Pediatric Skull LCH: FNB vs. Surgical Resection Outcomes
How Do I Diagnose Pediatric Patients’ Skull Langerhans Cell Histiocytosis Lesions?
Anat Ilivitzki MD 1, Michal Scolnik MD 2, Dana Ashkenazi MD 3, Myriam Ben-Arush Prof 3
- Department of Radiology Rambam Health Care Campus, Haifa, Israel.
- Department of Rehabilitation, Reuth Rehabilitation Hospital, Tel Aviv, Israel.
- Department of Pediatric Oncology, The Ruth Rappaport Children’s Hospital, Rambam Health Care Campus, Haifa, Israel.
OPEN ACCESS
PUBLISHED: 31 March 2025
CITATION: Ilivitzki, A., Scolnik, M., et al., 2025. How Do I Diagnose Pediatric Patients’ Skull Langerhans Cell Histiocytosis Lesions? Medical Research Archives, [online] 13(3).https://doi.org/10.18103/mra.v13i3.6370
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i3.6370
ISSN 2375-1924
Abstract
Background: Langerhans Cell Histiocytosis (LCH) commonly affects pediatric patients, with skull lesions being frequent. Diagnosis typically requires histopathologic confirmation. While surgical resection has been the standard approach, percutaneous fine needle biopsy (FNB) is emerging as a less invasive alternative.
Objective: To compare diagnostic performance, complications, procedure availability, hospital stay, and lesion regression outcomes between surgical resection and percutaneous FNB for pediatric skull LCH lesions.
Methods: We retrospectively reviewed 35 pediatric cases diagnosed with skull LCH between 2002 and 2024. Diagnostic procedures included surgical resection and percutaneous ultrasound-guided FNB. Data collected included demographics, procedure timing, complications, hospital stay, and follow-up outcomes, with particular focus on lesion regression post-biopsy.
Results: Of 35 cases, 26 underwent surgical resection and 9 underwent FNB. Diagnostic accuracy was 100% for surgery and 89% for FNB. The median time from biopsy request to procedure was significantly shorter for FNB (1 day) compared to surgery (12 days). Complications were more frequent in the surgical group (53.8%), including infections and cranial deformities, while no complications were observed in the FNB group. Notably, 4 cases (11%) showed lesion regression; 2 spontaneously regressed, and 2 others after biopsy, including a temporal bone lesion with central nervous system risk that remained disease-free after 17 years.
Conclusion: Percutaneous FNB is a safe, effective, and less invasive alternative to surgical resection for pediatric skull LCH lesions, offering faster procedure times, fewer complications, and the potential for lesion regression post-biopsy. This supports biopsy as the first-line diagnostic approach when feasible.
Keywords: Langerhans Cell Histiocytosis, biopsy, pediatric oncology, ultrasound, skull lesion
Introduction
Langerhans cell histiocytosis (LCH) is a rare disease, most often seen in children between the ages of 1 and 3 years old, and the most common manifestation is a skull lesion. Pediatric skull lesions are frequently detected and characterized through imaging modalities. The patient’s age and presenting symptoms are pivotal in refining the differential diagnosis. Among symptomatic older children, the primary differential considerations include LCH, osteomyelitis, Ewing’s sarcoma, osteosarcoma, lymphoma/leukemia, and metastatic neuroblastoma. Specifically, in the case of orbital lesions, the oncologic differential diagnosis includes LCH, rhabdomyosarcoma and neuroblastoma, as well as benign pathologies such as fibrous dysplasia. The broad differential diagnosis associated with pediatric skull lesions emphasizes the critical role of imaging in accurately differentiating these entities, and identifying which ones need further workup. In the case of malignancies, including LCH, pathologic confirmation is mandatory.
Langerhans cell histiocytosis is a difficult condition to characterize, indeed experts continue to debate whether it is a malignant process or an immune-mediated inflammatory state, with hyperactivity or hyperproliferation, respectively, of histiocytes. Recent research indicates that LCH is a clonal tumor originating from the unregulated proliferation and accumulation of immature myeloid dendritic cells that originate from the bone marrow. Regardless of the exact etiology, these aggregates of histiocytes form granulomatous infiltrative lesions that can mimic many other granulomatous conditions. Langerhans cell histiocytosis can manifest in almost any system in the body, in a wide variety of tissues, including but not limited to lung, skin, lymphoid, liver and subthalamic tissue, although, it has a strong predilection for bone, especially the skull. Additionally, more than 100 different subtypes of LCH have been identified, and it can present as a focal lesion or with multiorgan involvement.
The incidence of LCH in the pediatric population is reported to be approximately three to five cases per million. The clinical trajectory of LCH can range from self-limiting disease to aggressively progressive manifestations that may prove fatal. It is observed that between 30% and 40% of patients experience permanent sequelae. Skull bone LCH lesions, excluding the vault, are classified as central nervous system (CNS) risk lesions, carrying a potential risk for the onset of irreversible neurodegenerative CNS pathologies. Treatment options differ based on disease extent and severity at presentation.
Due to the wide variety of presentations of LCH, as well as the broad differential diagnosis associated with granulomatous lesions, and with lesions of the skull, diagnosis of LCH remains a challenge. The final diagnosis of LCH should always include histological and immunophenotypic analysis of lesional tissue, obtained from the most accessible yet representative lesion. Historically, surgical intervention with complete resection of the lesion and adjacent bone was the standard for obtaining biopsies in pediatric LCH cases involving the skull. In recent years, accumulating data have documented the efficacy of percutaneous fine-needle biopsy (FNB) for tissue diagnosis in pediatric lesions, leading to increasing popularity of FNB for various solid tumors. However, this shift has not been observed in the context of skull lesions.
The purpose of our research is to analyze the efficacy of ultrasound guided percutaneous biopsies, in pediatric lesions of the skull and face suspected to be Langerhans Cell Histiocytosis, by comparing this technique with traditional surgical biopsy. The parameters examined were whether a correct outcome was reached (diagnosis of LCH), time to procedure, complications associated with each procedure, availability of the procedure, the length of post procedure hospital stay, and whether lesion regression was observed, spontaneously or post procedural. We also discuss the limitations of percutaneous biopsies and suggest a logical approach to when tissue sampling is an appropriate option in pediatric skull lesions.
Material and Methods
We retrospectively studied all pediatric cases of skull lesions at our hospital between 2002 to 2024, that were pathologically confirmed as LCH. The diagnostic procedures performed were either craniotomy and lesion resection with subsequent pathology analysis or Ultrasound Guided Fine Needle Biopsy (US guided FNB) with pathology analysis; after 2019 on-site cytology was included as well. The charts of these patients were reviewed retrospectively. Data collection included demographics, clinical data, reason for the procedure, time lag between the request for a biopsy and the procedure (either surgery or percutaneous biopsy), sonographer, neurosurgeon, and anesthesiologist reports of the procedures, pathologic and cytological reports, and clinical follow-up immediately after the procedure. Long-term follow-up included clinical evaluation, imaging, and pathology reports. This work was approved by our local Institutional Review Board.
SURGICAL RESECTION OF THE LESIONS:
These procedures were performed under general anesthesia with intubation in the operating room. The lesion was removed and the surrounding abnormal bone tissue was sampled. During surgery, the neurosurgeon assessed for dural invasion. In earlier years a cranioplasty was performed as a second surgery and later on either cement or titanium mesh replaced the resected bone.
PER CUTANEOUS BIOPSIES:
The percutaneous biopsies were performed under sonographic guidance in the US suite. The team included an experienced pediatric radiologist, US technician, a nurse, and an anesthesiologist. All cases were carried out under deep sedation without intubation. We performed sonographic-guided biopsies of lytic bone lesions of the skull and facial bones with soft tissue mass. After a thorough examination of the child, the biopsy site, positioning of the child, and the probe were chosen. Percutaneous biopsy was an option whenever there was a soft tissue bulge above the skull level and the biopsy was tangential to the skull. For the core biopsy, we used a spring-loaded core biopsy needle (Super Core Biopsy Instrument; Angiotech), 16- to 18-G caliber. Core biopsies were sent for pathologic evaluation (3 cores in most cases), molecular biology, and genetic studies as needed. During 2019, we added an onsite smear cytology to some of our biopsies.
To assess diagnostic performance of both types of procedures, we identified and reviewed the charts of children who were diagnosed with LCH at our institution, checked whether they had undergone surgical resection or a percutaneous ultrasound-guided biopsy, and whether the pathological sample from each procedure had been diagnostic or not of LCH. We also reviewed each patient’s chart to ascertain the time lag between the request for diagnostic procedure until the procedure itself, as an assessment of procedure availability. Additionally, charts were analyzed for the length of the hospital stay surrounding the procedure, and the complications associated with the procedure itself. Imaging and charts were assessed for follow up to determine lesion recurrence. Skull contours on imaging were examined for closure of bone and esthetic outcomes, as areas of flattened, misshapen skull bone are very noticeable externally; this was relevant to surgical resection only. In cases of percutaneous biopsies, where lesions were sampled rather than removed, skull contours were not drastically changed, and the lesion was closely tracked on imaging enabling us to investigate lesion regression post biopsy. If the diagnostic procedure failed to provide a correct or conclusive diagnosis, we studied the reasons for the failure and the additional means taken to reach a final diagnosis according to the chart, imaging and pathological report, for example the pathology report indicated that the sample obtained was insufficient for diagnosis due to amount or quality, and the chart follow up revealed that a surgical biopsy was subsequently conducted and proved to be diagnostic.
Results
Between 2002 and 2024, 38 children presented to our institution with skull lesions and were pathologically diagnosed as Langerhans Cell Histiocytosis (LCH). In order to compare the surgical diagnostic procedure to the percutaneous biopsy we included only lytic bone lesion accompanied by a soft tissue component that enabled a percutaneous biopsy, and thus 3 cases were excluded. Diagnosis was obtained via surgical excision of the lesion in the majority of the cases (26 cases; 17 boys and 9 girls; age 3 months to 25 years, average 13 years and 4 months), while a minority underwent percutaneous biopsy (9 cases; 4 boys and 5 girls; age 1 month to 13 years, average 4 years and 3 months). All biopsies were performed to establish diagnosis following imaging findings on either X-ray, ultrasound (US), computed tomography (CT) or magnetic resonance imaging (MRI), suggestive of LCH.
Most patients were referred to surgical diagnostic procedures by general pediatricians (21) while only several were referred by pediatric oncologists (5), whereas referral for percutaneous biopsies were obtained exclusively from pediatric oncologists. The diagnostic workup alerting physicians to the suspicion of malignancy and the need of a biopsy, included clinical and imaging findings in all cases. Different, and sometimes multiple, imaging modalities were used including x-ray, US, CT, MRI and PET-CT. Tables 1 and 2 summarize the lesion parameters on imaging, including location, size of the lesion, the procedures performed, complications, length of stay, additional treatment and length of follow-up.
| Patient number | Location/Laterality | Size (cm) | Hospital stay (days) | Treatment | BRAF | Complication | Closure of bone defect: Mesh/Cement/Cranioplasty | Follow Up (years) | Recurrence | Multisystem/Multifocal | CNS Risk |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | F Lt | 3.5 | N/A | no | 2 | no | Not taken | no | 13 | no | no |
| 2 | T Rt | 5.8 | N/A | no | 2 | yes | yes | positive | no | 5 | no |
| 3 | F Lt | 3.6 | Delayed cranioplasty | no | 2 | no | Not taken | no | 12 | no | no |
| 4 | F Mid | 4 | no | Abscess | 3 | no | Not taken | no | 13 | no | no |
| 5 | P Lt | 1.5 | no | no | 2 | no | Not taken | no | 12 | no | no |
| 6 | F Rt | 4.5 | no | no | 4 | no | yes | 18 | no | yes | no |
| 7 | F Lt | 3 | no | no | 2 | no | yes | 20 | no | yes | no |
| 8 | F Rt | 3 | no | no | 2 | no | Not taken | no | 29 | no | no |
| 9 | O Mid | 3 | no | esthetic | 4 | no | Not taken | no | 10 | no | no |
| 10 | N/A | 4 | N/A | N/A | 2 | no | Not taken | no | 23 | no | no |
| 11 | N/A | 3.5 | Delayed cranioplasty | N/A | 2 | no | Not taken | no | 27 | no | no |
| 12 | O Rt | 4.2 | mesh* | no | 2 | no | Not taken | no | 5 | no | no |
| 13 | P Rt | 2.5 | mesh | no | 3 | no | Not taken | no | 19 | no | no |
| 14 | P Lt | 4.5 | cement | no | 2 | no | Not taken | no | 11 | no | no |
| 15 | F Lt | 4 | cement | no | 2 | no | Not taken | no | 12 | no | no |
| 16 | F Mid | 1.6 | cement | Hematoma | 3 | yes | no | 1 | no | yes | no |
| 17 | T Lt Orbit | 3 | mesh | 2 | no | yes | 1 | no | no | no | |
| 18 | P Rt | N/A | Surgical biopsy © | no | 2 | no | Not taken | no | 4 | no | no |
| 19 | F Lt | 4.7 | mesh | esthetic | 2 | no | Not taken | no | 10 | no | no |
| 20 | P Rt | 1.5 | mesh | no | 2 | no | Not taken | no | 12 | no | no |
| 21 | F Rt | 1.7 | no | esthetic | 4 | no | yes | 16 | no | no | yes |
| 22 | P Rt | 2.5 | no | Early | 1 | positive | yes | 3 | no | no | no |
| 23 | O Mid | 2 | no | no | 2 | no | Not taken | no | 3 | no | no |
| 24 | P Lt | 2.5 | no | no | 2 | no | Not taken | no | 3 | no | no |
| 25 | P Lt | 2.7 | cement | no | 2 | no | Not taken | no | 5 | no | no |
| 26 | F Lt | 3 | cement | no | 2 | no | Not taken | no | 5 | no | no |
| FNB Patient number | Location/Laterality | Biopsy Gun gauge (G) | Cytology | BRAF | Hospital stay (days) | Resection post biopsy | Recurrence/Regression | Follow Up (years) | Multisystem/Multifocal | CNS Risk |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | T Lt | 16 | diagnostic | positive | 2 | yes | Recurrence (twice) | 5 | no | no |
| 2 | O M | 16 | Not taken | no | 2 | no | Not taken | 15 | no | no |
| 3 | P Rt | 18 | Not taken | no | 3 | no | Not taken | 9 | no | no |
| 4 | NA | 18 | Not taken | Regression | no | 1 | yes¹ | 17 | no | no |
| 5 | T Lt | 18 | Not taken | no | no | no | Not taken | 6 | no | no |
| 6 | T Rt | 16 | diagnostic | Negative | 1 | yes | Recurrence | 4 | no | no |
| 7 | F orbit Lt | 18 | Not taken | no | no | yes | Not taken | 6 | no | no |
| 8 | P Rt | 18 | Not taken | no | no | no | Not taken | 14 | no | no |
| 9 | F Rt | N/A | Not taken | no | no | no | Not taken | no | no | no |
SURGICAL PROCEDURES
The median time between the biopsy request and procedure was 12 days (range 1–36 days). Surgical biopsy was diagnostic in all cases. Surgical interventions included resection in 24 cases and biopsy in 2 cases. Dural invasion was observed in only one instance. Bone replacement procedures were performed in 11 cases; one patient required a second operation for cranioplasty, 6 patients underwent cement replacement for skull defects, and 4 patients received mesh placement. Data were unavailable for 3 cases. The mean hospital stay following the procedure was 2.6 days (range 1–9 days), with 2 patients requiring intensive care on the first postoperative day.
Complications occurred in 14 patients (53.8%). Immediate postoperative complications occurred in 6 cases: three minor complications (two patients developed fluid collection at the site of resection, one with fever, another developed a hematoma at the surgical site) and three major complications (an infected titanium mesh, which was initially treated with antibiotics and later removed in a second operation, abscess at the surgical site, and a second surgery for completion of lesion resection in one patient). Late complications occurred in 8 cases and included headache in 2 patients, chronic sinusitis in one patient and esthetic cranial deformation in 5 patients. No anesthesia-related complications were reported.
PERCUTANEOUS BIOPSY
The median time between biopsy request and procedure was 1 day (range same day to 10 days), with 4 patients undergoing the biopsy within a day. Most procedures (8/9) were performed on hospitalized patients, with 1 performed on an outpatient basis. All procedures were conducted under deep sedation. The biopsy was diagnostic in 8 of 9 patients; the only non-diagnostic biopsy failed due to the absence of a soft tissue component adjacent to the lytic bone lesion, necessitating a subsequent surgical biopsy that was diagnostic. In two cases, on-site cytology was also performed and proved diagnostic. Core needle biopsy sizes were 18 G in 60% of cases and 16 G in 40%. The mean number of passes was 3. No complications related to anesthesia or the biopsy procedure were recorded. The mean hospital stay was 1.5 days (range same-day discharge to 3 days).
BRAF TESTING
Genetic testing for the BRAF mutation was incorporated into the oncologic protocol in recent years. A total of 9 patients were tested, with 5 returning positive results.
TREATMENT
The treatment protocol adhered to LCH protocol. Nine patients were deemed eligible for systemic chemotherapy, including 6 due to CNS involvement risk and 4 due to multiple recurrences.
REGRESSION OF LESIONS
Regression of lesions occurred in 4 cases. In 2 cases regression was spontaneous, one lesion regressed completely and the second decreased in size while the patient awaited surgery, yet it was nevertheless resected. Biopsy initiated complete regression in 2 cases, the first case post-surgical biopsy and the second post FNB. The FNB case was a temporal lesion with CNS risk, yet complete regression did not require further treatment and no recurrence was reported in 17 years of follow up.
Discussion
LCH can involve any organ or system, with bone involvement occurring in 80% of cases, many of which involve skull lesions. Clinical presentation and imaging alone are insufficient for a definitive diagnosis, as differential diagnoses vary depending on lesion location, age, and presentation; hence, histopathologic confirmation is required. Recent advancements have included the incorporation of BRAF mutation testing as part of the primary workup for LCH patients.
Pediatric LCH patients with bone lesions are categorized into four groups: Single-lesion Mono-system, Multi-lesion Mono-system, CNS risk, and Multisystem disease. Systemic treatment, typically steroids combined with vincristine, is indicated for all groups except for the Single-lesion Mono-system group. Accordingly, biopsy, rather than resection, would suffice for those treated systemically. In cases of isolated bone lesions without CNS risk, expectant management, topical treatment, surgery, and, in select cases, systemic therapy is acceptable according to the LCH protocol. This justifies the initial use of biopsy, particularly for the latter group.
Historically, surgical resection was considered the first step for skull lesions, providing both a histologic diagnosis and definitive treatment, especially in cases of solitary lesions. However, resection carries risks, including cranial defects, anesthesia complications, and unnecessary procedures in cases where the diagnosis is a systemic disease, such as metastatic neuroblastoma or lymphoma. Recent studies comparing open biopsy to fine needle biopsy (FNB) in tissue diagnosis in pediatric oncology, suggest that percutaneous biopsy, especially under ultrasound guidance, is a safer and more efficient option. Despite limited experience with percutaneous biopsy for skull lesions, two retrospective studies on CT-guided biopsies have demonstrated the procedure’s high diagnostic yield (91% in 23 cases in Betting et al. and 93% in 14 cases in Tomasian et al.), with minimal complications; Betting et al. reported one minor complication pertaining to sedation, and Tomasian et al. did not have any complications to report.
Our series of percutaneous ultrasound-guided skull biopsies represents the largest reported in the literature, with results that are comparable to those of CT-guided biopsies. In our cohort of LCH patients with skull and facial lesions, the diagnostic accuracy was 100% for the surgical group (all 26 cases) and 89% for the FNB group (8 out of 9 cases), with the non-diagnostic failure of one case in the FNB group due to a lack of soft tissue in the lytic lesion. This highlights the limitations of percutaneous biopsy in such cases. To avoid inadvertently sampling the vault cavity, we perform superficial biopsies, tangential to the bone, targeting any soft tissue protruding from the lesion. In cases lacking such tissue, percutaneous biopsy is not advised.
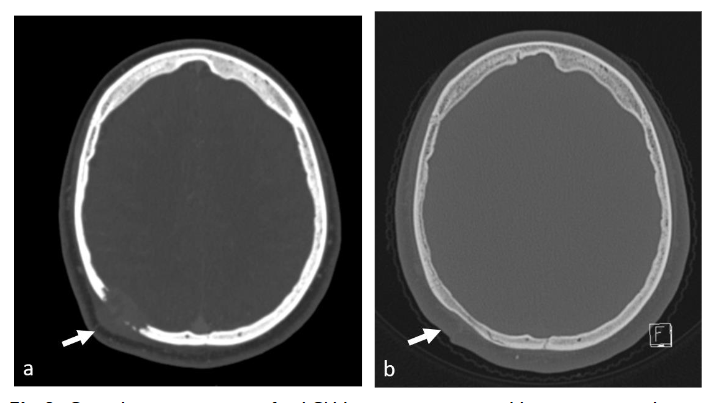
In two FNB cases, on-site cytology yielded a diagnosis, facilitating rapid treatment initiation. These results are consistent with previous findings demonstrating the diagnostic value of on-site cytology in pediatric oncology. No complications occurred in the FNB group, whereas the surgical group experienced post-operative complications occurred in more than half of the patients (14/26), some major complications that necessitated a second surgery and prolonged hospitalization, and in 8 cases (30%) long standing complications including chronic headache, chronic sinusitis and in 5 cases deformation of the skull.
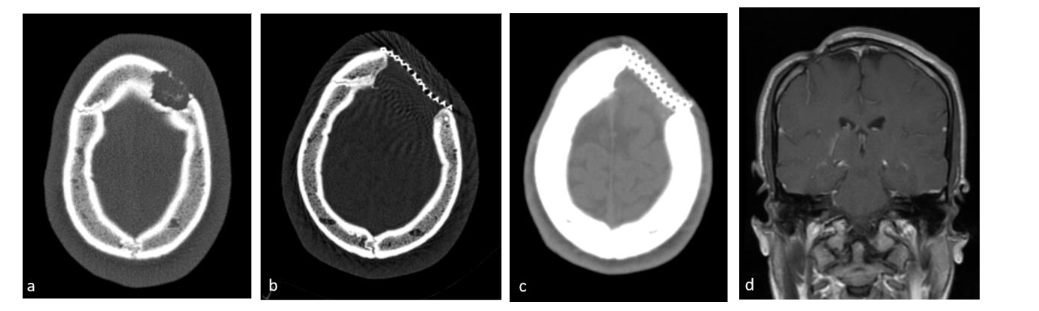
The surgical group also exhibited a higher rate of bone defect repair, with titanium mesh and cement replacement being used in some cases. The current consensus recommends biopsy over resection for lesions larger than 5cm to avoid unnecessary enlargement of the bony defect and complications such as delayed healing or permanent skeletal deformities. For lesions between 2–5cm, biopsy combined with partial curettage is an acceptable option. Anesthesia-related complications were not reported in either group.
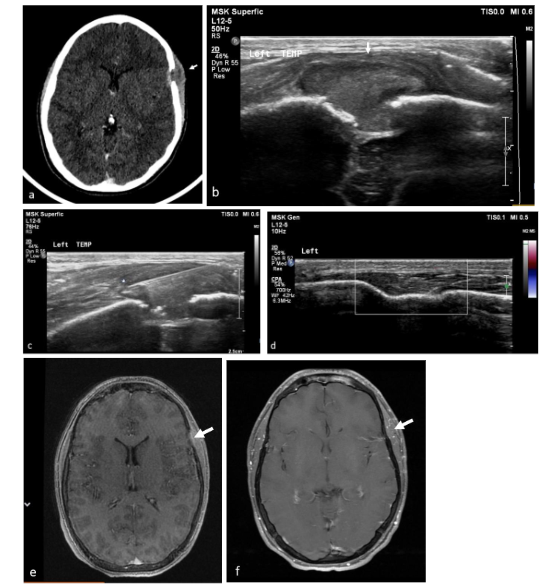
Conclusion
There are clear indications for systemic treatment in LCH, and in these cases, complete resection of skull lesions is unnecessary. The key value of percutaneous biopsy lies in its ability to obtain sufficient tissue for diagnostic purposes while avoiding the risks associated with surgery. As evidenced by numerous studies, percutaneous biopsy is a safe, effective, and low-complication technique for tissue diagnosis in pediatric oncology. This study demonstrates that, when feasible, percutaneous FNB is a reliable alternative to surgical resection for diagnosing skull and facial LCH lesions, with higher availability and no early or late complications. Regression post biopsy is an additional advantage precluding the need for treatment in some cases.
Although our study is limited by its retrospective nature and small sample size, the findings, in concordance with the broader literature, strongly support the utility of FNB in the management of skull lesions suspected of LCH and malignancy. Based on our experience and that of others, we recommend that for any new lesion in a pediatric patient that is suspected to be malignant in the skull or facial bones, the feasibility of percutaneous FNB should be assessed as the first approach, provided there is a safe window for tissue sampling under imaging guidance.
Declaration of Interest:
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this article.
References
- Sedain G, Khanal K, Pandey A, et al. Challenges in diagnosis and management of Langerhans Cell Histiocytosis in a 13-month-old child: a rare case report. Ann Med Surg. 2024;86(9):5611-5617. doi:10.1097/ms9.0000000000002430
- Orr T, Lesha E, Dugan JE, et al. Langerhans cell histiocytosis of the sella in a pediatric patient: case report with review of the literature. Child’s Nerv Syst. 2024;40(9):2947-2952. doi:10.1007/s00381-024-06456-y
- Hughes E, Rosenbaum D, Branson H, et al. Imaging approach to pediatric calvarial bulges. Pediatr Radiol. 2024;54(10):1603-1617. doi:10.1007/s00247-024-05967-9
- Choudhary G, Udayasankar U, Saade C, Winegar B, Maroun G, Chokr J. A systematic approach in the diagnosis of paediatric skull lesions: what radiologists need to know. Polish J Radiol. 2019;8(84):e92-e111. doi:10.5114/pjr.2019.83101.
- Haupt R, Minkov M, Astigarraga I et al. Langerhans Cell Histiocytosis (LCH): Guidelines for Diagnosis, Clinical Work-Up, and Treatment for Patients Till the Age of 18 Years. Pediatr Blood Cancer. 2013;60(2):175-184. doi:10.1002/pbc.24367
- Gerrie SK, Navarro OM, Lyons CJ, et al. Pediatric orbital lesions: bony and traumatic lesions. Pediatr Radiol. 2024;54(6):897-909. doi:10.1007/s00247-024-05882-z
- Yang J, Huang X, Bao Z, Xu J, Huang H, Huang H. Langerhans cell histiocytosis in children: the value of ultrasound in diagnosis and follow‑up. BMC Med Imaging. 2025;25(29):1-12. doi:10.1186/s12880-025-01563-x
- Wei M, Jiang W, Wang R, Xiao B, Zhan Q. Langerhans cell histiocytosis of the skull in 23 children. Eur J Med Res. 2024;29(1):1-6. doi:10.1186/s40001-024-01727-5
- Mavromati M, Caironi V, Saiji E, et al. Langerhans cell histiocytosis of the suprasellar region: diagnosis based on thyroid cytology. Eur Thyroid J. 2024;13(3):1-6. doi:10.1530/ETJ-24-0011
- Reisi N, Raeissi P, Harati Khalilabad T, Moafi A. Unusual sites of bone involvement in Langerhans cell histiocytosis: a systematic review of the literature. Orphanet J Rare Dis. 2021;16(1):1-13. doi:10.1186/s13023-020-01625-z
- Dharmaputra RK, Piesse CM, Chaubey S, Sinha AK, Chiam HC. A rare diagnosis of Langerhans cell histiocytosis made on thyroid histology with coexisting papillary thyroid cancer and AVP deficiency. Endocrinol Diabetes Metab Case Reports. 2024;2024(2). doi:10.1530/EDM-23-0050
- Alshadwi A, Nadershah M, AlBazie S. Langerhans cell histocytosis of the mandible in a pediatric patient. J Dent Child. 2013;80(3):145-149.
- Wadone MM, Masgal MM, Anita AM, Rajesh PS. A study of cytomorphological spectrum of head and neck lesions in pediatric age group. J Cytol. 2023;40(2):75-80. doi:10.4103/joc.joc_93_22
- Sokolovski B, Scolnik M, Malkin L, Postovsky S, Weyl Ben-Arush M, Ilivitzki A. Ultrasound-guided core biopsy with on-site cytology-immediate diagnosis in pediatric oncology. Diagn Cytopathol. 2021;49(7):817-821. doi:10.1002/dc.24746
- Goyal G, Tazi A, Go RS, et al. International expert consensus recommendations for the diagnosis and treatment of Langerhans cell histiocytosis in adults. Blood. 2022;139(17):2601-2621. doi:10.1182/blood.2021014343
- Allen CE, Ladisch S, McClain KL. How i treat Langerhans cell histiocytosis. Blood. 2015;126(1):26-35. doi:10.1182/blood-2014-12-569301
- Donadieu J, Henter J-I, Lehrnbecher T, et al. Standard Clinical Practice Recommendations for Pediatric-Onset Langerhans Cell Histiocytosis. Eur Soc Paediatr Oncol Eur Ref Netw Paediatr Oncol. 2020;(1.2).
- Yoon S, Park S. A study of 77 cases of surgically excised scalp and skull masses in pediatric patients. Child’s Nerv Syst. 2008;24(4):459-465. doi:10.1007/s00381-007-0523-2
- Ilivitzki A, Sokolovski B, Assalia A, et al. Ultrasound-Guided Core Biopsy for Tissue Diagnosis in Pediatric Oncology: 16-Year Experience With 597 Biopsies. Am J Roentgenol. February 2021:1-8. doi:10.2214/AJR.20.23196
- Sharan KC, Agarkar L, Jeyaraman R, Radhakrishnan S, Gochhait D. Langerhans cell histiocytosis of thyroid and bilateral parotid diagnosed on fine needle aspiration cytology. Cytopathol Off J Br Soc Clin Cytol. 2024;35(4):515-519. doi:10.1111/cyt.13376
- Betting T, Benson JC, Madhavan A et al. Safety and histopathologic yield of percutaneous CT-guided biopsies of the skull base, orbit, and calvarium. Neuroradiology. 2024;66(3):417-425. doi:10.1007/s00234-023-03266-y
- Tomasian A, Hillen TJ, Jennings JW. Percutaneous CT-guided skull biopsy: Feasibility, safety, and diagnostic yield. Am J Neuroradiol. 2019;40(2):309-312. doi:10.3174/ajnr.A5949
- Yanagawa T, Watanabe H, Shinozaki T, Ahmed A, Shirakura K, Takagishi K. The natural history of disappearing bone tumours and tumour-like conditions. Clin Radiol. 2001;56(11):877-886.
- Kamimura M, Kinoshita T, Itoh H, Yuzawa Y, Takahashi J, Ohtsuka K. Eosinophilic granuloma of the spine: early spontaneous disappearance of tumor detected on magnetic resonance imaging. Case report. J Neurosurg. 2000;93(2):312-316. doi:10.3171/spi.2000.93.2.0312
- Muscolo D, Slullitel G, Ranalletta M, Aponte-Tinao L, Ayerza M. Spontaneous remission of massive solitary eosinophilic granuloma of the femur. J Pediatr Orthop. 2023;23(6):763-765. doi:10.1097/00004694-200311000-00014
- Oliveira M, Steinbok P, Wu J, Heran N, Cochrane D. Spontaneous resolution of calvarial eosinophilic granuloma in children. Pediatr Neurosurg. 2003;38(5):247-252. doi:10.1159/000069828
- Zhang XH, Zhang J, Chen ZH, et al. Langerhans cell histiocytosis of skull: A retrospective study of 18 cases. Ann Palliat Med. 2017;6(2):159-164. doi:10.21037/apm.2016.11.04

