Outpatient Care in Systemic Lupus Erythematosus Complications
A Case Report: Outpatient Care & Recognizing the Complications of Systemic Lupus Erythematosus
Debra Penrod, DNP, RN, CRRN1
- School of Health Sciences—Nursing College of Health and Human Sciences Southern Illinois University Carbondale Carbondale, IL USA [email protected]
https://orcid.org/0000-0002-9288-6077
OPEN ACCESS
PUBLISHED: 31 October 2025
CITATION: Penrod, D., 2025. A Case Report: Outpatient Care & Recognizing the Complications of Systemic Lupus Erythematosus. Medical Research Archives, [online] 13(10).
https://doi.org/10.18103/mra.v13i10.7047
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i10.7047
ISSN 2375-1924
ABSTRACT
The objective of this case study is to study one patient’s trajectory from diagnosis to complications of Systemic Lupus Erythematosus that spanned 40 years. A discussion of the disorder will be explained as well as diagnosis and treatment. As an autoimmune disorder, this form of lupus triggers symptoms that make it difficult to pinpoint the diagnosis initially. Early diagnosis and treatment are paramount to managing the progression of lupus. Systemic Lupus Erythematosus can cause complications of infection, renal disease and heart failure. Treatment regimen themselves can cause untoward complications for patients if not managed closely. Through effective medical management of symptoms, regularly scheduled appointments which include lab evaluation and consulting specialists in monitoring these complications, patients can live an optimal quality of life. This case report is an example of a patient who received outpatient care which maintained her quality of life for many years with complications of renal failure and cardiogenic shock occurring later in life. This case serves as an example of one patient’s journey through systemic lupus erythematosus from a medical standpoint and the complications that can occur. Effective outpatient management of Systemic Lupus Erythematosus and when complications arise can help alleviate symptoms and promote optimal quality of life.
Keywords: Autoimmune disease, Systemic lupus erythematosus, acute and chronic renal disease, cardiovascular disease, pericardial effusion
Introduction
Systemic Lupus Erythematosus (SLE) is a chronic autoimmune disorder that can affect multiple systems that can manifest in life-threatening complications over time. SLE is described as a condition in which the body’s natural inflammatory response attacks healthy tissue as manifesting in a variety of ways from mild to severe. SLE affects women primarily who are diagnosed between the ages of 15 and 44 and who belong to the African American, Hispanic-Latino, Asian American, Native American, Native Hawaiian and Pacific Islander populations. Symptoms of arthritic pain in joints and morning stiffness, chronic fatigue, round, scaly rashes that can appear on any body part, photosensitivity and swelling in the legs are a few manifestations of SLE but every case can be different. This hyperinflammatory response can cause multiple system involvement including renal, neurological, and cardiovascular and respiratory complications. The mortality rate from SLE is two to three times higher than the general population globally. Causes of mortality are more predominantly from infection or cardiovascular disease. Patients with autoimmune disease also have a high probability of developing another autoimmune disorder as well which compounds treatment for the patient.
The pathophysiology of SLE indicates why multiple system involvement occurs. Autoantibodies are formed from the autoimmune response as large immune complexes deposit in the glomeruli and renal interstitium that causes scarring and cellular death. Anti-blood cell antibodies, anti-dsDNA antibodies and anti-phospholipid antibodies are specifically attributed to nephritis in addition to the large antibody-antigen immune complexes that lodge in organs. Minimizing lupus flares is paramount to successfully controlling these devastating complications. Chronic disease management is termed as “treat-to-target” by which the goal is to guide the patient into remission, control the remission and maintain this state with treatment without the use of glucocorticoids. Chloroquine and hydroxychloroquine are still used to block the autoimmune response given that patients adhere to the treatment regimen. Regular visits to monitor compliance are necessary to minimize complications from SLE. Research has shown that these antimalarial drugs can slow renal and retinopathy complications. As stated in their literature review, Katarzyna, Wiktor, Ewa, & Piotr reiterated that glucocorticoids are the mainstay of SLE treatment due to their ability to reduce inflammation. However, this drug class can cause irreversible organ damage which can lead to morbidity and mortality. The authors stated that glucocorticoids should be used cautiously and may be short term to minimize renal impairment.
The following case outlines the outpatient care and complications that arose from systemic lupus erythematosus which are not unusual for many patients.
The Case
OUTPATIENT CARE OF PATIENT
A Caucasian woman in her 30s living in rural midwestern United States was diagnosed with systemic lupus erythematosus after repeated breakouts of a red rash on her extremities as well as extreme fatigue. Once this diagnosis was established, the patient was started on prednisone therapy but could not tolerate the steroid long-term. The patient was prescribed hydroxychloroquine which maintained her exacerbations of SLE for many years. At the recommendation of her physician, this patient had a tubal ligation due to the lupus diagnosis in her mid-30s (around 1975). The patient was seen by an internal medicine physician and hematologist 45 minutes away from home for the first 15 years following diagnosis due to the absence of a rheumatologist in this rural area. In addition to quarterly lab orders, this patient’s disease process was managed with occasional days of extreme fatigue and mild skin eruptions. A rheumatologist became established in the area around 1990 so this patient began seeing him for specialized care. She worked as a certified nurse assistant at a local nursing home for 25 years and raised two children in the rural setting.
Following abnormal liver enzymes which led to a liver biopsy at age 65, this patient was diagnosed with autoimmune hepatitis and primary biliary cirrhosis which necessitated treatment with ursodiol. Celiac disease was discovered later at age 72 from a colonoscopy and Sjogren’s syndrome was diagnosed soon after. In addition to these autoimmune disorders, the patient also was treated for osteoporosis, hypothyroidism, hypertension and chronic renal failure. The patient’s glomerular filtration rate (GFR) was 58 and blood urea nitrogen (BUN) level was 20 at age 74 with a drop in GFR of 37 and an increase of BUN to 29 by age 76. The patient started seeing a cardiologist for blood pressure, atrial fibrillation and mild heart failure as well. The patient lived independently and was able to perform all activities of daily living up to the point of the following exacerbation of her disease.
ACUTE ONSET OF COMPLICATIONS
This patient, now age 77, was admitted to the emergency room with a gradual onset of weakness and some confusion. The patient was found at her home on the floor and brought to the emergency room by personal car. Her medical history was shared with the medical personnel, which included the above-mentioned diagnoses. This patient had recently been seen as an outpatient for pneumonia. The patient had no previous surgical history other than ankle fracture surgery, cataracts, cholecystectomy and right mastoidectomy and no other medical hospitalizations prior to this event.
At the time of this visit, the patient had been maintained on the following medications: allopurinol 300 mg daily, mycophenolate 500 milligrams (mg) twice (BID) a day, apixaban 5 mg BID, furosemide 40 mg BID, hydroxychloroquine 200 mg BID, levothyroxine 175 micrograms daily, metoprolol tartrate 50 mg daily, sacubitril-valsartan 24-26 mg BID, spironolactone 50 mg daily, ursodiol 500 mg BID and zoledronic acid 5 mg/100 milliliters (mL) yearly. Her labs a year before this date included the following: BUN 29, Creatinine 1.4, GFR 37.
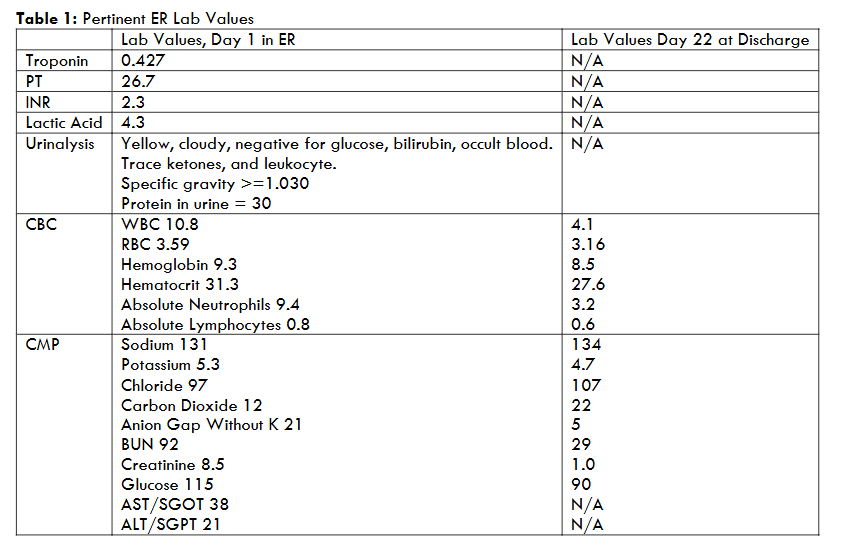
Upon assessment, the patient had a heart rate of 120 with distant heart sounds, blood pressure 76/51, and a pulse oximetry of 92% with tachypnea. Electrocardiogram was performed revealing atrial fibrillation with rapid ventricular response. She presented with edema as well as pallor but was alert and oriented to person, place, time and situation. The labs listed in Table 1, when compared to a year earlier, revealed worsening renal function. Computerized tomography scan of the chest revealed a moderate/large free-flowing pericardial effusion along the right heart border and a small left pleural effusion. The impression in the emergency room of objective and subjective data was in acute cardiogenic shock with possible septic shock, and acute respiratory failure. The patient had a history of Stage 3 Chronic Renal Failure but also was in acute renal failure superimposed on the already chronic state. An echocardiogram was also done which further confirmed a moderate to large free-flowing pericardial effusion circumferential to the heart with borderline evidence of cardiac tamponade at this time. Ejection fraction measured 60-65%. No two-dimensional evidence of hemodynamic compromise was noted. Active problems included cardiogenic shock, large pericardial effusion, pericarditis, troponin elevation, an elevated international normalized ratio of 2.3 and lactic acidosis. In this emergent state, treatment was aimed at decreasing the shock, acidosis and relieving the fluid pressing on the heart. After consideration, physicians attributed the cause to a severe lupus flare.
To relieve the pericardial effusion, a subxiphoid pericardial window procedure was performed which removed 500 mL of serosanguinous fluid from an area of moderate pericarditis. A trialysis catheter was placed to clear toxins post-procedure since the patient was uremic at this time with and dialysis anticipated a few days post-op. Two units of fresh frozen plasma were administered pre-procedure.
This patient was admitted to the intensive care unit (ICU) on mechanical ventilation following the pericardial window procedure. The trialysis line was created for dialysis pre and post procedure with the possibility of permanent dialysis. The patient’s last dialysis occurred on Day 11. The patient progressed the next two weeks in the ICU with mechanical ventilation the first two days post-operatively and 2 separate ultrasound-guided thoracentesis procedures a week later for right and left pleural effusions (675 mL removed from left and 250 mL removed from right lung). Other complications compounded this patient’s acute care stay which included difficulty swallowing and intermittent confusion. Kidney function continued to improve slowly post dialysis. Labs at discharge on Day 22 are listed in Table 1. The physician noted that there was a delay in the patient being weaned from the ventilator due to autoimmune hepatitis and primary biliary cirrhosis. A palliative care consult was requested by family due to patient’s slow progress and hospice was considered at intervals during the patient’s 3-week hospital stay. The physician noted that progress would be difficult to ascertain due to the autoimmune disease and to be prepared for hospice if improvement was not seen. The patient was discharged to a skilled care facility following hospitalization and spent the next 100 days participating in rehabilitation before discharging home with supervision.
Discussion
This case study shows outpatient care and acute complications that can occur because of SLE. When discussing her outpatient care, the story is familiar to other patients with this form of lupus. The patient’s lupus was controlled with the hydroxychloroquine, mycophenolate for nephroprotection and ursodiol which assisted in liver function due to the autoimmune hepatitis and primary biliary cirrhosis. It is not uncommon for patients with autoimmune disease to be diagnosed with other forms of autoimmune disease as well. In total, this patient had 5 different autoimmune disorders. Her history of osteoporosis aligns with 80-90% of patients with SLE. Chronic cardiac complications were managed with metoprolol tartrate, sacubitril-valsartan, apixaban, furosemide and spironolactone. Regular appointments with the primary internal medicine physician and the rheumatologist resulted in successful management of the patient’s lupus until her mid-70s.
Care for patients with SLE involves early intervention and timely referrals when needed. The scarcity of rheumatologists in rural areas along with the importance of early diagnosis and treatment, contributes to the severity of disease as well as complications. In this patient’s case, a rheumatologist was not available in the area for the first 15 years of care. Each patient with SLE has a unique path of disease that sometimes makes it difficult for providers to track and provide support. Research has suggested that “potential disruption points” interfere with optimal patient care. These include factors such as lack of healthcare provider training with SLE, delay of diagnosis, lack of specialty consults and management of the disorder, a management and referral strategy for lupus flares, interprofessional education and collaboration on cases, and lack of attention paid to SLE’s impact on activities of daily living. Other factors that play into the potential of complications include ethnicity, genetics, environmental factors such as smoking or pollutants, and socioeconomic status. Additionally, individual disease severity, coupled with any comorbidities, disease activity (remissions and flares), treatment regimen and access to care make an impact on health-related quality of life, organ damage and mortality.
The patient presented with symptoms that tend to be non-specific such as fatigue, flu-like symptoms, headache and joint pain, which is typical, making early SLE diagnosis difficult. When these symptoms combine with symptoms of inflammation, a light-sensitive rash, lymphadenopathy and abnormal labs, a diagnosis of lupus should be considered. The Arthritis Foundation lists the following labs that confirm the SLE diagnosis: an elevated erythrocyte sedimentation rate (ESR), elevated C-reactive protein (CRP), low hemoglobin and white blood cell counts, and positive levels of antinuclear antibodies. Baseline kidney function is a necessity following diagnosis as well as urine tests and imaging testing that includes chest x-ray and echocardiogram for baseline cardiac status. Biopsies may be necessary depending on how lupus has affected the patient. Ongoing labs that monitor lupus include anti-double-stranded DNA and complement proteins (C3, C4, and CH50).
Hydroxychloroquine therapy has been proven effective in treating SLE but not without its own potential complications. Frequent eye exams are important while on hydroxychloroquine therapy as well since its use long term can lead to retinal toxicity. A review of literature revealed the relationship between long-term use of hydroxychloroquine and cardiac complications which caused incidences of ventricular hypertrophy, hypokinesia, heart failure, pulmonary hypertension, and valvular dysfunction. Eighty-five percent of the patients who averaged 7 years of hydroxychloroquine therapy encountered cardiac conduction disorders. Monitoring the kidneys and protecting their function is paramount to managing lupus long-term. Nonsteroidal anti-inflammatory drugs should be avoided as well as iodinated radiocontrast, phosphorus containing laxatives, enemas and bowel preps that contribute to hyperphosphatemia. Aminoglycoside antibiotics and amphotericin B have the potential for nephrotoxicity as well as Gadolinium contrast. One concern with this patient could be that nephrology was not consulted earlier in this patient’s progression through chronic renal failure. Previous lab values revealed the changes in the patient’s GFR and BUN that may have necessitated a nephrology consult. The National Institute of Health recommends that patients see a nephrologist at any stage of Chronic Renal Disease and most certainly when the GFR drops below 60.
Pericardial effusions and pleuritis are actually common in patients with SLE. Symptomatic pericarditis occurred in 25% of patients in a 1999 study and asymptomatic cases were seen in over 50% of this study’s patients. A 2025 study revealed that 20% of patients with SLE experience pericardial effusions with reoccurrences also likely. Pleural effusions and bouts of pneumonia can occur as well. Some risk factors that could point to pericardial effusion development include hemolytic anemia, proteinuria, lymphadenopathy, and anti-Smith antibodies. Male gender and younger age can predict this likelihood. Patients with a pericardial effusion present with substernal pleuritic chest pain, dyspnea, fever, tachycardia and decreased or muffled heart sounds. A pericardial friction rub may be noted upon physician exam as well elevated ST segments with peaked T-waves. There is nothing to indicate that the patient in this case study had any of these symptoms prior to this acute event in the emergency room. This patient presented with cardiac tamponade which contributed to shock which, according to study from 1999, only 2% of SLE patients developed which was reiterated by the National Institutes of Health in 2023. Since pericardial effusion is the most common cardiac manifestation in SLE patients, early treatment is important and rarely results in cardiac tamponade as the patient in this case study experienced.
As was the case of the patient described in this study, prognosis is difficult with the patient’s history of autoimmune disease. SLE patients recovering from acute renal failure superimposed on chronic renal failure with autoimmune disorders involving the liver as well as declining heart function may not have a straight downhill trajectory which would make hospice decision-making unclear as was illustrated in this case study.
Conclusion
The goal of this case study was to provide the most current information that healthcare providers can use to effectively care for patients with chronic autoimmune disorders such as SLE and monitor for acute complications as they arise. The goal with this disorder is to diagnose early, refer to specialists when needed and monitor closely to prevent patients from life-threatening lupus flares. As was the situation with the patient case described in this article, several years of monitoring care while also supporting her unique presentation of lupus, helped this patient have years of independence and optimal quality of life. Careful monitoring of renal and cardiac function and recognizing when an acute lupus flare occurs is paramount to the patient’s survival living with this devastating disease.
Conflict of Interest & Funding Statement: I have no conflicts of interest to declare in the writing of this case study. I have no personal or financial relationships that influenced this work. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgments: I wish to thank Mary Ann Leggans for her contribution to this case study. She is a testimony of a person who enjoyed life to its fullest and did not allow a diagnosis steal her joy.
References
- Barber MRW, Drenkard C, Falasinnu T, et al. Global epidemiology of systemic lupus erythematosus. Nat Rev Rheumatol. 2021;17(9):515-532. doi:10.1038/s41584-021-00668-1
- National Resource Center on Lupus. Lupus Facts and Statistics. National Resource Center on Lupus. Published 2025. Accessed September 19, 2025. https://www.lupus.org/resources/lupus-facts-and-statistics
- Moulton VR, Suarez-Fueyo A, Meidan E, Li H, Mizui M, Tsokos GC. Pathogenesis of human systemic lupus erythematosus: A cellular perspective. Trends Mol Med. 2017;23(7):615-635. doi:10.1016/j.molmed.2017.05.006
- Justiz Vallant AA, Goyal A, Varacallo MA. Systemic Lupus Erythematosus. In: StatPearls. StatPearls Publishing; 2025. Accessed October 2, 2025. PMID: 30571026.
- Katarzyna PB, Wiktor S, Ewa D, Piotr L. Current treatment of systemic lupus erythematosus: a clinician’s perspective. Rheumatol Int. 2023;43(8):1395-1407. doi:10.1007/s00296-023-05306-5
- Alarcón GS, Ugarte-Gil MF, Pons-Estel G, Vila LM, Reveille JD, McGwin G. Remission and low disease activity state (LDAS) are protective of intermediate and long-term outcomes in SLE patients. Results from LUMINA (LXXVIII), a multiethnic, multicenter US cohort. Lupus. 2019;28:423-426. doi:10.1177/0961203319826693
- Pakchotanon R, Gladman DD, Su J, Urowitz M. Sustained complete renal remission is a predictor of reduced mortality, chronic kidney disease, and end-stage renal disease in lupus nephritis. Lupus. 2018;27:468-474. doi:10.1177/0961203317726376
- Alves P, Bashir MM, Wysocka M, Zeidi M, Feng R, Werth VP. Quinacrine suppresses tumor necrosis factor-α and IFN-α in dermatomyositis and cutaneous lupus erythematosus. J Invest Dermatol Symp Proc. 2017;18:S57-S63. doi:10.1016/j.jips.2016.11.001
- Chiavolini, D. Comorbidities in autoimmune disease and Multiple Autoimmune Syndrome. Global Autoimmune Institute: Empowering Solutions. 2025. Retrieved October 14, 2025 from https://www.autoimmuneinstitute.org/articles/comorbidities-in-autoimmune-disease-multiple-autoimmune-syndrome/
- Mosca M, Bruce IN, Andersen J, Ugarte-Gil MF, Arnaud L. Challenges and opportunities in access to care for systemic lupus erythematosus patients across Europe and worldwide. Rheumatology (Oxford). 2024;63(7):1772-1778. doi:10.1093/rheumatology/keae227
- Arnaud L, Tektonidou MG. Long-term outcomes in systemic lupus erythematosus: Trends over time and major contributors. Rheumatology (Oxford). 2020 Dec 5;59(Suppl5):v29-v38. doi: 10.1093/rheumatology/keaa382
- Dunkin M. Lupus tests. Arthritis Foundation. Published 2022. Accessed September 26, 2025. https://www.arthritis.org/health-wellness/about-arthritis/understanding-arthritis/lupus-testing
- Foundation of the American Society of Retina Specialists. Hydroxychloroquine-induced retinal toxicity. Retina Health Series. Published 2025. Accessed September 26, 2025. https://www.asrs.org/patients/retinal-diseases/45/hydroxychloroquine-induced-retinal-toxicity
- Chatre C, Roubille F, Vernhet H, Jorgensen C, Pers YM. Cardiac complications attributed to chloroquine and hydroxychloroquine: a systematic review of the literature. Drug Saf. 2018;41:919-931. doi:10.1007/s40264-018-0689-4
- Vaidya, SR, Aeddula, NR. Chronic Kidney Disease. In: StatPearls. StatPearls Publishing; 2025. Updated July 31, 2024. Accessed October 2, 2025. https://www.ncbi.nlm.nih.gov/books/NBK535404/
- Dein, E, Douglas, H, Petri, M, Law, G, Timlin, H. Pericarditis in Lupus. Cureus. 2019 Mar 1;11(3):e4166. doi: 10.7759/cureus.4166
- Kim, YJ, Lovell, J, Diab, A, Magder, LS, Goldman, D, Petri, M, Fava, A & Adamo, L. Incidence and Factors Associated With Recurrent Pericarditis in Lupus. JAMA Netw Open. 2025 Feb 3;8(2):e2461610. doi: 10.1001/jamanetworkopen.2024.61610
- Moder, KG, Miller, TD, & Tazelaar, HD: Cardiac involvement in systemic lupus erythematosus. Mayo Clin Proc. 1999, 74:275-284. 10.4065/74.3.275
- Amro, AM, Deeb, S, Rije, R, Deeb, N, Qunaibi, YY, Amr, B, Irzeqat, K, Alhadad, B, & Emar A. Systemic Lupus Erythematosus Presenting as Cardiac Tamponade and Pleural Effusion: A Case Report. Cureus. 2024 Jan 25;16(1):e52894. doi: 10.7759/cureus.52894




