Oral Biofilms: Impact on Diabetes and Systemic Health
Oral Biofilms and its Connection to Diabetes, Kidney Disease and Pancreatic Cancer
Gregori M. Kurtzman1, DDS; Robert A. Horowitz2, DDS; Richard Johnson3, MD; Zachariah Pedro4, BA
- Gregori M. Kurtzman, DDS General Dentistry and Implantology, General Dental Practice, Silver Spring, Maryland, USA
- Robert A. Horowitz, DDS Periodontology, New York University
- Richard Johnson, MD Internal Medicine, Retired, New York, USA
- Zachariah Pedro, BA Psychiatry, New York University, New York, USA
OPEN ACCESS
PUBLISHED: 30 June 2025
CITATION: Kurtzman, GM., Horowitz, RA., et al., 2025. Oral Biofilms and its Connection to Diabetes, Kidney Disease and Pancreatic Cancer. Medical Research Archives, [online] 13(6). https://doi.org/10.18103/mra.v13i6.6700
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i6.6700
ISSN 2375-1924
ABSTRACT
Systemic and oral health are interconnected via bacteria which may originate in the oral biofilm (plaque) which can circulate through the circulatory system. Oral biofilm and its associated periodontal health have not been frequently addressed in patients with systemic health issues. This is especially true for patients who do not respond to medical treatment from their physician for systemic issues. The clinical presence of periodontal disease (bleeding on probing, gingival inflammation) may have oral biofilm present. Periodontal reaction is dependent on the patient’s immune response to the associated bacteria and their byproducts present in the oral biofilm. Increasing evidence has emerged in recent years that are connecting oral biofilms with systemic conditions, either by initiating them or by complicating those medical conditions. Patient health needs to be considered as a whole-body system with connections that may originate in the oral cavity and have distant effects throughout the body. In order to maximize the patient’s total health, healthcare needs to be a coordinated effort between the physician and dentist to eliminate the oral biofilm and aid in the prevention of systemic disease or minimize these effects to improve the overall patient health and their quality of life. Various systemic health areas have been associated with the bacteria in oral biofilms and their byproducts. Specifically, diabetes, chronic kidney disease and pancreatic cancer have been connected to the oral biofilm. This article will discuss oral biofilm, its systemic effects related to diabetes, kidney disease, pancreatic issues and methods to control the oral biofilm and affect positive changes in the diabetic patient.
Keywords: Oral biofilm, chronic kidney disease, diabetes, pancreatic cancer
Introduction
The role of physicians in the management of systemic health is central to patient care. From early detection to long-term management, physicians are responsible for addressing a wide range of chronic conditions. However, an often-overlooked contributor to systemic disease is the presence of oral biofilm—complex microbial communities that reside in the oral cavity and play a pivotal role in the development and progression of systemic inflammation and disease. The identification and management of these oral pathogens typically fall within the scope of dental professionals. Consequently, effective management of patients with chronic systemic conditions necessitates a collaborative approach between medical and dental practitioners.
An expanding body of evidence has established associations between oral biofilm and various systemic diseases, particularly diabetes mellitus, chronic kidney disease, and pancreatic dysfunction. Oral pathogens, through mechanisms such as endotoxemia, immune modulation, and sustained inflammatory responses, have been shown to exacerbate metabolic dysregulation and impair systemic disease control. In individuals with underlying systemic conditions, the presence of chronic oral infection—particularly periodontitis—may not only initiate disease processes but also compromise the effectiveness of medical interventions.
A paradigm shift toward integrated, whole-body care is essential. Physicians and dentists must engage in coordinated care strategies to identify and reduce the burden of oral biofilm as a modifiable risk factor for systemic disease. Interdisciplinary collaboration can enhance disease prevention, improve therapeutic outcomes, and ultimately elevate the patient’s overall quality of life. This article explores the pathophysiological relationship between oral biofilm and systemic conditions, with a particular focus on diabetes, renal function, and pancreatic health. It also provides clinical considerations for incorporating oral health into systemic disease management frameworks.
What is an Oral Biofilm?
Oral biofilm—previously referred to simply as dental plaque—has gained increased scientific recognition as a complex, highly structured microbial ecosystem central to both oral and systemic health. Traditionally associated with periodontal disease and, to a lesser extent, dental caries, oral biofilm has historically been viewed as a localized dental concern. However, mounting evidence now implicates oral biofilm as a potential contributor to systemic disease, with the American Dental Association reporting over 200 documented links between oral and systemic health conditions.
This growing body of literature has demonstrated significant associations between periodontal disease, chronic oral inflammation, and a range of systemic disorders. These include cardiovascular disease, chronic kidney disease, diabetes mellitus, osteoporosis, respiratory illnesses, Alzheimer’s disease, and other inflammatory conditions. Such findings underscore the evolving understanding of oral biofilm as a dynamic and influential factor in whole-body health.
Oral biofilm is composed of complex microbial communities that adhere to the surfaces of teeth and reside within the gingival sulcus. These microorganisms are embedded in a self-produced extracellular polymeric matrix composed of both bacterial and host-derived components. Over 700 microbial species inhabit the oral cavity, most of which are commensal or benign under normal conditions. However, certain species have been identified as pathogenic, particularly in the context of ecological imbalance or dysbiosis. As bacterial load increases, these organisms form a multilayered biofilm with structural channels and protective barriers, significantly contributing to the development of periodontal disease.
Biofilm-associated bacteria exhibit heightened resistance to both host immune responses and antimicrobial treatments, whether administered topically or systemically. This resistance is attributed to several factors, including limited antimicrobial penetration, altered microenvironments within the biofilm, and the physiological heterogeneity of resident bacteria. In contrast to planktonic (free-floating) bacteria, biofilm-associated organisms display enhanced pathogenic synergy and resilience, complicating clinical management.
Furthermore, these microbial communities utilize quorum sensing—a cell-to-cell communication mechanism mediated by signaling molecules—to coordinate gene expression and collective behavior, including the production of virulence factors and enzymes. Oral fluids serve as a medium for this communication and facilitate the horizontal transfer of genetic material. The resulting multispecies biofilm exhibits emergent properties that surpass those of individual species alone, creating a highly adaptive and persistent microbial ecosystem.
The recognition of oral biofilm as a medically relevant factor underscores the importance of integrating dental and medical care, particularly for patients with chronic systemic conditions influenced by inflammatory or microbial pathways.
Quorum Sensing and Biofilm Maturation
Quorum sensing, a cell-to-cell communication mechanism used by bacteria, enables coordinated gene expression in response to population density. Within the context of oral biofilms, this system regulates a variety of critical functions, including the production of enzymes that modulate the activity and gene expression of neighboring bacterial species. These secreted byproducts can provoke a host immune response, attracting white blood cells (WBCs) to the site in an attempt to eliminate the microbial invaders. However, through quorum sensing, biofilm bacteria can interfere with chemotactic signaling, effectively disrupting WBC targeting and reducing the efficacy of the immune response. As a result, the localized gingiva becomes inflamed, setting the stage for periodontal tissue damage.
If WBCs fail to phagocytose and neutralize pathogens, they undergo apoptosis and subsequently lyse within approximately 72 hours. Upon lysis, the cytotoxic components originally intended for microbial destruction—such as reactive oxygen species, lysosomal enzymes, and proteases—are released into the surrounding tissue, inadvertently damaging host structures. This collateral tissue damage contributes to the degradation of connective tissue and alveolar bone, deepening periodontal pockets and exacerbating inflammation.
Oral biofilm is a highly diverse and site-specific microbial community. In the same patient, the microbial composition may differ significantly from one periodontal site to another. Once established, the biofilm community tends to maintain local ecological stability due to a dynamic balance of synergistic and antagonistic interspecies interactions. Subgingival regions, such as periodontal pockets, provide an ideal environment for biofilm maturation by offering protection from mechanical disruption during routine oral hygiene practices.
As biofilm matures, its microbial profile shifts dramatically. Initial colonization typically involves predominantly gram-positive cocci, including Streptococcus mutans, Streptococcus oralis, Streptococcus sanguis, Streptococcus mitis, Staphylococcus epidermidis, and Rothia dentocariosa. These are followed by gram-positive rods and filaments such as Actinomyces gerencseriae, Actinomyces viscosus, Actinomyces israelii, and Corynebacterium species. A small population of gram-negative cocci—such as Veillonella parvula and Neisseria species—also emerges in this early stage. These organisms, predominantly aerobic or facultative anaerobes, are highly resistant to natural clearance mechanisms such as mastication, salivary flow, and swallowing. Additionally, they are well-adapted to survive in oxygen-rich environments typical of the oral cavity.
This early-stage biofilm forms almost immediately after tooth cleaning, whether through daily brushing or professional prophylaxis. Over time, and particularly in protected environments like periodontal pockets, the microbial composition transitions to a more pathogenic, anaerobe-dominant structure. Mature biofilms in diseased sites are typically rich in gram-negative anaerobes, which are strongly associated with the pathogenesis of chronic periodontitis.
Biofilm Development, Structure, and Host Variability
The co-adhesion of secondary bacterial colonizers to the early oral biofilm is a critical phase in the formation of a mature and pathogenic microbial community. These interactions are mediated by highly specific receptor-ligand mechanisms between bacterial surface structures, facilitating not only adherence but also the integration of new species into the biofilm. As a result, the biofilm expands in volume and complexity, establishing a polymicrobial environment characterized by intricate biochemical interactions among its constituents. Within this network, bacterial species engage in both synergistic and antagonistic relationships, often supporting the metabolic needs of adjacent organisms through nutrient exchange and environmental conditioning.
Of particular importance is the interaction between obligate aerobes and anaerobes. The metabolic activity of aerobic bacteria can locally deplete oxygen, enabling the survival of obligate anaerobes even within the otherwise oxygen-rich milieu of the oral cavity. Continued bacterial proliferation and spatial organization ultimately result in a three-dimensional, functionally stratified biofilm. A hallmark of this mature structure is the development of an extracellular polymeric matrix composed of polysaccharides, proteins, lipids, and extracellular DNA. This matrix confers significant protective advantages, shielding the embedded microbial population from environmental insults, host immune defenses, and antimicrobial agents.
As the biofilm thickens, it fosters microenvironments that support the persistence of anaerobic pathogens deep within its structure—sites largely inaccessible to mechanical disruption and hostile to oxygen exposure. These microhabitats are crucial to the establishment of chronic periodontal infections and have systemic implications.
It is important to note that the presence of pathogenic oral biofilm does not always correlate with overt clinical signs such as gingival bleeding. While bleeding on probing, gingival inflammation, and tissue destruction are classic indicators of periodontal disease, their absence does not preclude microbial colonization or systemic impact. The identification and diagnosis of periodontal disease rely on comprehensive clinical evaluation by dental professionals, who assess both clinical markers and radiographic evidence of tissue breakdown.
The host immune response plays a central role in the progression and severity of periodontal disease. Variability in immunologic reactivity among patients results in differing clinical presentations. Some individuals exhibit pronounced inflammatory responses with visible signs of disease, while others may harbor significant pathogenic biofilm with minimal or no overt symptoms. This discrepancy underscores the importance of individualized risk assessment and early microbial screening to mitigate the potential systemic consequences of subclinical oral infections.
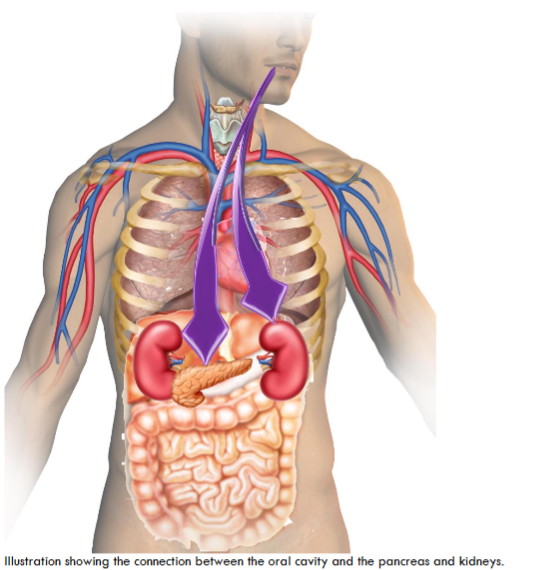
Systemic Implications of Oral Biofilm: Diabetes, Chronic Kidney Disease, and Pancreatic Cancer
DIABETES MELLITUS
Patients with diabetes mellitus face a significantly elevated risk—estimated at twofold—of developing periodontal disease compared to individuals without metabolic dysfunction. Moreover, both Type 1 and Type 2 diabetes are associated with increased prevalence, accelerated progression, and greater severity of periodontal pathology. A robust body of evidence supports a bidirectional relationship between diabetes and periodontal disease, wherein each condition can exacerbate the other. Periodontitis is now widely recognized as the sixth most common complication of diabetes and is an established risk factor for poor glycemic control and progression of diabetic complications.
Patients with chronic periodontal infections often demonstrate worsening glycemic control over time, contributing to the challenges of diabetes management. Importantly, multiple studies have shown that non-surgical periodontal therapy—including scaling and root planing—can lead to measurable improvements in hemoglobin A1c levels, suggesting that periodontal treatment may enhance metabolic outcomes. Accordingly, the management of oral biofilm and periodontal inflammation should be considered a standard component of diabetes care. Patients struggling to achieve glycemic control, whether through pharmacological or nutritional strategies, should be referred for periodontal evaluation. Integration of dental care has shown to improve diabetes outcomes and may serve as a critical adjunct in comprehensive disease management.
CHRONIC KIDNEY DISEASE (CKD)
Chronic kidney disease and cardiovascular disease share numerous modifiable risk factors, including chronic systemic inflammation, which has been linked to periodontal disease. Pathogenic bacteria present in oral biofilms have demonstrated the capacity to adhere to and invade endothelial cells, contributing to atherogenesis and impaired vasomotor function. These vascular effects are not limited to coronary circulation; similar endothelial dysfunction has been observed in renal vasculature.
The leading causes of CKD—diabetes mellitus, hypertension, and glomerulonephritis—account for approximately 75% of all adult cases. Patients with CKD often exhibit several risk factors for periodontal disease, including compromised immunity, limited oral hygiene capacity, and coexisting diabetes. Notably, there is a high prevalence of periodontal conditions among individuals undergoing dialysis, with reported rates of gingivitis and severe periodontitis at 46% and 35%, respectively. This association appears to be bidirectional: while CKD patients are more susceptible to oral disease, untreated periodontal inflammation may, in turn, exacerbate renal impairment, contributing to systemic inflammatory burden and disease progression.
PANCREATIC CANCER
Given the pancreas’s central role in insulin production and its close association with diabetes, it may also be indirectly influenced by chronic oral inflammation and microbial dysbiosis. Although established risk factors for pancreatic cancer include cigarette smoking and chronic pancreatitis, mounting evidence suggests that periodontal disease may also play a contributory role via sustained systemic inflammation.
In a collaborative study by the Harvard School of Public Health and Dana-Farber Cancer Institute, researchers found a significant association between periodontal disease and increased risk of pancreatic cancer. Specifically, men with periodontal disease were found to have a 63% higher risk of developing pancreatic cancer compared to those without such oral pathology. These findings have since been corroborated by subsequent research, underscoring the potential systemic consequences of untreated oral biofilm.
Managing Oral Biofilms: Approaches and Challenges
Effective management of oral biofilms involves a combination of professional dental treatments and patient-driven homecare strategies aimed at controlling biofilm levels and preventing the recurrence of periodontal disease, along with its associated systemic effects.
PROFESSIONAL CARE
Treatment of periodontal disease by a dental professional typically includes mechanical debridement to remove biofilm and restore periodontal tissues to a healthier state. However, this initial debridement alone is insufficient for long-term biofilm control. Despite the dentist’s efforts, without consistent patient involvement, biofilm re-growth can be rapid and challenging to manage.
CHALLENGES IN HOMECARE
Homecare is an essential component of biofilm management; however, standard methods of mechanical debridement, such as toothbrushing, are limited in their ability to effectively remove biofilm from deep periodontal pockets. Toothbrush bristles can only reach up to 3-4 mm subgingivally, making it difficult to eliminate bacteria in deeper pockets. Research indicates that within just three hours following cleaning, biofilm mass can increase by up to 400%. Additionally, other common at-home tools, such as dental floss and oral irrigators, are similarly ineffective at reaching beyond 1–2 mm or adequately irrigating the bottom of deeper pockets. As a result, biofilm regrowth occurs rapidly, often exceeding pre-cleaning levels within two days.
CHEMICAL AGENTS
Chlorhexidine is a widely used oral rinse that can impact young biofilms, but its efficacy diminishes in the face of mature or nutrient-limited biofilms. Furthermore, chlorhexidine has been shown to exhibit cytotoxic effects on fibroblasts and other gingival tissues, raising concerns regarding its long-term use.
An alternative, chlorine dioxide, has shown promising results as a more effective agent for biofilm management. Chlorine dioxide functions by degrading the biofilm matrix and disrupting bacterial cell walls, effectively peeling back the biofilm layer by layer. Unlike chlorhexidine, chlorine dioxide does not exhibit cytotoxicity against fibroblasts or other host tissues, making it safer for daily use. Studies have demonstrated that stabilized chlorine dioxide (e.g., Cloralstan) significantly reduces plaque, gingival indices, and bacterial counts in the oral cavity, with no reported toxicity even at concentrations of 40 ppm. This solution is highly efficient and safe for daily use, with additional benefits when used in conjunction with brushing. Using chlorine dioxide in an oral irrigator can enhance biofilm removal by facilitating deep flushing of periodontal pockets, further improving its effectiveness in reaching areas that standard rinsing cannot.
SPECIAL CONSIDERATIONS FOR DENTURES
Partial and full dentures are also susceptible to biofilm accumulation. Patients who wear dentures, particularly elderly individuals or those with physical limitations, may struggle with adequate denture hygiene. In such cases, caregivers should be instructed to clean the dentures daily with a toothbrush and chlorine dioxide rinse to remove biofilm effectively. This practice helps prevent aspiration of biofilm, which could introduce pathogens into the pulmonary system, thus reducing the risk of pulmonary infections, a common concern among older adults.
ANTIBIOTICS AND BIOFILM RESISTANCE
Oral biofilm bacteria are significantly more resistant to antibiotics than their planktonic counterparts—by as much as 1000 times. As a result, systemic or topical antibiotic therapies, including oral rinses and localized treatments, are largely ineffective in eradicating biofilm-associated bacteria. This resistance has implications for both natural teeth and dental implants, particularly in the context of peri-implantitis, where biofilm formation around implants may lead to chronic infections and complications.
PHOTOBIOMODULATION (PBM)
Photobiomodulation (PBM), also known as low-level laser therapy (LLLT), has emerged as an innovative adjunctive treatment for managing oral biofilms. PBM utilizes red and near-infrared light wavelengths (600-700 nm and 780-1100 nm) to penetrate gingival tissues, effectively targeting and disrupting the biofilm in periodontal pockets. PBM has shown antibacterial effects, particularly against Streptococcus mutans in biofilms, and has demonstrated the potential to reduce caries development. This therapy is especially beneficial for patients who may be unable to maintain routine oral care, such as the elderly, especially those with cognitive decline or dementia, who are at increased risk for oral health deterioration.
PHOTOBIOMODULATION (PBM) IN ORAL BIOFILM MANAGEMENT
Recent studies have demonstrated a significant reduction in bacterial populations when Photobiomodulation (PBM) is applied routinely to the skin. In an intraoral setting, PBM has proven to be an effective adjunctive treatment for various oral diseases, including tooth caries, pulpitis, periodontal diseases, peri-implantitis, and oral candidiasis. PBM is a convenient and user-friendly method to complement standard oral homecare practices such as brushing, flossing, and the use of oral irrigators. In addition to these common oral conditions, PBM has also shown potential in preventing and managing less common but significant conditions such as lichen planus and oral mucositis. These conditions, frequently encountered in the aging population, can be effectively managed through regular use of PBM, enhancing the patient’s overall oral health.
PATIENT COMPLIANCE AND EASE OF USE
One of the key advantages of PBM is its minimal time commitment and ease of use, making it an attractive option for patient compliance. The Accelite device (Accelite, Palm Beach Gardens, FL) is an intraoral PBM device designed for patient convenience. It is placed into the mouth, where the patient gently closes their mouth around it. For edentulous patients, the device should be used without dentures to ensure light penetration into the tissue. Upon activation, the device provides five minutes of irradiation, after which it automatically turns off. It is recommended that patients use this device once daily; however, increased frequency—up to twice daily (morning and evening)—has no adverse effects and may further improve the routine.
Conclusion:
Oral biofilm is far more complex than previously understood, functioning as a dynamic and coordinated community of bacteria protected by a slimy extracellular matrix. Research has shown that the bacteria within oral biofilms are directly responsible for periodontal disease, and more importantly, their effects can extend beyond the oral cavity, influencing a range of systemic conditions. These links are well-supported by extensive scientific literature, with studies showing that oral biofilm can contribute to a variety of systemic health issues.
It is important to note that the absence of visible oral symptoms, such as bleeding on probing or brushing, does not necessarily rule out the presence of periodontal disease or oral biofilm. Therefore, it is highly recommended that patients undergo a comprehensive dental evaluation to detect any underlying periodontal conditions and address any potential systemic effects of oral biofilm. During these evaluations, patients should be educated on improving their daily oral hygiene practices to help manage their biofilm levels effectively.
The Impact of Oral Biofilm on Diabetes Management
Diabetes is a prevalent systemic condition that affects a significant portion of the population. Management typically involves dietary modifications, oral medications to assist with glucose regulation, or insulin injections in cases where patients are resistant to oral medications. Physicians tailor treatment plans based on each patient’s unique needs, risk factors, and health conditions. However, some diabetic patients continue to struggle with glycemic control, even with medications or insulin therapy.
As highlighted in this article, the bacteria and their byproducts in oral biofilms have been shown to have systemic effects that can exacerbate the challenges in managing diabetes. By addressing and controlling oral biofilm, patients with diabetes—particularly those with difficulty stabilizing blood glucose levels—can experience improved outcomes. In such cases, managing oral biofilm can assist patients in achieving better control over their diabetes, supporting the efforts of their healthcare providers and contributing to overall better health management.
The presence of oral biofilm does not always correlate with visible signs such as gingival bleeding during brushing or soft tissue inflammation around the teeth. However, improving daily homecare as part of a consistent routine can significantly reduce the impact of oral biofilm and its associated complications. Addressing oral biofilm effectively not only enhances oral health but can also contribute positively to overall healthcare by preventing systemic issues linked to oral infections. Furthermore, encouraging patients with systemic health conditions to seek regular dental care and improve their oral hygiene practices can have a beneficial effect on their overall health, supporting the management and prevention of complications related to systemic diseases.
References:
- Loos BG. Systemic effects of periodontitis. Int J Dent Hyg. 2006;4(suppl 1):34-38; discussion 50-52. https://www.ada.org/resources/research/science-and-research-institute/oral-health-topics/periodontitis
- Hajishengallis G, Chavakis T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat Rev Immunol. 2021 Jul;21(7):426-440. doi: 10.1038/s41577-020-00488-6. Epub 2021 Jan 28. PMID: 33510490; PMCID: PMC7841384.
- Gilbert P, Maira-Litran T, McBain AJ, Rickard AH, Whyte FW: The physiology and collective recalcitrance of microbial biofilm communities. Adv Microb Physiol. 2002; 46():202-56.
- Kouidhi B, Al Qurashi YM, Chaieb K. Drug resistance of bacterial dental biofilm and the potential use of natural compounds as alternative for prevention and treatment. Microb Pathog. 2015 Mar;80:39-49. doi: 10.1016/j.micpath.2015.02.007. Epub 2015 Feb 21. PMID: 25708507.
- Huang R, Li M, Gregory RL. Bacterial interactions in dental biofilm. Virulence. 2011 Sep-Oct;2(5):435-44. doi: 10.4161/viru.2.5.16140. Epub 2011 Sep 1. PMID: 21778817; PMCID: PMC3322631.
- Wade W, Thompson H, Rybalka A, Vartoukian S.: Uncultured Members of the Oral Microbiome. J Calif Dent Assoc. 2016 Jul;44(7):447-56.
- Prazdnova EV, Gorovtsov AV, Vasilchenko NG, Kulikov MP, Statsenko VN, Bogdanova AA, Refeld AG, Brislavskiy YA, Chistyakov VA, Chikindas ML. Quorum-Sensing Inhibition by Gram-Positive Bacteria. Microorganisms. 2022 Feb 3;10(2):350. doi: 10.3390/microorganisms10020350. PMID: 35208805; PMCID: PMC8875677.
- Marsh PD, Featherstone A, McKee AS, et al.: A microbiological study of early caries of approximal surfaces in schoolchildren. J Dent Res. 1989 Jul; 68(7):1151-4. Larsen T, Fiehn NE. Dental biofilm infections – an update. APMIS. 2017 Apr;125(4):376-384. doi: 10.1111/apm.12688. PMID: 28407420.
- Hua, X, Cook, GS, Costerton, JW, et al.: Intergeneric Communication in Dental Plaque Biofilms. Journal of Bacteriology. 2000. Volume 182. p. 7067-7069.
- National Diabetes Statistics Report, 2014. Centers for Disease Control and Prevention website. www.cdc.gov/diabetes/pubs/statsreport14/national-diabetes-report-web.pdf.
- Stanko P, Izakovicova Holla L.: Bidirectional association between diabetes mellitus and inflammatory periodontal disease. A review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2014;158(1):35-8. doi: 10.5507/bp.2014.005. Epub 2014 Jan 27.
- Mealey BL, Rethman MP. Periodontal disease and diabetes mellitus. Bidirectional relationship. Dent Today. 2003 Apr;22(4):107-13. PMID: 12733412.
- Gurav AN. Periodontal therapy — an adjuvant for glycemic control. Diabetes Metab Syndr. 2012 Oct-Dec;6(4):218-23. doi: 10.1016/j.dsx.2012.09.007. Epub 2012 Oct 24. PMID: 23199544.
- Kumar M, Mishra L, Mohanty R, Nayak R. “Diabetes and gum disease: the diabolic duo”. Diabetes Metab Syndr. 2014 Oct-Dec;8(4):255-8. doi: 10.1016/j.dsx.2014.09.022. Epub 2014 Oct 13. PMID: 25450824.
- Paurobally N, Kruger E, Tennant M. Awareness About the Oral and Systemic Complications of Diabetes Among a Cohort of Diabetic Patients of the Republic of Mauritius. Int Dent J. 2021 Oct;71(5):438-448. doi: 10.1016/j.identj.2020.12.019. Epub 2021 Feb 25. PMID: 33640154; PMCID: PMC9275114.
- Pihlstrom BL, Michalowicz BS, Johnson NW. Periodontal diseases. Lancet. 2005;366(9499):1809-1820.
- Bascones-Martínez A, Muñoz-Corcuera M, Noronha S, et al. Host defense mechanisms against bacterial aggression in periodontal disease: Basic mechanisms. Med Oral Patol Oral Cir Bucal. 2009;14(12):e680-e685.
- Palmer BF.: Management of hypertension in patients with chronic kidney disease and diabetes mellitus. Am J Med. 2008 Aug;121(8 Suppl):S16-22. doi: 10.1016/j.amjmed.2008.05.018.
- DePaola DP, ed. Periodontitis and renal disease. Colgate Oral Care Report. 2007;17(4). Li L, Zhang YL, Liu XY, Meng X, Zhao RQ, Ou LL, Li BZ, Xing T. Periodontitis Exacerbates and Promotes the Progression of Chronic Kidney Disease Through Oral Flora, Cytokines, and Oxidative Stress. Front Microbiol. 2021 Jun 11;12:656372. doi: 10.3389/fmicb.2021.656372. PMID: 34211440; PMCID: PMC8238692.
- Buhlin K, Bárány P, Heimbürger O, et al. Oral health and pro-inflammatory status in end-stage renal disease patients. Oral Health Prev Dent. 2007;5(3):235-244.
- Colucci F.: An oral commensal associates with disease: chicken, egg, or red herring? Immunity. 2015 Feb 17;42(2):208-10. doi: 10.1016/j.immuni.2015.01.024.
- “Periodontal Disease and Pancreatic Cancer Linked” Article date: 1/19/07, www.medicalnewstoday.com https://www.health.harvard.edu/staying-healthy/gum-disease-may-signal-warning-for-pancreatic-cancer
- Michaud DS.: Role of bacterial infections in pancreatic cancer. Carcinogenesis. 2013 Oct;34(10):2193-7. doi: 10.1093/carcin/bgt249. Epub 2013 Jul 10.
- Palmer, RJ, Caldwell DE.: A flowcell for the study of plaque removal and regrowth. J Micro Methods 1995;24(2):171-82.
- Ng E, Lim LP. An Overview of Different Interdental Cleaning Aids and Their Effectiveness. Dent J (Basel). 2019 Jun 1;7(2):56. doi: 10.3390/dj7020056. PMID: 31159354; PMCID: PMC6630384.
- Teles FR, Teles RP, Sachdeo A.: Comparison of microbial changes in early redeveloping biofilms on natural teeth and dentures. J Periodontol. 2012 Sep;83(9):1139-48. doi: 10.1902/jop.2012.110506. Epub 2012 Mar 23.
- Shen Y, Stojicic S, Haapasalo M.: Antimicrobial efficacy of chlorhexidine against bacteria in biofilms at different stages of development. J Endod. 2011 May;37(5):657-61. doi: 10.1016/j.joen.2011.02.007. Epub 2011 Mar 23.
- Wyganowska-Swiatkowska M, Kotwicka M, Urbaniak P, Nowak A, Skrzypczak-Jankun E, Jankun J. Clinical implications of the growth-suppressive effects of chlorhexidine at low and high concentrations on human gingival fibroblasts and changes in morphology. Int J Mol Med. 2016 Jun;37(6):1594-600. doi: 10.3892/ijmm.2016.2550. Epub 2016 Apr 7. PMID: 27082817.
- Kerémi B, Márta K, Farkas K, et al. Effects of chlorine dioxide on oral hygiene–A systematic review and meta-analysis. Curr Pharm Des. 2020;26(25):3015-3025. doi:10.2174/1381612826666200515134450.
- Ma JW, Huang BS, Hsu CW, et al. Efficacy and safety evaluation of a chlorine dioxide solution. Int J Environ Res Public Health. 2017 Mar 22;14(3):329. doi:10.3390/ijerph14030329.
- Raghavendran K, Mylotte JM, Scannapieco FA. Nursing home-associated pneumonia, hospital-acquired pneumonia and ventilator-associated pneumonia: the contribution of dental biofilms and periodontal inflammation. Periodontology 2000. 2007;44:164.
- Kouidhi B, Al Qurashi YM, Chaieb K.: Drug resistance of bacterial dental biofilm and the potential use of natural compounds as alternative for prevention and treatment. Microb Pathog. 2015 Mar;80:39-49. doi: 10.1016/j.micpath.2015.02.007. Epub 2015 Feb 21.
- Rams TE, Degener JE, van Winkelhoff AJ.: Antibiotic resistance in human chronic periodontitis microbiota. J Periodontol. 2014 Jan;85(1):160-9. doi: 10.1902/jop.2013.130142. Epub 2013 May 20.
- Palma LF, Porfírio TDS, Serrano RV, Campos L, de Moraes LOC. Photobiomodulation Therapy for Managing Motor and Sensory Dysfunctions Following Temporomandibular Joint Surgery: A Case Report. J Lasers Med Sci. 2023 Dec 28;14:e67. doi: 10.34172/jlms.2023.67. PMID: 38318222; PMCID: PMC10843219.
- Balhaddad AA, AlQranei MS, Ibrahim MS, Weir MD, Martinho FC, Xu HHK, Melo MAS. Light Energy Dose and Photosensitizer Concentration Are Determinants of Effective Photo-Killing against Caries-Related Biofilms. Int J Mol Sci. 2020 Oct 15;21(20):7612. doi: 10.3390/ijms21207612. PMID: 33076241; PMCID: PMC7589159.
- Barroso RA, Navarro R, Tim CR, de Paula Ramos L, de Oliveira LD, Araki ÂT, Fernandes KGC, Macedo D, Assis L. Antimicrobial photodynamic therapy against Propionibacterium acnes biofilms using hypericin (Hypericum perforatum) photosensitizer: in vitro study. Lasers Med Sci. 2021 Aug;36(6):1235-1240. doi: 10.1007/s10103-020-03163-3. Epub 2020 Oct 21. PMID: 33083912.
- Huang S, Qi M, Chen Y. Photonics-based treatments: Mechanisms and applications in oral infectious diseases. Front Microbiol. 2023 Feb 10;14:948092. doi: 10.3389/fmicb.2023.948092. PMID: 36846804; PMCID: PMC9950554.
- Soh WK, Cheah KF, Veettil SK, Pandiar D, Nimbalkar S, Gopinath D. Photobiomodulation Therapy in the Management of Oral Lichen Planus: A Systematic Review and Meta-Analysis. Eur J Dent. 2024 May 14. doi: 10.1055/s-0044-1782213. Epub ahead of print. PMID: 38744337.
- Jabłoński P, Musiał M, Wiench R, Stefanik N, Olchowy C, Matys J, Skaba D, Grzech-Leśniak K. Photobiomodulation Therapy in the Treatment of Oral Mucositis-A Case Report. Medicina (Kaunas). 2022 Apr 29;58(5):618. doi: 10.3390/medicina58050618. PMID: 35630035; PMCID: PMC9147415.




