P2X3R Antagonist DT-0111 and Its Effect on Taste in Mice
THE SELECTIVE P2X3R ANTAGONIST DT-0111 DOES NOT AFFECT TASTE IN MICE
Amir Pelleg, Ph.D.¹, Anu Mahadevan, Ph.D.², Hillary Ellis³, Michael G. Tordoff, Ph.D.³
- Danmir Therapeutics, LLC, Newtown Square, Pennsylvania,
- Organix, Inc. Woburn, Massachusetts,
- Monell Chemical Senses Center, Philadelphia, Pennsylvania
OPEN ACCESS
PUBLISHED: 31 May 2025
CITATION: Pelleg, A., et al., 2025. THE SELECTIVE P2X3R ANTAGONIST DT-0111 DOES NOT AFFECT TASTE IN MICE. Medical Research Archives, [online] 13(5).
https://doi.org/10.18103/mra.v13i5.6537
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i5.6537
ISSN 2375-1924
ABSTRACT
Adenosine 5′-triphosphate (ATP) is found in every cell of the human body where it plays a critical role in cellular energetics and metabolism. ATP is released from cells under physiological and pathophysiological conditions. Extracellular ATP acts as an autocrine and paracrine molecule, the effects of which are mediated by cell surface purinergic receptors (P2R). The activation of P2X3R and P2X2/3R localized on vagal sensory nerve terminals has been implicated in the sensation of pain as well as multiple pulmonary disorders including chronic obstructive pulmonary disease (COPD), asthma and chronic cough. DT-0111 is a novel water-soluble molecule that is being developed as a drug candidate for cough suppression and bronchodilation. DT-0111 acts as a selective antagonist at P2X3R vs. P2X2/3R. Because the latter participates in taste sensation, its blockade could explain loss of taste sensation by certain drug candidates targeting P2X3R. Thus, we tested the hypothesis that DT-0111 does not affect taste sensation in a mouse model in vivo. Using the mice-gustometer model, we found that at doses up to x15 the targeted clinical dose (0.142 mg/kg), DT-0111 did not affect taste sensation.
Keywords
- ATP
- P2X3R
- taste perception
- DT-0111
- mice
INTRODUCTION
Adenosine 5′-triphosphate (ATP) is a ubiquitous nucleotide found in every cell of the human body at concentrations up to 4mM¹. ATP is released from cells through multiple mechanisms²,³ under physiologic and pathophysiologic conditions⁴–⁶. Extracellular ATP is rapidly degraded to ADP, AMP and eventually adenosine by ecto-nucleotidases (mainly CD39 and CD73)⁷,⁸. Extracellular ATP acts as an autocrine and paracrine agent, the effects of which are mediated by cell surface P2 purinergic receptors (P2R)⁹. P2R are classified into two families¹⁰, metabotropic P2YR, which are seven trans-membrane domain G protein coupled receptors¹¹, and ionotropic P2XR, cation channels¹²,¹³. The activation by ATP of P2X3R and P2X2/3R expressed by primary afferent neurons plays an important mechanistic role in the sensation of pain¹⁴. In 1996, Pelleg and Hurt showed for the first time that extracellular ATP stimulates P2XR localized on vagal sensory nerve terminals in the lungs¹⁵. In the same year it was shown that aerosolized ATP caused bronchoconstriction in healthy human subjects and more so in asthmatic patients¹⁶. A subsequent study demonstrated that these two phenomena are linked as cause and effect¹⁷.
ATP generates vagal afferent neural traffic in both slow conducting C-fibers¹⁵ and fast conduction Aδ fibers¹⁸ that are known to mediate the cough reflex. It also sensitizes airway sensory nerves to tussive stimuli¹⁹,²⁰, and triggers cough in free moving guinea-pigs²¹. Since ATP and the potent, non-hydrolysable analog alpha, beta methylene ATP (α,β m-ATP) activate neurons in the vagal ganglia expressing mRNA for both P2X2R and P2X3R, it seems that P2X3R and P2X2/3R are involved in the tussigenic action of ATP²²,²³. Accordingly, in recent years, P2X3R antagonists have been developed as novel drug candidates for the treatment of chronic cough²⁴,²⁵. During clinical trials with one of these candidates (AF-219), loss of taste sensation (ageusia) was identified as a major side effect²⁶. This has been confirmed using psychophysical testing of both humans and mice²⁷, and was explained by the potential blockade of P2X2/3R (additional to P2X3R), which is known to be involved in taste sensation²⁷,²⁸.
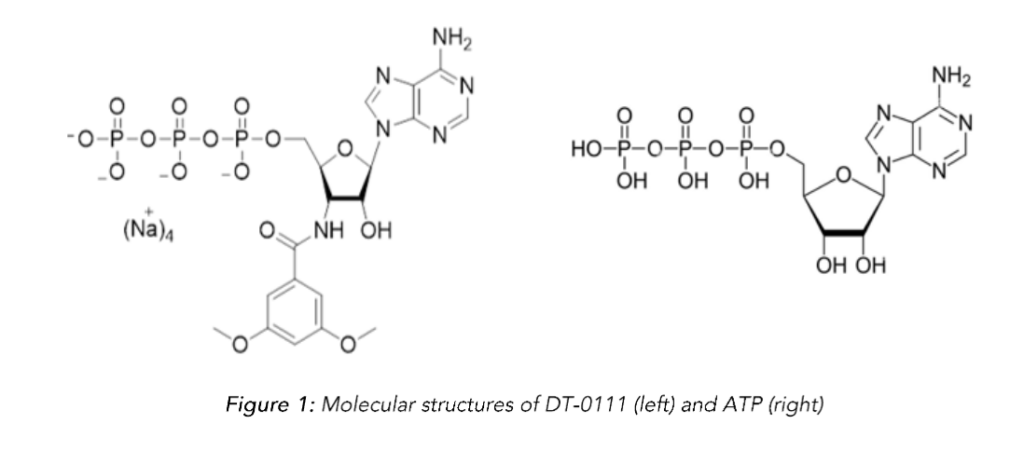
DT-0111 is a novel water-soluble small molecule (Fig. 1) that is being developed as a drug candidate for the treatment of COPD, asthma and chronic cough²⁹. Aerosolized DT-0111 suppressed aerosolized ATP-induced bronchoconstriction and cough in free moving guinea-pigs²¹. A recent study demonstrated that DT-0111 is a selective antagonist of P2X3R vs. P2X2/3R³⁰, thus, in the present study we tested the hypothesis that DT-0111 does not affect taste sensation in an animal model in vivo.
Materials and Methods
OVERVIEW:
A brief-access taste test was used to determine whether intraperitoneal injections of DT-0111 influenced taste responses. For this purpose, mice were first trained to drink in a gustometer, then quinine hydrochloride (QHCl), a prototypical bitter taste stimulus that is known to require ATP for successful transduction²⁷, was presented to the mice at different concentrations. The mice were treated with either a saline injection or with ascending doses of DT-0111.
ANIMALS AND MAINTENANCE:
The experiment involved 16 male C57BL/6J mice purchased from the Jackson Laboratory at age 7 weeks. Before commencement of the study, the mice were adapted for ~10 days to vivarium conditions; they were individually housed in tub-style cages with access to Teklad 8604 chow and deionized water; the vivarium was maintained at 23 C and kept on a 12:12 h light/dark cycle (as previously described²⁷).
ANIMAL TRAINING:
To train the mice to consume the taste solutions (i.e., either deionized water or water containing QHCl), they were first water-deprived for 22.5 h and then placed in a gustometer with its shutter open. Each gustometer consists of a 14.5 × 30 × 15 cm test chamber with a motorized shutter that controls access to a bottle with a drinking spout. Bottles containing taste solutions were mounted on a rack that was precisely positioned by a stepper motor such that any one of up to 16 taste solutions could be presented to a given mouse. The drinking spout of each bottle is part of a high frequency alternating current contact circuit so that each lick the mouse makes is detected and recorded.
During the first training session, each mouse had continuous access to water for 30 min from the time it first licked the drinking spout. It was then returned to its home cage and given water for 1 h. On the following two days, this procedure was repeated, except the shutter allowing access to water was closed 5 sec after each time the mouse began to lick, and it was reopened after a 7.5-sec interval. Once again, after 30 min, the mouse was returned to its home cage and given water for 1 h. By the 2nd test using these procedures, 14 of the 16 mice had learned to obtain water during the 5-sec access periods. The two recalcitrant mice were given two additional days of training.
TESTING ASSAY:
The mice then received a total of 18 tests, in which they drank either water or solutions of QHCl of various concentrations. Prior to each test, each mouse’s maintenance water supply was removed for 22.5 h. It was then given an intraperitoneal injection. Physiological saline solution (saline) was injected in the first two tests; subsequent pairs of tests involved injection of saline or 0.25, 0.5, 1, 2, 4, 8, and 16 mg/kg DT-0111; all injections were 0.0125 ml/g in volume. Accordingly, each mouse was tested 11 times after a saline injection and once with each of seven doses of DT-0111. Half of the mice received saline on the first test of each pair and DT-0111 on the second test; the other half received DT-0111 followed by saline.
Fifteen minutes after a given injection, the mouse was transferred from its home cage to a gustometer where it had access to water or a QHCl solution presented according to a repeating series of 5 concentrations (including water) of QHCl in a quasi-random order. The concentrations were 0 (deionized water), 0.032, 0.1, 0.32, or 1 mM QHCl, which prior work has shown span the range from indifference to strong avoidance in intact mice. Additional 1-sec “washout” trials with water were interposed between each test trial. Thus, a mouse received access to a QHCl solution for 5 sec followed by 7.5 sec with the shutter closed, then access to water for 1 sec followed by 7.5 sec with the shutter closed, followed by the next taste solution for 5 sec, and so on. The 1-sec “wash-out” with water helped maintain the mouse’s performance by “cleaning its palate” and preventing it from quitting because it expects only bad-tasting solutions. Test sessions lasted 20 min,
but mice generally stopped responding within the first 10 min of the test.
After the first of each pair of tests, each mouse received water for 1 h in its home cage and it was then water-deprived in preparation for the next session. After the second test of each pair, it received water for 24 h before being deprived for the next pair of tests.
BLOOD COLLECTION:
After the taste assays were completed, blood samples were withdrawn from the mice for the determination of DT-0111 plasma levels during the brief-access taste tests. Four blood samples were collected from each mouse, with 3–4 days between each sample collection. The mouse was water-deprived for 22.5 h (following the procedure for the behavioral tests). It then received an intraperitoneal injection of 0.25, 1, 4, or 16 mg/kg DT-0111, according to a counterbalanced order. At 15–20 min after the injection, ~50 μl blood was collected from the tip of the tail into a heparinized hematocrit tube. The blood was centrifuged at 2000 g for ~3 min to yield plasma, and the plasma stored at −78 C. Thus, we collected 16 samples of ~30 μl plasma after each of 4 doses of DT-0111: a total of 64 samples.
LEVELS OF DT-0111 IN PLASMA:
Waters Acquity UPLC and API4000 mass spectrometer were used by a contracted preclinical CRO.
Results
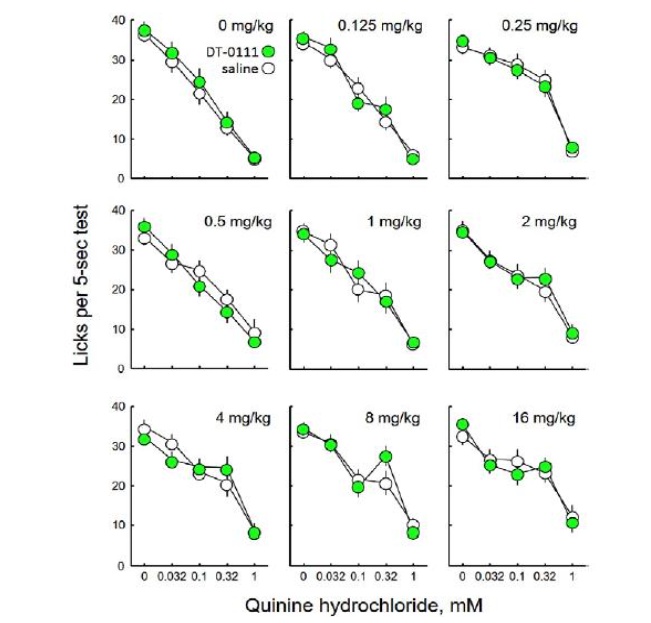
DT-0111 did not affect licking responses to QHCl; as can be seen in Figure 2, there was a little fluctuation in the gradient of the quinine concentration-response curve from test pair to test pair, which probably reflects subtle changes due to experience of the mice and their motivation to drink. However, responses on each pair of trials, one with saline and one with DT-0111, did not differ at any concentration of DT-0111 tested. This differs sharply from responses typically seen in mice that do not release ATP from taste cells²⁷,³¹,³². These mice, tested under nearly identical conditions, drank solutions containing various concentrations of QHCl (as were tested here) as if it was pure water, making 30–40 licks in each 5-sec access period even when the drinking solution contained 1 mM QHCl.
At the start and the end of the experiment, the mice weight was 21.6 ± 0.32 g (range 19.3 – 24.3 g), and 21.9 ± 0.35 g (range: 19.6 – 24.2 g) (mean ± SEM, n=14), respectively.
Unfortunately, the contracted CRO failed to establish a reliable methodology for DT-0111, i.e., one that yielded reproducible results. Because of these technical difficulties the amounts of DT-0111 were not detected in the plasma of blood samples withdrawn 15–20 min after its administration.
Discussion
In recent years there has been growing interest in the manipulation of signal transductions activated by extracellular ATP as new therapeutic modalities³³–³⁶. In particular, it is now well established that the activation of P2X3R is mechanistically involved in pain sensation³⁷, and chronic cough³⁸. Accordingly, several P2X3R antagonists have been have been developed as drug candidates for the treatment of chronic cough²⁴. During the relevant clinical trials, a side effect of ageusia was observed, and was attributed to undesirable antagonism of P2X2/3R, which is involved in taste sensation²⁸,³⁹.
Thus, in the present study we evaluated the effects of DT-0111, a novel potent P2X3R antagonist on taste sensation in mice. Our data show that in the current experimental model, at doses up to x15 the targeted clinical dose, DT-0111 did not affect taste sensation. In addition, there was no evidence for change in animals’ weights before vs. after experimentation indicating that the overall well being of the mice was stable. These results agree
well with results of our previous receptor selectivity study, which showed that DT-0111 is highly selective for homotrimeric P2X3R vs. the heterotrimeric P2X2/3R. The lack of BLU-5937’s (currently, GSK’s Camlipixant) effect on taste sensation was previously interpreted as significant selectivity for P2X3R vs. P2X2/3R⁴⁰. This explanation has been challenged by the recent findings that the taste buds of humans, unlike those of mice, are innervated by nerve fibers predominantly expressing homomeric P2X3R receptors and not heteromeric P2X2/3R³⁹.
It should be noted that a most recent study demonstrated that the binding site of Camlipixant localized on the P2X3R is different from that of ATP⁴¹, thus, indicating that Camlipixant is an allosteric modifier of P2X3R. Although the binding site of DT-0111 on P2X3R has not been determined, the similar molecular structure of DT-0111 and ATP (Fig. 2) strongly suggest that DT-0111 acts as a competitive antagonist at this receptor. Thus, although the binding sites of Camlipixant and DT-0111 are different, both manifest high selectivity for P2X3R vs. P2X2/3R³⁰,⁴¹.
STUDY LIMITATION:
The levels of DT-0111 at the taste buds were not determined, and therefore, it could be argued that due to the similarity between the molecular structure of DT-0111 and ATP (Fig. 1), which makes DT-0111 vulnerable to enzymatic degradation by ecto-phosphatases, these levels were below the threshold necessary to affect taste sensation. Accordingly, we are planning to conduct a confirmation study using a localized drug application model²⁷. That notwithstanding, the model used in the current study has been previously used for the similar evaluation of another P2X3R antagonist⁴⁰.
In conclusion, the present study strongly suggest that DT-0111 does not affect taste sensation in mice probably due to its preferential binding to P2X3R vs. P2X2/3R³⁰. Whether this binding preference holds in human P2X3R and P2X2/3R is yet to be determined.
References
1. Greiner JV, Glonek T. Intracellular ATP Concentration and Implication for Cellular Evolution. Biology (Basel) 2021;10(11). DOI: 10.3390/biology1 0111166.
2. Taruno A. ATP Release Channels. Int J Mol Sci 2018;19(3). DOI: 10.3390/ijms19030808.
3. Rahman M, Sun R, Mukherjee S, Nilius B, Janssen LJ. TRPV4 Stimulation Releases ATP via Pannexin Channels in Human Pulmonary Fibroblasts. Am J Respir Cell Mol Biol 2018;59(1):87-95. DOI: 10.1165/rcmb.2017-0413OC.
4. Pelleg A, Schulman ES. Adenosine 5′-Triphosphate Axis in Obstructive Airway Diseases. Am J Therap 2002;9(5):454-464. (http://dx.doi.org/10.1097/00045391-200209000-00014).
5. Bodin P, Burnstock G. Purinergic signalling: ATP release. Neurochem Res 2001;26(8-9):959-69. DOI: 10.1023/a:1012388618693.
6. Idzko M, Hammad H, van Nimwegen M, et al. Extracellular ATP triggers and maintains asthmatic airway inflammation by activating dendritic cells. Nature Med 2007;13(8):913-919.
(http://dx.doi.org/10.1038/nm1617).
7. Zimmermann H. Extracellular metabolism of ATP and other nucleotides. Naunyn-Schmiedeberg’s Arch Pharmacol 2000;362(4-5):299-309. (http://dx.doi.org/10.1007/s002100000309).
8. Zimmermann H, Zebisch M, Strater N. Cellular function and molecular structure of ecto-nucleotidases. Purinergic Signal 2012;8(3):437-502. DOI: 10.1007/s11302-012-9309-4.
9. Burnstock G. Purine and pyrimidine receptors. Cell Mol Life Sci 2007;64(12):1471-1483.
(http://dx.doi.org/10.1007/s00018-007-6497-0).
10. Kennedy C. The P2Y/P2X divide: How it began. Biochem Pharmacol 2021;187:114408. DOI: 10.1016/j.bcp.2021.114408.
11. von Kugelgen I, Hoffmann K. Pharmacology and structure of P2Y receptors. Neuropharmacology 2016;104:50-61. DOI: 10.1016/j.neuropharm.2015.1 0.030.
12. Jarvis MF, Khakh BS. ATP-gated P2X cation-channels. Neuropharmacology 2009;56(1):208-15. DOI: 10.1016/j.neuropharm.2008.06.067.
13. North RA. P2X receptors. Philos Trans R Soc Lond B Biol Sci 2016;371(1700). DOI: 10.1098/rstb.2 015.0427.
14. Inoue K. The Role of ATP Receptors in Pain Signaling. Neurochem Res 2022;47(9):2454-2468. DOI: 10.1007/s11064-021-03516-6.
15. Pelleg A, Hurt CM. Mechanism of action of ATP on canine pulmonary vagal C fibre nerve terminals. The Journal of physiology 1996;490 ( Pt 1):265-75. (http://www.ncbi.nlm.nih.gov/pubmed/8745294).
16. Pellegrino R, Wilson O, Jenouri G, Rodarte JR. Lung mechanics during induced bronchoconstriction. J Appl Physiol (1985) 1996;81(2):964-75. DOI: 10.11 52/jappl.1996.81.2.964.
17. Katchanov G, Xu J, Schulman ES, Pelleg A. ATP causes neurogenic bronchoconstriction in the dog. Drug Dev Res 1998;45(3-4):342-349.
(http://dx.doi.org/10.1002/(sici)1098-2299(199811/12)45:3/4<342::aid-ddr34>3.0.co;2-p).
18. Pelleg A, Schulman ES, Barnes PJ. Extracellular Adenosine 5′-Triphosphate in Obstructive Airway Diseases. Chest 2016;150(4):908-915. DOI: 10.1016/j.chest.2016.06.045.
19. Kamei J, Takahashi Y, Yoshikawa Y, Saitoh A. Involvement of P2X receptor subtypes in ATP-induced enhancement of the cough reflex sensitivity. Eur J Pharmacol 2005;528(1-3):158-161.
(http://dx.doi.org/10.1016/j.ejphar.2005.10.030).
20. Kamei J, Takahashi Y. Involvement of ionotropic purinergic receptors in the histamine-induced enhancement of the cough reflex sensitivity in guinea pigs. Eur J Pharmacol 2006;547(1-3):160-4. DOI: 10.1016/j.ejphar.2006.07.034.
21. Pelleg A, Xu F, Zhuang J, Undem B, Burnstock G. DT-0111: a novel drug-candidate for the treatment of COPD and chronic cough. Ther Adv Respir Dis 2019;13:1753466619877960. DOI: 10.1177/1753466619877960.
22. Pelleg A U, BJ. A-317491 Inhibits the activation of guinea-pig pulmonary vagal sensory nerve terminals by α,β methylene_ATP. Clin Immunol 2005;115(Suppl 1):S59-60.
23. Kwong K, Kollarik M, Nassenstein C, Ru F, Undem BJ. P2X2 receptors differentiate placodal vs. neural crest C-fiber phenotypes innervating guinea pig lungs and esophagus. AJP: Lung Cell Mol Physiol 2008;295(5):L858-L865.
(http://dx.doi.org/10.1152/ajplung.90360.2008).
24. Irwin R, Madison, JM. Uexplained or refractory chronic cough in adults. N Engl J Med 2025;392:1203-14. (Review). DOI: 10.1056/NEJMr a2309906.
25. Dicpinigaitis PV, McGarvey LP, Canning BJ. P2X3-Receptor Antagonists as Potential Antitussives: Summary of Current Clinical Trials in Chronic Cough. Lung 2020;198(4):609-616. DOI: 10.1007/s 00408-020-00377-8.
26. Smith JA. The Therapeutic Landscape in Chronic Cough. Lung 2024;202(1):5-16. DOI: 10.100 7/s00408-023-00666-y.
27. Flammer LJ, Ellis H, Rivers N, et al. Topical application of a P2X2/P2X3 purine receptor inhibitor suppresses the bitter taste of medicines and other taste qualities. Br J Pharmacol 2024;181(17):3282-3299. DOI: 10.1111/bph.16411.
28. Finger TE, Danilova V, Barrows J, et al. ATP signaling is crucial for communication from taste buds to gustatory nerves. Science 2005;310(5753): 1495-9. DOI: 10.1126/science.1118435.
29. Pelleg A, Barnes PJ, Schulman ES. Targeting the ATP-axis in lungs as a new therapeutic modality for COPD. Med Res Arch 2023;11(10). DOI: 10.18103/m ra.v11i10.4487.
30. Pelleg A, Sirtori E, Rolland JF, Mahadevan A. DT-0111: a novel P2X3 receptor antagonist. Purinergic Signal 2023. DOI: 10.1007/s11302-023-09930-5.
31. Ma Z, Taruno A, Ohmoto M, et al. CALHM3 Is Essential for Rapid Ion Channel-Mediated Purinergic Neurotransmission of GPCR-Mediated Tastes. Neuron 2018;98(3):547-561 e10. DOI: 10.1016/j.neuron.20 18.03.043.
32. Taruno A, Vingtdeux V, Ohmoto M, et al. CALHM1 ion channel mediates purinergic neurotransmission of sweet, bitter and umami tastes. Nature 2013;495(7440):223-6. DOI: 10.1038/natur e11906.
33. Jacobson KA, AP IJ, Muller CE. Medicinal chemistry of P2 and adenosine receptors: Common scaffolds adapted for multiple targets. Biochem Pharmacol 2021;187:114311. DOI: 10.1016/j.bcp.2 020.114311.
34. Kong Q, Quan Y, Tian G, Zhou J, Liu X. Purinergic P2 Receptors: Novel Mediators of Mechanotransduction. Front Pharmacol 2021;12: 671809. DOI: 10.3389/fphar.2021.671809.
35. Vultaggio-Poma V, Falzoni S, Salvi G, Giuliani AL, Di Virgilio F. Signalling by extracellular nucleotides in health and disease. Biochim Biophys Acta Mol Cell Res 2022;1869(5):119237. DOI: 10.101 6/j.bbamcr.2022.119237.
36. Pelleg A. Extracellular adenosine 5′-triphosphate in pulmonary disorders. Biochem Pharmacol 2021;187:114319. DOI: 10.1016/j.bcp.2 020.114319.
37. Huerta MA, Marcos-Frutos D, Nava J, Garcia-Ramos A, Tejada MA, Roza C. P2X3 and P2X2/3 receptors inhibition produces a consistent analgesic efficacy: A systematic review and meta-analysis of preclinical studies. Eur J Pharmacol 2024;984:177052. DOI: 10.1016/j.ejphar.2024.177 052.
38. Ramadan A, El-Samahy M, Elrosasy A, et al. Safety and efficacy of P2X3 receptor antagonist for the treatment of refractory or unexplained chronic cough: A systematic review and meta-analysis of 11 randomized controlled trials. Pulm Pharmacol Ther 2023;83:102252. DOI: 10.1016/j.pupt.2023.102252.
39. High B, Jette ME, Li M, et al. Variability in P2X receptor composition in human taste nerves: implications for treatment of chronic cough. ERJ Open Res 2023;9(2). DOI: 10.1183/23120541.000 07-2023.
40. Garceau D, Chauret N. BLU-5937: A selective P2X3 antagonist with potent anti-tussive effect and no taste alteration. Pulm Pharmacol Ther 2019;56: 56-62. DOI: 10.1016/j.pupt.2019.03.007.
41. Thach T, Dhanabalan K, Nandekar PP, et al. Mechanistic insights into the selective targeting of P2X3 receptor by camlipixant antagonist. The Journal of biological chemistry 2025;301(1):108109. DOI: 10.1016/j.jbc.2024.108109.




