Osteosarcoma Insights: Diagnosis and Clinical Challenges
Osteosarcoma or Not: Diagnostic Challenges, Differentials and a few Clinical Cases
Athema L. Etzioni1
- Department of Pathobiology, Tuskegee University College of Veterinary Medicine, 1200 West Montgomery Road, Tuskegee, Alabama, 36088
E-mail: [email protected]
OPEN ACCESS
PUBLISHED: 31 December 2024
CITATION: Etzioni, A.L., 2024. Osteosarcoma or Not: Diagnostic Challenges, Differentials and a few Clinical Cases. Medical Research Archives, [online] 12(12).
https://doi.org/10.18103/mra.v12i12.6059
COPYRIGHT: © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i12.6059
ISSN 2375-1924
ABSTRACT
Osteosarcoma, a mesenchymal tumor, arises from proliferation of malignant osteoblasts. It is important to observe clinical history, perform a thorough physical examination, and complete appropriate diagnostic testing to differentiate osteosarcoma from other conditions; whether neoplastic, trauma-induced or inflammatory in nature with similar compiled information and background. Cytology may serve as a screening tool to determine whether a swelling represents a neoplastic lesion or an inflammatory process.
Benign processes may include inflammatory lesions such as an abscess; reactive conditions, such as hypersensitivity reactions; hematomas or seromas. Neoplastic conditions may also be benign, such as cysts, fibromas or adenomas or may be malignant. Malignant conditions may include, but are not limited to soft tissue sarcomas, fibrosarcomas or other sarcomas, adenocarcinomas or mast cell tumors, for example.
Histopathology is generally considered the gold standard for determining the definitive diagnosis of a swelling. Additional diagnostics, such as immunohistochemistry, special staining, flow cytometry and PARR may be explored. The latter two diagnostics are especially useful to evaluate lymphoid neoplasia, whether lymphoma or leukemia.
The goal of this manuscript is to discuss osteosarcoma, elucidate differentials that may appear cytologically similar and to briefly mention diagnostic tools that may help to arrive at a definitive diagnosis, as well as to rule out other causes of similar findings.
Keywords: bone disease, diagnostic, lameness, neoplasia, osteolysis.
Introduction
One-Health is important for everyone and everything. This means the health of humans, animals and the environment. Many of the diseases and other types of ailments that humans suffer with are also diagnosed in dogs, cats and other species of animals. Environmental challenges have and still include control of waste production from humans and industrial factories. Pollution has been linked to the increased occurrence of certain types of cancers, including those of bone (García-Pérez et al., 2017), liver (Gant et al., 2023), breast and have been recognized in humans (Robinson, 2002) and animals (Cassali et al., 2012), including marine life (Baines et al., 2021).
There is an increased occurrence of people and animals living in close proximity to industrial plants and in urban areas that have a higher percentage of cancer cases (García-Pérez et al., 2017; Mitchell et al., 2021). The aim of this article is to discuss a few canine cases to provide some background information, diagnostics, clinical presentation, caveats and differential diagnoses for osteosarcoma. Differentials for osteosarcoma cytologically include other mesenchymal tumors, especially when they are poorly-differentiated, plasma cell tumors and histiocytic sarcomas (the author’s personal experience).
Background
Cancer, malignant neoplasia, may occur in any aged individuals (Withrow and Vail, 2007). There are leukemias (Evans, 2022) and embryonic tumors that may be found in newborn babies and animal alike (Withrow and Vail, 2007). Osteosarcomas occur in pediatric patients (Gadwal, et al., 2001; Simpson, and Brown, 2018), but are more commonly diagnosed in older dogs (Pool, 2022).
In cases of bony lesions, fractures may be more common than neoplastic. However, it is important to embrace various diagnostic modalities to definitively determine the underlying cause of lameness. In dogs and humans, it has been determined that fracture fixation devices have served as a nidus for development of osteosarcoma (Sinibaldi, et al., 1976; Harrison, et al., 1976; Etzioni, 2022; Withrow, and Vail, 2007).
Also of note are that certain parasites, such as Spirocerca lupi, can cause lesions that may develop into osteosarcoma (Withrow, and Vail, 2007). It is important to thoroughly examine each case and perform appropriate diagnostic and complimentary testing to then arrive at a definitive diagnosis, to instill the proper treatment.
This manuscript will highlight a few osteosarcoma cases, briefly discuss some differential diagnoses that could appear similar and discuss diagnostics that aid in definitive determination of osteosarcoma.
Origin
Osteosarcoma is a malignant mesenchymal tumor of bone, arising from the proliferation of malignant osteoblasts. It is the most common bone tumor, and may originate from long bones in the limbs, vertebrae and ribs in dogs. Having the affliction may result in lameness, especially when involving long bones.
Away from the elbow and toward the knee is a generalization for the usual distribution of this neoplastic process. However, these tumors may also occur in soft tissues, such as mammary glands. It is unknown why extraskeletal or extraosseous osteosarcomas occur. Most extraskeletal osteosarcomas occur in the soft tissues of humans and visceral organs of dogs (Patnaik, 1990; Makielski, 2019).
Whether these tumors, when occurring in the mammary gland are associated with spay/neuter status is unknown. Although there are associations of sex (humans and animals), spay/neuter status (for animals), and breed (for animals) (Dhein, et al., 2024). Mixed mammary tumors, whether benign or malignant forms, may contain bone elements, thus, variable osteoclasts, osteoblasts and even bone matrix may be identified on cytology and histopathology (Cassali, et al., 2012).
There are many studies associating a more frequent occurrence of mammary carcinomas and mixed mammary tumors in intact dogs and those spayed after the first estrous cycle. It is noteworthy that intact dogs, whether male or female, are at an increased risk for development of mammary gland tumors. They can be malignant, with osteosarcoma being among the differential diagnoses.
Diagnosis
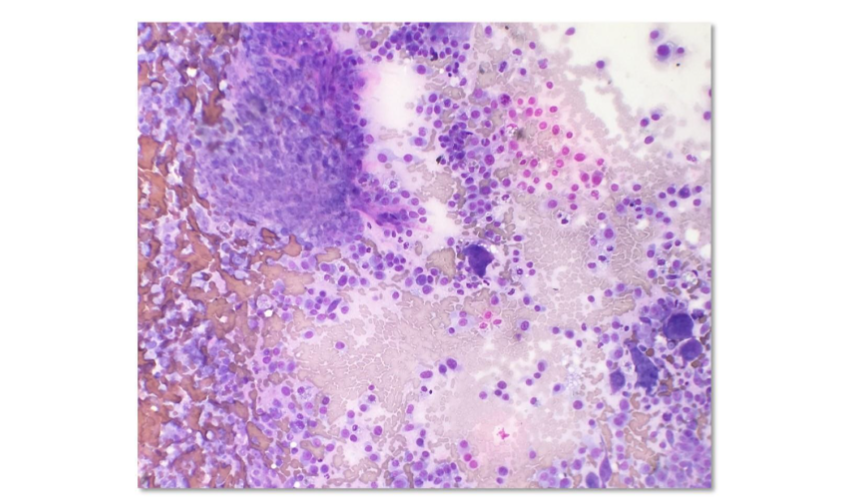
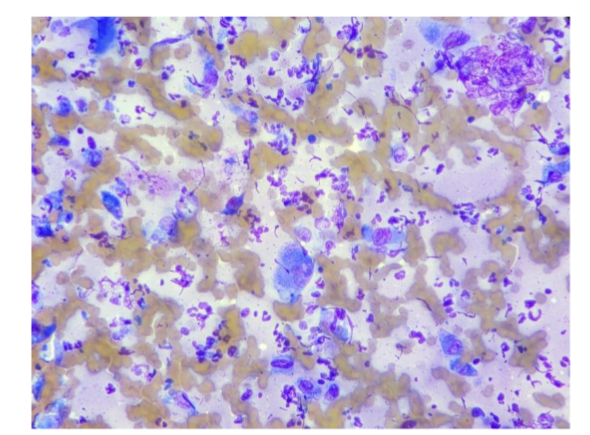
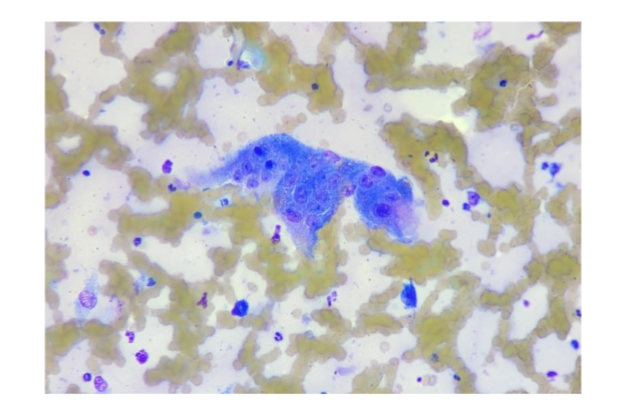
Diagnostics may include immunohistochemistry, special staining for ALP activity, flow cytometry and PARR. The latter two diagnostics are especially useful to evaluate lymphoid neoplasia, whether lymphoma or leukemia. In contrast to some mesenchymal cells, osteoblasts (the cell of origin for osteosarcomas) tend to exfoliate well (Fig. 1 & 2).
Classic cytologic descriptions of osteosarcomas may include moderate to high numbers of plump mesenchymal cells in a densely granular eosinophilic background with varying amounts of an extracellular pink wispy or amorphous matrix material in association with the neoplastic cells (Raskin and Meyer, 2016; Etzioni, 2022).
Individual neoplastic cells are oval to rounded with moderately basophilic cytoplasm, with/without a perinuclear clearing/Golgi zone and flecks of deeply magenta material in their cytoplasm. The nucleus is extremely eccentric, often appearing as if it is leaving the cell. They have coarse chromatin with usually one to two prominent nucleoli.
Variable numbers of mitotic figures may be noted, as well as variable amounts of background blood. Because of the perinuclear clearing area, some of these tumors may cytologically resemble or be mistaken for plasma cell tumors. Thus, further diagnostics may be necessary to make the distinction of osteosarcoma, especially if the tumor is poorly differentiated. Poorly differentiated tumors often don’t resemble the normal counterparts.
For a definitive diagnosis (Etzioni, 2022), fine-needle aspiration biopsy was performed and deposited into formalin and processed for a routine histopathology sample. An initial pre-Wright-stained cytology smear was stained alkaline phosphatase (ALP) activity positive with the special stain (Raskin and Meyer, 2016; Aguinaga, 2018; Etzioni, 2022; Nehlhaus et al., 2018; Cagle et al., 2024). Additionally, immunohistochemical (IHC) and minimal residual disease testing (MRD) may support the interpretation of neoplasia however, the interpretation of results can be limited by the number of cells sampled by this method may be diminished. When multiple diagnostic tools were used to overcome this limitation, fine-needle aspiration biopsy requires quick execution for diagnostic purposes.
Clinical Presentation
This neoplastic condition, osteosarcoma, amongst other causes of lameness, require diagnostic testing to elucidate. When lameness presents, diagnostic imaging is a common recommendation to pinpoint if the bone is affected by fracture, lysis or proliferative lesions (Cheverko, 2017).
This may also show, especially with digital radiography, increased densities of soft tissues associated with inflammatory lesions, especially of the muscles. These inflammatory lesions may be of infectious or noninfectious causes. Osteoarthritis, synovitis, autoimmune joint disease, cruciate or meniscal tears are also differentials for lameness.
With inflammatory lesions, peripheral blood evaluation for white blood cell counts, fine needle aspirate and subsequent cytologic and chemical assessment of synovial fluid are necessary to
evaluate for infectious and inflammatory joint disease (Raskin and Meyer, 2016). Evaluation of patients often requires multiple modalities. There is no one for all diagnostic tool applicable in any given case. To investigate the cause of disease in patients, whether they are human or animal, diagnostic testing is necessary. Testing may include, but are not limited to diagnostic imaging, hematology, clinical chemistry, urinalysis, fecal, skin testing, PCR or serological testing for infectious agents. Discussion of the subtypes (Cagle, et al., 2024) and metastatic nature of osteosarcoma (Regan, et al., 2022) are beyond the scope of this manuscript.
Case 1
12-year-old, intact male, Collie dog that presented with a right hind limb lameness. Pain therapy was initiated with minimal effect. This patient had a mid-femoral swelling that by radiographs centered on cerclage wiring put in place following a femoral fracture sustained at 2.5 years of age. The patient began showing right hindlimb lameness several months prior to presentation, which progressed in severity. Clinical signs were mildly alleviated by pain medication.
Owner recently noticed a swelling/mass of the cranial aspect of the right hindlimb (femur). Osteosarcoma was suspected. Fine needle aspirate cytology (Fig. 1 & 2) revealed abundant nucleated cells interpreted as malignant mesenchymal neoplasia with evidence of acute and chronic hemorrhage. Differentials included plasma cell tumor or osteosarcoma due to location.
DESCRIPTION:
Several slides were examined and were of good diagnostic quality. There was high nucleated cellularity consisting mainly of large pleomorphic mesenchymal cells. These cells measured approximately 30–45 mm in diameter, displayed marked anisokaryosis, anisocytosis, and had moderately variable nuclear to cytoplasmic ratios.
The cytoplasm was medium to light blue and abundant. The nuclei were medium purple, large, round and central to eccentrically-located. Frequently, binucleate and trinucleate cells were noted with an occasional multinucleated cell also seen. Nuclear molding was observed in some binucleate and multinucleate cells.
Nuclei measured about 20 mm in diameter. Chromatin was finely to coarsely granular and contained one to two prominent, large nucleoli. Frequently, nucleoli were larger than a red blood cell. There were 2–3 mitotic figures seen per 50x field.
The background infrequently contained a small amount of amorphous extracellular medium pink material associated with neoplastic cells. There were mild to moderate numbers of erythrocytes, the occasional nondegenerate neutrophil and macrophage noted. A rare erythrophagocytic macrophage was seen that infrequently also contained hemosiderin.
No infectious agents were observed. Furthermore, the morphology of the cells are reminiscent of plasma cell origin, but also consider an osteosarcoma. The pink extracellular material noted may be consistent with immunoglobulin or amyloid if this is a plasma cell tumor or osteoid if this is an osteosarcoma. Histopathology was needed for further characterization.
Bony lysis may be seen with both of these neoplasms, as well as pink extracellular matrix. In cases of plasma cell tumors, they can produce immunoglobulins and amyloid. With osteosarcoma, the matrix was osteoid, the precursor to bone formation. Leukocytes are consistent with the amount of peripheral blood observed on the smears.
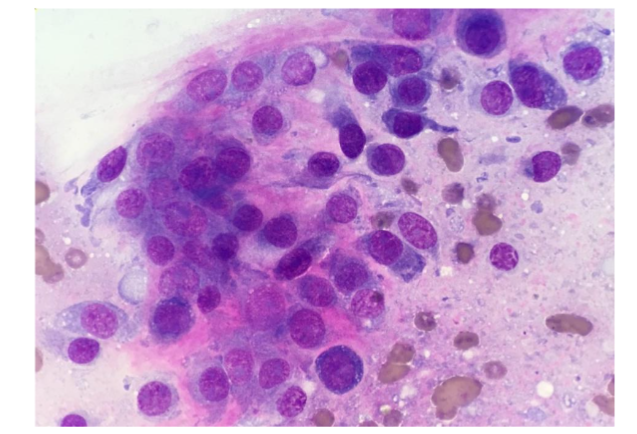
Figure 2: 12-year-old, intact male, Collie dog with osteosarcoma (Case 1). Note, the nucleated mesenchymal cells do not resemble the classic cell morphology of osteoblasts. High nucleated cellularity. Low power view at 60x objective, Wrights stain.
For a definitive diagnosis (Etzioni, 2022), fine-needle aspiration biopsy was performed and deposited into formalin and processed as a routine histopathology sample. An initial pre-Wright-stained cytology smear was stained alkaline phosphatase (ALP) activity positive with the special stain, nitroblue tetrazolium/5-Bromo-4-chloro-3-indolyl phosphate (NBT/BCIP) (Thermo Scientific) (Raskin and Meyer, 2016; Agustina, 2018; Etzioni, 2022; Neihuas, et al., 2011; Cagle, et al., 2024).
Additionally, vimentin, CD18 and MUM1 immunohistochemical (IHC) stains were performed supporting the interpretation of a mesenchymal neoplasm (vimentin+) and ruling out leukocytic origin (CD18) and plasma cell tumor (MUM1) respectively. The additive findings of the various diagnostics including the IHC confirmed a diagnosis of osteosarcoma.
Case 2
7-year-old, spayed female, Bull Mastiff with a distal tibial bone lesion (Fig. 3). She had an intermittent toe touching lameness of the right hindlimb. Initially, bone biopsies with histopathology were performed to only arrive at reactive bone (Fig. 3, blue arrow). This case drives home that if the tissue sampled does not encompass the root cause or nidus, the definitive diagnosis may be missed.
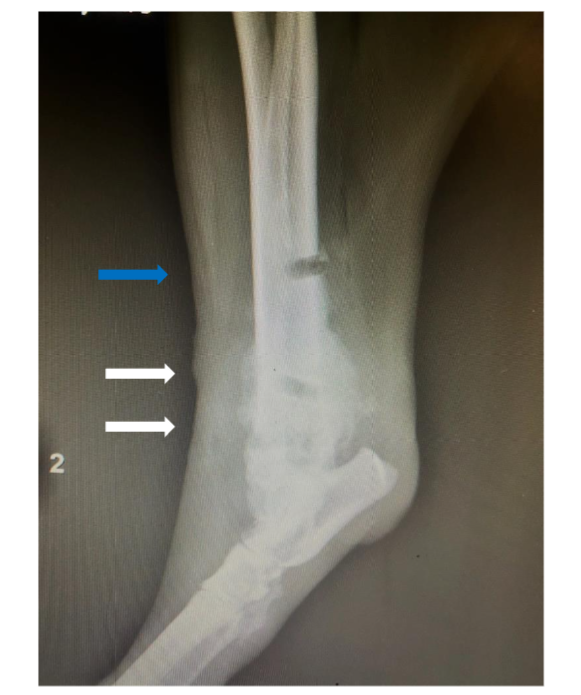
Figure 3: 7-year-old, spayed female, Bull Mastiff, plain lateral radiograph of the right hock. A lucent area in the bone is present where a biopsy revealed reactive bone (blue arrow). Subsequent to this, fine-needle aspirate biopsy was performed distally (white arrows). While cytology supported an interpretation of osteosarcoma, core biopsy with histopathology from this site confirmed a diagnosis of osteosarcoma.
Caveats
Case 2 (Fig. 3) demonstrates the difficulties of getting a diagnostic sample. Employing multiple diagnostic tools were useful to overcome this limitation. Fine-needle aspiration biopsy requires quick execution for diagnostic quality retention. There are times when interpretation of cells sampled by this method may be diminished.
When cells are either delayed in extraction from tissues or delayed in spreading them onto a glass slide, the nucleated cells may clot or fail to spread (Fig. 4A). The cells may remain in a clot or rupture with the spreading action during smear preparation. Case 2 also demonstrates that the area sampled may vary with the types of cells represented (Fig. 4B).
With neoplastic and inflammatory lesions, they often result in a mixed population of cell types. With a predominance of dysplastic cells and minimal to no inflammatory cells being seen, it may result in a more likely diagnosis of neoplasia. However, when the opposite occurs and an abundance of inflammatory cells are seen (Fig. 4B) with minimal numbers of tissue cells, the interpretation here is as an inflammatory lesion.
Inflammation causes neighboring cells to sometimes become dysplastic and in other cases may be the nidus of irreparable tissue damage, which may result in neoplasia. In this case, evaluation of additional areas resulted in a collection of cells by cytology to support an interpretation of osteosarcoma, which was subsequently confirmed by core biopsy with histopathology.
Figure 4A: 7-year-old, spayed female, Bull Mastiff. Fine-needle aspirate biopsy cytology displaying clotted nucleated cells, nondiagnostic sample. Low power view at 20x objective, Wrights stain.
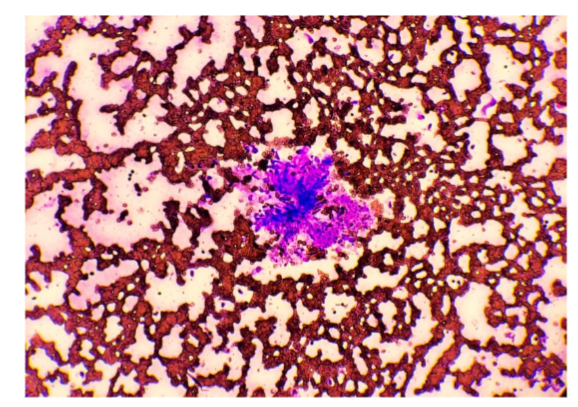
Osteoclasts
The presence of osteoclasts must not be overinterpreted. When osteoclasts (Fig. 5) are seen, this indicates bony lysis is likely occurring. However, bony lysis may occur in neoplastic and non-neoplastic lesions. With inflammation of the bone, lysis may occur. Thus, seeing this cell type does not constitute a diagnosis of neoplasia.
Since osteoclasts are charged with cleaning up bone, variable numbers of them may be seen in osteosarcomas, mixed mammary tumors and osteomyelitis. They may also be a part of the process of remodeling bone during fracture healing and during normal growth in animals and humans.
Additionally, giant cells may be mistaken for osteoclasts and may be a feature of feline and less commonly canine osteosarcomas cytologically and histologically (Pool, 1978).
Case 3
8-year-old, spayed female, Yorkshire Terrier dog that presented with a caudal/inguinal mammary swelling. Fine-needle aspiration cytology revealed cells similar to those described as general cytologic findings. Cytologically, these cells appear as classic osteoblasts.
With the presence of a perinuclear clearing area, they may resemble plasma cells. Plasma cells may produce an extracellular amorphous pink matrix that could represent immunoglobulin or amyloid. On cytology, these substances may appear similar.
The histopathologic diagnosis was osteosarcoma. The formation of osteoid, bone matrix, is a histologically feature of osteosarcoma. MUM1 is an immunohistochemical stain expected to be positive for plasma cell tumors.
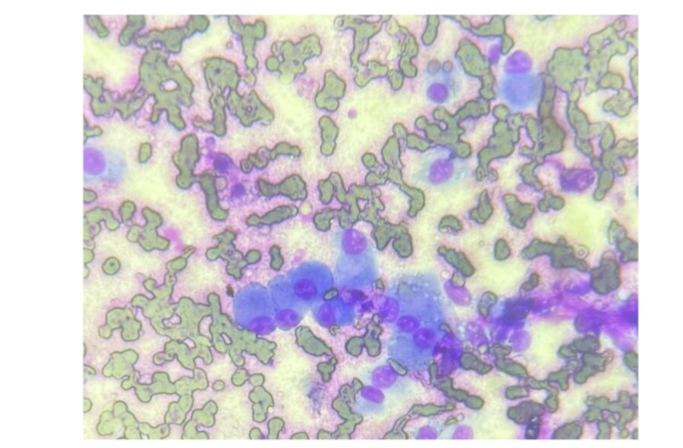
Figure 6: 8-year-old, spayed female, Yorkshire Terrier dog that presented with a caudal/inguinal mammary swelling. Fine-needle aspirate biopsy cytology displaying moderate nucleated cellularity with plump mesenchymal cells consistent with osteoblasts in a background of blood. 100x objective, Diff Quik stain. Photo credit: Dr. Ginger Bailey.
Case 4
5-year-old, intact male, Bulldog that presented for a left front limb lameness, which by plain radiographs was centered at the shoulder joint. This patient was managed with supportive therapy, which included minimal exercise, pain and anti-inflammatory medications.
When presenting to the teaching hospital at Tuskegee University repeat survey radiography and fine needle aspiration were performed. With an interpretation of osteosarcoma likely, the patient was humanely euthanized. The lesion was not further biopsied with histopathology due to the extensiveness of the lesion and inability to completely surgically remove the tumor.
Discussion
Osteosarcoma is one of the most common malignant neoplastic conditions that occur in
humans and animals. It is the most common bony tumor in both animals and humans. The use of diagnostics are necessary to investigate the cause of disease and confirm a diagnosis; therefore, specific therapies may be instilled for better patient care.
As discussed here, tools included diagnostic imaging, cytology, histopathology, special staining, immunohistochemistry (immunocytochemistry may also be useful). A definitive diagnosis may require these tests in succession or in tandem (Raskin and Meyer, 2016; Etzioni, 2022).
Special staining is commonly used to highlight infectious agents, such as bacteria, higher bacteria, protozoa, and fungus (Raskin and Meyer, 2016). In veterinary medicine, we may not always have the liberty to perform diagnostic testing. Cost may be a limiting factor to a client’s ability to accept diagnostic testing. Therefore, empirical or symptomatic treatment may be requested.
At times this may result in resolve of clinical signs, whether temporary or permanent. With lameness, which can be debilitating, the employment of diagnostic imaging is desired to help elucidate the cause. With neoplasia, often clinical signs continue to worsen, despite administering anti-inflammatory and pain medications.
With inflammatory lesions such as muscle strain, sprain and osteomyelitis, these medications may placate the clinical signs. Communication may be a factor involved in a client’s willingness to allow diagnostic testing or understanding of the disease process their pet may be afflicted with.
With complete disclosure regarding the possibilities and the necessity of diagnostic testing, the client would be armed with the necessary information to make an informed decision moving forward.
Conflict of Interest:
None.
Funding Statement:
None.
Acknowledgements:
None.
ORCID Number:
https://orcid.org/0000-0002-1363-8957
References
References
1. Dhein ES, Heikkilä U, Oevermann A, et al. Incidence rates of the most common canine tumors based on data from the Swiss Canine Cancer Registry (2008 to 2020). PLoS One. 2024;19(4):e03 02231. Published 2024 Apr 18. doi: 10.1371/journa l.pone.0302231.
2. García-Pérez, J, Morales-Piga, A, Gómez-Barroso, et al. Risk of bone tumors in children and residential proximity to industrial and urban areas: New findings from a case-control study. Sci Total Environ, 2017; 579: 1333-1342. doi.org/10.1016/j. scitotenv.2016.11.131
3. Makielski KM, Mills LJ, Sarver AL, et al. Risk Factors for Development of Canine and Human Osteosarcoma: A Comparative Review. Vet Sci. 2019; 6(2):48. Published 2019 May 25. doi:10.3390 /vetsci6020048
4. Evans SJM. Flow Cytometry in Veterinary Practice. Vet Clin North Am Small Anim Pract. 2023;53(1): 89-100. doi:10.1016/j.cvsm.2022.07.008
5. Robinson D. Cancer clusters: findings vs feelings. MedGenMed. 2002;4(4):16. Published 2002 Nov 6.
6. Sinibaldi K, Rosen H, Liu SK, DeAngelis M. Tumors associated with metallic implants in animals. Clin Orthop Relat Res. 1976;(118):257-266.
7. Harrison JW, McLain DL, Hohn RB, Wilson GP 3rd, Chalman JA, MacGowan KN. Osteosarcoma associated with metallic implants. Report of two cases in dogs. Clin Orthop Relat Res. 1976; (116):253-257.
8. Dantas Cassali G, Cavalheiro Bertagnolli A, Ferreira E, Araújo Damasceno K, de Oliveira Gamba C, Bonolo de Campos C. Canine mammary mixed tumours: a review. Vet Med Int. 2012;2012: 274608. doi: 10.1155/2012/274608. Epub 2012 Oct 21. PMID: 23193497; PMCID: PMC3485544.
9. Mitchell PD, Dittmar JM, Mulder B, et al. The prevalence of cancer in Britain before industrialization. Cancer. 2021;127(17):3054-3059. doi:10.1002/cncr.33615
10. Gadwal SR, Gannon FH, Fanburg-Smith JC, Becoskie EM, Thompson LD. Primary osteosarcoma of the head and neck in pediatric patients: a clinicopathologic study of 22 cases with a review of the literature. Cancer. 2001;91(3):598-605.
11. Etzioni AL. Osteosarcoma diagnosed in a dog using a formalin-fixed fine-needle aspirate biopsy. Vet Clin Pathol. 2022;51:349-355. doi: 10.1111/ vcp.13056
12. Cheverko CM, Bartelink EJ. Resource intensification and osteoarthritis patterns: changes in activity in the prehistoric Sacramento-San Joaquin Delta region. Am J Phys Anthropol. 2017; 164(2):331-342. doi:10.1002/ajpa.23272
13. Patnaik AK. Canine extraskeletal osteosarcoma and chondrosarcoma: a clinicopathologic study of 14 cases. Vet Pathol. 1990;27(1):46-55. doi:10.117 7/030098589002700107
14. Simpson E, Brown HL. Understanding osteosarcomas. JAAPA. 2018;31(8):15-19. doi:10.1 097/01.JAA.0000541477.24116.8d
15. Regan DP, Chow L, Das S, et al. Losartan Blocks Osteosarcoma-Elicited Monocyte Recruitment, and Combined With the Kinase Inhibitor Toceranib, Exerts Significant Clinical Benefit in Canine Metastatic Osteosarcoma. Clin Cancer Res. 2022; 28(4):662-676. doi:10.1158/10 78-0432.CCR-21-2105
16. Agustina H, Asyifa I, Aziz A, Hernowo BS. The Role of Osteocalcin and Alkaline Phosphatase Immunohistochemistry in Osteosarcoma Diagnosis. Patholog Res Int. 2018;2018:6346409. Published 2018 May 3. doi:10.1155/2018/6346409
17. Raskin, RE. Meyer DJ. Canine and Feline Cytology: A Color Atlas and Interpretation Guide. 3rd Ed. Elsevier, Inc. St. Louis, Missouri, 2016, PAGES.
18. Withrow SJ, Vail DM. Small Animal Clinical Oncology. 4th Ed. Saunders and Elsevier. St. Louis, Missouri. 2007, PAGES.
19. Cagle LA, Maisel M, Conrado FO, et al. Telangiectatic osteosarcoma in four dogs: Cytologic, histopathologic, cytochemical, and immunohistochemical findings. Vet Clin Pathol. 2024;53(1):85-92. doi:10.1111/vcp.13338
20. Neihaus SA, Locke JE, Barger AM, et al. A novel method of core compared to for J Am Anim Hosp Assoc. 2011;47:317-23.
21. Gan T, Bambrick H, Tong S, Hu W. Air pollution and liver cancer: A systematic review. J Environ Sci (China). 2023 Apr;126:817-826. doi: 10.1016/j.jes.2022.05.037. Epub 2022 Jun 2. PMID: 36503807.
22. Baines C, Lerebours A, Thomas F, Fort J, Kreitsberg R, Gentes S, Meitern R, Saks L, Ujvari B, Giraudeau M, Sepp T. Linking pollution and cancer in aquatic environments: A review. Environ Int. 2021 Apr;149:106391. doi: 10.1016/j.envint.2021. 106391. Epub 2021 Jan 27. PMID: 33515955.
23. Pool RR. Tumors of Bone and Cartilage. In: Moulton, editor. Tumors in Domestic Animals. Univ. California Press; Berkeley: 1978. pp. 89–149.

