Personalized Health: Epigenetics and Lifestylopathy Approach
Personalized Health Through Epigenetics: The Lifestylopathy Approach
Jawad Alzeer, PhD1
- College of Medicine and Health Sciences, Palestine Polytechnic University, Hebron, Palestine. Swiss Scientific Society for Developing Countries, Zürich, Switzerland.
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: Alzeer, J., 2025. Personalized Health Through Epigenetics: The Lifestylopathy Approach. Medical Research Archives, [online] 13(4). https://doi.org/10.18103/mra.v13i4.6435
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6435
ISSN 2375-1924
Abstract
The interaction between epigenetics and lifestyle has revolutionized the understanding of personalized health, shifting the focus from genetic determinism to modifiable gene expression. This manuscript explores Lifestylopathy, a holistic framework that integrates biological, psychological, and environmental factors to optimize gene regulation, metabolic balance, and long-term health outcomes. By leveraging epigenetic mechanisms, such as DNA methylation, histone modifications, and non-coding RNA interactions, this approach highlights how structured lifestyle interventions can actively shape gene expression to enhance immune resilience, cognitive stability, and disease prevention. Lifestylopathy classifies potential energy into four key domains; chemical, elastic, mental, and voluntary energy, which collectively influence cellular function, stress adaptation, and healing processes. Furthermore, this model proposes a balance between potential energy, represented by structure, and entropy, represented by adaptive flexibility, both of which are essential for optimal biological function. While the therapeutic potential of lifestyle-driven epigenetic modulation is promising, further research is needed to develop measurable biomarkers for non-physical energy domains, assess the long-term impact of voluntary behaviors on genetic expression, and address socioeconomic disparities affecting health optimization. By integrating Lifestylopathy principles into preventive healthcare and public policy, this framework has the potential to redefine personalized medicine, offering a sustainable and inclusive model for long-term well-being.
Keywords:
Lifestylopathy, Epigenetics, Personalized Medicine, Gene Expression, Potential Energy, Entropy, Health Optimization
Introduction
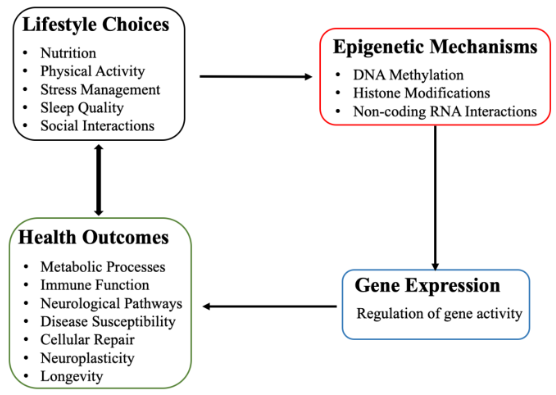
The relationship between lifestyle choices and genetic expression has emerged as a crucial area of interest in modern biomedical research. Traditionally, genetic inheritance was viewed as an unchangeable determinant of health, with predispositions to diseases largely dictated by DNA sequences. However, advancements in epigenetics have fundamentally transformed this perspective by demonstrating that gene expression is highly dynamic and can be modulated by environmental and behavioral factors. This paradigm shift underscores the idea that health is not solely predetermined by genetics but is actively shaped by daily choices, including diet, physical activity, stress levels, sleep quality, and even spiritual and emotional well-being. Research indicates that lifestyle behaviors can significantly influence health outcomes, as seen in studies linking lifestyle choices to genetic predispositions for conditions such as obesity and cardiovascular disease.
Epigenetic mechanisms, such as DNA methylation, histone modifications, and non-coding RNA interactions, regulate the activation or suppression of specific genes in response to environmental inputs. These mechanisms explain how external factors influence metabolic processes, immune function, neurological pathways, and disease susceptibility. For instance, poor dietary habits, chronic stress, and exposure to environmental toxins have been shown to activate genes associated with inflammation, insulin resistance, and mental health disorders, whereas nutrient-rich diets, physical exercise, and mindfulness practices promote the expression of genes linked to cellular repair, neuroplasticity, and longevity. This interplay between lifestyle and gene regulation offers an evidence-based framework for disease prevention and health optimization, highlighting the importance of personalized approaches to health that consider both genetic and environmental factors.
Lifestylopathy expands on this understanding by proposing a holistic model of health that integrates biological, psychological, emotional, and spiritual dimensions. In contrast to conventional approaches that focus solely on treating symptoms, Lifestylopathy emphasizes the importance of lifestyle and behavior as well as the importance of balancing potential energy and entropy, recognizing that health is a dynamic balance rather than a fixed state. By fostering nutritional optimization, structured behavioral patterns, and stress resilience living, Lifestylopathy offers a framework that aligns closely with epigenetic principles, ensuring that lifestyle choices actively enhance physiological stability rather than contribute to disorder. This holistic approach is supported by evidence that lifestyle modifications can significantly impact gene expression and overall health outcomes, particularly in populations at risk for chronic diseases.
This manuscript explores the intersection of Lifestylopathy and epigenetics as a foundational approach to personalized health. By examining how specific lifestyle interventions such as targeted nutrition, stress management techniques, structured and physical activity modulate gene expression, this work aims to provide an integrative model for long-term well-being. In addition, the effects of entropy reduction, mental balance and metabolic homeostasis on gene regulation will be examined, suggesting that structured lifestyle changes may serve as a preventative and therapeutic tool to optimize health, reduce disease risk and improve biological resilience across generations.
Epigenetics: How Environment Shapes Our Genes
DNA serves as a genetic blueprint, encoding instructions for every biological function in the body. However, genes do not operate in isolation; they are regulated by epigenetic mechanisms that determine whether a gene is expressed (active) or silenced (inactive) without altering the actual DNA sequence. Epigenetics involves reversible chemical modifications, such as DNA methylation and histone acetylation, which influence how DNA is packaged and accessed, thereby controlling gene activity. These modifications act as biological switches, shaping cell differentiation, function, and disease susceptibility in response to environmental signals, psychological states, and lifestyle choices.
Unlike genetic mutations, which are permanent, epigenetic changes are dynamic and adaptable, meaning that factors like nutrition, physical activity, stress levels, toxin exposure, and social interactions continuously shape our gene expression. This interplay between biology and behavior highlights that while our genetic code remains constant, our epigenome is dynamic, making it possible to influence long-term health and disease outcomes through conscious lifestyle decisions.
The environment provides the raw material that interacts with behavior and psychology, guiding epigenetic modifications that determine health outcomes. Nutrition, for example, is a direct epigenetic modulator supplies essential methyl groups that regulate genes involved in inflammation, metabolism, and cognitive function. In contrast, poor dietary habits, excessive sugar intake, and exposure to endocrine-disrupting chemicals can lead to abnormal DNA methylation, disrupting metabolic processes and increasing disease susceptibility.
Similarly, chronic stress alters the methylation of the FKBP5 gene, which regulates cortisol sensitivity, leading to prolonged stress responses, anxiety, and immune dysfunction. Psychological resilience, cultivated through practices like mindfulness, gratitude, and social support, has been shown to counteract stress-induced epigenetic damage, reinforcing stability in both mental and physical health.
Lifestyle choices further modulate the epigenetic environment. Regular physical activity enhances histone acetylation, thereby protecting against cardiovascular disease, neurodegenerative disorders, and mood disturbances. Sleep quality, exposure to natural light, and daily rhythm regulation also play critical roles, ensuring that genes responsible for cellular repair and immune function remain active. Social environments shape epigenetic outcomes as well, with studies demonstrating that positive social connections enhance oxytocin-driven gene expression, promoting emotional resilience and stress adaptability. In contrast, social isolation has been linked to inflammatory gene activation, increasing the risk of chronic diseases.
Epigenetic modifications are reversible and can be influenced by intentional lifestyle changes. Research indicates that positive habits can reshape epigenetic patterns, enhancing gene expression to promote longevity and prevent disease. Understanding that daily choices actively impact genetic function empowers individuals to take control of their health, not only for themselves but for future generations. However, while epigenetics plays a crucial role in well-being, it is not the sole determinant of health. Genetic predispositions, such as metabolic disorders and neurodegenerative diseases, create biological limitations that lifestyle changes alone cannot fully overcome. Additionally, socioeconomic disparities present significant barriers to optimal health. Limited access to nutritious food, safe living conditions, and quality healthcare restricts the ability of many individuals to implement positive health choices.
Lifestylopathy and Epigenetics
Lifestylopathy is a comprehensive healthcare model designed to create optimal conditions for disease prevention and healing by harmonizing environmental and behavioral factors with the body’s natural biological processes. It is founded on four core principles: homeostasis, which ensures internal stability and the self-regulation of biological functions in response to external influences; duality, which acknowledges the dynamic balance between health and disease; energy balance, which emphasizes maintaining potential energy to sustain cellular function while minimizing entropy, the natural tendency toward disorder; and lifestyle-therapeutic drug compatibility, which underscores that medical treatments are most effective when aligned with an individual’s biological state and lifestyle, ensuring that interventions work synergistically for optimal healing.
By integrating these principles, Lifestylopathy offers a personalized approach to medicine that goes beyond treating symptoms, addressing the epigenetic and metabolic foundations of long-term health. Lifestylopathy aligns closely with the principles of epigenetics, which explores how environmental and lifestyle behaviors influence gene expression without altering the underlying DNA sequence. By actively shaping both internal and external conditions, Lifestylopathy aims to foster genetic expressions that promote healing, resilience, and longevity. Through this integrative approach, it seeks to create a biological environment that supports long-term health and well-being, reflecting the holistic nature of integrative medicine that emphasizes the interconnectedness of various health determinants.
Beyond its biological and environmental dimensions, Lifestylopathy also recognizes the importance of human sciences in understanding health holistically. By integrating principles from chemistry and physics, it seeks to establish a deeper connection between human health and the life sciences. However, a truly comprehensive approach must also incorporate insights from human psychology, as mental and emotional well-being are fundamental to overall health, disease prevention, and the effectiveness of therapeutic interventions. This holistic perspective is essential for addressing complex health issues, as it acknowledges the multifaceted nature of health and the need for a collaborative approach that includes both conventional and complementary therapies.
Lifestylopathy and Energy
The Second Law of Thermodynamics, which dictates the natural increase of entropy, a measure of disorder, within closed systems, finds a considerable parallel in biological processes. In living organisms, this translates to a gradual decline of cellular efficiency, metabolic stability, and overall physiological resilience. Without continuous energy input, entropy accumulates, leading to cellular dysfunction, and disease progression. This relationship between entropy and biological systems underscores the necessity for organisms to maintain energy input to counteract the inevitable drift towards disorder, a concept that is particularly relevant in the context of chronic diseases and aging.
Lifestylopathy, guided by the principle of duality, recognizes health as a dynamic balance between potential energy and entropy. Potential energy, representing the body’s stored biological, chemical, and neurological reserves, provides the essential strength and rigidity necessary to maintain cellular repair, immune defense, and neurocognitive function, thereby resisting physiological collapse, the natural tendency towards disorder. Conversely, while entropy represents disorder, it also imparts a degree of flexibility and adaptability, enabling the body to respond and adjust to changing conditions for survival and efficiency. Furthermore, it contributes to the thermal homeostasis by generating heat, maintaining a stable body temperature. The challenge lies in the inherent asymmetry of this balance: potential energy requires conscious, deliberate enrichment, while entropy accumulates spontaneously. Therefore, Lifestylopathy strategically prioritizes the active maximization of potential energy to effectively counteract the inevitable, entropic drift towards disorder, ensuring sustained well-being.
Lifestylopathy integrates the concept of potential energy as a fundamental driver of health, classifying it into four key domains that influence epigenetic regulation, metabolic stability, and overall well-being. These energy sources shape gene expression, cellular resilience, and the body’s ability to mitigate the risk of chronic diseases. By strategically enhancing these domains, individuals can adjust their lifestyle to optimize biological function, reinforcing both therapeutic efficacy and long-term health outcomes.
| Energy Source | Definition | Role in Epigenetics | Ways to Enhance | Key Gene(s) |
|---|---|---|---|---|
| Chemical Energy | Energy derived from biochemical reactions and metabolic processes | Regulates gene expression through nutrient availability, methylation, and ATP synthesis | Optimize diet with essential nutrients (e.g., folate, B vitamins, polyphenols) | MTHFR, DNMT3A, TERT (DNA methylation, telomere maintenance, longevity) |
| Elastic Energy | Energy stored and released through movement, exercise, and mechanical function | Enhances DNA repair mechanisms, mitochondrial function, and histone modifications | Engage in regular physical activity (e.g., aerobic, resistance training, yoga) | SIRT1, PGC-1α, TERT (Mitochondrial function, energy metabolism, telomere lengthening) |
| Mental Energy | Cognitive and emotional energy used in decision-making, learning, and mindfulness | Influences neuroplasticity, stress resilience, and neurotransmitter-related gene expression by promoting cognitive flexibility and emotional stability | Practice mindfulness, meditation, and learning-based cognitive stimulation; enrich with supportive information (reading, intellectual discussions, exposure to diverse perspectives); engage in lifelong learning and problem-solving activities to strengthen brain-derived neurotrophic factor (BDNF) expression | BDNF, COMT, FKBP5 (Neuroplasticity, stress response, cognitive resilience) |
| Voluntary Energy | Energy driven by conscious choices, ethical actions, and purpose-driven behavior | Modulates epigenetic markers through long-term behavior, ethical alignment, and goal-setting, reinforcing neurobiological resilience and emotional well-being | Cultivate purposeful living, ethical behaviors, and positive social engagement; practice praying, fasting, and self-discipline; engage in helping others, volunteering, and community involvement to strengthen oxytocin-driven gene expression linked to trust and social bonding | OXTR, SLC6A4, FKBP5 (Social bonding, stress resilience, emotional regulation) |
1. Chemical Potential Energy (Nutritional Epigenetics): Nutrition plays a pivotal role in epigenetic regulation, influencing gene expression through mechanisms like DNA methylation, histone modifications, and chromatin remodeling. Essential nutrients act as cofactors for these processes, impacting key cellular functions such as inflammation, metabolism, and repair. Methyl donors, including folate, choline, and vitamin B12, are crucial for maintaining DNA methylation patterns, preventing global hypomethylation associated with chronic inflammation, metabolic dysfunction, and cancer. Antioxidants, such as polyphenols, protect against oxidative stress-induced epigenetic damage, supporting immune resilience, neuroprotection, and cellular longevity. Thus, a nutrient-rich diet acts as a powerful epigenetic modulator, fostering physiological balance and enhancing the body’s capacity to resist disease.
2. Elastic Potential Energy (Physical Activity & Movement): Physical activity serves as a potent epigenetic regulator, influencing gene expression through histone acetylation and chromatin restructuring, leading to enhanced mitochondrial function, energy metabolism, and neuroplasticity. Both aerobic and resistance training activate protective genes, mitigating inflammation, lowering cardiovascular risk, and improving insulin sensitivity. Exercise-induced epigenetic modifications also downregulate pro-inflammatory markers, stabilizing neuroprotective gene expression and promoting mental clarity, emotional resilience, and cognitive longevity. Thus, integrating regular movement into daily life allows individuals to proactively shape their epigenetic landscape, bolstering defenses against chronic disease and neurological decline.
3. Mental Potential Energy (Neuroepigenetics & Stress Regulation): Chronic psychological stress disrupts DNA methylation patterns in stress-response genes, increasing susceptibility to systemic inflammation, immune dysfunction, and mental health disorders. However, mindfulness, meditation, and cognitive restructuring can counteract these maladaptive epigenetic shifts by stabilizing genes for emotional regulation and modulating neuroepigenetic pathways, ultimately preserving cognitive function and protecting against age-related mental decline.
4. Voluntary Potential Energy (Spirituality & Purpose): Unlike passive or spontaneous biological processes, voluntarily acquired potential energy is intentionally cultivated through behaviors such as self-discipline, ethical decision-making, and persistence. When human behavior aligns with a voluntarily chosen lifestyle, principles, values, or core beliefs, it creates consistency, compatibility, and harmony. This alignment raises the intrinsic value of behavior, as it reflects a deep coherence between body and mind. The resulting connection fosters a sense of enriched spirituality, which, in turn, manifests as potential energy, enhancing one’s capacity to engage in actions that promote construction, productivity, and overall well-being. Research in epigenetics has shown that spiritual practices regulate stress-response genes, reducing cortisol levels and pro-inflammatory cytokine activity. Engaging in ethical behavior, gratitude, and purpose-driven living fosters positive epigenetic modifications that enhance mental well-being and psychological resilience, while spiritual engagement has been linked to increased telomerase activity, a marker of cellular longevity. Through these mechanisms, spirituality serves as a biological modulator, reinforcing genetic and metabolic stability while promoting overall physiological harmony.
Through the enrichment of these four domains of potential energy, Lifestylopathy highlights key elements of epigenetic regulation, promotes cellular integrity, and reduces disease risk. This holistic model highlights the interconnected relationship between lifestyle choices, environmental influences, and gene expression, reinforcing the need for personalized, proactive healthcare in achieving optimal health and longevity.
Epigenetics and Physics
The activation of potential energy serves as the primary force that counteracts entropy, the natural drift toward disorder. Unlike kinetic energy, which is dissipative, potential energy represents stored possibilities that, when harnessed, bring structure, stability, and organization into a system. In human behavior, this energy appears as latent personality traits, cognitive abilities, and emotional capacities that, if left undeveloped, contribute to psychological decline and environmental entropy. Just as the absence of physical exercise leads to laziness and weakness, neglecting the activation of potential energy results in stagnation and decline. In our understanding, potential energy remains the only known force capable of resisting the universal pull toward disorder, emphasizing its vital role in sustaining both biological and psychological balance. This understanding is consistent with the Second Law of Thermodynamics, demonstrating that entropy naturally increases unless countered by an external energy source.
Without deliberate effort to activate and direct potential energy, human behavior and personality risk devolving into chaotic and self-destructive patterns. While many sources of potential energy have been extensively studied, the role of voluntarily acquired potential energy remains largely unexplored. Understanding this form of potential energy requires the inclusion of quantum mechanics. The superposition, in quantum mechanics, describes the ability of a system to exist in multiple potential states simultaneously until an observation collapses it into a singular reality. Similarly, human personality is dynamic, consisting of potential traits that remain dormant until an individual consciously chooses to activate one over the others. Each trait has both a constructive and a destructive aspect. The deliberate cultivation of constructive traits is essential for fostering positive outcomes. Neglecting this process increases the risk of destructive traits emerging, resulting in disordered behavior and negative environmental effects.
This suggests that human behavior and the surrounding environment can be deliberately shaped through the activation of constructive traits. By consciously enriching potential energy through constructive actions, individuals create an environment conducive to stability and growth. The environment acts as both a trigger and a regulator of gene activity, meaning that individuals shape their biological expression through lifestyle choices. This bidirectional relationship between environment and behavior forms a self-reinforcing cycle in which personal development influences external conditions, and in turn, these conditions further modulate genetic expression. Constructive traits, when activated voluntarily, create a structured behavioral framework that enriches potential energy and stabilizes the surrounding environment.
Research in epigenetics has shown that voluntary actions directly influence gene expression, particularly in regulating stress-related pathways. For instance, mindfulness and meditation modify DNA methylation patterns in genes associated with cortisol regulation, reducing chronic stress responses and enhancing emotional resilience. Similarly, ethical decision-making and disciplined behavior support neuroplasticity, improving cognitive adaptability while lowering the risk of anxiety and depression. Purpose-driven actions further regulate inflammatory gene expression, minimizing the effects of stress-related chronic illnesses and promoting long-term health. Conversely, neglecting the cultivation of these traits allows entropy to take over, leading to behavioral instability, psychological distress, and gene expression patterns that increase vulnerability to disease.
The connection between conscious choices, personality development, and epigenetic regulation highlights that personality is not merely an inherited trait but a dynamic, evolving construct shaped by intentional living. When actions are deeply rooted in personal values and beliefs, they create a self-reinforcing cycle of stability, motivation, and resilience. This consistency nurtures a profound sense of purpose, elevating potential energy and empowering individuals to engage in constructive, goal-oriented pursuits. As a result, positive lifestyle habits strengthen neurocognitive, metabolic, and immune resilience, cultivating an adaptive state that not only mitigates stress but also promotes holistic well-being. In contrast, a lack of voluntarily cultivated potential energy can lead to internal disharmony, increasing susceptibility to emotional instability, physiological decline, and stress-related disorders.
Lifestyle-Therapeutic Drug Compatibility
Establishing a strong compatibility between a patient’s lifestyle and their prescribed medication is crucial for maximizing therapeutic effectiveness. Just as soil quality determines how well a seed grows, the body’s internal environment influences how effectively a drug is absorbed, metabolized, and utilized. When a treatment plan is aligned with an individual’s lifestyle orientations such as Vegan, Halal, Kosher, Vegetarian, Gluten free, Glucose free, and Lactose free, the drug transitions from being a mere chemical intervention to becoming a catalyst for long-term healing. Conversely, when lifestyle behaviors contradict the intended effects of a medication, they can diminish its efficacy, introduce complications, and create imbalances that hinder recovery.
Lifestylopathy transforms this understanding into a structured, systematic approach to healthcare. It underscores the premise that mere medical treatment is insufficient if not complemented by supportive lifestyle choices that promote healing. The body is not a passive recipient of pharmaceuticals; rather, it actively participates in the healing process, which is shaped by overall well-being. A lifestyle behavior that prioritizes nutrient-dense foods, restorative sleep, regular physical activity, and emotional balance creates an optimal biological framework in which medications can function at their highest potential. Additionally, when individuals perceive their treatment as aligned with their lifestyle orientations, values, beliefs, and daily routines, their trust in the healing process deepens. This psychological alignment triggers positive physiological responses, such as the placebo effect, which has been shown to enhance treatment outcomes by activating the body’s intrinsic healing mechanisms.
On the other hand, lifestyle behaviors that conflict with prescribed treatment can impair the body’s ability to integrate and respond effectively to medications. Incompatibility increases biological entropy, leading to greater disorder and conflict within physiological systems. Conversely, compatibility fosters biological potential, where physiological processes merge toward recovery. When coherence is achieved, medications can function optimally, and natural healing mechanisms are reinforced rather than disrupted. The interaction between medication, lifestyle, and behaviors culminates in a favorable internal environment that significantly enhances therapeutic outcomes through several mechanisms: direct pharmacological efficacy, lifestyle alignment effects, and psychological and neurophysiological benefits, including the placebo effect that amplifies therapeutic responses.
When these three elements converge, they create a synergistic enhancement of treatment outcomes. This synergy not only improves immediate therapeutic efficacy but also influences long-term health by regulating epigenetic mechanisms. The alignment of lifestyle with medication also has profound implications for gene expression. The body’s internal state serves as a biochemical signaling environment that influences epigenetic modifications dictating gene activation or suppression. Conditions that promote low stress and support healing are more likely to activate genes related to cellular repair and immune function while suppressing those linked to inflammation and disease progression. Favorable lifestyle conditions, including proper nutrition and regular physical activity, can modulate epigenetic markers, thus regulating gene expression effectively. When lifestyle and medication work in harmony, they consistently signal the body toward recovery and reduce inflammation-related stresses, fostering a form of cellular memory that enhances the body’s proficiency in maintaining advantageous gene expression patterns and engendering long-term health benefits, including reduced relapse rates and bolstered resilience against potential illnesses.
Conclusion
The integration of Lifestylopathy and epigenetics offers a novel framework for personalized medicine, emphasizing the dynamic interaction between lifestyle choices, gene expression, and biological resilience. By recognizing potential energy as a modifiable determinant of health, this approach shifts the focus from reactive disease management to proactive health optimization, where structured interventions, nutrition, exercise, mindfulness, and ethical living, actively shape epigenetic landscapes to enhance metabolic stability, immune function, and cognitive longevity. However, the full clinical application of this model requires further empirical validation, particularly in measuring non-physical energy domains, quantifying epigenetic responses to voluntary behaviors, and addressing socioeconomic disparities in health accessibility. Future research should focus on developing biomarkers for potential energy (e.g., oxytocin, stress-adaptive epigenetic markers), conducting longitudinal studies on lifestyle-based epigenetic modifications, and exploring the balance between entropy reduction and adaptive flexibility in biological systems. Additionally, integrating Lifestylopathy principles into public health policies can ensure that these insights extend beyond individualized wellness to population-wide health strategies, fostering a more inclusive, preventive, and sustainable model of medicine. With rigorous scientific refinement, Lifestylopathy has the potential to bridge the gap between genetics, environment, and human behavior, positioning itself as a foundational discipline in the future of holistic and integrative medicine.
Conflicts of interests: None
Funding statement: The authors have no funding to report.
References
- Zhang M, Ward J, Strawbridge R, Celis‐Morales C, Pell J, Lyall D, et al. How do lifestyle factors modify the association between genetic predisposition and obesity-related phenotypes? A 4-way decomposition analysis using UK Biobank. BMC Med. 2024;22(1). https://doi.org/10.1186/s12916-024-03436-6.
- Liu J. Unveiling the dominant influence: genetic status and lifestyle factors in cardiovascular health. Theor Nat Sci. 2024;54(1):116-121. https://doi.org/10.54254/2753-8818/54/2024au0146.
- Norwitz N, Saif N, Ariza I, Isaacson R. Precision nutrition for Alzheimer’s prevention in APOE4 carriers. Nutrients. 2021;13(4):1362. https://doi.org/10.3390/nu13041362.
- Zhang T, Meaney M. Epigenetics and the environmental regulation of the genome and its function. Annu Rev Psychol. 2010;61(1):439-466. https://doi.org/10.1146/annurev.psych.60.110707.163625.
- Dauncey M. Genomic and epigenomic insights into nutrition and brain disorders. Nutrients. 2013;5(3):887-914. https://doi.org/10.3390/nu5030887.
- Rajpathak S, Liu Y, Ben‐David O, Reddy S, Atzmon G, Crandall J, et al. Lifestyle factors of people with exceptional longevity. J Am Geriatr Soc. 2011;59(8):1509-1512. https://doi.org/10.1111/j.1532-5415.2011.03498.x.
- Dauncey M. Recent advances in nutrition, genes and brain health. Proc Nutr Soc. 2012;71(4):581-591. https://doi.org/10.1017/s0029665112000237.
- Alzeer J. Lifestylopathy as personalized medicine: a holistic approach to health. Med Res Arch. 2025;13(1). https://doi.org/10.18103/mra.v13i1.6209.
- Nutt D. Addiction: lifestyle choice or medical diagnosis? J Eval Clin Pract. 2013;19(3):493-496. https://doi.org/10.1111/jep.12045.
- Gillsjö C, Karlsson S, Ståhl F, Eriksson I. Lifestyle’s influence on community-dwelling older adults’ health: a mixed-methods study design. Contemp Clin Trials Commun. 2021;21:100687. https://doi.org/10.1016/j.conctc.2020.100687.
- Reddy O, Werf Y. The sleeping brain: harnessing the power of the glymphatic system through lifestyle choices. Brain Sci. 2020;10(11):868. https://doi.org/10.3390/brainsci10110868.
- Dauncey M. Nutrition, the brain and cognitive decline: insights from epigenetics. Eur J Clin Nutr. 2014;68(11):1179-1185. https://doi.org/10.1038/ejcn.2014.173.
- Meng W, Adams M, Deary I, Palmer C, McIntosh A, Smith B. Genetic correlations between pain phenotypes and depression and neuroticism. 2018. https://doi.org/10.1101/362574.
- Moore L, Le T, Fan G. DNA methylation and its basic function. Neuropsychopharmacology. 2012;38(1):23-38. https://doi.org/10.1038/npp.2012.112.
- Li E, Zhang Y. DNA methylation in mammals. Cold Spring Harb Perspect Biol. 2014;6(5):a019133-a019133. https://doi.org/10.1101/cshperspect.a019133.
- Reddy M, Natarajan R. Recent developments in epigenetics of acute and chronic kidney diseases. Kidney Int. 2015;88(2):250-261. https://doi.org/10.1038/ki.2015.148.
- Lima R, Hayashi D, Lima K, Gomes N, Ribeiro M, Prada P, et al. The role of epigenetics in the etiology of obesity: a review. J Clin Epigenetics. 2017;3(4). https://doi.org/10.21767/2472-1158.100075.
- Tarale P, Chakrabarti T, Sivanesan S, Naoghare P, Bafana A, Krishnamurthi K. Potential role of epigenetic mechanism in manganese-induced neurotoxicity. Biomed Res Int. 2016;2016:1-18. https://doi.org/10.1155/2016/2548792.
- Fransquet P, Wrigglesworth J, Woods R, Ernst M, Ryan J. The epigenetic clock as a predictor of disease and mortality risk: a systematic review and meta-analysis. Clin Epigenetics. 2019;11(1). https://doi.org/10.1186/s13148-019-0656-7.
- Zannas AS, Wiechmann T, Gassen NC, Binder EB. Gene-Stress-Epigenetic Regulation of FKBP5: Clinical and Translational Implications. Neuropsychopharmacology. 2016;41(1):261-274. https://doi.org/10.1038/npp.2015.235.
- Brivio P, Sbrini G, Tarantini L, Parravicini C, Gruca P, Łasoń M, et al. Stress modifies the expression of glucocorticoid-responsive genes by acting at epigenetic levels in the rat prefrontal cortex: modulatory activity of lurasidone. Int J Mol Sci. 2021;22(12):6197. https://doi.org/10.3390/ijms22126197.
- Kim M, Costello J. DNA methylation: an epigenetic mark of cellular memory. Exp Mol Med. 2017;49(4):e322-e322. https://doi.org/10.1038/emm.2017.10.
- MacKinnon A, Feeley N, Gold I, Hayton B, King L, Nagy C, et al. The interaction between oxytocin receptor gene methylation and maternal behavior on children’s early theory of mind abilities. Dev Psychopathol. 2019;32(2):511-519. https://doi.org/10.1017/s0954579419000257.
- Hing B, Braun P, Cordner Z, Ewald E, Moody L, McKane M, et al. Chronic social stress induces DNA methylation changes at an evolutionary conserved intergenic region in chromosome X. Epigenetics. 2018;13(6):627-641. https://doi.org/10.1080/15592294.2018.1486654.
- Fernandes J, Arida RM, Gomez-Pinilla F. Physical exercise as an epigenetic modulator of brain plasticity and cognition. Neurosci Biobehav Rev. 2017;80:443-456. https://doi.org/10.1016/j.neubiorev.2017.06.012.
- Nilsson E, Skinner M. Environmentally induced epigenetic transgenerational inheritance of disease susceptibility. Transl Res. 2015;165(1):12-17. https://doi.org/10.1016/j.trsl.2014.02.003.
- Alzeer J. Integrating medicine with lifestyle for personalized and holistic healthcare. J Public Health Emerg. 2023;7:33. https://doi.org/10.21037/jphe-23-71.
- Alzeer J. Lifestylopathy: unlocking potential by embracing duality and homeostasis for improved healthcare. Int J Regen Med. 2023;1-6. https://doi.org/10.31487/j.rgm.2023.02.02.
- Alzeer J. Entropy and potential energy as a key role of halalopathy in disease prevention and cure. Longhua Chin Med. 2020;3:20-20. https://doi.org/10.21037/lcm-20-40.
- Alzeer J. Halalopathy: stimulation of the immune system through enrichment of potential energy. Int J Regen Med. 2022;1-5. https://doi.org/10.31487/j.rgm.2022.01.02.
- Alzeer, J. (2023). The role of buffers in establishing a balance of homeostasis and maintaining health. American Journal of Medical Chemistry, 1, 1–6. https://doi.org/10.31487/j.ajmc.2023.01.01.
- Horrigan B, Lewis S, Abrams D, Pechura C. (2012). Integrative medicine in America—how integrative medicine is being practiced in clinical centers across the United States. Global Advances in Health and Medicine, 1(3), 18–52. https://doi.org/10.7453/gahmj.2012.1.3.006.
- Waterland R. (2012). Nutritional epigenetics. N/A, 14–26. https://doi.org/10.1002/9781119946045.ch2.
- Park L, Friso S, Choi S. (2011). Nutritional influences on epigenetics and age-related disease. Proceedings of the Nutrition Society, 71(1), 75–83. https://doi.org/10.1017/s0029665111003302.
- Decourcelle A, Leprince D, Dehennaut V. (2019). Regulation of polycomb repression by O-GlcNAcylation: Linking nutrition to epigenetic reprogramming in embryonic development and cancer. Frontiers in Endocrinology, 10. https://doi.org/10.3389/fendo.2019.00117.
- Haggarty P. (2013). Epigenetic consequences of a changing human diet. Proceedings of the Nutrition Society, 72(4), 363–371. https://doi.org/10.1017/s0029665113003376.
- Ideraabdullah F, Zeisel S. (2018). Dietary modulation of the epigenome. Physiological Reviews, 98(2), 667–695. https://doi.org/10.1152/physrev.00010.2017.
- Hino S, Nagaoka K, Nakao M. (2013). Metabolism–epigenome crosstalk in physiology and diseases. Journal of Human Genetics, 58(7), 410–415. https://doi.org/10.1038/jhg.2013.57.
- Tiffon C. (2018). The impact of nutrition and environmental epigenetics on human health and disease. International Journal of Molecular Sciences, 19(11), 3425. https://doi.org/10.3390/ijms19113425.
- Lewis C, Olive M. (2014). Early-life stress interactions with the epigenome. Behavioural Pharmacology, 25(5–6), 341–351. https://doi.org/10.1097/fbp.0000000000000057.
- Dick A, Chen A. (2021). The role of TET proteins in stress-induced neuroepigenetic and behavioural adaptations. Neurobiology of Stress, 15, 100352. https://doi.org/10.1016/j.ynstr.2021.100352.
- Kawatake-Kuno A, Murai T, Uchida S. (2021). The molecular basis of depression: Implications of sex-related differences in epigenetic regulation. Frontiers in Molecular Neuroscience, 14. https://doi.org/10.3389/fnmol.2021.708004.
- Badaeva A, Danilov A, Clayton P, Moskalev A, Karasev A, Tarasevich A. et al. (2023). Perspectives on neuronutrition in prevention and treatment of neurological disorders. Nutrients, 15(11), 2505. https://doi.org/10.3390/nu15112505.
- Potter C, Moorman A, Relton C, Ford D, Mathers J, Strathdee G, et al. (2018). Maternal red blood cell folate and infant vitamin B12 status influence methylation of genes associated with childhood acute lymphoblastic leukemia. Molecular Nutrition & Food Research, 62(22). https://doi.org/10.1002/mnfr.201800411.
- Wu S, Zhang J, Li F, Du W, Zhou X, Wan M, et al. (2019). One-carbon metabolism links nutrition intake to embryonic development via epigenetic mechanisms. Stem Cells International, 2019, 1–8. https://doi.org/10.1155/2019/3894101.
- Alzeer J, Arafeh R, Al-Gubory K H. (2017). Antioxidants in the prevention and treatment of cancer. In K. Al-Gubory & I. Laher (Eds.), Nutritional Antioxidant Therapies: Treatments and Perspectives (pp. N/A). Springer, Cham. https://doi.org/10.1007/978-3-319-67625-8_19.
- Chango A, Pogribny I. (2015). Considering maternal dietary modulators for epigenetic regulation and programming of the fetal epigenome. Nutrients, 7(4), 2748–2770. https://doi.org/10.3390/nu7042748.
- Shields A E, Zhang Y, Argentieri M A, Warner E T, Cozier Y C, Liu C, et al. (2021). Stress and spirituality in relation to HPA axis gene methylation among US Black women. Epigenomics, 13(21), 1711–1734. https://doi.org/10.2217/epi-2021-0275.
- Xue J, Zempleni J. (2013). Epigenetic synergies between biotin and folate in the regulation of pro-inflammatory cytokines and repeats. Scandinavian Journal of Immunology, 78, 419–425.
- Teixeira M Z. (2021). Telomere and telomerase: Biological markers of organic vital force state and homeopathic treatment effectiveness. Homeopathy, 110(4), 283–291. https://doi.org/10.1055/s-0041-1726008.
- Alzeer J. (2024). Harnessing the power of choice: How to thrive in a universe of entropy. American Journal of Medical Chemistry, 5(1), 2–6. https://doi.org/10.31487/j.AJMC.2024.01.02.
- Alzeer J. (2024). Beyond disorder: A new perspective on entropy in chemistry. American Journal of Medical Chemos, 5, 1–5.
- Hazard D, Plisson‐Petit F, Moreno‐Romieux C, Fabre S, Drouilhet L. (2020). Genetic determinism exists for the global DNA methylation rate in sheep. Frontiers in Genetics, 11. https://doi.org/10.3389/fgene.2020.616960.
- Sanders A, Bhongir N, vonHoldt B, Pellegrini M. (2022). Association of DNA methylation with energy and fear-related behaviors in canines. Frontiers in Psychology, 13. https://doi.org/10.3389/fpsyg.2022.1025494.
- Alzeer J, Benmerabet H. (2024). Exploring the intersection of quantum mechanics and human psychology. Psychological Disease Research, 7(1), 1–6. https://doi.org/10.31487/j.pdr.2024.01.
- Kumsta R, Schlotz W, Golm D, Moser D, Kennedy M, Knights N, et al. (2017). HPA axis dysregulation in adult adoptees twenty years after severe institutional deprivation in childhood. Psychoneuroendocrinology, 86, 196–202. https://doi.org/10.1016/j.psyneuen.2017.09.021.
- Alzeer J, Benmerabet H. (2025). Potentiality to actuality: Quantum physics inspires creative innovation. Jurnal Pijar Mipa, 20(1), 1–6. https://doi.org/10.29303/jpm.v20i1.8176.
- Chaix R, Fagny M, Cosín‐Tomás M, Álvarez‐López M, Lemée L, Regnault B, et al. (2020). Differential DNA methylation in experienced meditators after an intensive day of mindfulness-based practice: Implications for immune-related pathways. Brain, Behavior, and Immunity, 84, 36–44. https://doi.org/10.1016/j.bbi.2019.11.003.
- Lester B, Conradt E, Marsit C. (2016). Introduction to the special section on epigenetics. Child Development, 87(1), 29–37. https://doi.org/10.1111/cdev.12489.

