Placental Biomarkers in Pre-eclampsia and BPD
Placental biomarkers – A mechanistic review of their pathophysiology and their association with bronchopulmonary dysplasia
R. Chokshi¹, K. McMullen², S. Amatya³, M. Hoffman⁴, J. O’Brien¹*
- Division Maternal Fetal Medicine – Pennsylvania State College of Medicine, Hershey PA, USA.
- Department of OBGYN – Pennsylvania State College of Medicine, Hershey PA, USA.
- Department of Neonatology – Pennsylvania State College of Medicine, Hershey PA, USA.
- Center for Women’s Health and Research – Christiana Care, Newark DE, USA.
OPEN ACCESS
PUBLISHED: 31 October 2024
CITATION: Chokshi, R., McMullen, K., et al., 2024. Placental biomarkers – A mechanistic review of their pathophysiology and their association with bronchopulmonary dysplasia. Medical Research Archives, [online] 12(10).https://doi.org/10.18103/mra.v12i10.5991
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5991
ISSN 2375-1924
Abstract
Placental biomarkers have been studied extensively for their role in development and progression of preeclampsia. Two biomarkers in particular, soluble fms-like tyrosine kinase (sFlt-1) and placental growth factor (PlGF), are part of the angiogenic profile of preeclampsia and serve as a useful measure for placental dysfunction. Alterations in their level are responsible for the pathophysiology of preeclampsia, and their measurements can help predict and predate clinical signs and symptoms of preeclampsia, allowing for risk assessment and escalation in care. Given that these biomarkers measure placental dysfunction, it is reasonable to suspect that they may be useful in the prediction of other placentally derived disease processes, such as fetal growth restriction and adverse neonatal outcomes. Recent studies have shown that low plasma PlGF in maternal serum is more reliable in distinguishing fetal growth restriction secondary to placental dysfunction versus constitutionally small fetuses when compared to typical ultrasound indicators such as abdominal circumference and umbilical artery doppler studies. Altered PlGF has predicted adverse neonatal outcomes, such as development of bronchopulmonary dysplasia and even perinatal deaths. High cord blood levels of sFlt-1 have been shown to correlate with degree of neonatal thrombocytopenia, indicative of endothelial disruption in the newborn. In the era of personalized and precision medicine, placental biomarkers may assist in triaging fetal surveillance and delivery, as well as neonatal care. This review covers the known pathophysiology of these biomarkers, our current understanding of their signaling pathways and downstream effects and includes recent research in their association with a significant complication of prematurity – bronchopulmonary dysplasia.
Introduction
Placental biomarkers have been studied extensively for their role in development and progression of preeclampsia¹. Two biomarkers in particular; soluble fms-like tyrosine kinase (sFlt-1), and placental growth factor (PlGF) are part of the angiogenic profile of preeclampsia and serve as a useful measure for placental dysfunction. Alterations in their level are responsible for the pathophysiology of preeclampsia, and their measurements can help predict and predate clinical signs and symptoms of preeclampsia, allowing for risk assessment and escalation in care². First approved in Europe in 2009, and recently in the United States by the FDA, the biomarker ratio has found its way into clinical areas as an adjunct to current testing for preeclampsia.
Given that these biomarkers measure placental dysfunction, it is reasonable to suspect that they may be useful in the prediction of other placentally derived disease processes such as fetal growth restriction (FGR) and adverse neonatal outcomes. Benton et al.³ noted that low plasma PlGF (<5th percentile for gestational age) in maternal serum was more reliable in distinguishing FGR secondary to placental dysfunction versus constitutionally small fetuses when compared to typical ultrasound indicators such as abdominal circumference and umbilical artery doppler studies. Griffin et al.⁴ in the UK utilized the ‘PELICAN’ cohort study to show low PlGF levels outperformed ultrasound and 46 other biomarkers for the prediction of delivery of a small for gestational age (SGA) newborn less than the 3rd percentile.
Regarding neonatal outcomes, Parchem et al.⁵ as part of a secondary analysis of the PETRA (Preeclampsia Triage by Rapid Assay Trial) data noted that a low PlGF value was independently associated with an adverse neonatal outcome (aRR 17.2, 95% CI 5.2–56.3) with a sensitivity of 95.8% and a negative predictive value of 99.2%. All perinatal deaths in their study were predicted with a low PlGF.
While maternal biomarkers may have a role in predicting poor neonatal outcomes, they are not currently used to risk assess or triage delivery for suspected FGR. It is also unclear whether neonatal serum elevation of these markers after delivery has an impact on long term adverse outcomes. This review will focus on the pathophysiology of these particular biomarkers and their known interactions with the developing fetus and potential for downstream adverse effects, in particular bronchopulmonary dysplasia (BPD).
Pathophysiology
sFLT-1 and PlGF are involved in signaling via VEGF (Vascular Endothelial Growth Factor) and its receptor family, which play an essential role in angiogenesis and vascular maturation. Null mutations of VEGF receptors are known to be embryonically lethal⁶ ⁷. The balance of pro (PlGF) and anti-angiogenesis (sFLT-1) factors are essential for normal vascular development and tissue growth, with alterations in their balance responsible for dysregulation and a host of pathologies including cancer and preeclampsia⁸.
Shibuya⁷ first described the VEGF receptor-1 (VEGFR-1) in 1990, designating it as Fms-like tyrosine kinase-1 (Flt-1) due to similarities with a previously known tyrosine kinase receptor. It was later found that VEGF as a ligand bound to and activated Flt-1⁹, solidifying its function in angiogenesis regulation. Further research has shown that the VEGF system has three receptors (VEGFR 1,2 and 3), with VEGFR-1 and 2 present in vascular endothelial cells and VEGFR-3 seen primarily in the lymphatic endothelium⁸. VEGFR-2 also known as Flk-1 and KDR in older studies is shown to have much lower affinity for VEGF but significantly higher biologic activity¹⁰ (see Figure 1).
PlGF is a member of the VEGF family and while primarily expressed by the human placenta¹¹ is also seen in other microvascular environments¹². PlGF is a homolog of VEGF sharing 44% of the amino acid sequence and has significant structural similarity to VEGF¹³ allowing it to bind to VEGFR-1, but not to VEGFR-2. PlGF influences endothelial cell chemotaxis and proliferation and potentiates the pro-angiogenic effects of VEGF (possibly by displacing VEGF from VEGFR-1 and allowing it to activate VEGFR-2¹⁰. PlGF is produced by the placental villous cytotrophoblasts and syncytiotrophoblasts, with maternal serum levels peaking around 30 weeks gestation in normal pregnancies¹⁴. It appears to play a vital role in placental vasculogenesis and angiogenesis and its production has been shown to correlate with oxygen tension, with PlGF expression significantly down-regulated by hypoxia¹⁵ ¹⁶.
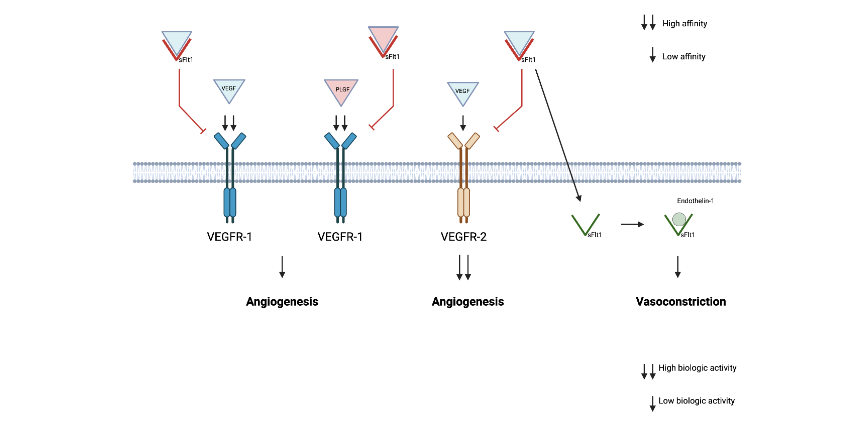
Figure 1 – VEGF – Vascular Endothelial Growth Factor, VEGFR-1 (Flt-1), sFLt-1 (soluble version of Flt-1), VEGFR-2 (Flk1, KDR). PLGF can bind to VEGFR-1 but not to VEGFR-2, and likely modulates VEGF activity by displacing it from VEGFR-1. sFLT-1 binds to circulating VEGF and PLGF blocking its ligand activity.
Conversely sFLT-1 expression is increased with hypoxia, and is a major anti-angiogenic factor produced by the placenta during pregnancy, especially in the setting of preeclampsia¹⁸ ¹⁷ ¹⁸. The Flt-1 gene is known to encode for two mRNA’s – the full length transmembrane receptor (Flt1, VEGFR-1) and a truncated version that lacks the cell membrane anchoring domain and the intracellular tyrosine kinase¹⁹ ²⁰ (see Figure 2 and Figure 3). This soluble version of the Flt-1 receptor (sFlt-1) acts as a decoy molecule by binding to VEGF and PLGF with high affinity and acting as a competitive inhibitor. It also binds directly with VEGFR-1 and 2 and acts as a potent inhibitor of angiogenesis in vivo⁸ ¹⁹ ²⁰.
sFLT-1 is primarily produced by the trophoblast and its expression is significantly upregulated in hypoxic conditions, possibly via the hypoxia-inducible factor (HIF) ¹²¹. It has also been shown to be expressed in vascular endothelial cells and monocytes¹⁹. Because it lacks anchoring domains to the cell membrane it is free to circulate and is known to induce endothelial dysfunction by adhering to and antagonizing VEGF and PlGF receptors altering angiogenesis and vascular maturation¹. (see figure 1)
sFLT-1 levels are known to be elevated in healthy pregnancies compared to non-pregnant controls (mean serum levels of 1762 vs. 85 pg/mL)²². In normal pregnancy, sFLT-1 levels remain fairly stable till 33 to 36 weeks gestation where they begin to rise by ~145 pg/mL per week till delivery. However in pregnancies complicated with preeclampsia the levels are noted to steeply increase about five weeks prior to overt symptomatology, and are markedly elevated compared to non-preeclamptic controls (8150 vs 2466 pg/mL)¹⁸. Conversely PlGF concentration decreases fairly early during pregnancy, peaking at ~32 weeks with steady near linear decrease thereafter. Patients who developed preeclampsia had decreased levels of PlGF as early as 13 weeks compared to controls. and remained lower at time of delivery (73 vs. 370 pg/mL). VEGF levels did not starkly vary with gestational age, but did show reduction in preeclamptic patients¹⁸.
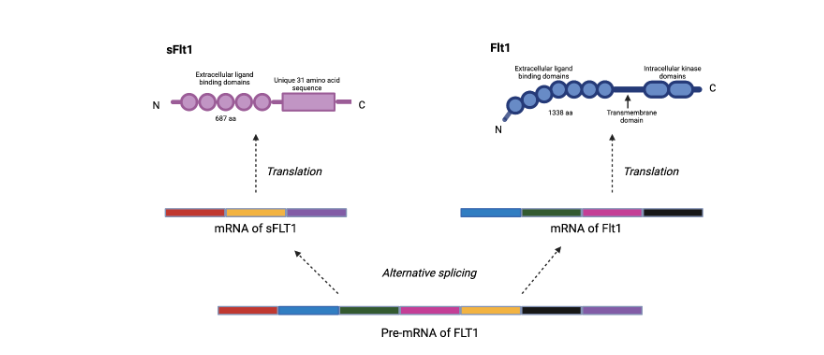
Figure 2 – alternative splicing of Flt-1 mRNA producing the soluble fms-like tyrosine kinase (sFLT-1)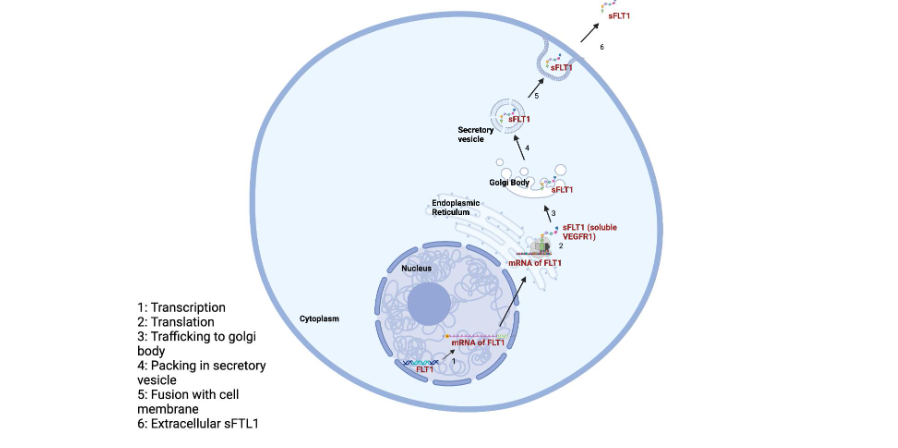
Figure 3 – cellular trafficking of sFLT-1
Biomarkers and Bronchopulmonary Dysplasia
Bronchopulmonary dysplasia (BPD) is a common complication of prematurity, with significant neonatal impact and healthcare costs. Characterized as a chronic lung disorder secondary to abnormal alveolarization and pulmonary vascular development, it affects almost 50% of extremely preterm newborns born at less than 28 weeks gestational age with long term respiratory and neurodevelopmental impairment²³ ²⁴. Infants with BPD suffer from long term respiratory insufficiency, pulmonary hypertension, exercise intolerance, frequent lung infections and recurrent hospitalization²⁵.
Multiple epidemiologic studies have noted an increased risk of BPD in preterm newborns born to preeclamptic mothers²⁶–²⁸. The underlying hypothesis being that preeclampsia is a known anti-angiogenic state, and that lung development similar to vascular formation requires an appropriate balance of pro and anti-angiogenic factors²⁷,²⁹.
BPD is characterized by aberrant lung development, with histological studies noting a pattern of decreased alveolarization (fewer and larger alveoli with fewer septae), decreased capillary density and overall dysmorphic pulmonary vasculature²⁵,²⁹. VEGF is known to be expressed in the distal airspace and its signaling effects are essential to develop the alveolar structure of the lungs³⁰–³⁴. Given the proximity of the airways and blood vessels in the embryonic lung, it is hypothesized that angiogenesis plays an important role in normal alveolarization, with VEGF intricately linked to this process³⁵,³⁶.
Kasahara³⁰ noted that chronic blockade of VEGF caused emphysema in a rat model, highlighting the importance of the VEGF signaling pathway in normal airway development and maintenance. Hasan et al³⁷. noted low levels of VEGF along with elevated levels of sFLT-1 in the tracheal aspirate of preterm infants that developed BPD, concluding that their levels could be utilized as biological predictors for the development of this disorder. Bhatt³⁸ showed disrupted pulmonary vasculature and decreased VEGF in newborns dying from BPD concluding that impaired angiogenesis likely contributed to their pathology. Thebaud et al. not only showed that VEGF blockade in newborn rats impaired alveolar development mimicking BPD, but that postnatal intra-tracheal VEGF gene therapy (to induce overexpression of VEGF) was able to preserve and restore normal alveolarization²⁹.
Utilizing umbilical cord blood, Yang et al.²³ noted that elevated PlGF levels could be used as a predictor for development of bronchopulmonary dysplasia (BPD) in preterm infants. The mechanism of this is unclear at this time, but in a prior paper they also showed that PlGF overexpression caused emphysematous change in a mice model, and that PlGF could inhibit proliferation of pulmonary Type II epithelial cells in vitro³⁹. As previously noted, PlGF is known to modulate VEGF activity by displacing VEGF from VEGFR-1 allowing more binding of VEGFR-2, and there is some initial research noting a larger role for PlGF in the intra molecular receptor cross talk⁴⁰.
To study the possible mechanistic impact of maternal preeclampsia on fetal lung development, Tang et al²⁵. injected sFLT-1 versus saline into the amniotic sac of pregnant Sprague-Dawley rats during the late canalicular and early saccular stage of their fetal lung development (gestational age 20 days, term being 22 days). They noted that the rats injected with intraamniotic sFLT-1 showed decreased alveolar number and reduced pulmonary vessel density consistent with BPD and also evidence of right and left ventricular hypertrophy. They also noted extremely high concentrations of free sFLT-1 in amniotic fluid levels of sFLT-1 secondary to maternal preeclampsia increases the risk of abnormal lung development by impeding VEGF signaling.
Conclusion
In summary, there is now abundant evidence that proper angiogenesis is crucial to early lung development with the VEGF signaling pathway playing an important role. Given the known interaction of sFLT-1 and PlGF with VEGF/VEGF-R, it is reasonable to expect alterations in their levels to influence neonatal adverse outcomes. Maternal and cord blood levels of these biomarkers have shown predictive value for the development of maternal and neonatal adverse outcomes¹⁴,²³,⁴¹–⁴⁵; but further research is needed to evaluate if these biomarkers persist in neonatal bood and quantify their complex downstream effects.
The possible future impact of this research is that maternal serum level of these biomarkers may influence optimal delivery timing, especially in pregnancies complicated by preeclampsia or fetal growth restriction. Given the potential deleterious effects of abnormal placental signaling on the
developing fetus and neonate, earlier delivery may be protective in selected cases. Therapeutic interventions to mitigate adverse perinatal outcomes are also being studied. Early research in extracorporeal apheresis to reduce circulating sFLT-1 in pregnant patients affected with preeclampsia has been promising⁴⁶. A study by McLaughlin et al. found the addition of low molecular weight heparin in the early second trimester could correct low circulating PlGF levels⁴⁷. As an example of the future potential of precision medicine, Turanov et al. developed a short interfering RNA (siRNA) sequence that selectively silenced sFLT-1 mRNA in a preeclamptic animal model prolonging the pregnancy without adverse effects to the developing fetus⁴⁸.
While these above approaches are groundbreaking, the described molecular pathways of placental dysfunction remain incomplete, and further ongoing research is needed. A better understanding of the complex interplay between placenta, fetus and eventual neonate will help elucidate more targeted therapeutics and optimize clinical management of these serious maternal and neonatal conditions.
Conflict of Interest
None
Funding Statement
None
References
1. Burwick RM, Rodriguez MH. Angiogenic Biomarkers in Preeclampsia. Obstetrics and Gynecology. 2024; 143(4):515-523. doi:10.1097/AOG.0000000000005532
2. Thadhani R, Lemoine E, Rana S, et al. Circulating Angiogenic Factor Levels in Hypertensive Disorders of Pregnancy. NEJM Evidence. 2022;1 (12). doi:10.1056/evidoa2200161
3. Benton SJ, McCowan LM, Heazell AEP, et al. Placental growth factor as a marker of fetal growth restriction caused by placental dysfunction. Placenta. 2016;42:1-8. doi:10.1016/j.placenta.2016.03.010
4. Griffin M, Seed PT, Duckworth S, et al. Predicting delivery of a small-for-gestational-age infant and adverse perinatal outcome in women with suspected pre-eclampsia. Ultrasound in Obstetrics and Gynecology. 2018;51(3):387-395. doi:10.1002/uog.17490
5. Parchem JG, Brock CO, Chen HY, Kalluri R, Barton JR, Sibai BM. Placental Growth Factor and the Risk of Adverse Neonatal and Maternal Outcomes. Obstetrics and Gynecology. 2020;135(3):665-673. doi:10.1097/AOG.0000000000003694
6. Kinghorn K, Gill A, Marvin A, et al. A defined clathrin-mediated trafficking pathway regulates sFLT1/VEGFR1 secretion from endothelial cells. Angiogenesis. 2024;27(1):67-89. doi:10.1007/s104 56-023-09893-6
7. Shibuya M. Involvement of Flt-1 (VEGF receptor-1) in cancer and preeclampsia. Proc Jpn Acad Ser B Phys Biol Sci. 2011;87(4):167-178. doi:10.2183 /pjab.87.167
8. Liao L, Zhao X, Zhou M, Deng Y, Li Y, Peng C. sFlt-1: A Double Regulator in Angiogenesis-related Diseases. Curr Pharm Des. 2021;27(40):4160-4170. doi:10.2174/1381612827666210902155015
9. Vries C De, Escobedo JA, al et. The fms-Like Tyrosine Kinase, a Receptor for Vascular Endothelial Growth Factor. Science (1979). 1992;255(5047):989. https://ezaccess.libraries.psu.edu/login?url=https://www.proquest.com/scholarly-journals/fms-like-tyrosine-kinase-receptor-vascular/docview/213550860/se-2?accountid=13158
10. Hiratsuka S, Minowa O, Kuno J, Noda T, Shibuya M. Flt-1 Lacking the Tyrosine Kinase Domain Is Sufficient for Normal Development and Angiogenesis in Mice. Vol 95.; 1998. www.pnas.org .
11. Yang W, Ahn H, Hinrichs M, Torry RJ, Torry DS. Evidence of a novel isoform of placenta growth factor (PlGF-4) expressed in human trophoblast and endothelial cells. J Reprod Immunol. 2003;60( 1):53-60. doi:https://doi.org/10.1016/S0165-0378(03)00082-2
12. Torry D. Expression and function of placenta growth factor: implications for abnormal placentation. J Soc Gynecol Investig. 2003;10(4):17 8-188. doi:10.1016/S1071-5576(03)00048-0
13. Iyer S, Leonidas DD, Swaminathan GJ, et al. The Crystal Structure of Human Placenta Growth Factor-1 (PlGF-1), an Angiogenic Protein, at 2.0 Å Resolution. Journal of Biological Chemistry. 2001; 276(15):12153-12161. doi:10.1074/jbc.M008055200
14. Sherrell H, Dunn L, Clifton V, Kumar S. Systematic review of maternal Placental Growth Factor levels in late pregnancy as a predictor of adverse intrapartum and perinatal outcomes. European Journal of Obstetrics and Gynecology and Reproductive Biology. 2018;225:26-34. doi:10 .1016/j.ejogrb.2018.03.059
15. Makrydimas G, Sotiriadis A, Savvidou MD, Spencer K, Nicolaides KH. Physiological distribution of placental growth factor and soluble Flt-1 in early pregnancy. Prenat Diagn. 2008;28(3) :175-179. doi:10.1002/pd.1916
16. Gobble RM, Groesch KA, Chang M, Torry RJ, Torry DS. Differential Regulation of Human PlGF Gene Expression in Trophoblast and Nontrophoblast Cells by Oxygen Tension. Placenta. 2009;30 (10):869-875. doi:10.1016/j.placenta.2009.08.003
17. Kluivers ACM, Biesbroek A, Visser W, et al. Angiogenic imbalance in pre-eclampsia and fetal growth restriction: enhanced soluble fms-like tyrosine kinase-1 binding or diminished production of placental growth factor? Ultrasound in Obstetrics and Gynecology. 2023;61(4):466-473. doi:10.1002 /uog.26088
18. Levine RJ, Maynard SE, Qian C, et al. Circulating Angiogenic Factors and the Risk of Preeclampsia. Vol 12.; 2004. www.nejm.org
19. Kendall RL, Wang G, Thomas KA. Identification of a Natural Soluble Form of the Vascular Endothelial Growth Factor Receptor, FLT-1, and Its Heterodimerization with KDR. Vol 226.; 1996.
20. Kendall RL, Thomas KA. Inhibition of Vascular Endothelial Cell Growth Factor Activity by an Endogenously Encoded Soluble Receptor (Endothelial Ceils/Mitogenic Inhibitor/Alternative Transcription/Angiogenesis). Vol 90.; 1993. https://www.pnas.org
21. Nevo O, Soleymanlou N, Wu Y, et al. Increased expression of sFlt-1 in in vivo and in vitro models of human placental hypoxia is mediated by HIF-1. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 2006;291 (4):R1085-R1093. doi:10.1152/ajpregu.00794.2005
22. Ahmed AR, Khan MD, Batool H, Chughtai O, Chughtai AS, Sarwar S. Reference Interval of Soluble FMS-like Tyrosine Kinase-1 in Non-Pregnant and Pregnant Females: A Novel Biomarker for Pre-eclampsia. Journal of the College of Physicians and Surgeons Pakistan. 2023;33(12):1395-1399. doi:10. 29271/jcpsp.2023.12.1395
23. Yang WC, Chen CY, Chou HC, Hsieh WS, Tsao PN. Angiogenic Factors in Cord Blood of Preterm Infants Predicts Subsequently Developing Bronchopulmonary Dysplasia. Pediatr Neonatol. 2015;56(6):382-385. doi:10.1016/j.pedneo.2015.02.001
24. Jobe AJ. The New BPD: An Arrest of Lung Development. Pediatr Res. 1999;46(6):641-641. doi:10.1203/00006450-199912000-00007
25. Tang JR, Ananth Karumanchi S, Seedorf G, Markham N, Abman SH. Excess soluble vascular endothelial growth factor receptor-1 in amniotic fluid impairs lung growth in rats: linking preeclampsia with bronchopulmonary dysplasia. Am J Physiol Lung Cell Mol Physiol. 2012;302:36-46. doi:10.1152/ajplung.00294.2011.-Epidemiological
26. Gortner L, Misselwitz B, Milligan D, et al. Rates of bronchopulmonary dysplasia in very preterm neonates in Europe: Results from the MOSAIC cohort. Neonatology (Basel, Switzerland). 2011;99 (2):112-117. doi:10.1159/000313024
27. Hansen AR, Barnés CM, Folkman J, McElrath TF. Maternal Preeclampsia Predicts the Development of Bronchopulmonary Dysplasia. Journal of Pediatrics. 2010;156(4):532-536. doi:10. 1016/j.jpeds.2009.10.018
28. Korhonen P, Tammela O, Koivisto AM, Laippala P, Ikonen S. Frequency and Risk Factors in Bronchopulmonary Dysplasia in a Cohort of Very Low Birth Weight Infants. Vol 54.; 1999.
29. Thébaud B, Ladha F, Michelakis ED, et al. Vascular endothelial growth factor gene therapy increases survival, promotes lung angiogenesis, and prevents alveolar damage in hyperoxia-induced lung injury: Evidence that angiogenesis participates in alveolarization. Circulation. 2005;11 2(16):2477-2486. doi:10.1161/CIRCULATIONAHA.105.541524
30. Kasahara Y, Tuder RM, Taraseviciene-Stewart L, et al. Inhibition of VEGF receptors causes lung cell apoptosis and emphysema. Journal of Clinical Investigation. 2000;106(11):1311-1319. doi:10.11 72/JCI10259
31. KASAHARA Y, TUDER RM, COOL CD, LYNCH DA, FLORES SC, VOELKEL NF. Endothelial Cell Death and Decreased Expression of Vascular Endothelial Growth Factor and Vascular Endothelial Growth Factor Receptor 2 in Emphysema. Am J Respir Crit Care Med. 2001;163( 3):737-744. doi:10.1164/ajrccm.163.3.2002117
32. Role_of_the_Flt-1_receptor_tyr.
33. LETTERS TO NATURE.; 1996.
34. Abnormal_blood_vessel_developm.
35. Healy AM, Morgenthau L, Zhu X, Farber HW, Cardoso W V. VEGF Is Deposited in the Subepithelial Matrix at the Leading Edge of Branching Airways and Stimulates Neovascularization in the Murine Embryonic Lung. Published online 2000. doi:10.10 02/1097-0177(2000)9999:9999
36. abman-2012-bronchopulmonary-dysplasia.
37. Hasan J, Beharry KD, Valencia AM, Strauss A, Modanlou HD. Soluble vascular endothelial growth factor receptor 1 in tracheal aspirate fluid of preterm neonates at birth may be predictive of bronchopulmonary dysplasia/chronic lung disease. Pediatrics. 2009;123(6):1541-1547. doi:10.1542/ peds.2008-1670
38. bhatt-et-al-2012-disrupted-pulmonary-vasculature-and-decreased-vascular-endothelial-growth-factor -flt-1-and-tie-2-in.
39. Tsao PN, Yi-Ning S, Li H, Huang PH, al et. Overexpression of Placenta Growth Factor Contributes to the Pathogenesis of Pulmonary Emphysema. Am J Respir Crit Care Med. 2004;169 (4):505-511. https://ezaccess.libraries.psu.edu/login?url=https://www.proquest.com/scholarly-journals/overexpression-placenta-growth-factor-contributes/docview/199583015/se-2?accountid=13158
40. Autiero M, Waltenberger J, Communi D, et al. Role of PlGF in the intra- and intermolecular cross talk between the VEGF receptors Flt1 and Flk1. Nat Med. 2003;9(7):936-943. doi:10.1038/nm884
41. Voller SB, Chock S, Ernst LM, et al. Cord blood biomarkers of vascular endothelial growth (VEGF and sFlt-1) and postnatal growth: A preterm birth cohort study. Early Hum Dev. 2014;90(4):195-200. doi:10.1016/j.earlhumdev.2014.01.003
42. Tsao PN, Wei SC, Su YN, et al. Placenta Growth Factor Elevation in the Cord Blood of Premature Neonates Predicts Poor Pulmonary Outcome.; 2004. http://publications.aap.org/pediatrics/article-pdf/113/5/1348/1003883/zpe00504001348.pdf
43. Witwicki J, Chaberek K, Szymecka-Samaha N, Krysiak A, Pietruski P, Kosińska-Kaczyńska K. Sflt-1/plgf ratio in prediction of short-term neonatal outcome of small for gestational age neonates. Children. 2021;8(8). doi:10.3390/children8080718
44. Tsao PN, Wei SC, Su YN, Chou HC, Chen CY, Hsieh WS. Excess soluble fms-like tyrosine kinase 1 and low platelet counts in premature neonates of preeclamptic mothers. Pediatrics. 2005;116(2):468-472. doi:10.1542/peds.2004-2240
45. Villalaín C, Herraiz I, Valle L, et al. Maternal and perinatal outcomes associated with extremely high values for the sflt-1 (Soluble fms-like tyrosine kinase 1)/plgf (placental growth factor) ratio. J Am Heart Assoc. 2020;9(7). doi:10.1161/JAHA.119.015548
46. Thadhani R, Kisner T, Hagmann H, et al. Pilot Study of Extracorporeal Removal of Soluble Fms-Like Tyrosine Kinase 1 in Preeclampsia. Published online 2011. doi:10.1161/CIRCULATIONAHA
47. McLaughlin K, Hobson SR, Chandran AR, et al. Circulating maternal placental growth factor responses to low-molecular-weight heparin in pregnant patients at risk of placental dysfunction. Am J Obstet Gynecol. 2022;226(2):S1145-S1156 .e1. doi:10.1016/j.ajog.2021.08.027
48. Turanov AA, Lo A, Hassler MR, et al. RNAi modulation of placental sFLT1 for the treatment of preeclampsia. Nat Biotechnol. 2018;36(12):1164-1173. doi:10.1038/nbt.4297

