Pharmacodynamic vs Pharmacokinetic Assays for DOACs
Measuring Direct Oral Anticoagulants: Pharmacodynamic vs Pharmacokinetic Assays – A New Pharmacodynamic Assay
Chad Kimmitt¹, Deborah Siegal², Jack Ansell³
- Chad Kimmitt, MD; Alberta Health Services; [email protected]
- Deborah Siegal, MD; University of Ottawa and Ottawa Hospital Research Institute. [email protected]
- Jack Ansell, MD; Hofstra Northwell Zucker School of Medicine; [email protected]
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Kimmitt, C., Siegal, D., et al., 2026. Measuring Direct Oral Anticoagulants: Pharmacodynamic vs Pharmacokinetic Assays – A New Pharmacodynamic Assay. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Direct oral anticoagulants need no routine monitoring, but measurement of drug levels or activity can be essential in emergent and selected situations. Pharmacokinetic assays measure drug concentration and pharmacodynamic assays measure drug activity on coagulation. However, drug effect on coagulation as measured by a pharmacodynamic assay does not always correlate with drug efficacy as measured by a pharmacokinetic assay. To measure antithrombotic effect, routinely available pharmacodynamic assays are too insensitive to reliably assess low levels of drug concentration where critical decisions need to be made, thus pharmacokinetic assays are used. For emergent situations, however, a readily available and rapid turnaround pharmacokinetic assay is not available. This review discusses the qualities and value of pharmacokinetic and pharmacodynamic assays, summarizes the various assays currently in use, and presents data on a recently developed, whole blood, point of care coagulometer that provides a pharmacodynamic assessment of blood coagulation with sensitivity to the direct oral anticoagulants.
Keywords
Direct oral anticoagulants, pharmacodynamic assays, pharmacokinetic assays, coagulation, point of care testing.
Introduction
Direct oral anticoagulants (DOACs) are the preferred anticoagulants for the prevention and treatment of venous thromboembolism (VTE) and ischemic stroke prevention in the setting of non-valvular atrial fibrillation. These include apixaban, rivaroxaban, and edoxaban (direct inhibitors of factor Xa), and dabigatran (direct inhibitor of thrombin [factor IIa]). Numerous studies in the fields of VTE and atrial fibrillation have demonstrated comparable efficacy with superior bleeding profiles of DOACs compared to vitamin K antagonists (VKAs; e.g. warfarin)1-6. Advantages of DOACs include minimal drug and food interactions, predictable pharmacokinetics, and short drug half-lives in the absence of significant renal or hepatic impairment which preclude the need for routine monitoring of anticoagulant activity7. However, it may be desirable to measure anticoagulation status in emergencies such as serious bleeding, urgent surgery, and breakthrough thrombotic events to make informed decisions about the management of any residual anticoagulation. An important challenge is to obtain accurate, reliable, and timely measurement of anticoagulant activity to establish therapeutic thresholds and apply the results to clinical practice.
Clinical Scenarios Where DOAC Measurement May Be Beneficial
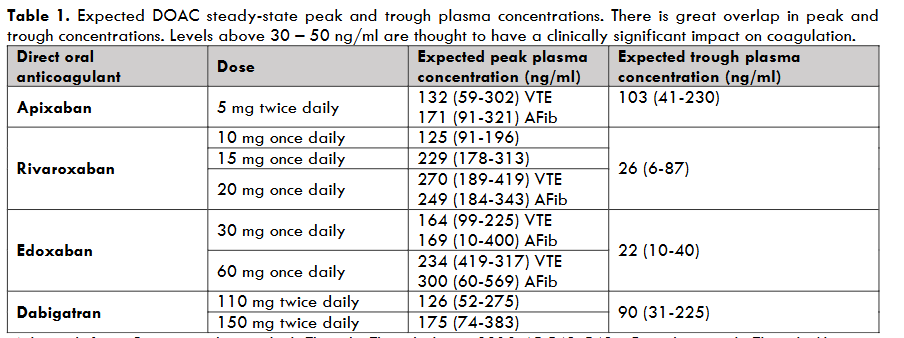
While there is no established therapeutic range for plasma DOAC concentration, ascertaining the presence and amount of circulating DOACs may be beneficial for urgent and non-urgent situations if it would change management7. For urgent scenarios such as severe bleeding, need for emergency high bleeding risk surgery, neuraxial procedures (e.g. lumbar puncture, neuraxial anesthesia), or systemic thrombolysis in the setting of acute ischemic stroke, the rapid detection and quantification of clinically significant DOAC activity using a sensitive assay could optimize management and expedite treatment8-11. For example, major bleeding occurs in about 2% to 4% of DOAC-treated patients annually12 and a sensitive assay with rapid turnaround time can guide the use of reversal or hemostatic therapies8. Similarly, in DOAC-treated patients requiring urgent surgery, including one-third of patients with major bleeding, DOAC measurement can guide the timing of surgery (i.e. proceed or delay) and/or the use of reversal or hemostatic therapies13,14. For breakthrough thrombosis on treatment which occurs in up to 20% of DOAC-treated patients with atrial fibrillation, measurement of DOAC concentration may help ascertain DOAC adherence or absorption, and the safety of administering a time-sensitive administration of systemic thrombolysis15-19. While the threshold DOAC concentration or activity which corresponds to a clinically important hemostatic defect is not known, expert consensus guidance suggests that levels above 30 ng/mL to 50 ng/mL are likely clinically significant (Table 1)14,20,21. Therefore, to be clinically useful, DOAC assays need to be sensitive at the lower end of drug concentration. Optimal management DOAC-treated patients with emergencies includes knowledge of anticoagulant presence or activity using a readily available, accurate, rapid turnaround assay.
Measurement of DOACs may also be considered for non-urgent determination. This includes assessing for excessively low drug levels in the setting of high body weight or body mass index, recurrent VTE or ischemic stroke while on treatment, confirmation of drug adherence, potential drug-drug interactions, and absorption in patients with previous bowel resection or bariatric surgery. Alternatively, assessment of excessively high drug levels may be helpful in settings for which there is a concern where patients are at risk of supratherapeutic DOAC concentrations due to reduced drug clearance such as those with concurrent renal or liver impairment, low body weight, or suspected drug-drug interactions9. For these scenarios, accurate quantification of DOAC levels may change management by selecting an alternative DOAC, dose adjustment, or switch to VKA.
Pharmacodynamic Vs Pharmacokinetic Assays for DOAC Measurement
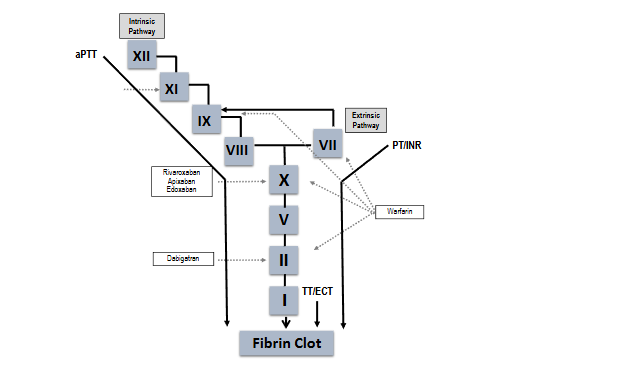
Two general types of coagulation assays can be used to assess an anticoagulant’s presence or activity – functional, pharmacodynamic (PD) assays that measure the dynamics of clot formation as its endpoint (Figure 1), and quantitative or pharmacokinetic (PK) assays that measure drug concentration9. The value of a PK assay is dependent on drug mechanism of action and the assay sensitivity at various levels of drug concentration22. The value of a PD assay is more complex and is dependent on which specific coagulation pathway the assay measures, reagents used in the assay, as well as types of instrumentation7,9. For example, vitamin K antagonists (VKAs) indirectly impair coagulation by inhibiting VK-dependent coagulation factor synthesis, an effect which has slow onset and offset of action and has little correlation with drug concentration, making a PK assay of little value23. Therefore, a PD assay is used to assess and guide anticoagulation (the prothrombin time [PT]/international normalized ratio [INR]). In contrast, the DOACs have a direct immediate effect on their specific target (either factor Xa or IIa), have a rapid onset and offset of anticoagulation, and quickly reach maximum plasma concentration7. The plasma concentration of DOACs correlates with anticoagulant efficacy and a PK assay (e.g., drug-specific calibrated anti-Xa activity assay for factor Xa inhibitors) or, in the case of the thrombin inhibitor, dabigatran, a PD assay, the thrombin time (TT) or dilute thrombin time (dTT), are the measures of choice7. Routine PD (coagulation) assays such as the PT/INR or activated partial thromboplastin time (aPTT) are highly variable depending on the assay, reagents, and instrument, with low sensitivity especially at lower drug concentrations, and are therefore not accurate nor reliable for quantifying DOAC anticoagulant effect7,24-27.
Assessment of anticoagulant effect in urgent situations (e.g. serious bleeding or urgent surgery) requires a rapid turnaround assay24. For the VKAs, widely available, rapid turnaround, PD assays (PT/INR) are available. For the DOACs, the quantitative drug-specific calibrated PK assays are not widely available and often have prolonged turnaround times. This leaves a major gap in managing patients on DOACs who present with major bleeding (eligibility for anticoagulant reversal or hemostatic therapy), need urgent surgery (perioperative management), need thrombolysis for acute ischemic stroke (breakthrough on treatment), or other situations where clinicians need immediate information to make critical decisions. A rapid turnaround PD or PK assay would fill this gap24.
Currently Available Assays for DOAC Measurement And Their Limitations
Pharmacodynamic Assays
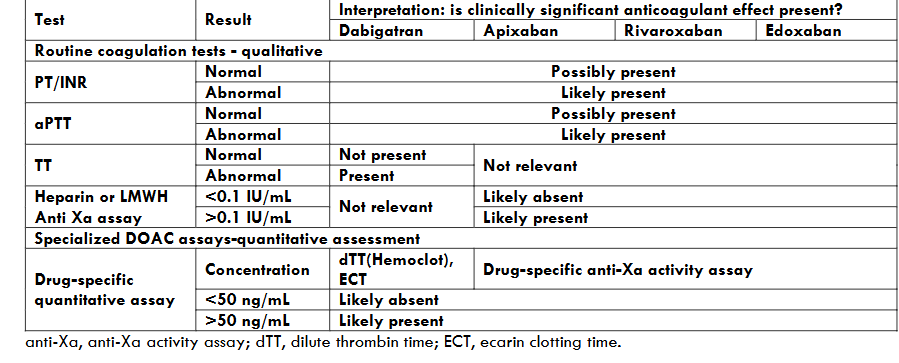
Prothrombin time/International Normalized Ratio, activated partial thromboplastin time, and thrombin time The effect of DOACs on PT/INR and aPTT varies according to the DOAC administered, and the assay, reagents, and analyzer used7,9. Associations between DOAC concentration and the magnitude of PT/INR or aPTT prolongation varies across DOACs. For the factor Xa inhibitors, the PT/INR is generally more reflective of anticoagulant effect than is the aPTT; rivaroxaban has a greater effect on the PT/INR than apixaban, although the effect varies across assays28-30. In general, the PT/INR and aPTT are not sufficiently sensitive to rule out clinically significant or on-therapy range DOAC levels (Table 2)25,31,32. Both assays may have non-linear associations with DOAC concentration particularly at higher levels and cannot be used for quantification. This means that an otherwise unexplained prolonged PT/INR or aPTT in a DOAC-treated patient supports the presence of clinically significant DOAC concentrations but does not provide an assessment of actual drug levels31.
Dabigatran, in contrast, can prolong the aPTT suggesting clinically significant concentrations, but this is also variable across assays. However, the TT, a PD assay, is very sensitive for dabigatran such that a normal result rules out clinically significant dabigatran concentrations24,30, although the TT may be prolonged even in the presence of very low dabigatran levels and cannot be used for quantitation. A dilute TT (dTT) increases the sensitivity of the measurement, and when calibrated to dabigatran levels, can provide quantitative information. The TT or dTT cannot be used to determine the presence or absence of DOACs other than dabigatran. Lastly, the ecarin clotting time (ECT) or ecarin chromogenic assay (ECA) employs a snake venom that is sensitive to direct thrombin inhibitors similar to thrombin, but not to heparin or antiphospholipid antibodies. With calibration it can be used to determine dabigatran levels. As a snake venom it is only available in limited quantities and is not widely available for clinical use24.
In summary, routine PD coagulation tests such as PT/INR and aPTT have a limited role in DOAC measurement due to poor sensitivity as part of a clinical assessment of the likelihood of clinically significant levels in conjunction with timing of last dose and estimated drug clearance (Table 2). It is important to consider that coagulation tests can be abnormal due to other conditions such as hepatic impairment, disseminated intravascular coagulation, coagulopathy of trauma or massive transfusion, or vitamin K deficiency.
Thromboelastogram / Rotational Thromboelastogram
Thromboelastogram/rotational thromboelastogram (TEG and ROTEM) are point of care viscoelastic assays that assess clot formation and lysis in real time using different techniques. ROTEM measures clot formation time (CFT) and TEG measures reaction time (R time)24,33,34. While associations are seen between CFT and R time and increasing peak concentrations of rivaroxaban and dabigatran, results with apixaban are heterogeneous. Additionally, these tests are not sensitive for assessing trough DOAC levels, which is important for determining the presence of clinically significant DOAC concentrations in emergencies33. These assays are not currently recommended by the International Society of Hemostasis and Thrombosis for evaluating the presence of DOAC due to insufficient supporting data21.
Thrombin Generation Assay
The thrombin generation assay (TGA) is a pharmacodynamic assay that assesses hemostasis through a continuous and simultaneous measurement of thrombin formation and inhibition35. The assay reflects the activity of both factor Xa inhibitors and thrombin inhibitors with each class of inhibitors affecting the parameters of the TGA in different ways36,37. While the TGA is the only PD assay to correlate clinical outcomes with drug effect on hemostasis for both bleeding and thrombosis37,38, other studies have shown substantial inter-individual variability in DOAC plasma concentrations and TGA parameters39. Unfortunately, the assay is more complex than routine PD assays (i.e., PT/INR and aPTT or TT) and it is not used clinically but used predominantly as a research assay.
Pharmacokinetic Assays
DOAC-specific anti-Xa assays for factor Xa and IIa inhibitors Drug-specific, pharmacokinetic, calibrated anti-Xa activity assays are accurate and reliable assays for quantifying plasma concentrations of factor Xa inhibitors. Yet, important challenges for interpretation include significant intra- and inter-individual variability reflected by high coefficients of variation22, and a lack of established therapeutic ranges9. ISTH guidance endorses levels above 30 to 50 ng/mL, as the threshold above which clinically significant impairment of hemostasis is present, and reversal may be indicated30. It is important to note that this threshold is based on current expert consensus, and a precise concentration threshold for effect on hemostasis has not been evaluated in clinical studies. This quantification method does bear limitations. The turnaround time for anti-Xa activity assay results reporting can delay treatment, especially when needed in time sensitive clinical emergencies. An American-based retrospective study assessing the turnaround time for DOAC levels using anti-Xa activity assays in the emergency department demonstrated an average result time of 45.9 minutes (interquartile range of 35.3-54.7 minutes)40. Unfortunately, many sites do not have the access or ability to perform DOAC-calibrated anti-Xa activity testing. Lastly, the presence of other agents that inhibit factor Xa, such as heparin or low molecular weight heparin, will falsely elevate the anti-Xa level which leads to difficulty in result interpretation.
Since DOAC-calibrated and specific anti-Xa assays are not widely implemented in hospitals, many institutions have utilized a more commonly available heparin-calibrated anti-Xa activity assay as a substitute. While some studies show a good correlation between an unfractionated heparin or low molecular weight heparin-calibrated anti-Xa activity assay and other more specific measures of drug concentration including mass spectroscopy, the gold standard for determining DOAC concentration, caution is advised because of differences in chromogenic substrates and other variables, and local laboratories must validate assays before their general use9,41-43. Further, other studies have shown variable and limited sensitivity and specificity for detecting consensus-based cutoffs of 30 ng/mL and 50 ng/mL using different assays44.
Dabigatran is measured with a pharmacodynamic assay, the TT (qualitative) or dilute thrombin time (dTT), the latter of which can be calibrated to provide dabigatran-specific concentrations45. The ecarin clotting time (ECT) using ecarin, a thrombin-like enzyme from snake venom, also provides drug specific concentrations, but neither the dTT or the ECT are routinely available in most hospitals9,34,45.
Urine Dipstick
DOASENSE is a urine based, qualitative, point of care test, that detects the presence of DOACs and differentiates between anti-Xa inhibitors and direct thrombin inhibitors. A test strip is dipped into urine and then placed to rest for 10 minutes at which point the final colour is then compared to reference colours for the test result46. This test distinguishes between the presence or absence of a DOAC at a threshold of 30 ng/mL. A meta-analysis demonstrated a mean sensitivity of 97.8% for DOAC detection with a negative predictive value (NPV) of 86.6%. The NPV varied based on the DOAC in question with edoxaban at 100%, rivaroxaban at 97.9%, and apixaban at 81.8%47. Urinary excretion of DOACs varies with apixaban being the least renally cleared at 25%, rivaroxaban at 30%, edoxaban at 50% and dabigatran at 80%48. This may explain the discrepancies in negative predictive value. Certain limitations do exist with this qualitative test. First, test performance is unknown in the setting of acute renal injury or chronic kidney disease and there are no validated creatinine clearance cutoffs for when this test can and cannot be used. False positives may occur if the test strip is read either earlier or later than the 10-minute rest period. Additionally, abnormal urine colour may deem this visually-based test invalid. Lastly, obtaining a urine sample may not always be feasible depending on the clinical setting.
A Novel, Clot-Based Pharmacodynamic Assay for DOACS
A new point-of-care (PoC) coagulometer, ClotChek® (Perosphere Technologies, Inc, Danbury, CT), provides a rapid turnaround PD assessment of global coagulation in whole blood49. In vitro spiked whole blood validation studies have shown it to have high sensitivity and precision, especially at trough factor Xa inhibitor levels, a critical decision point in patients with major bleeding or those needing emergent surgery or intervention. The ClotChek coagulometer is a handheld, rechargeable, battery-operated device as shown in Figure 2. It employs an optical measurement scheme based on transmitted near infrared light to detect fibrin assembly, the final step in the clotting cascade. It measures clotting in whole blood similar to the traditional manual whole blood clotting time (mWBCT).
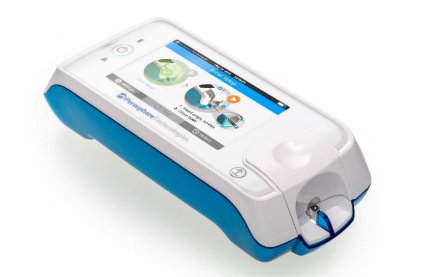
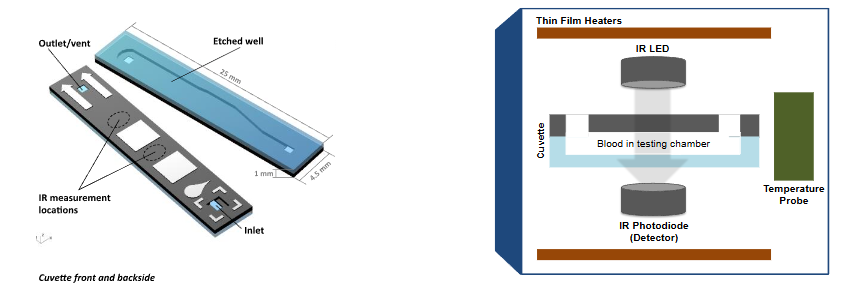
Figure 2. Image of ClotChek® instrument and schematic of ClotChek cuvette and IR imaging system.
The mWBCT, developed over 100 years ago to assess general blood coagulation50, has been modified multiple times to minimize technical difficulties and standardize results. In the 1960’s it was used routinely to monitor and adjust unfractionated heparin therapy51, but was replaced by the plasma-based aPTT in the 1970s52. The mWBCT employs no reagents and coagulation is activated through the intrinsic pathway of coagulation (factor XII, kallikrein, factor XI) by blood contact with the negatively charged glass surface of collection tubes. A normal mWBCT is approximately 7 minutes. The ClotChek device mirrors the mWBCT in that it employs no chemical or biological reagents. It uses disposable, microfluidic cuvettes made of silicon and glass for clotting time measurements and clotting is initiated through contact of a blood sample with the glass surfaces inside a cuvette. The assay requires 14 µL of fresh whole blood which is transferred into an inserted cuvette. The coagulometer is activated as blood enters by capillary action and starts recording the intensity of infrared light transmitted across cuvette and blood sample at 0.5 second intervals. An algorithm automatically detects peak fibrin formation and computes a clotting time associated with the peak in the infrared signal corresponding to peak fibrin clot formation. The instrument performs numerous tests to confirm its proper function and to monitor the quality of the blood sample and collected data. Test results are available in minutes and reported as clotting times in seconds. The coagulometer is broadly sensitive to anticoagulants and disease mechanisms that interfere with blood clotting processes.
In Vitro Validation
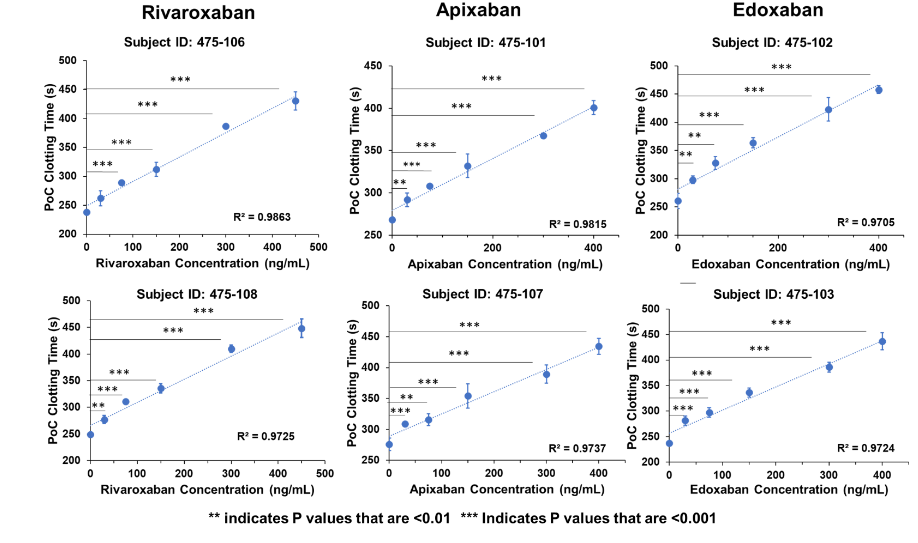
Initial studies demonstrated accuracy of the instrument against the manual WBCT using in vitro spiked human whole blood samples containing increasing concentrations of apixaban, rivaroxaban, edoxaban and enoxaparin53. Mean baseline clotting times were in the range of 221.2 – 281 sec (normal range) for the coagulometer, while mean baseline clotting times for manual WBCT were approximately twice as high, in the range of 456 – 498 sec. All coefficient of variation (CV) values for both methods were within the target range of CV<10%, with most of the values being in the range of CV<5%, indicating uniformly acceptable repeatability of both methods. For both clotting time and percent rise of clotting time from baseline, R > 0.98 indicated a strong correlation between the two methods for each individual subject of each anticoagulant. Subsequent in vitro studies testing the analytic measurement range and linearity of the DOACs (Figure 3) showed a strong linear dose response across the tested concentration ranges with R2 values of 0.9705 to 0.9863 for the three DOACs and precision values ranging between 0.85 – 3.84 CVs.
In Vivo Validation
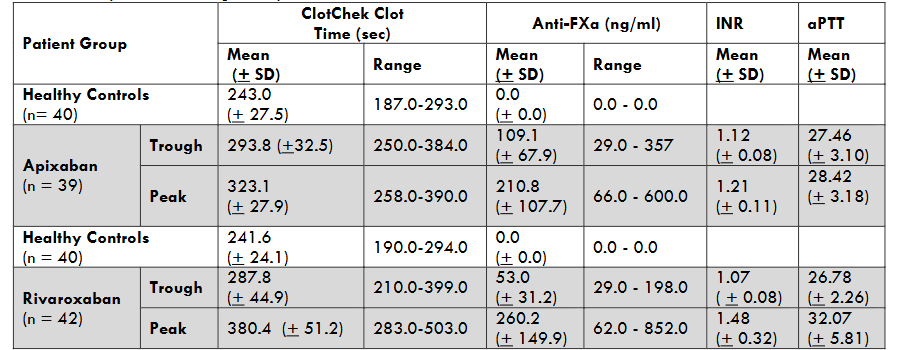
A recent clinical study defined the normal clotting range in 141 healthy volunteers. Mean normal ClotChek clotting time in healthy volunteers was 244.2 (+ 25.8) seconds with a range of 183 – 296 seconds. Trough and peak measurements in patients taking apixaban or rivaroxaban were compared to simultaneous anti-Xa activity measurements, INR, and aPTT values54 (Table 3).
Finally, a sensitivity-specificity analysis was performed to determine the ability of the ClotChek clotting time to predict a DOAC level above 75 ng/mL, a parameter used in a recent DOAC reversal study representing sufficient anticoagulant effect to warrant reversal (N Engl J Med 2019;380:1326-1335). The calculated area under the curve, representing the confidence with which a single measurement can discriminate a sample being above or below the threshold, was 0.911 for apixaban and 0.961 for rivaroxaban. For apixaban, sensitivity was 83.3% and specificity was 86.2%, while for rivaroxaban sensitivity was 90.5% and specificity was 90.0%. These findings indicate that the ClotChek assay can discriminate between individuals with meaningful anticoagulation from those with none or less significant degrees of anticoagulation with a high level of confidence.
Conclusion
In selecting an assay to assess the antithrombotic efficacy and safety of a DOAC, one must consider the value of a pharmacokinetic vs pharmacodynamic assay. Presently, routine PD assays are too variable and insensitive and a pharmacokinetic assay measuring drug concentration is the assay of choice for the DOACs. Such assays, however, are not widely available and have turnaround times insufficient for important clinical decisions in emergent situations. A new automated, POC, rapid turnaround, whole blood clotting time assay has been shown to have good sensitivity at the lower end of drug concentration, filling a gap not addressed by existing PK assays. Further studies are necessary to confirm the value of this assay.
Conflict of Interest
DS and JA are advisors to Perosphere Tech, Inc. JA has stock options in Perosphere Tech. CK has no conflict of interest.
Funding
Partial funding was received from Perosphere Tech, Inc.
Acknowledgments
None
References
- López-López JA, Sterne JAC, Thom HHZ, et al. Oral anticoagulants for prevention of stroke in atrial fibrillation: systematic review, network meta-analysis, and cost effectiveness analysis. BMJ. 2017;359:j5058.
- Agnelli G, Buller HR, Cohen A. et al. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. 2013;369:799-808.
- Büller HR, Décousus H, Grosso MA. et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N Engl J Med. 2013;369:1406-1415.
- Bauersachs R, Berkowitz SD, Brenner B, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363:2499–510.
- Büller HR, Prins MH, Lensin AW, et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012;366:1287–97.
- Schulman S, Kearon C, Kakkar AK. et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361:2342-2352.
- Eikelboom JW, Quinlan DJ, Hirsh J, Connolly SJ, Weitz JI. Laboratory monitoring of non-vitamin K antagonist oral anticoagulant use in patients with atrial fibrillation: a review JAMA Cardiol. 2017;2:566-574.
- Gendron N, Billoir P, Siguret V, et al. Is there a role for the laboratory monitoring in the management of specific antidotes of direct oral anticoagulants? Thromb Res. 2024;237:171-180.
- Mithoowani S, Tan CW, Siegal DM. Measuring direct oral anticoagulant (DOAC) levels: Applications, limitations, and future directions. Int J Lab Hematol. 2025;22 Apr 2025. doi: 10.1111/ijlh.14483.
- Mithoowani S, Bungard T, Castellucci L, et al. Multidisciplinary expert guidance for the management of severe bleeding on oral anticoagulation: An algorithm for practicing clinicians. Thromb Haemost. 2025;125:717-732.
- Ebner M, Birschmann I, Peter A, et al. Emergency coagulation assessment during treatment with direct oral anticoagulants: Limitations and solutions. Stroke. 2017;48:2457-2463.
- Ruff CT, Giugliano RP, Braunwald E, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet. 2014;383:955–962.
- Siegal DM, Freedman D, Ansell J. Urgent procedures or surgeries among patients receiving oral anticoagulants: A systematic literature review. J Thromb Thrombolys. 2023;55:197-202.
- Douketis JD, Li Na, St. John M, et al. Perioperative management of direct oral anticoagulants in patients having a high bleed risk surgery or neuraxial procedure: the Perioperative Anticoagulant Use for Surgery Evaluation (PAUSE-2) pilot randomized trial. J Thromb Haemost. 2025;23:2226-2234.
- McCabe JJ, Cheung Y, Foley M, et al. Residual risk of recurrent stroke despite anticoagulation in patients with atrial fibrillation: A systematic review and meta-analysis. . 2025:82:696-705.
- Choi Y, Kim JS. Anticoagulation failure in stroke: Causes, risk factors, and treatment. J Stroke. 2025;27:195-206.
- Probeck K, Elitharp D, Condon C, Casparis A, Labropoulos N. True failures of direct oral anticoagulants at a university medical center. J Vas Surg. 2022;10:561 (abst).
- Seiffge DJ, De Marchis GM, Koga M, et al. Ischemic stroke despite oral anticoagulant therapy in patients with atrial fibrillation. Ann Neruol. 2020;87:677-687.
- Meinel TR, Branca M, De Masrchis GM, et al. Prior anticoagulation in patients with ischemic stroke and atrial fibrillation. Ann Neurol. 2021;90:517.
- Connolly SJ, Crowther M, Eikelboom JW, et al. Full study report of Andexanet alfa for bleeding associated with factor Xa inhibitors. N Engl J Med. 2019;380:1326-1335.
- Levy JH, Shaw JR, Castellucci LA, et al. Reversal of direct oral anticoagulant: guidance from the SSC of the ISTH. J Thromb Haemost. 2024;22:2889-2899.
- Baker RM, Selby R, Moffat KA, et al. Investigating interassay variability between direct oral anticoagulant calibrated anti-factor Xa assays: a substudy of the perioperative anticoagulation use for surgery evaluation (PAUSE). Res Pract Thromb Haemost. 2025:9:e102899.
- Ansell, J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G. The pharmacology and management of the vitamin K antagonists: ACCP evidence-based clinical practice guidelines. . 2008;133:160S-198S.
- Connors JM. Testing and monitoring direct oral anticoagulants. Blood.2018;132:2009-2015.
- Testa S, Legnani C, Tripodi A, et al. Poor comparability of coagulation screening test with specific measurement in patients receiving direct oral anticoagulants: results from a multicenter/multiplatform study. J Thromb Haemost. 2016;14:2194-2201.
- Shaw JR, Li N, Nixon J, et al. Coagulation assays and direct oral anticoagulant levels among patients having an elective surgery or procedure. J Thromb Haemost. 2022;20:2953-2963.
- Van Edom CJ, Fiorelli F, Balthazar T, et al. Discrepancy between parallel anti-Xa and activated partial thromboplastin time is related to mortality in patients with Impella-supported cardiogenic shock. J Thromb Haemost. 2025;23:1810-1816.
- Baker P, Platton S, Arachchillage DJ, et al.. Measurement of heparin, direct oral anticoagulants and other non-coumarin anticoagulants and their effects on haemostasis assays: A British Society for Haematology Guideline. Br J Haematol. 2024;205:1302-1318.
- Kim PY, Yeh CH, Dale BJ, et al.. Mechanistic basis for the differential effects of rivaroxaban and apixaban on global tests of coagulation. TH Open 2018:2:e190-e201.
- Levy JH, Ageno W, Chan NC, Crowther M, Verhamme P, Weitz JI; Subcommittee on Control of Anticoagulation. When and how to use antidotes for the reversal of direct oral anticoagulants: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14:623-7.

