Periadjuvant ADT for High-Risk Prostate Cancer Outcomes
Peri-Adjuvant Androgen Deprivation Therapy for Patients with High-Risk Prostate Cancer Undergoing Radical Prostatectomy – The Concept Behind and Initial Outcomes
Deerush Kannan Sakthivel., MD., DrNB 1; Sindhu Sankaran., MRCS 2; Madhav Tiwari., Mch 3; Sandeep Bafna., DrNB 4; Devanathan Mch 5; Sanjai Addla., FRCS 6; Narasimhan Ragavan., FRCS 4
- Deerush Kannan Sakthivel, MD., DrNB(Urology), Fellow in Uro Oncology, Miami Cancer Institute, Baptist Health South Florida, Miami, USA
- Sindhu Sankaran, MRCS (Edinburgh), Registrar, Department of Urology, Apollo Hospitals, Chennai, India
- Madhav Tiwari, Mch (Urology), Associate Consultant, Department of Urology, Apollo Hospitals, Chennai, India.
- Sandeep Bafna, DrNB (Urology), Consultant, Department of Urology, Apollo Hospitals, Chennai, India.
- Devanathan -, Mch (Urology), Senior consultant Urologist Paduka hospital
- Sanjai Addla, FRCS (Urology), Consultant Urologist, Apollo Hospitals, Hyderabad
- Narasimhan Ragavan, FRCS (Urology), Consultant, Department of Urology, Apollo Hospitals, Chennai, India.
OPEN ACCESS
PUBLISHED: 20 April 2025
CITATION: SAKTHIVEL, Deerush Kannan et al. Peri-Adjuvant Androgen Deprivation Therapy for Patients with High-Risk Prostate Cancer Undergoing Radical Prostatectomy – The Concept Behind and Initial Outcomes. Medical Research Archives, [S.l.], v. 13, n. 4, apr. 2025. Available at: <https://esmed.org/MRA/mra/article/view/6497>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i4.6497
ISSN 2375-1924
Abstract
Background: Radical prostatectomy (RP) is increasingly employed as part of a multimodal strategy for treating locally advanced and/or high-risk prostate cancer (LA-HR CaP), though its oncological adequacy alone is limited due to the risk of micrometastatic disease. While androgen deprivation therapy (ADT) with radiation remains the traditional standard, the optimal integration of ADT with RP is yet to be defined. This study introduces the concept of periadjuvant ADT, which combines neoadjuvant ADT, RP, and adjuvant ADT for a total duration of 18 months.
Methods: A retrospective study was conducted on 127 patients with LA-HR CaP who received neoadjuvant ADT (Degarelix ± Abiraterone/Apalutamide) for 3–6 months, followed by RP and continuation of ADT postoperatively. Treatment regimens were individualized based on patient preference and affordability. Data were collected on clinical, operative, pathological, and follow-up parameters. Kaplan-Meier analysis was used for survival outcomes.
Results: The majority (67.7%) of patients were staged as T3, with nodal involvement in 26%. Neoadjuvant therapy was well tolerated, with no significant intraoperative complications (99.2% complication-free). Positive surgical margins and nodal positivity were seen in 24.4% and 26.8% of patients, respectively. Adjuvant radiation was required in 29.1% of cases. At a median follow-up of 12 months, overall survival was 97.6%, with a cumulative Kaplan-Meier survival rate of 94.6%.
Conclusion: Periadjuvant ADT combining neoadjuvant and adjuvant hormonal therapy with RP is a feasible and safe approach for LA-HR CaP in a resource-limited setting. Early results suggest acceptable surgical morbidity and promising short-term oncological outcomes. Longer follow-up is warranted to evaluate survival benefits and refine treatment duration guidelines.
Keywords
- Prostate Cancer
- Androgen Deprivation Therapy
- Radical Prostatectomy
- Neoadjuvant Therapy
- Adjuvant Therapy
Introduction
Radical prostatectomy (RP) has now become the standard of care for patients with localized prostate cancer (CaP).¹ The procedure has been widely adopted since the advent of robotic surgery.² A better understanding of surgical techniques has allowed this surgery to be performed with minimal complications and excellent functional recovery.
Localized CaP is defined as a tumor confined to the prostate on digital rectal examination. The European Association of Urology (EAU) further subdivides localized cancer into three different risk groups—low, intermediate, and high-risk—based on PSA levels and Gleason scores.3,4 Locally advanced and/or high-risk prostate cancer (LA-HR CaP) includes tumors that extend beyond the prostatic capsule (cT3, cT4, or N1 disease) or cases where PSA levels exceed 20 ng/mL, or the Gleason score is greater than 7.3,4
Although RP remains a standard treatment option for clinically localized high-risk prostate cancer, providing excellent local control, patients with high-risk disease remain at considerable risk for recurrence after surgery. Patients with LA-HR CaP have a higher risk of local and systemic recurrence due to possible microscopic disease in the local area or metastatic sites.5 Hence, multimodal therapy for these patients aims to address both the primary tumor and microscopic disease.6
Traditionally, LA-HR CaP patients have been treated with multimodal therapy, which includes androgen deprivation therapy (ADT) along with radiation therapy (RT) to the prostate.7 The duration of ADT in such patients is typically 2–3 years.7,9 This approach has resulted in excellent overall survival (OS) and disease-free survival (DFS).7,9
However, patients with locally advanced CaP often experience troublesome local symptoms such as bladder outflow obstruction and ureteric obstruction, which frequently require additional operative procedures for symptom relief.10,11 On the other hand, upfront RP for LA-HR CaP carries a risk of multifocal positive margins, necessitating additional therapy such as ADT or early salvage RT to the prostatic bed.¹²,¹³ Limited expertise in performing RP in LA-HR CaP patients in the past contributed to ADT and RT becoming the standard of care for this cohort.14,15
According to EAU guidelines, RP can be considered for patients with locally advanced and/or high-risk CaP only as part of a multimodal therapy approach.16 The components of multimodal therapy include the use of ADT and RT in appropriate circumstances.17 However, the timing and duration of these treatments remain undefined.18
In the past, most studies have been conducted in patients with low-risk CaP, where the probability of extraprostatic microscopic disease is minimal.19 Neoadjuvant ADT and RP have been tested recently in patients with high-risk CaP, demonstrating an improvement in negative surgical margins but not in OS.20-23 This may be because of the fact that patients received neoadjuvant ADT and no additional treatment in the postoperative setting was contemplated. In comparison to patients undergoing RT for similar disease specifications, the ADT has not been continued in the postoperative period in patients undergoing RP.
Given the recent trend toward surgical management of LA-HR CaP, long-term outcomes and guideline recommendations on ADT dosage and duration in this subset remain loosely defined. Although the impact of ADT has primarily been observed in margin status rather than overall oncological outcomes, ongoing studies highlight the need to address this gap before establishing definitive guideline recommendations regarding the dosage, duration, and timing of ADT in combination with RP for LA-HR CaP patients.
On the other hand, as mentioned above, combining systemic ADT with local therapy (RT) has improved overall survival in these patients. However, this approach fails to address issues related to local symptoms.24 Furthermore, in patients with long life expectancy, addressing both the local and micrometastatic components using RP and ADT, respectively, may be more beneficial, as the long-term side effects of radiation therapy become more pronounced after a decade.25
In recent years, significant advancements have been made in ADT with the introduction of androgen receptor pathway inhibitors (ARPIs) such as Abiraterone acetate, Enzalutamide, and Apalutamide.26 These drugs have been well-established in the metastatic setting.26 Ongoing trials are evaluating the role of these drugs (in addition to standard ADT) in the neoadjuvant setting.28 ADT for LA-HR CaP can be administered in the form of luteinizing hormone-releasing hormone (LHRH) agonists (e.g., Goserelin, Leuprolide) or LHRH antagonists.29,30
We propose the introduction of a new term, Periadjuvant ADT, for patients undergoing RP. This regimen would include neoadjuvant ADT for 3–6 months, followed by RP, with the continuation of ADT as adjuvant therapy for a total duration of 18 months. This study aims to assess the safety and short-term oncological efficacy of periadjuvant therapy, which includes neoadjuvant and adjuvant ADT (with or without ARPI), in patients undergoing radical prostatectomy for locally advanced high-risk prostate cancer.
Materials and Methods
A retrospective study was conducted following ethics committee approval (AMH-C-S-023/03-23). Data were collected from a prospectively maintained database that included details of all patients with locally advanced and/or high-risk CaP. As this was a preliminary study in a resource-limited setting, patients were offered Degarelix, Degarelix + Abiraterone, or Degarelix + Apalutamide, with treatment selection based on preference and affordability.
All included patients had their diagnosis confirmed via transrectal ultrasound (TRUS)-guided biopsy, and staging was performed using PSMA PET MRI. Disease status was assessed through clinical and radiological evaluation, with imaging and pathology reviewed by a single radiology and pathology team for consistency, as per routine practice.
Periadjuvant therapy, which combines neoadjuvant therapy (3–6 months), RP and adjuvant ADT for a total of 18 months, was initiated after obtaining informed consent. Patients were allocated to one of the treatment arms based on their choice. Collected data included demographics, pre-treatment cancer status, operative and post-operative outcomes, pathology, functional and oncological follow-up, and the need for further treatment.
STATISTICAL ANALYSIS
Data were tabulated in Microsoft Excel and analyzed using SPSS (IBM, version 28.0). Categorical variables were presented as frequency (percentage), continuous variables as Mean ± SD, and skewed data as Median (IQR). Kaplan-Meier survival analysis was performed to assess survival probabilities.
Results
A total of 127 patients were included in the study. The majority (48.8%) were in the 61–70 years age group, with 30.7% aged above 70 years. Comorbidities were present in 70.1% of patients, and all cases (100%) were histologically confirmed as adenocarcinoma (Table 1).
| Parameters (n=127) | n (%) |
|---|---|
| Age (In years) | |
| 51 – 60 | 26 (20.5) |
| 61 – 70 | 62 (48.8) |
| >70 | 39 (30.7) |
| Co-morbidities | |
| Yes | 89 (70.1) |
| No | 38 (29.9) |
| Biopsy results | |
| Adenocarcinoma | 127 (100.0) |
Preoperative evaluation showed that 31.5% had PSA levels ≥40 ng/mL, while 22.8% had PSA <10 ng/mL. PSMA with MRI staging classified 67.7% as T3, with nodal involvement in 15% and distant metastasis in 7.1%. Preoperative biopsy-based Gleason grading (GG) identified 29.1% as GG5, with similar distributions for GG2 (23.6%), GG3 (21.3%), and GG4 (21.3%) (Table 2).
| Parameters (Pre operative) (n=127) | n (%) |
|---|---|
| PSA | |
| <10 | 29 (22.8) |
| 10 – 19 | 31 (24.4) |
| 20 – 29 | 19 (15) |
| 30 – 39 | 8 (6.3) |
| ≥40 | 40 (31.5) |
| PSMA with MRI | |
| T2 | 35 (27.6) |
| T3 | 86 (67.7) |
| T4 | 6 (4.7) |
| N | |
| 0 | 108 (85.0) |
| 1 | 19 (15.0) |
| M (Oligometastatic disease) | |
| 0 | 118 (92.9) |
| 1 | 9 (7.1) |
| Gleason grading (Pre operative biopsy) | |
| GG1 | 6 (4.7) |
| GG2 | 30 (23.6) |
| GG3 | 27 (21.3) |
| GG4 | 27 (21.3) |
| GG5 | 37 (29.1) |
The mean operative time was 248.9 ± 57.9 minutes, with a median docking time of 180 minutes (IQR: 150–225). Blood loss was <250 mL in 93.7% of cases, and intraoperative complications were rare (0.8%) (Table 3).
| Parameters (n=127) | n (%) |
|---|---|
| Operative time (In Mins) | Mean ± SD 248.9 ± 57.9 |
| Range | 120 – 480 |
| Dock time (In Mins) | Mean ± SD 192.9 ± 52.7 |
| Median (IQR) | 180 (150 – 225) |
| Range | 100 – 375 |
| Blood loss | |
| <250 ml | 119 (93.7) |
| >250 ml | 8 (6.3) |
| Complications | |
| No | 126 (99.2) |
| Yes | 1 (0.8) |
Final histopathology revealed that 65.35% had T3b disease, with nodal metastasis in 26%. Postoperative GG showed 30.0% as GG5, and 31.5% exhibited treatment-related histologic effects. Surgical margins were positive in 24.4% of cases, while 26.8% had positive lymph nodes (Tables 4–6).
| Parameters (n=127) | n (%) |
|---|---|
| HPE Results | |
| T0 | 1(0.8) |
| T2 | 22(17.32) |
| T3a | 19(14.96) |
| T3b | 83(65.35) |
| T4 | 2(1.57) |
| N | |
| N0 | 94(74.0) |
| N1 | 33 (26.0) |
| Gleason grading (post operative) | * |
| GG1 | 2 (1.6) |
| GG2 | 14 (11.0) |
| GG3 | 14 (11.0) |
| GG4 | 19 (15.0) |
| GG5 | 38 (30.0) |
| Not available due to ADT effect | 40 (31.5) |
| Margin (n=127) | n (%) |
|---|---|
| Margin positivity (multifocal) | |
| Yes | 31 (24.4) |
| No | 96 (75.6) |
| Tumor (n=127) | n (%) |
|---|---|
| Nodes positivity | |
| Yes | 34 (26.8) |
| No | 93 (73.2) |
Regarding preoperative treatment, 63.8% received Degarelix alone, and 30.7% received Degarelix with Abiraterone. The majority (85.7%) were on an 18-month Degarelix regimen. Radiation therapy was administered to 29.1% of patients, with 22.8% receiving prostate bed radiation (Table 7). Although all the patients were advised a 18-month course of ADT, based on the tumor board recommendations, in patients (n=18) who had excellent response, ADT was discontinued in 12 months.
| Parameters (n=127) | n (%) |
|---|---|
| Treatment given (Pre operative) | |
| Degarelix | 81 (63.8) |
| Degarelix + Abiraterone | 39 (30.7) |
| Degarelix + Apalutamide | 7 (5.5) |
| Duration of Degarelix | |
| 12 Months | 18 (14.2) |
| 18 Months | 109 (85.7) |
| Radiation therapy | |
| Yes | 37 (29.1) |
| No | 90 (70.9) |
| Radiation therapy | |
| RT metastatic site | 8 (6.3) |
| RT Prostate bed | 29 (22.8) |
| Not given | 90 (70.9) |
Last follow-up (In months) Median (IQR) 12 (6 – 48) Range 4 – 60. At a median follow-up of 12 months (IQR: 6–48 months), overall survival was 97.6%, with three deaths recorded—two due to neuroendocrine small cell carcinoma and one due to stroke (Table 8).
| Parameters (n=127) | n (%) |
|---|---|
| Survival status | |
| Alive | 124 (97.6) |
| Expired | 3 (2.4) |
| Reason for death | |
| Neuroendocrine small cell carcinoma | 2 (1.6) |
| Stroke | 1 (0.8) |
| Last follow-up (In months) | |
| Median (IQR) | 12 (6 – 48) |
| Range | 4 – 60 |
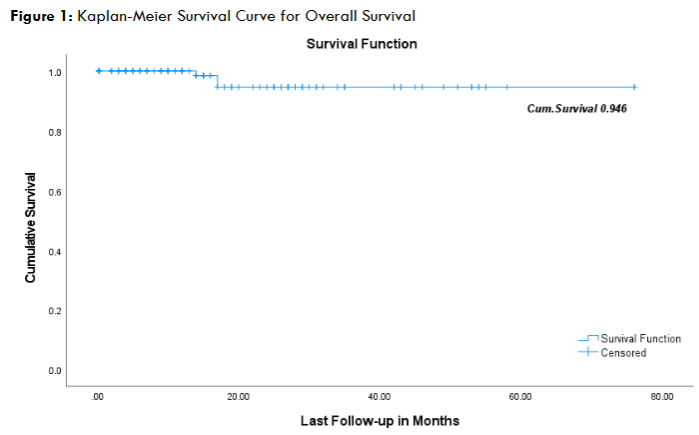
The Kaplan-Meier survival curve (Figure 1) demonstrates a cumulative survival rate of 94.6% at the last follow-up, indicating favourable overall survival outcomes with a gradual decline over time.
Discussion
The treatment landscape for LA-HR CaP is evolving, with an increasing emphasis on multimodal strategies. While ADT with RT has been the traditional standard of care,7,9 RP is now being reconsidered for its role in local tumor control and symptom relief.30 However, RP alone is often inadequate for oncological control due to the high risk of microscopic extraprostatic disease, necessitating systemic therapy to optimize outcomes.5,6
Our study introduces the novel concept of periadjuvant ADT, which integrates neoadjuvant therapy (3–6 months), RP, and adjuvant ADT (total of 18 months). This structured approach aims to maximize oncological control by addressing both local and micrometastatic disease.16,17
Neoadjuvant ADT can downstage tumors, improve surgical margin status, and reduce tumor burden, facilitating a better surgical outcome.19-23 Post-RP adjuvant ADT aims to suppress residual microscopic disease, potentially delaying recurrence.18 The combination of RP and ADT thus offers a more comprehensive treatment strategy compared to either modality alone.34,35
Our findings suggest that periadjuvant ADT is safe and feasible, with no major adverse effects related to Degarelix-based neoadjuvant therapy.29,30 Importantly, there were no intraoperative complications, reaffirming that neoadjuvant ADT does not compromise the surgical field or increase technical difficulty.12,13 Postoperative outcomes demonstrated acceptable morbidity, supporting the viability of RP in this subset of patients.³¹ There was one patient who developed a postoperative pelvic hematoma and was managed conservatively. The pathological outcomes, including margin and nodal status, further validate the oncological safety of this approach.32,33 With the advent of new robotic platforms and the increasing use of robotic surgery for complex oncological procedures, surgical expertise has improved. Consequently, the trend of LA-HR CaP through surgery is expected to rise in the future. Therefore, the oncological management protocol with ADT needs to be better defined and standardized.34-36
Compared to conventional strategies, periadjuvant ADT offers several advantages. RP alone, while effective in controlling local symptoms, does not adequately address micrometastatic disease, which remains the principal driver of recurrence.5 Conversely, ADT with RT, though oncologically effective, does not offer the same degree of symptom relief in patients with significant obstructive symptoms.10,11 Integrating ADT before and after RP within a periadjuvant framework bridges this gap, ensuring both local and systemic control.16 Furthermore, with ARPI agents such as Abiraterone and Apalutamide demonstrating survival benefits in metastatic settings,26,27 their incorporation into earlier disease stages within a periadjuvant framework may further enhance oncological control.28 In this study, the Kaplan-Meier survival analysis demonstrated a cumulative survival rate of 94.6% at the last follow-up, reflecting favorable overall survival outcomes with a gradual decline over time (Figure 1). These findings support the potential role of intensified periadjuvant therapy in optimizing disease control and long-term survival in high-risk prostate cancer patients.
Despite its promise, our study has certain limitations. Being a retrospective study and a lack of a control arm limits direct comparison to standard treatments such as ADT + RT or RP alone.14,15 The short follow-up duration prevents definitive conclusions on long-term survival outcomes, and the heterogeneous treatment selection based on patient preference and affordability introduces potential selection bias. Additionally, as guidelines on ADT in the surgical setting remain undefined, the optimal duration and intensity of ADT in combination with RP require further investigation.18
The findings from this study support the safety and feasibility of periadjuvant ADT, but its long-term oncological impact remains to be fully understood. Larger prospective, randomized trials are essential to validate its effectiveness and refine its role in treatment guidelines.17 Future studies should focus on defining the ideal duration and intensity of ADT, evaluating the role of ARPIs in this setting, and identifying biomarkers for patient selection.
Periadjuvant ADT represents a paradigm shift in the management of LA-HR CaP, offering a structured, multimodal strategy that maximizes oncological control while preserving the benefits of RP.16,17,37,38 Our preliminary findings indicate that this approach is safe and feasible, with the potential to redefine standard treatment protocols for high-risk prostate cancer. However, further long-term studies are warranted to establish its definitive role in clinical practice.17,18
Conclusion
Periadjuvant ADT with RP for LA-HR CaP is a viable and safe option, demonstrating promising short-term oncological and functional outcomes. The absence of significant perioperative complications and acceptable morbidity suggests its feasibility in clinical practice. However, long-term follow-up and randomized controlled trials are essential to validate its oncological benefits and define its optimal role in treatment protocols for LA-HR CaP.
Conflicts of Interest Statement:
None.
Funding Statement:
No funding was received.
Acknowledgments:
We thank Ms. Pavithra for data collection and Mr. Logesh for statistical analysis.
References
- Würnschimmel C, Wenzel M, Wang N, et al. Radical prostatectomy for localized prostate cancer: 20-year oncological outcomes from a German high-volume center. Urol Oncol. 2021;39(12):830.e17-830.e26. doi:10.1016/j.urolonc.2021.04.031
- Esperto F, Cacciatore L, Tedesco F, et al. Impact of Robotic Technologies on Prostate Cancer Patients’ Choice for Radical Treatment. J Pers Med. 2023;13(5):794. Published 2023 May 5. doi:10.3390/jpm13050794
- Mottet N, van den Bergh RCN, Briers E, et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2021;79(2):243-262. doi:10.1016/j.eururo.2020.09.042
- Amin MB, Greene FL, Edge SB, et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67(2):93-99. doi:10.3322/caac.21388
- Meng MV, Elkin EP, Latini DM, Duchane J, Carroll PR. Treatment of Patients With High Risk Localized Prostate Cancer: Results From Cancer of the Prostate Strategic Urological Research Endeavor (CaPSURE). J Urol (2005) 173:1557–61. doi:10.1097/01.ju.0000154610.81916.81
- Eastham J.A., Auffenberg G.B., Barocas D.A., Chou R., Crispino T., Davis J.W., Eggener S., Horwitz E.M., Kane C.J., Kirkby E., et al. Clinically Localized Prostate Cancer: AUA/ASTRO Guideline, Part I: Introduction, Risk Assessment, Staging, and Risk-Based Management. J. Urol. 2022;208:10–18. doi: 10.1097/JU.0000000000002757.
- Bolla M, Collette L, Blank L, et al. Long-term results with immediate androgen suppression and external irradiation in patients with locally advanced prostate cancer (an EORTC study): a phase III randomised trial. Lancet. 2002;360(9327):103-106. doi:10.1016/s0140-6736(02)09408-4
- Bolla M, de Reijke T, Van Tienhoven G, et al. Duration of androgen suppression in the treatment of prostate cancer. New Eng Jour of Med. 2009;360(24):2516–2027. doi: 10.1056/NEJMoa0810095.
- Zapatero A, Gurerrero A, Maldonado X, et al. High-dose radiotherapy with short-term or long-term androgen deprivation in localized prostate cancer (DART 01/05 GICOR): a randomised, controlled phase 3 trial. The Lancet Oncology. 16(3):320–327. doi: 10.1016/S1470-2045(15)70045-8. 3/15.
- Cahill TP, Withey SJ, Hazell S, Cahill D, Kinsella N. Does bladder outflow obstruction obfuscate the traditional clinical factors that are used to assess the risk of prostate cancer at rapid-access diagnostic clinics? BJUI Compass. 2024;6(1):e478. Published 2024 Dec 20. doi:10.1002/bco2.478
- Friedlander JI, Duty BD, Okeke Z, Smith AD. Obstructive uropathy from locally advanced and metastatic prostate cancer: an old problem with new therapies. J Endourol. 2012;26(2):102-109. doi:10.1089/end.2011.0227
- Srivatsa N, Nagaraja H, Shweta S, Raghunath SK. Radical Prostatectomy for Locally Advanced Prostate Cancers-Review of Literature. Indian J Surg Oncol. 2017;8(2):175-180. doi:10.1007/s13193-016-0599-9
- Calleris G, Filleron T, Kesch C, et al. Surgery with or Without Darolutamide in High-risk and/or Locally Advanced Prostate Cancer: The SUGAR (CCAFU-PR2) Phase 2 Trial Rationale and Protocol. Eur Urol Oncol. 2024;7(3):494-500. doi:10.1016/j.euo.2023.09.020
- Bastian, P.J., et al. Clinical and pathologic outcome after radical prostatectomy for prostate cancer patients with a preoperative Gleason sum of 8 to 10. Cancer, 2006. 107: 1265. https://pubmed.ncbi.nlm.nih.gov/16900523
- Yossepowitch, O., et al. Radical prostatectomy for clinically localized, high risk prostate cancer: critical analysis of risk assessment methods. J Urol, 2007. 178: 493. https://pubmed.ncbi.nlm.nih.gov/17561152
- EAU Guidelines. Edn. presented at the EAU Annual Congress Paris April 2024. ISBN 978-94-92671-23-3.
- Lawton CAF, Lin X, Hanks GE, et al. Duration of Androgen Deprivation in Locally Advanced Prostate Cancer: Long-Term Update of NRG Oncology RTOG 9202. Int J Radiat Oncol Biol Phys. 2017;98(2):296-303. doi:10.1016/j.ijrobp.2017.02.004
- Parker CC, Kynaston H, Cook AD, et al. Duration of androgen deprivation therapy with postoperative radiotherapy for prostate cancer: a comparison of long-course versus short-course androgen deprivation therapy in the RADICALS-HD randomised trial. Lancet. 2024;403(10442):2416-2425. doi:10.1016/S0140-6736(24)00549-X
- Meng MV, Grossfeld GD, Sadetsky N, Mehta SS, Lubeck DP, Carroll PR. Contemporary patterns of androgen deprivation therapy use for newly diagnosed prostate cancer. Urology. 2002;60(3 Suppl 1):7-12. doi:10.1016/s0090-4295(02)01560-1
- Gómez Rivas J, Ortega Polledo LE, De La Parra Sánchez I, et al. Current Status of Neoadjuvant Treatment Before Surgery in High-Risk Localized Prostate Cancer. Cancers (Basel). 2024;17(1):99. Published 2024 Dec 31. doi:10.3390/cancers17010099
- Ashrafi A.N., Yip W., Aron M. Neoadjuvant Therapy in High-Risk Prostate Cancer. Indian J. Urol. 2020;36:251–261. doi: 10.4103/iju.IJU_115_20.
- Kumar S., Shelley M., Harrison C., Coles B., Wilt T.J., Mason M.D. Neo-adjuvant and adjuvant hormone therapy for localised and locally advanced prostate cancer. Cochrane Database Syst. Rev. 2006;2006:CD006019. doi:10.1002/14651858.CD006019.pub2.
- Sankaran S, Govindaswamy TG, Dholakia K, Selvaraj N, Kanan D, Tiwari M, Ragavan N. Efficacy of Degarelix in the Neoadjuvant Setting in Managing Locally Advanced Prostate Cancer. Cureus. 2023 Jun 21;15(6):e40752. doi: 10.7759/cureus.40752. PMID: 37363117; PMCID: PMC10285566.
- Bolla M, Henry A, Mason M, Wiegel T. The role of radiotherapy in localised and locally advanced prostate cancer. Asian J Urol. 2019;6(2):153-161. doi:10.1016/j.ajur.2019.02.001
- Pinkawa M, Holy R, Piroth MD, et al. Consequential late effects after radiotherapy for prostate cancer – a prospective longitudinal quality of life study. Radiat Oncol. 2010;5:27. Published 2010 Apr 8. doi:10.1186/1748-717X-5-27
- Bastos DA, Soares A, Schutz FAB, et al. Androgen Receptor Pathway Inhibitor Therapy for Advanced Prostate Cancer: Secondary Analysis of a Randomized Clinical Trial. JAMA Netw Open. 2025;8(1):e2454253. Published 2025 Jan 2. doi:10.1001/jamanetworkopen.2024.54253
- Rathkopf DE, Antonarakis ES, Shore ND, et al. Safety and Antitumor Activity of Apalutamide (ARN-509) in Metastatic Castration-Resistant Prostate Cancer with and without Prior Abiraterone Acetate and Prednisone. Clin Cancer Res. 2017;23(14):3544-3551. doi:10.1158/1078-0432.CCR-16-2509
- PROTEUS: A randomized, double-blind, placebo (PBO)-controlled, phase 3 trial of apalutamide (APA) plus androgen deprivation therapy (ADT) versus PBO plus ADT prior to radical prostatectomy (RP) in patients (pts) with localized or locally advanced high-risk prostate cancer (PC). Journal of Clinical Oncology Volume 40, Number 6_suppl. https://doi.org/10.1200/JCO.2022.40.6_suppl.TPS285
- Clinton TN, Woldu SL, Raj GV. Degarelix versus luteinizing hormone-releasing hormone agonists for the treatment of prostate cancer. Expert Opin Pharmacother. 2017;18(8):825-832. doi:10.1080/14656566.2017.1328056
- Sethi R, Sanfilippo N. Six-month depot formulation of leuprorelin acetate in the treatment of prostate cancer. Clin Interv Aging. 2009;4:259-267. doi:10.2147/cia.s4885
- Veeratterapillay R, Goonewardene SS, Barclay J, Persad R, Bach C. Radical prostatectomy for locally advanced and metastatic prostate cancer. Ann R Coll Surg Engl. 2017;99(4):259-264. doi:10.1308/rcsann.2017.0031
- Gandaglia G, Abdollah F, Hu J, et al. Is robot-assisted radical prostatectomy safe in men with high-risk prostate cancer? Assessment of perioperative outcomes, positive surgical margins, and use of additional cancer treatments. J Endourol. 2014;28(7):784-791. doi:10.1089/end.2013.0774
- Sooriakumaran P, Karnes J, Stief C, et al. A Multi-institutional Analysis of Perioperative Outcomes in 106 Men Who Underwent Radical Prostatectomy for Distant Metastatic Prostate Cancer at Presentation. Eur Urol. 2016;69(5):788-794. doi:10.1016/j.eururo.2015.05.023
- Raghavan D, Kannan D, Sekaran PG, et al. A Novel Technique of Renorrhaphy in Difficult Partial Nephrectomies by Single-Layered Parenchymal Imbrication. Cureus. 2023;15(7):e42702. Published 2023 Jul 30. doi:10.7759/cureus.42702
- Kannan D, Sekaran PG, Panneerselvam A, Soni J, Jain N. Robotic transabdominal graft nephrectomy for renal mass in the transplant kidney. Videourology. 2023;37(1). https://doi.org/10.1089/vid.2023.0028
- Tiwari M, Kannan D, Addla S, Ragavan M, Ragavan N. Robot-assisted repair of neobladder-vaginal fistula. Videourology. 2024. https://doi.org/10.1089/vid.2024.0055
- Anast, J.W., Andriole, G.L. & Grubb, R.L. Managing the local complications of locally advanced prostate cancer. Curr prostate rep 6, 5–10 (2008). https://doi.org/10.1007/s11918-008-0002-6
- Van Poppel H, Vekemans K, Da Pozzo L, et al. Radical prostatectomy for locally advanced prostate cancer: results of a feasibility study (EORTC 30001). Eur J Cancer. 2006;42(8):1062-1067. doi:10.1016/j.ejca.2005.11.030

