient education. Persisting efforts to strengthen self-care capacities in both patients and their family members and broader utilisation of novel digital health technologies can also lead to improved uptake and adherence to medical treatment. Yet, gaps remain between guideline recommendations and their application in clinical practice. Despite numerous challenges, it is incumbent upon healthcare providers to close these gaps and enhance the quality of HF care.
References
- Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. Nov 10 2018;392(10159):1789-1858. doi:10.1016/s0140-6736(18)32279-7
- Seferović PM, Vardas P, Jankowska EA, et al. The Heart Failure Association Atlas: Heart Failure Epidemiology and Management Statistics 2019. Eur J Heart Fail. Jun 2021;23(6):906-914. doi:10.1002/ejhf.2143
- Greene SJ, Fonarow GC, Vaduganathan M, Khan SS, Butler J, Gheorghiade M. The vulnerable phase after hospitalization for heart failure. Nat Rev Cardiol. Apr 2015;12(4):220-9. doi:10.1038/nrcardio.2015.14
- McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. Sep 21 2021;42(36):3599-3726. doi:10.1093/eurheartj/ehab368
- McDonagh TA, Metra M, Adamo M, et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Developed by the task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) With the special contribution of the Heart Failure Association (HFA) of the ESC. European Heart Journal. 2023;44(37):3627-3639. doi:10.1093/eurheartj/ehad195
- Mebazaa A, Davison B, Chioncel O, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure (STRONG-HF): a multinational, open-label, randomised, trial. Lancet. Dec 3 2022;400(10367):1938-1952. doi:10.1016/s0140-6736(22)02076-1
- Voors AA, Angermann CE, Teerlink JR, et al. The SGLT2 inhibitor empagliflozin in patients hospitalized for acute heart failure: a multinational randomized trial. Nature Medicine. 2022/03/01 2022;28(3):568-574. doi:10.1038/s41591-021-01659-1
- Cunningham JW, Vaduganathan M, Claggett BL, et al. Dapagliflozin in Patients Recently Hospitalized With Heart Failure and Mildly Reduced or Preserved Ejection Fraction. Journal of the American College of Cardiology. 2022;80(14):1302-1310. doi:doi:10.1016/j.jacc.2022.07.021
- Chioncel O, Mebazaa A, Maggioni AP, et al. Acute heart failure congestion and perfusion status-impact of the clinical classification on in-hospital and long-term outcomes; insights from the ESC-EORP-HFA Heart Failure Long-Term Registry. Eur J Heart Fail. Nov 2019;21(11):1338-1352. doi:10.1002/ejhf.1492
- Malgie J, Wilde MI, Clephas PRD, et al. Contemporary guideline-directed medical therapy in de novo, chronic, and worsening heart failure patients: First data from the TITRATE-HF study. European Journal of Heart Failure. 2024;26(7):1549-1560. doi:https://doi.org/10.1002/ejhf.3267
- Greene SJ, Butler J, Albert NM, et al. Medical Therapy for Heart Failure With Reduced Ejection Fraction: The CHAMP-HF Registry. J Am Coll Cardiol. Jul 24 2018;72(4):351-366. doi:10.1016/j.jacc.2018.04.070
- Biegus J, Cotter G, Davison BA, et al. The Effects of Burst Steroid Therapy on Short-term Decongestion in Acute Heart Failure Patients With Pro-inflammatory Activation: A Post Hoc Analysis of the CORTAHF Randomized, Open-label, Pilot Trial. J Card Fail. Feb 2025;31(2):354-366. doi:10.1016/j.cardfail.2024.09.002
- Martens P, Nijst P, Mullens W. Current Approach to Decongestive Therapy in Acute Heart Failure. Curr Heart Fail Rep. Dec 2015;12(6):367-78. doi:10.1007/s11897-015-0273-5
- Ambrosy AP, Pang PS, Khan S, et al. Clinical course and predictive value of congestion during hospitalization in patients admitted for worsening signs and symptoms of heart failure with reduced ejection fraction: findings from the EVEREST trial. Eur Heart J. Mar 2013;34(11):835-43. doi:10.1093/eurheartj/ehs444
- Pagnesi M, Staal L, Ter Maaten JM, et al. Decongestion and Outcomes in Patients Hospitalized for Acute Heart Failure: Insights From the RELAX-AHF-2 Trial. JACC Heart Fail. Mar 2025;13(3):414-429. doi:10.1016/j.jchf.2024.09.013
- Odajima S, Fujimoto W, Kuroda K, et al. Association of congestion with worsening renal function in acute decompensated heart failure according to age. ESC Heart Fail. Dec 2022;9(6):4250-4261. doi:10.1002/ehf2.14157
- Chioncel O, Benson L, Crespo-Leiro MG, et al. Comprehensive characterization of non-cardiac comorbidities in acute heart failure: an analysis of ESC-HFA EURObservational Research Programme Heart Failure Long-Term Registry. Eur J Prev Cardiol. Sep 20 2023;30(13):1346-1358. doi:10.1093/eurjpc/zwad151
- Metra M, Adamo M, Tomasoni D, et al. Pre-discharge and early post-discharge management of patients hospitalized for acute heart failure: A scientific statement by the Heart Failure Association of the ESC. Eur J Heart Fail. Jul 2023;25(7):1115-1131. doi:10.1002/ejhf.2888
- Salah K, Kok WE, Eurlings LW, et al. A novel discharge risk model for patients hospitalised for acute decompensated heart failure incorporating N-terminal pro-B-type natriuretic peptide levels: a European coLlaboration on Acute decompeNsated Heart Failure: ELAN-HF Score. Heart. Jan 2014;100(2):115-25. doi:10.1136/heartjnl-2013-303632
- Núñez J, Llàcer P, García-Blas S, et al. CA125-Guided Diuretic Treatment Versus Usual Care in Patients With Acute Heart Failure and Renal Dysfunction. Am J Med. Mar 2020;133(3):370-380.e4. doi:10.1016/j.amjmed.2019.07.041
- Feola M, Testa M, Ferreri C, et al. Role of Response-to-Diuretic in Predicting Prognosis in Discharged Heart Failure Patients After an Acute Decompensation. Archives of Medical Research. 2018/04/01/ 2018;49(3):198-204. doi:https://doi.org/10.1016/j.arcmed.2018.08.007
- Chen Y, Ji X-w, Zhang A-y, Lv J-c, Zhang J-g, Zhao C-h. Prognostic Value of Plasma Ghrelin in Predicting the Outcome of Patients with Chronic Heart Failure. Archives of Medical Research. 2014/04/01/ 2014;45(3):263-269. doi:https://doi.org/10.1016/j.arcmed.2014.01.004
- Collins SP, Lindsell CJ, Storrow AB, Abraham WT. Prevalence of negative chest radiography results in the emergency department patient with decompensated heart failure. Ann Emerg Med. Jan 2006;47(1):13-8. doi:10.1016/j.annemergmed.2005.04.003
- Kobayashi M, Watanabe M, Coiro S, et al. Mid-term prognostic impact of residual pulmonary congestion assessed by radiographic scoring in patients admitted for worsening heart failure. Int J Cardiol. Aug 15 2019;289:91-98. doi:10.1016/j.ijcard.2019.01.091
- Thavendiranathan P, Yingchoncharoen T, Grant A, et al. Prediction of 30-day heart failure-specific readmission risk by echocardiographic parameters. Am J Cardiol. Jan 15 2014;113(2):335-41. doi:10.1016/j.amjcard.2013.09.025
- Öhman J, Harjola VP, Karjalainen P, Lassus J. Assessment of early treatment response by rapid cardiothoracic ultrasound in acute heart failure: Cardiac filling pressures, pulmonary congestion and mortality. Eur Heart J Acute Cardiovasc Care. Jun 2018;7(4):311-320. doi:10.1177/2048872617708974
- Goonewardena SN, Gemignani A, Ronan A, et al. Comparison of hand-carried ultrasound assessment of the inferior vena cava and N-terminal pro-brain natriuretic peptide for predicting readmission after hospitalization for acute decompensated heart failure. JACC Cardiovasc Imaging. Sep 2008;1(5):595-601. doi:10.1016/j.jcmg.2008.06.005
- Picano E, Pellikka PA. Ultrasound of extravascular lung water: a new standard for pulmonary congestion. Eur Heart J. Jul 14 2016;37(27):2097-104. doi:10.1093/eurheartj/ehw164
- Coiro S, Rossignol P, Ambrosio G, et al. Prognostic value of residual pulmonary congestion at discharge assessed by lung ultrasound imaging in heart failure. Eur J Heart Fail. Nov 2015;17(11):1172-81. doi:10.1002/ejhf.344
- Gargani L, Pang PS, Frassi F, et al. Persistent pulmonary congestion before discharge predicts rehospitalization in heart failure: a lung ultrasound study. Cardiovasc Ultrasound. Sep 4 2015;13:40. doi:10.1186/s12947-015-0033-4
- Mullens W, Dauw J, Martens P, et al. Acetazolamide in Acute Decompensated Heart Failure with Volume Overload. N Engl J Med. Sep 29 2022;387(13):1185-1195. doi:10.1056/NEJMoa2203094
- Trullàs JC, Morales-Rull JL, Casado J, et al. Combining loop with thiazide diuretics for decompensated heart failure: the CLOROTIC trial. European Heart Journal. 2022;44(5):411-421. doi:10.1093/eurheartj/ehac689
- ter Maaten JM, Beldhuis IE, van der Meer P, et al. Natriuresis-guided diuretic therapy in acute heart failure: a pragmatic randomized trial. Nature Medicine. 2023/10/01 2023;29(10):2625-2632. doi:10.1038/s41591-023-02532-z
- Palin V, Drozd M, Garland E, et al. Reduction of heart failure guideline-directed medication during hospitalization: prevalence, risk factors, and outcomes. ESC Heart Fail. Oct 2022;9(5):3298-3307. doi:10.1002/ehf2.14051
- Kimmoun A, Cotter G, Davison B, et al. Safety, Tolerability and efficacy of Rapid Optimization, helped by NT-proBNP and GDF-15, of Heart Failure therapies (STRONG-HF): rationale and design for a multicentre, randomized, parallel-group study. Eur J Heart Fail. Nov 2019;21(11):1459-1467. doi:10.1002/ejhf.1575
- Biegus J, Mebazaa A, Davison B, et al. Effects of Rapid Uptitration of Neurohormonal Blockade on Effective, Sustainable Decongestion and Outcomes in STRONG-HF. J Am Coll Cardiol. Jul 23 2024;84(4):323-336. doi:10.1016/j.jacc.2024.04.055
- Čelutkienė J, Čerlinskaitė-Bajorė K, Cotter G, et al. Impact of Rapid Up-Titration of Guideline-Directed Medical Therapies on Quality of Life: Insights From the STRONG-HF Trial. Circ Heart Fail. Apr 2024;17(4):e011221. doi:10.1161/circheartfailure.123.011221
- Adamo M, Pagnesi M, Mebazaa A, et al. NT-proBNP and high intensity care for acute heart failure: the STRONG-HF trial. Eur Heart J. Aug 14 2023;44(31):2947-2962. doi:10.1093/eurheartj/ehad335
- Biegus J, Voors AA, Collins SP, et al. Impact of empagliflozin on decongestion in acute heart failure: the EMPULSE trial. Eur Heart J. Jan 1 2023;44(1):41-50. doi:10.1093/eurheartj/ehac530
- Vardeny O, Claggett B, Kachadourian J, et al. Reduced loop diuretic use in patients taking sacubitril/valsartan compared with enalapril: the PARADIGM-HF trial. Eur J Heart Fail. Mar 2019;21(3):337-341. doi:10.1002/ejhf.1402
- Solomon SD, McMurray JJV, Vaduganathan M, et al. Finerenone in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. Oct 24 2024;391(16):1475-1485. doi:10.1056/NEJMoa2407107
- Vaduganathan M, Claggett BL, Desai AS, et al. Time to Significant Benefit of Finerenone in Patients With HeartFailure. Journal of the American College of Cardiology. 2025/01/21/ 2025;85(2):199-202. doi:https://doi.org/10.1016/j.jacc.2024.09.018
- Vaduganathan M, Claggett BL, Jhund P, et al. Time to Clinical Benefit of Dapagliflozin in Patients With Heart Failure With Mildly Reduced or Preserved Ejection Fraction: A Prespecified Secondary Analysis of the DELIVER Randomized Clinical Trial. JAMA Cardiology. 2022;7(12):1259-1263. doi:10.1001/jamacardio.2022.3750
- Voors AA, Dittrich HC, Massie BM, et al. Effects of the adenosine A1 receptor antagonist rolofylline on renal function in patients with acute heart failure and renal dysfunction: results from PROTECT (Placebo-Controlled Randomized Study of the Selective Adenosine A1 Receptor Antagonist Rolofylline for Patients Hospitalized with Acute Decompensated Heart Failure and Volume Overload to Assess Treatment Effect on Congestion and Renal Function). J Am Coll Cardiol. May 10 2011;57(19):1899-907. doi:10.1016/j.jacc.2010.11.057
- Gattis WA, O’Connor CM, Gallup DS, Hasselblad V, Gheorghiade M. Predischarge initiation of carvedilol in patients hospitalized for decompensated heart failure: results of the Initiation Management Predischarge: Process for Assessment of Carvedilol Therapy in Heart Failure (IMPACT-HF) trial. J Am Coll Cardiol. May 5 2004;43(9):1534-41. doi:10.1016/j.jacc.2003.12.040
- Sanam K, Bhatia V, Bajaj NS, et al. Renin-Angiotensin System Inhibition and Lower 30-Day All-Cause Readmission in Medicare Beneficiaries with Heart Failure. Am J Med. Oct 2016;129(10):1067-73. doi:10.1016/j.amjmed.2016.05.008
- Velazquez EJ, Morrow DA, DeVore AD, et al. Angiotensin-Neprilysin Inhibition in Acute Decompensated Heart Failure. N Engl J Med. Feb 7 2019;380(6):539-548. doi:10.1056/NEJMoa1812851
- Wachter R, Senni M, Belohlavek J, et al. Initiation of sacubitril/valsartan in haemodynamically stabilised heart failure patients in hospital or early after discharge: primary results of the randomised TRANSITION study. Eur J Heart Fail. Aug 2019;21(8):998-1007. doi:10.1002/ejhf.1498
- Berg DD, Braunwald E, DeVore AD, et al. Efficacy and Safety of Sacubitril/Valsartan by Dose Level Achieved in the PIONEER-HF Trial. JACC Heart Fail. Oct 2020;8(10):834-843. doi:10.1016/j.jchf.2020.06.008
- Fonarow GC, Abraham WT, Albert NM, et al. Prospective Evaluation of Beta-Blocker Use at the Time of Hospital Discharge as a Heart Failure Performance Measure: Results From OPTIMIZE-HF. Journal of Cardiac Failure. 2007;13(9):722-731. doi:10.1016/j.cardfail.2007.06.727
- Fonarow GC, Abraham WT, Albert NM, et al. Influence of beta-blocker continuation or withdrawal on outcomes in patients hospitalized with heart failure: findings from the OPTIMIZE-HF program. J Am Coll Cardiol. Jul 15 2008;52(3):190-9. doi:10.1016/j.jacc.2008.03.048
- Lee KK, Yang J, Hernandez AF, Steimle AE, Go AS. Post-discharge Follow-up Characteristics Associated With 30-Day Readmission After Heart Failure Hospitalization. Med Care. Apr 2016;54(4):365-72. doi:10.1097/mlr.0000000000000492
- McAlister FA, Youngson E, Kaul P, Ezekowitz JA. Early Follow-Up After a Heart Failure Exacerbation: The Importance of Continuity. Circ Heart Fail. Sep 2016;9(9) doi:10.1161/circheartfailure.116.003194
- Savarese G, Lund LH, Dahlström U, Strömberg A. Nurse-Led Heart Failure Clinics Are Associated With Reduced Mortality but Not Heart Failure Hospitalization. J Am Heart Assoc. May 21 2019;8(10):e011737. doi:10.1161/jaha.118.011737
- Logeart D, Berthelot E, Bihry N, et al. Early and short-term intensive management after discharge for patients hospitalized with acute heart failure: a randomized study (ECAD-HF). European Journal of Heart Failure. 2022;24(1):219-226. doi:https://doi.org/10.1002/ejhf.2357
- Savarese G, Kishi T, Vardeny O, et al. Heart Failure Drug Treatment-Inertia,Titration, and Discontinuation: A Multinational Observational Study (EVOLUTION HF). JACC: Heart Failure. 2023/01/01/ 2023;11(1):1-14. doi:https://doi.org/10.1016/j.jchf.2022.08.009
- Tomasoni D, Pagnesi M, Colombo G, et al. Guideline-directed medical therapy in severe heart failure with reduced ejection fraction: An analysis from the HELP-HF registry. Eur J Heart Fail. Feb 2024;26(2):327-337. doi:10.1002/ejhf.3081
- Seferović PM, Polovina M, Adlbrecht C, et al. Navigating between Scylla and Charybdis: challenges and strategies for implementing guideline-directed medical therapy in heart failure with reduced ejection fraction. Eur J Heart Fail. Dec 2021;23(12):1999-2007. doi:10.1002/ejhf.2378
- Khan MS, Segar MW, Usman MS, et al. Frailty, Guideline-Directed Medical Therapy, and Outcomes in HFrEF: From the GUIDE-IT Trial. JACC Heart Fail. Apr 2022;10(4):266-275. doi:10.1016/j.jchf.2021.12.004
- Arrigo M, Biegus J, Asakage A, et al. Safety, tolerability and efficacy of up-titration of guideline-directed medical therapies for acute heart failure in elderly patients: A sub-analysis of the STRONG-HF randomized clinical trial. Eur J Heart Fail. Jul 2023;25(7):1145-1155. doi:10.1002/ejhf.2920
- Savarese G, Lindberg F, Christodorescu RM, et al. Physician perceptions, attitudes, and strategies towards implementing guideline-directed medical therapy in heart failure with reduced ejection fraction. A survey of the Heart Failure Association of the ESC and the ESC Council for Cardiology Practice. European Journal of Heart Failure. 2024;26(6):1408-1418. doi:https://doi.org/10.1002/ejhf.3214
- Jalloh MB, Granger CB, Fonarow GC, Van Spall HGC. Multi-level implementation strategies to improve uptake of evidence-based therapies in heart failure. Eur Heart J. Jun 20 2023;44(23):2055-2058. doi:10.1093/eurheartj/ehad150
- van der Wal MH, Jaarsma T, van Veldhuisen DJ. Non-compliance in patients with heart failure; how can we manage it? Eur J Heart Fail. Jan 2005;7(1):5-17. doi:10.1016/j.ejheart.2004.04.007
- Jonkman NH, Westland H, Groenwold RH, et al. What Are Effective Program Characteristics of Self-Management Interventions in Patients With Heart Failure? An Individual Patient Data Meta-analysis. J Card Fail. Nov 2016;22(11):861-871. doi:10.1016/j.cardfail.2016.06.422
- Unverzagt S, Meyer G, Mittmann S, Samos FA, Unverzagt M, Prondzinsky R. Improving Treatment Adherence in Heart Failure. Dtsch Arztebl Int. Jun 24 2016;113(25):423-30. doi:10.3238/arztebl.2016.0423
- Wang L, Zhao Y, Han L, et al. Pharmacist-Led Management Model and Medication Adherence Among Patients With Chronic Heart Failure: A Randomized Clinical Trial. JAMA Netw Open. Dec 2 2024;7(12):e2453976. doi:10.1001/jamanetworkopen.2024.53976
- Goldstein CM, Gathright EC, Dolansky MA, et al. Randomized controlled feasibility trial of two telemedicine medication reminder systems for older adults with heart failure. J Telemed Telecare. Sep 2014;20(6):293-9. doi:10.1177/1357633×14541039
- Stolfo D, Iacoviello M, Chioncel O, et al. How to handle polypharmacy in heart failure. A clinical consensus statement of the Heart Failure Association of the ESC. European Journal of Heart Failure. n/a(n/a) doi:https://doi.org/10.1002/ejhf.3642
- Pandey A, Keshvani N, Wang TJ. Should Polypills Be Used for Heart Failure With Reduced Ejection Fraction? Circulation. 2022;146(4):276-278. doi:doi:10.1161/CIRCULATIONAHA.122.059661
- Ruppar TM, Cooper PS, Mehr DR, Delgado JM, Dunbar-Jacob JM. Medication Adherence Interventions Improve Heart Failure Mortality and Readmission Rates: Systematic Review and Meta-Analysis of Controlled Trials. J Am Heart Assoc. Jun 17 2016;5(6) doi:10.1161/jaha.115.002606
- Tomasoni D, Vitale C, Guidetti F, et al. The role of multimorbidity in patients with heart failure across the left ventricular ejection fraction spectrum: Data from the Swedish Heart Failure Registry. Eur J Heart Fail. Apr 2024;26(4):854-868. doi:10.1002/ejhf.3112
- Denfeld QE, Winters-Stone K, Mudd JO, Gelow JM, Kurdi S, Lee CS. The prevalence of frailty in heart failure: A systematic review and meta-analysis. Int J Cardiol. Jun 1 2017;236:283-289. doi:10.1016/j.ijcard.2017.01.153
- Seferović PM, Piepoli MF, Lopatin Y, et al. Heart Failure Association of the European Society of Cardiology Quality of Care Centres Programme: design and accreditation document. Eur J Heart Fail. May 2020;22(5):763-774. doi:10.1002/ejhf.1784
- Bonilla-Palomas JL, Gámez-López AL, Castillo-Domínguez JC, et al. Nutritional Intervention in Malnourished Hospitalized Patients with Heart Failure. Archives of Medical Research. 2016/10/01/ 2016;47(7):535-540. doi:https://doi.org/10.1016/j.arcmed.2016.11.005
- Holland R, Battersby J, Harvey I, Lenaghan E, Smith J, Hay L. Systematic review of multidisciplinary interventions in heart failure. Heart. Jul 2005;91(7):899-906. doi:10.1136/hrt.2004.048389
- Kitzman DW, Whellan DJ, Duncan P, et al. Physical Rehabilitation for Older Patients Hospitalized for Heart Failure. N Engl J Med. Jul 15 2021;385(3):203-216. doi:10.1056/NEJMoa2026141
- Nadruz W, Jr., West E, Sengeløv M, et al. Prognostic Value of Cardiopulmonary Exercise Testing in Heart Failure With Reduced, Midrange, and Preserved Ejection Fraction. J Am Heart Assoc. Oct 31 2017;6(11) doi:10.1161/jaha.117.006000
- Swank AM, Horton J, Fleg JL, et al. Modest increase in peak VO2 is related to better clinical outcomes in chronic heart failure patients: results from heart failure and a controlled trial to investigate outcomes of exercise training. Circ Heart Fail. Sep 1 2012;5(5):579-85. doi:10.1161/circheartfailure.111.965186
- Resurrección DM, Moreno-Peral P, Gómez-Herranz M, et al. Factors associated with non-participation in and dropout from cardiac rehabilitation programmes: a systematic review of prospective cohort studies. Eur J Cardiovasc Nurs. Jan 2019;18(1):38-47. doi:10.1177/1474515118783157
- Lee CS, Bidwell JT, Paturzo M, et al. Patterns of self-care and clinical events in a cohort of adults with heart failure: 1 year follow-up. Heart Lung. Jan-Feb 2018;47(1):40-46. doi:10.1016/j.hrtlng.2017.09.004
- Jaarsma T, Hill L, Bayes-Genis A, et al. Self-care of heart failure patients: practical management recommendations from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. Jan 2021;23(1):157-174. doi:10.1002/ejhf.2008

