Point of Care Echocardiography in Pediatric Emergencies
Point of Care Echocardiography in the Paediatric Emergency Department
Beirne N¹, Conlon E², Barrett M³, Mullen S⁴, Quinn N¹, O’Connor G² Kavanagh K¹
- Department of Paediatric Emergency Medicine, Children’s Health Ireland at Temple Street, Dublin
- Department of Emergency Medicine, Mater Misericordiae University Hospital, Dublin
- Department of Paediatric Emergency Medicine, Children’s Health Ireland at Crumlin, Dublin
- Department of Paediatric Emergency Medicine, Royal Belfast Hospital for Sick Children
OPEN ACCESS
PUBLISHED:28 February 2025
CITATION: Beirne, N., et al., 2025. Point of Care Echocardiography in the Paediatric Emergency Department. Medical Research Archives, [online] 13(2).
https://doi.org/10.18103/mra.v13i2.6332
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i2.6332
ISSN 2375-1924
ABSTRACT
Background: Point of care echocardiography (POCE) provides real time haemodynamic information, crucial in the Emergency Department (ED) where decision-making is required to be both timely and correct in order to establish the most appropriate lifesaving therapy. This case series will discuss several scenarios where the use of POCE directly affected management of critically ill patients.
Aims: To demonstrate the appropriate use of POCE in the Paediatric Emergency Department (PED).
Methods: This is a descriptive study incorporating a series of 5 case reports. A five-part case series derived from tertiary level paediatric emergency departments on the island of Ireland.
Results:
Case 1: describes the use of POCE to confirm the absence or presence of regular organised contractions during pulse checks and to reduce time off the chest in paediatric cardiac arrest.
Case 2: describes the use of POCE to assess left ventricle function and to guide fluid resuscitation in paediatric sepsis.
Case 3: describes the use of POCE to clarify diagnosis of myocarditis in the setting of undifferentiated shock and thereafter to guide fluid management.
Case 4: describes the use of POCE to clarify diagnosis and to guide fluid management, differentiating low flow cardiac output from no-flow cardiac arrest, in an infant with septic emboli.
Case 5: describes the use of POCE in the setting of a new murmur in an unwell adolescent presenting with Transient Ischaemic Attack (TIA). As well as the importance of repeat examination in an evolving clinical picture to clarify the diagnosis of an atrial mass and assist in directing on-going management.
Conclusion: It is knowing when and why to use POCE that ensures it is a tool that is used for the benefit of patient management and outcome. Point of care echocardiography is not a screening tool and must answer a clinical question based on the compilation of the presenting signs and symptoms.
Keywords: Point of Care Echocardiogram, Paediatric Emergency Department, Point of Care Ultrasound.
Introduction
The first recorded use of ultrasound to examine cardiac anatomy was in 1954 by Hertz and Edler(1). From this seminal point in cardiac diagnostics, the field of echocardiography has evolved. There has been significant investment into advancing technology and expanding the scope of utility of echocardiography since then. Paediatric echocardiography has widespread and essential utility in the field of paediatric cardiology. Identification of structural lesions, assessment of cardiac function and diagnosis of congenital heart defects are among the most vital indications for trans-thoracic echocardiography. However, since the 1990s, it has been recognised that there is a role for the point of care echocardiography (POCE). While POCE may not provide the same detail as formal echocardiography, it can be argued that its evolving use in Emergency Medicine (EM) provides certain advantages. The use of POCE by non-cardiac physicians has been explored in detail, and studies have shown that EM physicians have a high degree of both accuracy and inter-observer agreement in the diagnosis of left ventricular function, pericardial effusion, and right ventricular preload(2–4). The facility of point of care ultrasound has been a mainstay of trauma assessment for decades(5–7). It has been found that after a short duration of training EM trainees could reliably perform and interpret focused echocardiography at the bedside(8). In fact, many international training bodies have incorporated bedside ultrasound as a core component of the EM training schemes(9,13).
One of the advantages of POCE is the provision of real time information of haemodynamic physiology(14). This becomes crucial in the Emergency Department (ED) where decision-making is required to be both timely and correct in order to establish the most appropriate lifesaving therapy. In this sense, the access to formal echocardiography is typically limited, whereas POCE provides dynamic immediate data to assist in the clinical decision making process. The traditional clinical determination of subcategories of shock have been somewhat simplified through POCE. The identification of cardiogenic, obstructive, distributive, or hypovolemic shock is made more feasible with ultrasound assessment of the left and right ventricular function and size, inferior vena cava size and the pericardium(15–17).
The use of POCE has been described as “problem orientated, physiology base imaging” thereby providing answers to specific clinical questions. The American Society of Echocardiography has proposed recommendations for the provision of POCE including indications for its use, technical considerations, and imaging views(18). The five conventional imaging views are:
• subcostal view (SC)
• subcostal inferior vena cava (IVC) view
• parasternal long-axis view (PLAX)
• parasternal short-axis view (PSAX)
• apical four-chamber view (A4C)
Rather than an in-depth interpretation of the images, dichotomous or binary questions may be answered readily in an emergency situation. An example of this is the presence or absence of a pericardial effusion, or whether the IVC is collapsed or not. This information then informs the clinical findings and influences management. This case series will discuss several scenarios where the use of POCE directly affected management of critically ill patients.
Methods
This is a descriptive study incorporating a series of 5 case reports.
Cases were selected and submitted via Consultants in Emergency Medicine across 4 emergency departments incorporating 3 paediatric emergency departments and 1 adult emergency department across the island of Ireland. All point of care echocardiograms were completed by senior emergency medicine clinicians.
Prior to submission informed consent was obtained for potentially identifiable cases and, where required, accessed case information independently via retrospective review of their notes and
echocardiogram images. All data was anonymised prior to submission to the authors.
A standardised dataset was extracted from each case submitted including anonymised demographic data, presenting complaint and history thereof, initial management in the paediatric emergency department, use of point of care echocardiogram, interim diagnosis and initial management. Information relating to formal diagnosis and on-ward therapeutic interventions were also gathered, where available. All cases were reviewed by 4 authors (first, second, fifth and senior author). Cases are presented in a narrative descriptive style with accompanying echocardiogram images.
Results: 5-Part Case Series
1. CARDIAC ARREST
An 8-month-old previously well infant boy presented to the ED in cardiac arrest. His mother had found him cold and unresponsive in his cot that morning. She called emergency services and commenced chest compressions.
On arrival, he demonstrated no signs of life and CPR was on-going. At the next pulse check, a clinician trained in POCE used a subcostal view to visualise the heart, in place of palpating the femoral pulses. This reduced the time off the chest and allowed compressions to be recommenced more quickly.
The POCE confirmed the absence of any regular organised contractions. This assisted the team’s decision to stand-down their resuscitation efforts, in accordance with the Advanced Paediatric Life Support (APLS) algorithm. During the debrief, members of the resuscitation team verbalised that being able to see that the heart wasn’t beating, helped them to accept cessation of resuscitative efforts and felt that it removed any doubt about whether the pulse was present or not. The child’s case was referred to the coroner for post-mortem with a provisional diagnosis of Sudden Infant Death Syndrome (SIDS) in the absence of any identifiable risk factors or preceding events.
2. SEPSIS
A 6-year-old boy presented with a 3 day history of fever, lethargy, vomiting, headache and a 12-hour history of a progressive diffuse non-blanching rash. His medical history was unremarkable however he had not received his routine childhood vaccinations. On assessment, he was tachycardic and drowsy. His blood samples showed a lactic acidosis and raised inflammatory markers.
In an effort to guide both sepsis management and fluid resuscitation, POCE was performed which showed a hyper-dynamic left ventricle, a small pericardial effusion, and a collapsing inferior vena cava (IVC).
This supported the clinical requirement for further fluid resuscitation following the initial 20ml/kg bolus. Recurrent monitoring of left ventricular function and IVC collapsibility during inspiration were used as surrogates for the central venous pressure and intravascular volume.
He was transferred to the Paediatric Intensive Care Unit (PICU) within 90 minutes of presenting to ED, following 60ml/kg Hartmann’s solution, IV antibiotics, and a plan for controlled rapid sequence induction of anaesthesia and intubation. His cerebrospinal fluid samples later returned a positive growth of Neisseria meningitidis confirming the diagnosis.
Figure 2: PLAX view showing a hyperdynamic left ventricle. There is a pericardial effusion present.
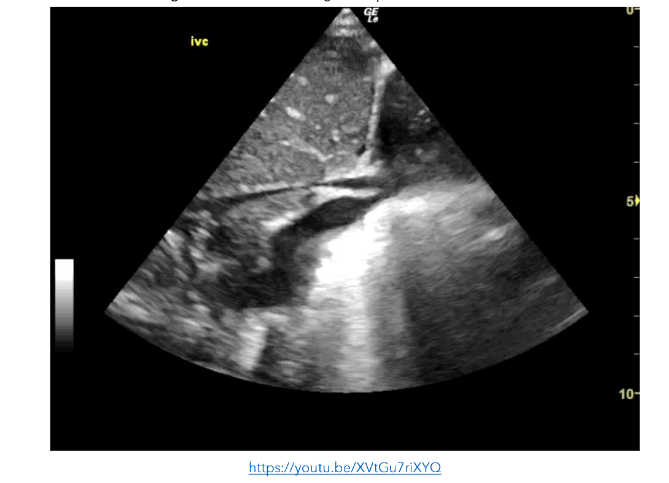
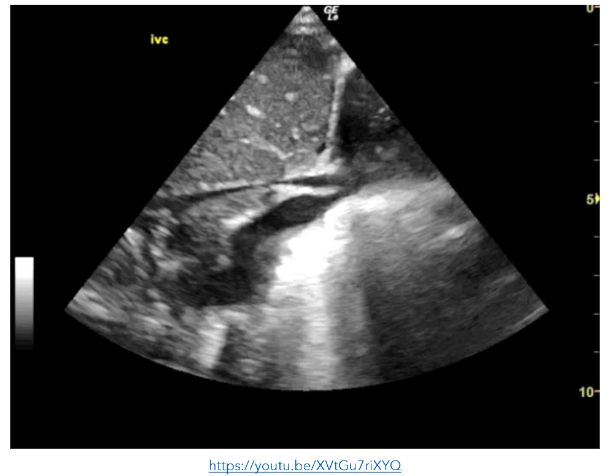
3. SHOCK
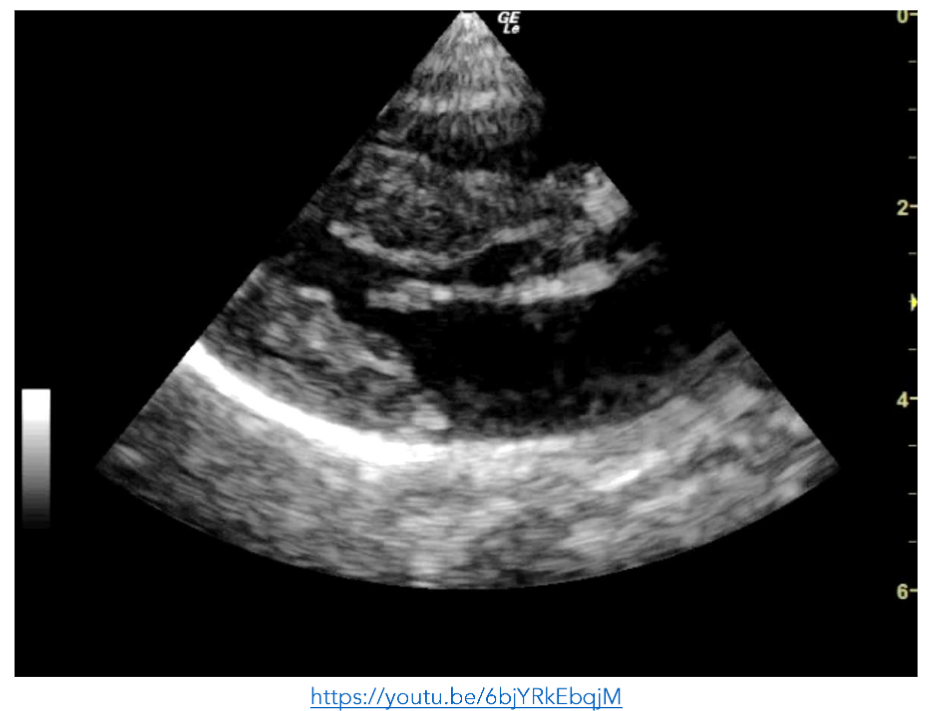
A previously well 9 month-old was brought to the ED by her mother with a recent history of bronchiolitis in the preceding 10 days. Although her initial upper respiratory symptoms had resolved, she now had reduced oral intake and urinary output. On assessment, the infant was tachypnoeic, tachycardic and had bilateral patchy infiltrates on CXR. As the baby was unwell with an undifferentiated shock, POCE was performed. It demonstrated very poor contractility of the left ventricle which contributed to a working diagnosis of myocarditis.
Early use of POCE demonstrated cardiovascular compromise and directed care appropriately to ensure judicious fluid management and avoidance of fluid overload, initiation of continuous cardiac monitoring to promptly alert to and manage arrhythmias, diuresis, and afterload reducing agents, with an option to escalate to inotropic support and mechanical ventilation if needed.
Figure 4: PLAX view showing and enlarged left ventricle with very poor contractility
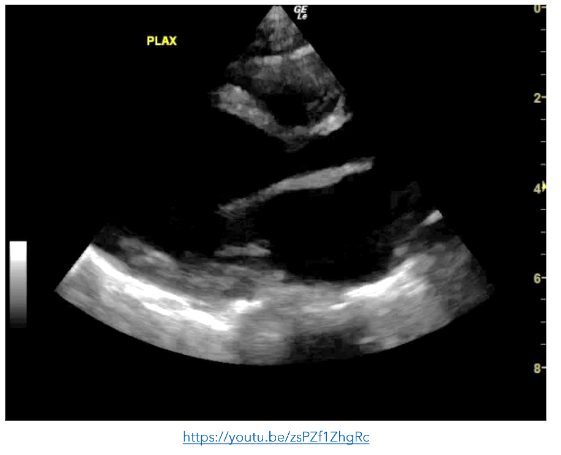
4. POOR CARDIAC OUTPUT
A 9 day-old neonate presented to the ED with his parents who were concerned about progressive lethargy, work of breathing and grunting in the preceding 24 hours. On assessment he was hypotensive, hypoxic and febrile. His neonatal course to date included treatment for Group B-Streptococcus infection following delivery.
On arrival he rapidly deteriorated. His pulse could not be effectively palpated, and cardiac monitoring noted a bradycardia of 50 beats per minute (bpm). As per resuscitative guidelines, chest compressions were commenced. During a rhythm check POCE was ongoing at the time and while contractility was very poor, there were regular organised contractions at a rate of 80 bpm. Compressions were discontinued and the patient was commenced on inotropic support. Another finding from the POCE was the presence of multiple small mobile masses in the right atrium and ventricle, consistent with multiple septic emboli.
Point of care echocardiogram findings helped to guide fluid resuscitation, antibiotics, chronotropic and inotropic support, and rapid sequence induction for hypoxia and cardiogenic shock, including transfer to the paediatric intensive care unit (PICU) within 70 minutes.
Figure 6: PLAX view showing reduced contractility of the left ventricle
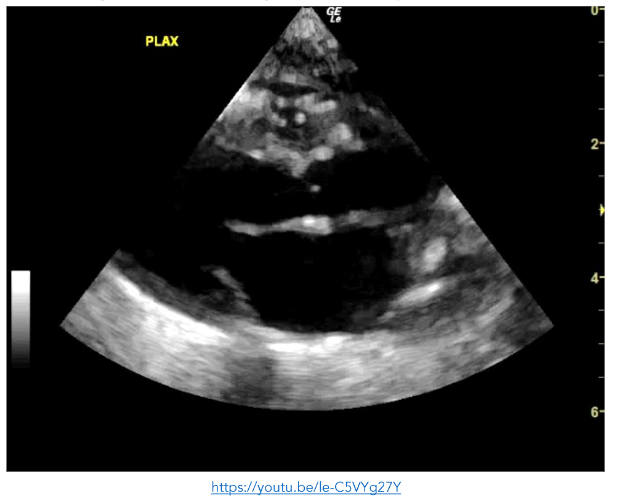
Figure 7: A4C view showing small echogenic masses in the left ventricle, right ventricle and right atrium consistent with septic emboli
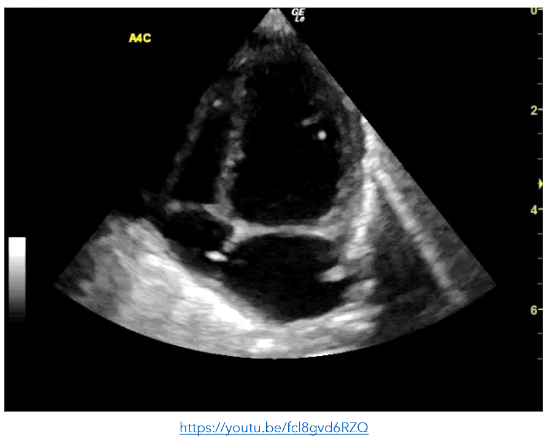
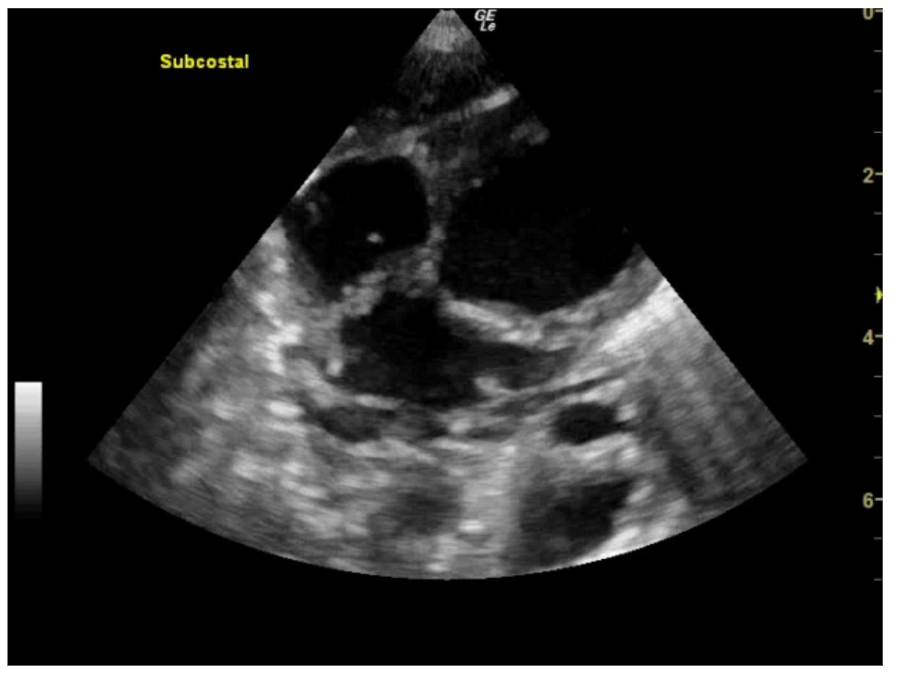
Figure 8: SC view showing small echogenic masses in the left ventricle, right ventricle and right atrium consistent with septic emboli. The right atrium is poorly visualised and cannot be assessed https://youtu.be/awqHN68tV2g
5. HAEMODYNAMIC INSTABILITY WITH NEW CLINICAL FINDINGS
A previously well 15 year-old adolescent was brought to the ED by ambulance. He had been found collapsed in the bathroom by his mother with slurred speech, right sided facial droop and ipsilateral hemiparesis. A working diagnosis of stroke was made, however after resolution of symptoms within one hour and nil acute pathology noted on CT brain, this was revised to a transient ischaemic attack (TIA).
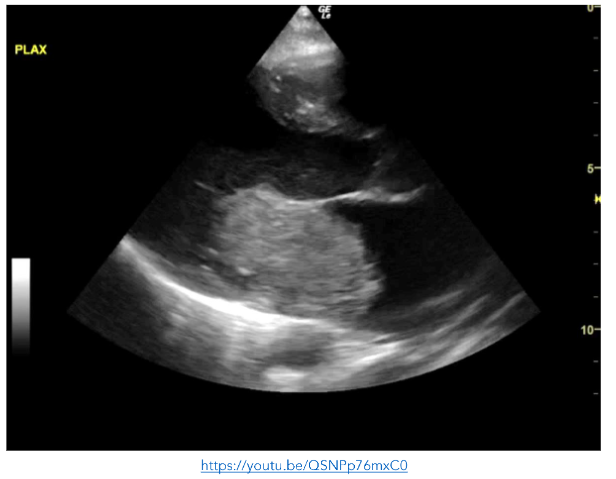
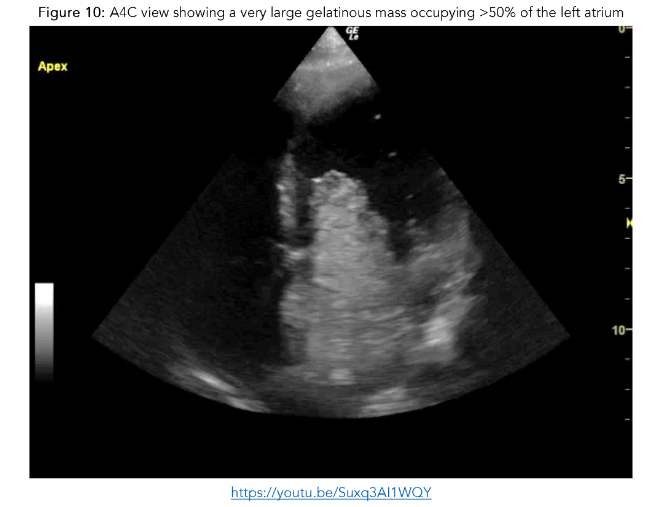
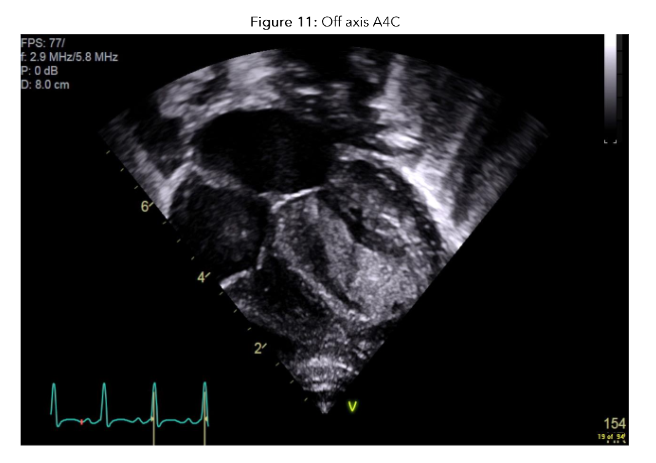
The boy was re-examined by a senior clinician following the initial survey and was noted to have a grade 3/6 ejection systolic murmur. Point of care echocardiogram was performed revealing a very large gelatinous mass occupying >50% of the left atrium.
While in ED he developed a work of breathing and became hypoxic. Point of care echocardiography was repeated and showed a distended and plethoric IVC, with innumerable B-lines anteriorly.
His symptoms rapidly resolved with a stat dose of intravenous furosemide. On transfer to PICU, he became hypotensive secondary to cardiac obstruction. Due to his clinical instability, he proceeded to theatre where he underwent resection of a cardiac myxoma, the mass initially identified on POCE in the ED. Use of POCE rapidly clarified the broader issues in this critically unwell patient with a relatively rare presentation for his age-group, additionally it guided his resuscitation and onward care accurately.
Similarly, a 4-month-old infant was undergoing resuscitation due to his peri-arrest status, POCE demonstrated a likely cardiac rhabdomyoma with three-quarters the left ventricular cavity occupied and extended into the LV outflow tract. Point of care echocardiography quickly clarified the cause of deterioration and directed care appropriately with expedited transfer to the PICU for extracorporeal membrane oxygenation.
Figure 9: PLAX view showing a very large gelatinous mass occupying >50% of the left atrium