Outcomes of ASCT for Relapsed Hodgkin Lymphoma
Outcomes of Autologous Stem Cell Transplant for relapsed or refractory classic Hodgkin lymphoma in the era of PD-1 inhibitors
Sanjal H. Desai, MD 1; Ivana N. M. Micallef, MD 2
- Division of Hematology, Mayo Clinic, Rochester, MN, USA
- Division of Hematology, Oncology, Transplantation, University of Minnesota, Minneapolis, MN, USA
OPEN ACCESS
PUBLISHED: 28 Febuary 2025
CITATION: MICALLEF, Ivana N. M.; DESAI, Sanjal H.. Outcomes of Autologous Stem Cell Transplant for relapsed or refractory classic Hodgkin lymphoma in the era of PD-1 inhibitors. Medical Research Archives. Available at: <https://esmed.org/MRA/mra/article/view/6234>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i2.6234
ISSN 2375-1924
ABSTRACT
Classic Hodgkin lymphoma (cHL) is associated with excellent cure rates with front line therapy; however, relapses occur in about 10-20% of patients. Salvage therapy and autologous stem cell transplant is the standard of care treatment approach for relapsed/refractory cHL resulting in cure rates of 50-60%. Relapse rates are higher for patients with certain high-risk features. Addition of post-transplant consolidation brentuximab vedotin (BV) has led to significantly higher 5 year progression free survival rates of 60%. Patients who progressed after autologous stem cell transplant had overall survival of 2-3 years. cHL is remarkably sensitive to PD-1 inhibitors, due to overexpression of PD-L1 on Hodgkin Reed Sternberg cells that interact with PD-1 on immune effector cells in the tumor microenvironment. Studies have also shown that PD-1 inhibitors may increase naïve T cells in peripheral blood and recruit them to cHL tumor microenvironment to facilitate anti-tumor responses. PD-1 inhibitors containing salvage regimens have led to response rates approaching 90-100% in relapsed/refractory cHL. Patients who undergo autologous transplant immediately after PD-1 inhibitor-based therapy have 2 year progression free survival of ~93%, which is significantly higher than patients who undergo autologous transplant after chemotherapy or BV based regimens. Even in patients who undergo autologous transplant in complete response, PD-1 inhibitor-based therapy prior to autologous transplant is associated with post-transplant PFS of 97%, significantly higher than chemotherapy or BV. This finding raises the possibility that PD-1 inhibitors may induce cure in relapsed/refractory cHL, not just by improving depth of response but also by affecting post-transplant immune reconstitution. The remarkable outcomes of PD-1 inhibitors in relapsed/refractory cHL raises a question whether these patients can be cured without autologous transplant. Future large randomized clinical trials are needed to answer this question.
Keywords:
cHL, ASCT, PD-1 inhibitors, lymphoma
Introduction:
In 2023, ~9000 Americans were diagnosed with classic Hodgkin lymphoma (cHL), 95% of newly diagnosed patients were young adults. Frontline treatment chemo immunotherapy cured up to 70-80% of patients with cHL, unfortunately 5-10% of patients are refractory to frontline chemo immunotherapy and 10-20% relapse after achieving an initial remission. For transplant-eligible patients with relapsed/refractory classic Hodgkin lymphoma (R/R cHL), salvage chemotherapy and autologous stem cell transplant are considered standard of care. Prior to the advent of novel agents, this approach cured 50-60% of patients with R/R cHL. Patients who are refractory to frontline therapy, relapse within a year, have extra nodal disease, who require more than 1 line of salvage therapy and those who undergo transplant in partial response have a higher risk of relapse. The risk of relapse in this population approaches 50-60%. Various platinum based salvage chemotherapy regimens such as ifosfamide, carboplatin and etoposide (ICE); dexamethasone, cytarabine, cisplatin (DHAP); or etoposide, cytarabine, cisplatin (ESHAP) lead to complete response rates of 40% as assessed by CT scan and 3 year PFS of ~50% post autologous stem cell transplant (ASCT). The addition of an anti-CD30 antibody-drug-conjugate brentuximab vedotin (BV) to ICE resulted in an improved overall response rate of 69% with 2 year event free survival (EFS) of 91%. Combination of BV with bendamustine led to complete response rates of 75% and 3 year EFS of 67%. The addition of BV maintenance for 1 year post-ASCT in high-risk patients improved 5 year PFS from 40% in the placebo treated patients to 60% in the BV treated patients. Outcomes of patients who progress after autologous stem cell transplant were dismal in the era prior to advent of PD-1 inhibitors and BV. Prior to advent of PD-1 inhibitors and BV, multiple studies have reported median overall survival of 2-3 years in patients of cHL who progress after ASCT. PD-1 inhibitors, such as pembrolizumab and nivolumab, have shown remarkable activity in R/R cHL. Studies incorporating PD-1 inhibitors have shown encouraging outcomes. Here, we review mechanism of action of PD-1 inhibitors, key studies incorporating PD-1 inhibitors in R/R cHL and outcomes of R/R cHL in the era of PD-1 inhibitors.
Mechanism of action of PD-1 inhibitors:
PD-1/PDL-1 axis and role of CD4+ helper T cells in pathobiology of cHL:
Programmed cell death- 1 (PD-1) is a transmembrane protein receptor that is expressed on T cells. PD-1 mediates negative regulation of T cells, suppresses T cell activation and promotes T cell exhaustion. Programmed cell death-ligand 1 (PD-L1) is expressed on malignant cells, including Hodgkin Reed Sternberg (HRS) cells. PD-1/PD-L1 interaction results in reduced phosphorylation of T cell receptor signaling molecules, decreased cytokine production and consequent T cell suppression.
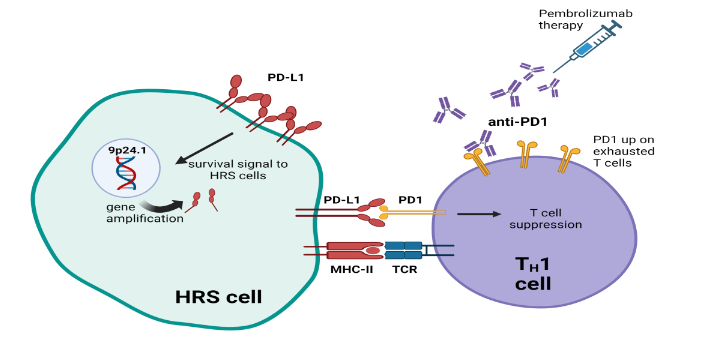
Hodgkin Reed Sternberg (HRS) cells is the malignant cell of cHL, and comprises <5% of tumor tissue in cHL. HRS cell is surrounded by immune effector cells such as macrophages and tumor infiltrating lymphocytes. HRS cells have high surface expression of PD-L1 due to amplification of the 9p24 gene locus. Interaction of PD-L1 and PD-1 leads to suppression of T cell antitumor responses and sends retrograde signals of growth and survival to HRS cells. This PD-1/PD-L1-mediated T cell suppression in cHL appears to involve CD4-positive helper T cells. An evaluation of the immune infiltrate in cHL tumor biopsies has revealed high numbers of PD-1 positive T-helper (TH) cells surrounding HRS cells. PD-1/PD-L1 interaction promotes these cells to differentiate into regulatory T cells (Treg) rather than T-helper 1 (TH1) polarized cells. Engagement of PD-L1 with surface PD-1 promotes plasticity of fully differentiated TH1 towards regulatory T cells (Treg) that drive immune exhaustion, immune tolerance, and anti-tumor effects. By binding to PD-1 on immune effector cells, PD-1 inhibitors interrupt these signals and promote antitumor effects.
Effect of PD-1 inhibitors on peripheral blood immune repertoire to facilitate antitumor response:
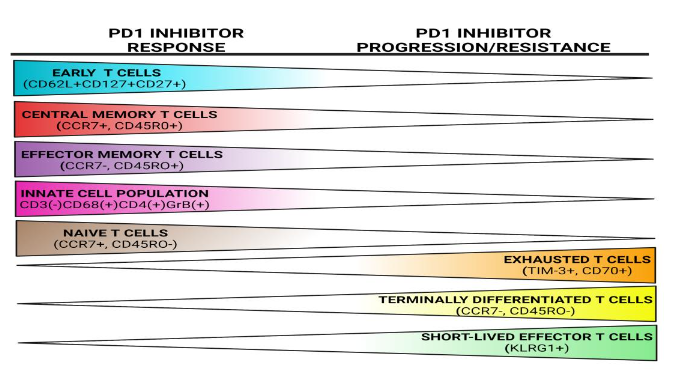
Translational studies on patients receiving PD-1 inhibitors show that antitumor effects are not generated by reversing immune exhaustion of pre-existing TH cells but by recruitment of new, naïve, unexhausted TH cells to the tumor microenvironment, which then drive anti-tumor response. Mass cytometry (CyTOF) and single cell RNA sequencing (scRNAseq) conducted on tumor tissue and peripheral blood samples of patients receiving PD-1 inhibitors show recruitment of newly immigrant CD4+ TH cells from peripheral blood to the tumor microenvironment, rather than expansion of pre-existing intratumoral CD4+ TH cells. These naïve, tumor specific CD4+ TH cells are present in the peripheral blood pre-treatment and are recruited to the tumor microenvironment during PD-1 inhibitor therapy. In peripheral blood of lymphoma patients receiving PD-1 inhibitors, CyTOF analyses evaluating CD4+ T cells differentiation has shown increasing population of CD62L+CD127+CD27+ early stage T cells, CCR7+CD45RO- naïve T cells, CCDR7+CD45RO+ central memory T cells and CCR7-CD45RO+ effector memory T cells and this expansion correlates with PD-1 responsiveness. On the other hand, patients who progress on PD-1 inhibitors were more likely to have CD45RO-CCR7- terminally differentiated T cells, TIM-3+, CD70+ exhausted T cells or KLRG1+ short-lived effector T cells. T cell receptor (TCR) diversity, which reflects the number of individual T cell clones capable of recognizing distinct antigens also increases in the peripheral blood of patients receiving PD-1 inhibitors and correlates with response. Expansion of these new singleton T cell clones correlate with expansion of naïve, early and central memory T cells, suggesting that new T cell clones are more likely to be naïve, unexhausted T cells. Thus, the population of naïve, unexhausted T cells in the peripheral blood increases during PD-1 inhibitors treatment and these cells are then recruited to the cHL tumor microenvironment to exert antitumor effects.
Clinical Trials of PD-1 inhibitors in classic Hodgkin lymphoma (cHL):
PD-1 inhibitors such as nivolumab and pembrolizumab are the most extensively studied checkpoint inhibitors in cHL and are FDA approved for this indication. Single agent nivolumab was first studied in patients with R/R cHL who had received 2 or more prior lines of therapy including ASCT. In this heavily pre-treated patient population, which included patients who underwent ASCT, nivolumab led to overall response rate (ORR) of 87% and 24 week PFS rates of 86%. Single agent pembrolizumab was studied in a similar population of R/R cHL patients and led to ORR of 71.4% and CR rate of 27.6%. Median PFS was 13.7 months. Pembrolizumab was compared to brentuximab vedotin (BV) in a randomized phase 3 trial in patients of R/R cHL who has received 2 or more prior lines of therapy including ASCT. In this study, pembrolizumab led to significantly longer median PFS of 13.4 months compared to BV which had median PFS of 8.3 months. These early trials led to the FDA approval of nivolumab and pembrolizumab in R/R cHL.
PD-1 inhibitors in treatment-eligible relapsed/refractory classic Hodgkin lymphoma (cHL): changing treatment paradigm
Encouraging results in heavily pre-treated patients who relapsed after or are ineligible for ASCT led to clinical trials assessing efficacy of pembrolizumab and nivolumab in second-line salvage setting for transplant-eligible patients of cHL who are refractory to or relapsed after frontline treatment. Table-1 presents details of study design, patient characteristics and outcomes of the major clinical trials in this setting. In these clinical trials, response rates to PD1- inhibitors based salvage therapy ranged from 70-100% and 60-90% patients achieved CR. In patients who successfully underwent ASCT after PD1- inhibitor-based therapy the PFS approached 97% at 2 years. Thus, PD-1 inhibitor-based salvage therapies have remarkable efficacy in transplant-eligible patients, although, randomized trials comparing them to chemotherapy based or BV based regimen are lacking at this point.
| Study | Reference | Design | Regimen | Study population | Response rates (N, %) | % underwent ASCT | Survival |
|---|---|---|---|---|---|---|---|
| Advani et al. | Ref 38 | Single arm, phase 1/2 | BV/Nivo | R/R cHL after 1 prior line of therapy | ORR 77/91 (85%), CR 61/91 (67%) | 67/91 (73%) | 3-year PFS 77%, 91% in patients who underwent ASCT, 3-year OS was 93% |
| Moskowitz et al. | Ref 39 | Single arm, phase 1/2 | Pembro-GVD | R/R cHL after 1 prior line of therapy | ORR 38/38, 100%; CR 36/38 (95%) | All patients | All alive and in remission over median follow up of 13.5 months |
| Mei et al. | Ref 40 | Single arm, phase 2 | Nivo-ICE | R/R cHL after 1 prior line of therapy | ORR 39/42 (93%); CR 38/42 (91%) | 33/42 (78.5%) | 2-year PFS 72%, 2-year PFS 95% in patients who directly underwent ASCT, 2-year OS 95% |
| Locke et al. | Ref 41 | Single arm, phase 2 | Pembro-ICE | R/R cHL after 1 prior line of therapy | Total 42 patients, 37 evaluable. ORR 36/37, 97.3%, CR 26/37, 89.2% | 40/42, 95.2% | 2-year PFS 87.2%, 2-year OS 95.1% |
| Diefenbach et al. | Ref 42 | Two-arm, randomized, phase 2 | BV/Nivo/ipi vs BV/Nivo | R/R cHL after 1 prior line of therapy | ORR 88% in both arms, CR 60.7% in BV/Nivo and 66.7% in BV/Nivo/Ipi | — | 24 months DOR rates 72.9% for BV/Nivo, 82.4% for BV/Nivo/Ipi (p=0.554) |
Abbreviations: N; number, %, percentages, BV; brentuximab vedotin, Nivo; nivolumab, Ipi; ipilimumab, R/R cHL; relapsed or refractory classic Hodgkin lymphoma, ICE; chemotherapy combination of ifosfamide, carboplatin, etoposide; GVD; chemotherapy combination of gemcitabine, vinorelbine and doxil, ORR; overall response rates, CR; complete response rates, DOR; duration of response, PFS; progression free survival, OS; overall survival.
PD-1 inhibitors in treatment-naïve classic Hodgkin lymphoma (cHL): changing treatment paradigm
Due to remarkable activity in R/R cHL, PD-1 inhibitors were investigated in treatment-naïve cHL. In a randomized phase 3 trial of treatment-naïve cHL, nivolumab in combination with adriamycin, vinblastine and dacarbazine (N+AVD) has shown excellent PFS of 94% at 1 year, significantly higher than BV in combination with AVD (86% PFS at 1 year). Movement of PD-1 inhibitors in frontline treatment of classic Hodgkin lymphoma creates an investigative need for patients who have progressive disease on PD-1 inhibitors. Therefore, novel strategies will be needed to improve outcomes of patients who progress on PD-1 inhibitors.
Outcomes of relapsed/refractory classic Hodgkin lymphoma (cHL) in the era of PD-1 inhibitors: real-world evidence
In the absence of randomized controlled trials, real-world evidence provides some insights into the comparative efficacy of PD-1 inhibitor-based salvage therapy and other approaches. In a multicenter study of 946 R/R cHL patients who underwent ASCT, PD1- inhibitor-based first salvage therapy was associated with significantly higher 2-year event free survival (EFS: 79.7%), compared to chemotherapy based (2 year EFS 49.6%) or BV+chemotherapy (2 year EFS 62.3%). In patients who underwent ASCT after first salvage therapy, PD-1 inhibitor-based therapy was associated with significantly higher 2-year PFS of 98% compared to patients who received chemotherapy (2-year PFS 68.8%, or BV+chemotherapy (2-year PFS 84 %). In another cohort of 986 R/R cHL with a larger population of PD-1 inhibitor treated patients, PD-1 inhibitor-based salvage therapy prior to ASCT was associated with significantly higher 2-year PFS of 93.1% compared to chemotherapy based (2-year PFS 71.6%) or BV-based salvage therapy (2-year PFS 73.9%). Although the type of salvage chemotherapy was associated with an improved EFS and PFS, the type of salvage therapy was not significantly associated with overall survival in either of these studies. Overall survival exceeded 90% in all treatment arms. Notwithstanding inherent limitations such as exclusion of patients who could not be bridged to transplant and the retrospective nature, both these studies generated an important hypothesis and provided practice-informing insights into treatment paradigm of R/R cHL in the absence of randomized clinical trials.
Outcomes of patients who progress after autologous stem cell transplant in PD-1 era:
R/R cHL patients who progress after ASCT or are ineligible to receive ASCT have incurable disease. Before the advent of novel agents including PD-1 inhibitors, these patients had median overall survival of 2-3 years. The improvement in disease response and duration of response with PD-1 inhibitors and BV has led to the improvement in overall survival in this population. In two independent multicenter studies of R/R cHL mentioned above, median OS of patients who progressed after ASCT approached 10 years and 5 year OS was approaching 60%. In addition, sequencing of therapies mattered. Patients who received PD-1 inhibitors first at the time of progression had a significantly higher 5-year post-progression overall survival (PPS) of 77% compared to patients who received BV (5-year PPS of 63%) or chemotherapy (5-year PPS of 49%). Thus, the outcomes of patients who progress after ASCT are remarkably better in the era of PD1-inhibitors.
Future Directions:
Role of autologous stem cell transplant in relapsed/refractory classic Hodgkin lymphoma (cHL):
As PFS rates approach 90% at 2 years for R/R cHL patients who receive PD-1 inhibitors prior to ASCT, question arise whether some of these patients could be cured without ASCT. This question was addressed in a single arm phase 2 trial that assessed efficacy of pembrolizumab in combination with GVD (Pembro-GVD) chemotherapy followed by pembrolizumab maintenance in transplant-eligible R/R cHL patients. In this study, R/R cHL patients who progressed after 1 prior line of therapy were enrolled to receive 4 cycles of Pembro-GVD followed by 1 year of pembrolizumab maintenance. Pembro-GVD resulted in 90% CR rates in this population. Two-year PFS in all patients was 51%. All but 1 patient who progressed were able to successfully be bridged to ASCT. Stage IV disease at enrollment was associated with higher risk of progression. Although not confirmed in a large randomized trial, it seems that ASCT is necessary to achieve optimal cure rates in R/R cHL.
PD-1 inhibitor-resistant classic Hodgkin lymphoma (cHL):
The movement of PD-1 inhibitors in frontline will result in a population of R/R cHL patients with PD-1 inhibitors resistant disease. Therefore, studies evaluating novel strategies and outcomes in patients who progress after PD1-inhibitors based therapy are more relevant now than ever. In a real-world retrospective study of patients who are refractory or intolerant to BV and PD-1 inhibitors, median overall survival was 7.4 years after progression or intolerance to BV and PD-1 inhibitors. 75% of patients were refractory to both BV and PD-1 inhibitors. Retreatment with PD1 inhibitors can also be efficacious. Outcomes of R/R cHL patients who achieved CR after pembrolizumab monotherapy, discontinued pembrolizumab in CR and experienced disease relapse, were assessed in a long term follow up analyses of KEYNOTE-087 study. In 20 patients who were re-treated with pembrolizumab, median duration of first CR to pembrolizumab was 27.2 months. 19 patients were evaluable for response at data cut off and ORR was 73.7% (14 out 19) with pembrolizumab retreatment. The median duration of response was 15.2 months. Seven (36.8%) patients achieved CR. These findings highlight feasibility of re-treatment with pembrolizumab in R/R cHL patients who relapse after prior remission. Incorporate investigational agents with PD-1 inhibitors in order to re-sensitize cHL microenvironment to PD-1 inhibitors is being studies in ongoing clinical trials. Results of these studies will be practicing informing.
Conclusion
PD-1 inhibitors have changed the treatment landscape of cHL. PD-1 inhibitor-based treatment is now considered standard of care both in the frontline, salvage and post ASCT setting. Their use not only improves the depth of response but is also associated with improvement in PFS. This improvement is felt to be associated with the increase of naïve unexhausted T cell in the peripheral blood being recruited to the cHL tumor microenvironment to exert an antitumor effect. Despite the use of PD-1 inhibitors, ASCT is still necessary to achieve optimal cures.
References:
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. Jan 2022;72(1):7-33. doi:10.3322/caac.21708
- Ansell SM, Radford J, Connors JM, et al. Overall Survival with Brentuximab Vedotin in Stage III or IV Hodgkin’s Lymphoma. N Engl J Med. Jul 28 2022;387(4):310-320. doi:10.1056/NEJMoa2206125
- Connors JM, Jurczak W, Straus DJ, et al. Brentuximab Vedotin with Chemotherapy for Stage III or IV Hodgkin’s Lymphoma. N Engl J Med. Jan 25 2018;378(4):331-344. doi:10.1056/NEJMoa1708984
- Desai S, Ansell SM. Future directions in Hodgkin lymphoma: checkpoint inhibitors and beyond. Leuk Lymphoma. Aug 2021;62(8):1795-1804. doi:10.1080/10428194.2021.1885667
- Josting A, Franklin J, May M, et al. New prognostic score based on treatment outcome of patients with relapsed Hodgkin’s lymphoma registered in the database of the German Hodgkin’s lymphoma study group. J Clin Oncol. Jan 1 2002;20(1):221-30. doi:10.1200/JCO.2002.20.1.221
- Moskowitz CH, Nimer SD, Zelenetz AD, et al. A 2-step comprehensive high-dose chemoradiotherapy second-line program for relapsed and refractory Hodgkin disease: analysis by intent to treat and development of a prognostic model. Blood. Feb 1 2001;97(3):616-23. doi:10.1182/blood.v97.3.616
- Linch DC, Winfield D, Goldstone AH, et al. Dose intensification with autologous bone-marrow transplantation in relapsed and resistant Hodgkin’s disease: results of a BNLI randomised trial. Lancet. Apr 24 1993;341(8852):1051-4. doi:10.1016/0140-6736(93)92411-l
- Schmitz N, Pfistner B, Sextro M, et al. Aggressive conventional chemotherapy compared with high-dose chemotherapy with autologous haemopoietic stem-cell transplantation for relapsed chemosensitive Hodgkin’s disease: a randomised trial. Lancet. Jun 15 2002;359(9323):2065-71. doi:10.1016/S0140-6736(02)08938-9
- Shah GL, Yahalom J, Matasar MJ, et al. Risk factors predicting outcomes for primary refractory hodgkin lymphoma patients treated with salvage chemotherapy and autologous stem cell transplantation. Br J Haematol. Nov 2016;175(3):440-447. doi:10.1111/bjh.14245
- Moskowitz CH, Walewski J, Nademanee A, et al. Five-year PFS from the AETHERA trial of brentuximab vedotin for Hodgkin lymphoma at high risk of progression or relapse. Blood. Dec 20 2018;132(25):2639-2642. doi:10.1182/blood-2018-07-861641
- Sureda A, Constans M, Iriondo A, et al. Prognostic factors affecting long-term outcome after stem cell transplantation in Hodgkin’s lymphoma autografted after a first relapse. Ann Oncol. Apr 2005;16(4):625-33. doi:10.1093/annonc/mdi119
- Constans M, Sureda A, Terol MJ, et al. Autologous stem cell transplantation for primary refractory Hodgkin’s disease: results and clinical variables affecting outcome. Ann Oncol. May 2003;14(5):745-51. doi:10.1093/annonc/mdg206
- Josting A, Muller H, Borchmann P, et al. Dose intensity of chemotherapy in patients with relapsed Hodgkin’s lymphoma. J Clin Oncol. Dec 1 2010;28(34):5074-80. doi:10.1200/JCO.2010.30.5771
- Aparicio J, Segura A, Garcera S, et al. ESHAP is an active regimen for relapsing Hodgkin’s disease. Ann Oncol. May 1999;10(5):593-5. doi:10.1023/a:1026454831340
- Moskowitz AJ, Schoder H, Yahalom J, et al. PET-adapted sequential salvage therapy with brentuximab vedotin followed by augmented ifosfamide, carboplatin, and etoposide for patients with relapsed and refractory Hodgkin’s lymphoma: a non-randomised, open-label, single-centre, phase 2 study. Lancet Oncol. Mar 2015;16(3):284-92. doi:10.1016/S1470-2045(15)70013-6
- LaCasce AS, Bociek RG, Sawas A, et al. Three-year outcomes with brentuximab vedotin plus bendamustine as first salvage therapy in relapsed or refractory Hodgkin lymphoma. Br J Haematol. May 2020;189(3):e86-e90. doi:10.1111/bjh.16499
- Tun AM, Wang Y, Matin A, et al. Outcomes of Patients With Classic Hodgkin Lymphoma Who Relapsed After Autologous Stem Cell Transplant. Hemasphere. Apr 2023;7(4):e869. doi:10.1097/HS9.0000000000000869
- Badar T, Epperla N, Szabo A, et al. Trends in postrelapse survival in classic Hodgkin lymphoma patients after experiencing therapy failure following auto-HCT. Blood Adv. Jan 14 2020;4(1):47-54. doi:10.1182/bloodadvances.2019000736
- Baumeister SH, Freeman GJ, Dranoff G, Sharpe AH. Coinhibitory Pathways in Immunotherapy for Cancer. Annu Rev Immunol. May 20 2016;34:539-73. doi:10.1146/annurev-immunol-032414-112049
- Wu R, Sattarzadeh A, Rutgers B, Diepstra A, van den Berg A, Visser L. The microenvironment of classical Hodgkin lymphoma: heterogeneity by Epstein-Barr virus presence and location within the tumor. Blood Cancer J. May 6 2016;6(5):e417. doi:10.1038/bcj.2016.26
- Green MR, Monti S, Rodig SJ, et al. Integrative analysis reveals selective 9p24.1 amplification, increased PD-1 ligand expression, and further induction via JAK2 in nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Blood. Oct 28 2010;116(17):3268-77. doi:10.1182/blood-2010-05-282780
- Jalali S, Price-Troska T, Bothun C, et al. Reverse signaling via PD-L1 supports malignant cell growth and survival in classical Hodgkin lymphoma. Blood Cancer J. Feb 19 2019;9(3):22. doi:10.1038/s41408-019-0185-9
- Ansell SM. PD-1 Blockade in Classic Hodgkin Lymphoma. JCO Oncol Pract. Feb 2021;17(2):72-73. doi:10.1200/OP.20.01020
- Taylor JG, Truelove E, Clear A, Calaminici M, Gribben JG. PDL1 shapes the classical Hodgkin lymphoma microenvironment without inducing T-cell exhaustion. Haematologica. Apr 1 2023;108(4):1068-1082. doi:10.3324/haematol.2022.280014
- Amarnath S, Mangus CW, Wang JC, et al. The PDL1-PD1 axis converts human TH1 cells into regulatory T cells. Sci Transl Med. Nov 30 2011;3(111):111ra120. doi:10.1126/scitranslmed.3003130
- Kanamori M, Nakatsukasa H, Ito M, Chikuma S, Yoshimura A. Reprogramming of Th1 cells into regulatory T cells through rewiring of the metabolic status. Int Immunol. Jul 24 2018;30(8):357-373. doi:10.1093/intimm/dxy043
- Chen X, Fosco D, Kline DE, et al. PD-1 regulates extrathymic regulatory T-cell differentiation. Eur J Immunol. Sep 2014;44(9):2603-16. doi:10.1002/eji.201344423
- Francisco LM, Salinas VH, Brown KE, et al. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med. Dec 21 2009;206(13):3015-29. doi:10.1084/jem.20090847
- Yost KE, Satpathy AT, Wells DK, et al. Clonal replacement of tumor-specific T cells following PD-1 blockade. Nat Med. Aug 2019;25(8):1251-1259. doi:10.1038/s41591-019-0522-3
- Cader FZ, Hu X, Goh WL, et al. A peripheral immune signature of responsiveness to PD-1 blockade in patients with classical Hodgkin lymphoma. Nat Med. Sep 2020;26(9):1468-1479. doi:10.1038/s41591-020-1006-1
- Yang ZZ, Kim HJ, Villasboas JC, et al. Mass Cytometry Analysis Reveals that Specific Intratumoral CD4(+) T Cell Subsets Correlate with Patient Survival in Follicular Lymphoma. Cell Rep. Feb 19 2019;26(8):2178-2193 e3. doi:10.1016/j.celrep.2019.01.085
- Yang ZZ, Grote DM, Xiu B, et al. TGF-beta upregulates CD70 expression and induces exhaustion of effector memory T cells in B-cell non-Hodgkin’s lymphoma. Leukemia. Sep 2014;28(9):1872-84. doi:10.1038/leu.2014.84
- Yang ZZ, Grote DM, Ziesmer SC, et al. IL-12 upregulates TIM-3 expression and induces T cell exhaustion in patients with follicular B cell non-Hodgkin lymphoma. J Clin Invest. Apr 2012;122(4):1271-82. doi:10.1172/JCI59806
- Wu H, Tang X, Kim HJ, et al. Expression of KLRG1 and CD127 defines distinct CD8(+) subsets that differentially impact patient outcome in follicular lymphoma. J Immunother Cancer. Jul 2021;9(7) doi:10.1136/jitc-2021-002662
- Ansell SM, Lesokhin AM, Borrello I, et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N Engl J Med. Jan 22 2015;372(4):311-9. doi:10.1056/NEJMoa1411087
- Armand P, Chen YB, Redd RA, et al. PD-1 blockade with pembrolizumab for classical Hodgkin lymphoma after autologous stem cell transplantation. Blood. Jul 4 2019;134(1):22-29. doi:10.1182/blood.2019000215
- Kuruvilla J, Ramchandren R, Santoro A, et al. Pembrolizumab versus brentuximab vedotin in relapsed or refractory classical Hodgkin lymphoma (KEYNOTE-204): an interim analysis of a multicentre, randomised, open-label, phase 3 study. Lancet Oncol. Apr 2021;22(4):512-524. doi:10.1016/S1470-2045(21)00005-X
- Advani RH, Moskowitz AJ, Bartlett NL, et al. Brentuximab vedotin in combination with nivolumab in relapsed or refractory Hodgkin lymphoma: 3-year study results. Blood. Aug 12 2021;138(6):427-438. doi:10.1182/blood.2020009178
- Moskowitz AJ, Shah G, Schoder H, et al. Phase II Trial of Pembrolizumab Plus Gemcitabine, Vinorelbine, and Liposomal Doxorubicin as Second-Line Therapy for Relapsed or Refractory Classical Hodgkin Lymphoma. J Clin Oncol. Oct 1 2021;39(28):3109-3117. doi:10.1200/JCO.21.01056
- Mei MG, Lee HJ, Palmer JM, et al. Response-adapted anti-PD-1-based salvage therapy for Hodgkin lymphoma with nivolumab alone or in combination with ICE. Blood. Jun 23 2022;139(25):3605-3616. doi:10.1182/blood.2022015423
- Bryan LJ, Casulo C, Allen PB, et al. Pembrolizumab Added to Ifosfamide, Carboplatin, and Etoposide Chemotherapy for Relapsed or Refractory Classic Hodgkin Lymphoma: A Multi-institutional Phase 2 Investigator-Initiated Nonrandomized Clinical Trial. JAMA Oncol. May 1 2023;9(5):683-691. doi:10.1001/jamaoncol.2022.7975
- Diefenbach CS, Jegede O, Ansell SM, et al. Results from an Intergroup Randomized Phase II Study of the Combinations of Ipilimumab, Nivolumab and Brentuximab Vedotin in Patients with Relapsed/Refractory Classic Hodgkin Lymphoma: A Trial of the ECOG-ACRIN Research Group (E4412). Blood. 2023;142(Supplement 1):607-607. doi:10.1182/blood-2023-184531
- Herrera AF, LeBlanc M, Castellino SM, et al. Nivolumab+AVD in Advanced-Stage Classic Hodgkin’s Lymphoma. N Engl J Med. Oct 17 2024;391(15):1379-1389. doi:10.1056/NEJMoa2405888
- Desai SH, Merryman RW, Shah H, et al. PD-1 Blockade before Autologous Stem Cell Transplantation Improves Outcomes in Relapsed/Refractory Classic Hodgkin Lymphoma: Results from a Multicenter Cohort. Blood. 2023/11/02/ 2023;142:182. doi:https://doi.org/10.1182/blood-2023-179573
- Desai SH, Spinner MA, David K, et al. Checkpoint inhibitor-based salvage regimens prior to autologous stem cell transplant improve event-free survival in relapsed/refractory classic Hodgkin lymphoma. Am J Hematol. Mar 2023;98(3):464-471. doi:10.1002/ajh.26827
- Bazarbachi A, Boumendil A, Finel H, et al. The outcome of patients with Hodgkin lymphoma and early relapse after autologous stem cell transplant has improved in recent years. Leukemia. Jun 2022;36(6):1646-1653. doi:10.1038/s41375-022-01563-8
- Desai SH, Spinner MA, Evens AM, et al. Overall survival of patients with cHL who progress after autologous stem cell transplant: results in the novel agent era. Blood Adv. Dec 12 2023;7(23):7295-7303. doi:10.1182/bloodadvances.2023011205
- Stuver R, Drill E, Ganesan N, et al. Outcomes for Patients with Classical Hodgkin Lymphoma Who Relapse after Autologous Stem Cell Transplant in the Era of Novel Therapies. Blood. 2023/11/02/ 2023;142:3069. doi:https://doi.org/10.1182/blood-2023-182143
- Moskowitz A, Shah GL, Ganesan N, et al. Pembrolizumab Maintenance Instead of Autologous Hematopoietic Cell Transplantation for Patients with Relapsed or Refractory Hodgkin Lymphoma in Complete Response after Pembrolizumab, Gemcitabine, Vinorelbine, and Liposomal Doxorubicin. Blood. 2024/11/05/ 2024;144:569. doi:https://doi.org/10.1182/blood-2024-202537
- Voorhees T, McLaughlin E, Torka P, et al. Outcomes in Patients with Classical Hodgkin Lymphoma Refractory or Intolerant to Brentuximab Vedotin and Anti-PD-1 Therapy: Real World Analysis from 14 U.S. Academic Centers. Blood. 2023/11/02/ 2023;142:3061. doi:https://doi.org/10.1182/blood-2023-181056
- Armand P, Zinzani PL, Lee HJ, et al. Five-year follow-up of KEYNOTE-087: pembrolizumab monotherapy for relapsed/refractory classical Hodgkin lymphoma. Blood. Sep 7 2023;142(10):878-886. doi:10.1182/blood.2022019386
- Timmerman J, Lavie D, Johnson NA, et al. Favezelimab in Combination with Pembrolizumab in Patients with Heavily Pretreated Anti-PD-1-Refractory Classical Hodgkin Lymphoma: Updated Analysis of an Open-Label Phase 1/2 Study. Blood. 2023;142(Supplement 1):4440-4440. doi:10.1182/blood-2023-182019
- Herrera AF, Chen L, Budde LE, et al. Pembrolizumab Plus Vorinostat Induces Responses in Patients with Hodgkin Lymphoma Who Are Refractory to Prior PD-1 Blockade. Blood. 2021;138(Supplement 1):234-234. doi:10.1182/blood-2021-150031
- Bachanova V, Hegerova L, Cao Q, et al. Ruxolitinib Plus Nivolumab in Patients with R/R Hodgkin Lymphoma after Failure of Check-Point Inhibitors: Preliminary Report on Safety and Efficacy. Blood. 2021;138(Supplement 1):230-230. doi:10.1182/blood-2021-149675

