Pomalidomide vs. Proteasome Inhibitors in Myeloma Care
Pomalidomide-based vs. Proteasome Inhibitor-based Regimens for Multiple Myeloma: What Is the Optimal Sequence after First Relapse
Issam S. Hamadeh1, Nagham Youssef2, Shebli Atrash2
- Clinical Pharmacy Services, Memorial Sloan Kettering Cancer Center, New York, NY.
- Department of Hematologic Oncology and Blood Disorders, Levine Cancer Institute/Atrium Health, Charlotte, NC.
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Hamadeh, IS., Youssef, N., et al., 2025. Pomalidomide-based vs. Proteasome Inhibitor-based Regimens for Multiple Myeloma: What Is the Optimal Sequence after First Relapse. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7081
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v13i11.7081
ISSN 2375-1924
ABSTRACT
While significant advances have been made over the past two decades, treatment of multiple myeloma remains a challenge owing to the clinical trajectory of the disease. Following each relapse, disease remission periods tend to become progressively shorter as drug-resistant clones continually emerge. Lenalidomide is an immunomodulatory agent that is considered the backbone of frontline therapy for newly diagnosed multiple myeloma. Because of its widespread use and continuous administration until disease progression, lenalidomide refractoriness has become increasingly prevalent in clinical practice. Findings from several landmark clinical trials (such as CANDOR, IKEMA, IKARIA and APOLLO) provided ample evidence that helped establish pomalidomide and carfilzomib-based regimens as the mainstay of treatment in the early relapsed settings. Of note, clinical trials with novel agents such as T-cell engaging bispecific antibodies and antibody-drug conjugates are currently underway. Hence, navigating this crowded space of existing and emerging therapies for the purpose of selecting the optimal regimen to treat early disease relapse, might become increasingly complex over time. The primary objectives of this review article were to provide a succinct overview of the current literature and highlight the substantial heterogeneity in patient- and disease-specific characteristics that existed in the aforementioned trials which evaluated the efficacy of pomalidomide and proteasome-based regimens in relapsed/refractory multiple myeloma. Based on the available evidence, a treatment algorithm was developed which could be utilized as a practical tool to help guide treatment selection based on patient comorbidities, intolerance and refractoriness to prior therapies.
Keywords: relapsed/refractory multiple myeloma, pomalidomide, carfilzomib, daratumumab, isatuximab.
I. Introduction/background
Multiple myeloma (MM) is a hematologic malignancy characterized by uncontrolled proliferation of monoclonal plasma cells predominantly within the bone marrow and occasionally in extramedullary sites. It accounts for approximately 13% of all hematologic cancers and 1% of all malignancies worldwide. The past two decades have witnessed significant advances in the treatment of MM which translated into a substantial improvement in patient outcomes as evidenced by the increase in both progression free survival (PFS) and overall survival. Nevertheless, MM remains an incurable disorder where all patients will inevitably experience disease progression or relapse.
The International Myeloma Working Group (IMWG) has refined the definitions of disease progression and relapse in 2015 to guide clinical decision-making. Generally, prognosis worsens with each successive line of therapy with remission periods becoming progressively shorter secondary to selection or evolution of drug-resistant clones which portend an aggressive disease phenotype. This underscores the importance of effective therapeutic strategies at the time of early relapse.
Lenalidomide, an immunomodulatory agent (IMiD), has long been a backbone of frontline therapy. It is widely used in treating transplant eligible or ineligible patients with newly diagnosed MM either in combination with other agents or as monotherapy during the maintenance phase of treatment following an autologous stem cell transplant. Consequently, a growing number of patients will be deemed lenalidomide-refractory at the time of disease relapse or progression. This has created a clear unmet need for strategies that exclude lenalidomide, as these patients have limited options and worse outcomes.
Proteasome inhibitor (PI)–based combinations, such as daratumumab with carfilzomib or bortezomib, and IMiD-based combinations using pomalidomide with anti-CD38 or anti-SLAMF7 monoclonal antibodies, have demonstrated significant improvements in response depth and progression-free survival. These regimens have become cornerstones in managing early relapsed MM, particularly for lenalidomide-exposed or refractory patients.
Beyond these established backbones, novel therapeutic classes are reshaping the treatment paradigm of relapsed/refractory MM (RRMM). Antibody–drug conjugates such as belantamab mafodotin have shown promising efficacy when combined with proteasome inhibitors or pomalidomide. Furthermore, T-cell–redirecting bispecific antibodies have also demonstrated deep and durable responses in heavily pretreated RRMM, paving the way for their integration into earlier lines of therapy.
Given the wide range of available drug regimens and the heterogeneity in patient characteristics at relapse, an in-depth understanding of comparative efficacy and safety data is critical for optimizing treatment sequencing.
This review aims to outline the key findings from clinical trials which evaluated proteasome inhibitor- and IMiD-based triplet drug combinations in early relapse, describe their efficacy in lenalidomide-refractory patients and discuss emerging data on novel agents such as belantamab mafodotin and T-cell engaging bispecific antibodies. By contextualizing these findings, we seek to provide practical recommendations that would help guide or navigate optimal regimen selection in light of the evolving therapeutic landscape of RRMM.
II. Combinations with anti-CD38 monoclonal antibodies for relapsed/refractory multiple myeloma
A. Proteasome inhibitor-based regimens with daratumumab or isatuximab.
All three approved PIs (bortezomib, carfilzomib and ixazomib) were studied in combination with the anti-CD38 monoclonal antibody, daratumumab. The following section recapitulates the clinical efficacy of each of these combinations.
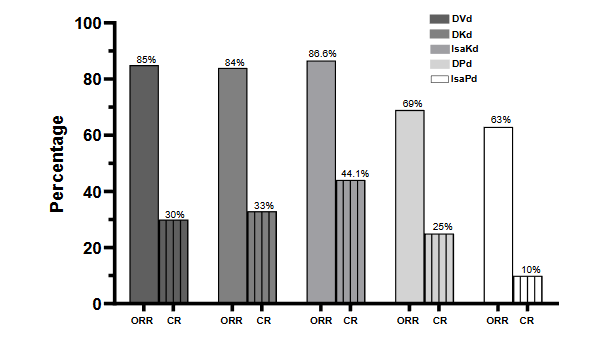
In the CANDOR study, the triplet regimen of daratumumab, carfilzomib and dexamethasone (DKd) was compared to carfilzomib and dexamethasone (Kd) in patients with RRMM. The median number of prior lines of therapy was about 2; high risk cytogenetic abnormalities [defined as deletion 17p, t(4;14), t(14;16), t(14;20), or 1q gain/amplification] were present in 15% of the patients and 20% had ISS (international staging system) stage III at baseline; 32% and 28% of the patients were refractory to lenalidomide and bortezomib, respectively. The triplet regimen was associated with a higher overall response rate (ORR) of 84%; of which 33% were at least complete responses (CRs) per IMWG criteria. Response stratification by prior lines of therapy indicated an ORR of 90% among patients whose disease relapsed after first line therapy; in the subgroup of patients who had 2 to 3 prior lines of therapy, the ORR ranged from 76-84%. DKd significantly extended PFS where the median was 28 months (95% CI: 22.7-NE). Overall, the regimen was well tolerated; upper respiratory tract infections were quite prevalent with an overall incidence (any grade) of 78%; incidence of pneumonia was 23% and neutropenia was 15%. Carfilzomib discontinuation rate was low (5%) and was primarily attributable to infections.
The phase II study by Delimpasi et al set out to discern the efficacy of ixazomib when added to daratumumab and dexamethasone in RRMM where more than half of the patients (59%) experienced disease progression after first-line therapy. About one-third of the patients (34.4%) were refractory to lenalidomide; 24.6% had high risk cytogenetics and 14.8% had stage III disease. With this triplet regimen, a clinical response (per IMWG) was observed in 64.4% of the patients and at least a CR in 13.6%. The median PFS was 16.8 months (range: 10.1-23.7 months) in the entire study population; subgroup analysis based on lenalidomide refractoriness and high-risk cytogenetics showed a median PFS of 8.7 months (95% CI: 2.9–22.1 months) and 12.6 months (95% CI:5.6–NR), respectively. About one-third of the patients (36.1%) developed infections of any grade with pneumonia (of any grade) occurring in 13.1% of the patients.
The CASTOR study enrolled RRMM patients who received at least 1 prior line of therapy to daratumumab, bortezomib and dexamethasone (DVd); 22.7% of the patients had high risk cytogenetics and 24% had stage III disease. About one-fourth of the patient (24%) were refractory to lenalidomide. DVd resulted in an ORR of 85% and at least a CR rate (CRR) of 30%. Fourteen percent of the patients achieved a measurable residual disease (MRD) negative status with the triplet regimen. PFS was 16.7 months. Further analyses of PFS among the subgroup of patients who had 1 previous line of therapy and those with lenalidomide refractory disease revealed a median PFS of 27 months and 7.8 months, respectively. Neutropenia occurred 20% (grade 3-4: 14%) of the patients and pneumonia in 16% (grade 3-4: 10%).
Isatuximab is another FDA approved anti-CD38 monoclonal antibody; its efficacy and safety was also evaluated in combination with carfilzomib and dexamethasone (IKd) in the early relapse setting where the median of prior lines of therapy was 2; high risk cytogenetic abnormalities were present in 23% of the patients and 15% had ISS stage III disease at the time of the study. Almost one third of the patients were refractory to either lenalidomide (32%) or a PI (31%). Eighty seven percent of the patients responded to the triplet with 44% achieving at least a CR. Furthermore, 33.5% of the patients achieved an MRD negative status at 10-5. IKd significantly prolonged PFS where the median was 35.7 months (95% CI: 25.8-44.0). Infections were one of the most common side effects where 29% of the patients developed pneumonia and 36% had upper respiratory tract infections; neutropenia (any grade) occurred in 55% of the patients treated with IKd.
B. Pomalidomide-based regimens with daratumumab or isatuximab.
The APOLLO study investigated the efficacy of daratumumab with pomalidomide and dexamethasone (DPd). The median number of prior lines of therapy was 2 (range: 2-3) with 79% and 47% of the patients having lenalidomide and bortezomib refractory disease, respectively. Stage III disease was present in 22% of the patients and 38% had high risk cytogenetics. DPd yielded a median PFS of 12.4 months (95% CI: 8.3-19.3); 69% of the patients had a response with 25% achieving at least a CR. An MRD negative status was reached in 9% of the patients. The incidence of neutropenia was 72% (grade 3-4: 69%) and pneumonia was 18% (grade 3-4: 12%).
In the ICARIA study, isatuximab was added to the pomalidomide-dexamethasone backbone (IPd) to treat patients with RRMM. The median number of previous therapies was 3 (range: 2-4); almost all the patients were refractory to lenalidomide (94%) and about three-quarters (77%) were refractory to a PI. At study entry, 16% of the patients had high risk cytogenetic abnormalities and 27% had stage III disease. The triplet combination led to a PFS of 11.5 months (95% CI: 8.9-13.9). The ORR and CRR were 63% and 5%, respectively. MRD negativity was observed or noted in 7% of the patients who received IPd. Grade 3 neutropenia occurred in 50% of the patients, pneumonia in 23% and upper respiratory tract infection (grade 3) in 3%.
It is worth noting that a new subcutaneous formulation of isatuximab delivered via an on-body injector device is being investigated with pomalidomide and dexamethasone for RRMM in the IRAKLIA phase III clinical trial. Primary results from this study indicated that subcutaneous IPd was non-inferior (ORR: 71.1%) to intravenous IPd (ORR: 70.5%). Notably, infusion reactions occurred at a significantly lower rate with subcutaneous isatuximab (1.5%) compared to intravenous isatuximab (25%) with a relative risk of 0.061 (95% CI: 0.022-0.164).
Findings from all the landmark clinical trials described herein insinuate that PI-based regimens particularly those that contain carfilzomib might be more effective than those that were pomalidomide-based as typified by the higher ORRs and longer PFS associated with the former. It is essential to emphasize that most of the patients enrolled in the clinical trials with pomalidomide were lenalidomide-refractory and heavily pre-treated (with at least 3 prior lines of therapy). To address this issue, Zayad et al compared pomalidomide plus daratumumab head-to-head with carfilzomib plus daratumumab in a real-world setting where about 80% of the patients (in either arm) were lenalidomide refractory. The median PFS in the pomalidomide arm (14.8 months, 95% CI: 11.47-18.13) was comparable to that observed in the carfilzomib arm (11.7 months, 95% CI: 8.03-16.90) with a hazard ratio of 0.83 (95% CI: 0.62-1.12, p=0.22). Thereby, both regimens are viable options in the RR setting especially among patients who are refractory to lenalidomide.
III. Combinations with anti-SLAMF7 monoclonal antibodies for relapsed/refractory multiple myeloma
In addition to anti-CD38 monoclonal antibodies, elotuzumab which is a monoclonal antibody targeting SLAMF7 was also studied in combination with either a PI or an IMiD in RRMM. Nevertheless, only the IMiD-based combinations with elotuzumab were approved in this setting. The Eloquent-3 trial reported an ORR of 53% (95% CI: 40-66%) and a CRR or better of 8% among patients who received 3 prior lines of therapy; of which 68% were refractory to both lenalidomide and a PI. Elotuzumab with pomalidomide and dexamethasone (EPd) resulted in a PFS of 10.3 months (95% CI: 6.5 to not reached). The overall incidence of neutropenia 36% (grade 3-4: 13%), pneumonia 7% (grade 3-4:5%) and infections 65% (grade 3-4:13%).
In the study by Jakubiak et al, elotuzumab was used with bortezomib and dexamethasone (EVd) to treat patients with RRMM where the majority (about two thirds, 65%) included in the study experienced disease progression after first line therapy. After a follow-up period of 15.9 months, 66% (95% CI: 55-77%) of the patients who were treated the triplet regimen of EVd achieved a response with a median PFS of 9.7 months (95% CI: 7.4-12.2 months). Infections occurred at a frequency of 67%, of which 21% were at least grade 3.
Elotuzumab was also combined with carfilzomib plus dexamethasone (EKd) in patients who received a median of 2 prior lines of therapy (range: 1-3); 47% were refractory to either lenalidomide or bortezomib and 20% were double refractory (i.e. refractory to both bortezomib and lenalidomide). High risk cytogenetic abnormalities were present in about three-fourths of the patients (73% n=11/15). The ORR was 87% and the estimated PFS was 22 months (95% CI: 17.4-27.1 months). Grade 3-4 neutropenia was noted in 40% of the patients and respiratory tract infections occurred in 66% of patients who received EKd.
IV. Novel combinations for relapsed/refractory multiple myeloma.
A. Belantamab mafoditin- based regimens
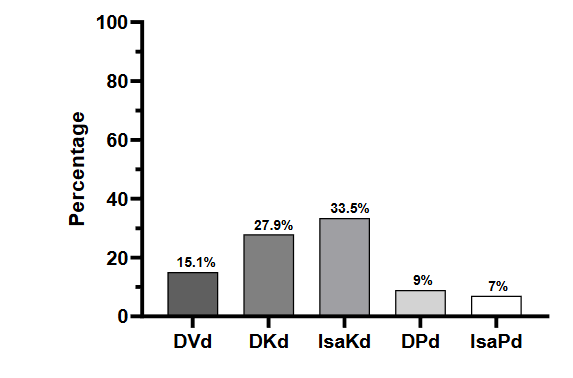
In addition to naked monoclonal antibodies, antibody drug conjugates targeting B-cell maturation antigen (BCMA) have also been evaluated in combination with either a PI (bortezomib) or an IMiD (pomalidomide) in the RR setting. The ongoing phase III clinical trial, DREAMM-7, enrolled adult patients with MM that progressed after at least one prior line of therapy. Patients who met the study inclusion criteria were randomized to receive either the investigational triplet regimen comprising belantamab mafoditin, bortezomib and dexamethasone (BVd) or the standard of care, daratumumab with bortezomib and dexamethasone (DVd). After a follow up period of about 28 months, BVD demonstrated an improvement in clinical efficacy compared to DVd, typified by a higher ORR [83% (95% CI:77-87%) in BVd group vs. 71% (95% CI:65-77%) DVd group] as well as CRR or better (35% in the BVd group as compared with 17% in the DVd group). Additionally, BVD resulted in a significantly longer PFS (median: 36.6 months, 95% CI: 28.4- NR) when compared to DVd (median: 13.4 months, 95% CI:11.1 to 17.5); thereby translating into a hazard ratio of 0.41 for disease progression or death (95% CI: 0.31-0.53; p<0.001).
B. T-cell engaging bispecific antibody-based regimens.
T-cell engaging bispecific antibodies have recently emerged as a promising class of therapeutics that profoundly transformed the treatment paradigm of RRMM. Currently, there are four agents that received regulatory approval; three (teclistamab, elranatamab and linvoseltamab) target the BCMA antigen, whereas talquetamab is the only approved T-cell engaging bispecific antibody that targets the GPRC5D antigen. All these agents showed unprecedented efficacy as monotherapy in heavily pre-treated MM patients. This provided the impetus to evaluate their clinical efficacy in combination with various agents in earlier lines of therapy.
Table 1. Summary of the clinical efficacy of proteasome inhibitor-based and immunomodulatory-based combination regimens in relapsed/refractory multiple myeloma
| Regimen | Number of patients | Prior lines of therapy | Lenalidomide refractoriness | Proteasome inhibitor refractoriness | High risk cytogenetics | Median PFS |
|---|---|---|---|---|---|---|
| DVd | 251 | Median: 2 (range: 1-9) | – 1: 48.6% – 2: 27.9% – 3: 14.7% – ≥4: 8.8% |
24.0% | Not reported | 20.4 months |
| DKd | 312 | – 1: 46.0% – 2-3: 54.0% |
32.0% | 28.0% | 15.0% | 28.6 months (range: 22.7-NE) |
| IKd | 179 | – 1: 44.1% – 2: 35.8% – 3: 18.4% |
31.8% | 31.3% | 23.5% | 35.7 months (range: 25.8-44.0) |
| DPd | 151 | – 1: 11.0% – 2-3: 75.0% – ≥4: 14.0% |
79.0% | 47.0% | 38.0% | 12.4 months (range: 8.3-19.3) |
| IPd | 154 | Median: 3 (range: 2-4) | 94.0% | 77.0% | 16.0% | 11.1 months (range: 7.8-13.8) |
| BVd | 243 | – 1: 51.0% – 2-3: 36.0% – ≥4: 12.0% |
Not reported | Not reported | 28.0% | 36.6 months (range: 28.4-NE) |
| BPd | 155 | – 1: 53.0% – 2-3: 35.0% – ≥4: 12.0% |
81.0% | 26.0% | 34.0% | 12-month PFS: 71.0% (95% CI: 63.0-78.0) |
Table 2. Novel combinations with either an immunomodulatory agent or a proteasome inhibitor
| Clinical trial name | Combination | Median of prior lines of therapy | High risk cytogenetics | Triple class refractoriness | Overall response | Complete response | Measurable residual disease negativity |
|---|---|---|---|---|---|---|---|
| CAMMA 1 | Cevostamab + pomalidomide | 2 (range: 1-6) | – | 70 mg dose: 15.0% 105 mg dose: 36.4% |
– 70 mg: 31.0% – 105 mg: 24.0% |
– 70 mg: 86.2% – 105 mg: 88.0% |
– 70 mg: 44.8% (95% CI: 21.4-71.9) – 105 mg: 38.5% (95% CI: 20.0-64.9) |
| MonumenTAL-2 | Talquetamab + pomalidomide | 3 | – | 0.4 mg: 41.7% 0.8 mg/kg: 33.3% |
0.4 mg/kg: 25.0% 0.8 mg/kg: 21.1% |
0.4 mg/kg: 86.7% 0.8 mg/kg: 83.5% |
0.4 mg/kg: 60.0% 0.8 mg/kg: 44.4% |
| MagnetisMM-20 | Elranatamab + carfilzomib | 2 (range: 1-3) | 33.3% | 41.7% | 100% | Not reported | Not reported |
| Linker-MM2 | Livoseltamab + bortezomib | 3 (range: 2-9) | Not reported | 41.0% | 79.0% | Not reported | Not reported |
| Linker-MM2 | Livoseltamab + carfilzomib | 3 (range: 2-6) | 17.0% | 33.0% | – DL1 (100 mg): 91.0% – DL1b (150 mg): 100% |
Not reported | Not reported |
V. Conclusion
The choice between an IMiD-based and a PI-based regimen poses so many challenges in clinical practice which primarily stem from the everchanging treatment landscape of RRMM; this decision-making process will even become more complicated if belantamab mafoditin-based regimens become approved to treat patients in this setting. Hence, it is imperative that patient specific and disease-related factors be considered to streamline or guide the selection of the optimal regimen such as pre-existing comorbidities, tolerability of prior regimens, last line of therapy at the time of progression as well as type of disease progression (biochemical vs. clinical). As illustrated in Figure 3, the presence of cardiovascular diseases (heart failure, pulmonary hypertension, myocardial infection) favor pomalidomide-based regimens. The clinical efficacy of these regimens hinges on disease sensitivity to anti-CD38 monoclonal antibodies (daratumumab and isatuximab). Although elotuzumab targets a different antigen (SLAMF7), the study by Parrondo et al demonstrated modest activity in patients who were daratumumab refractory. However, an elotuzumab-based regimen can still be utilized as a bridge to chimeric antigen receptor (CAR) T-cell therapy in patients who are triple-class refractory (PI, IMiD and anti-CD38 monoclonal antibodies).
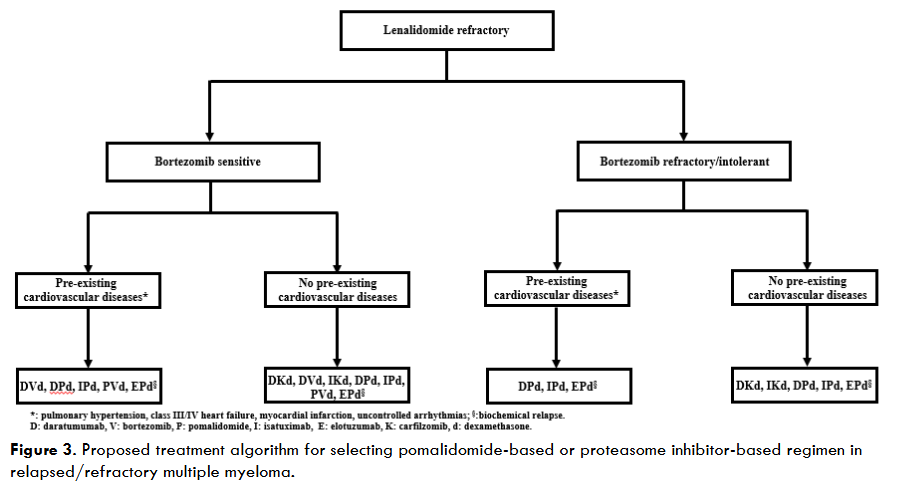
V. Conclusion
The choice between an IMiD-based and a PI-based regimen poses so many challenges in clinical practice which primarily stem from the everchanging treatment landscape of RRMM; this decision-making process will even become more complicated if belantamab mafodotin-based regimens become approved to treat patients in this setting. Hence, it is imperative that patient specific and disease-related factors be considered to streamline or guide the selection of the optimal regimen such as pre-existing comorbidities, tolerability of prior regimens, last line of therapy at the time of progression as well as type of disease progression (biochemical vs. clinical). As illustrated in Figure 3, the presence of cardiovascular diseases (heart failure, pulmonary hypertension, myocardial infection) favor pomalidomide-based regimens. The clinical efficacy of these regimens (listed in Figure 3) hinges on disease sensitivity to anti-CD38 monoclonal antibodies (daratumumab and isatuximab). Although elotuzumab targets a different antigen (SLAMF7), the study by Parrondo et al demonstrated modest activity in patients who were daratumumab refractory. However, an elotuzumab-based regimen can still be utilized as a bridge to chimeric antigen receptor (CAR) T-cell therapy in patients who are triple-class refractory (PI, IMiD and anti-CD38 monoclonal antibodies)³².
Contributions: All authors contributed equally to writing and revising this manuscript.
Conflicts of Interest: The authors have no competing conflicts of interest to declare that are directly or indirectly related to this work.
Funding: No specific grant from any funding agency
References
- Sonneveld P. Management of multiple myeloma in the relapsed/refractory patient. Hematology Am Soc Hematol Educ Program. 2017 Dec 8;2017(1):508-517. doi: 10.1182/asheducation-2017.1.508.
- de Arriba de la Fuente F, Montes Gaisán C, de la Rubia Comos J. How to Manage Patients with Lenalidomide-Refractory Multiple Myeloma. Cancers (Basel). 2022 Dec 27;15(1):155. doi: 10.3390/cancers15010155.
- Dimopoulos MA, Moreau P, Terpos E, et al. Multiple Myeloma: EHA-ESMO Clinical Practice Guidelines for Diagnosis, Treatment and Follow-up. Hemasphere. 2021;5(2):e528. Published 2021 Feb 3. doi:10.1097/HS9.0000000000000528
- Kumar SK, Dispenzieri A, Lacy MQ, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia. 2014;28(5):1122-1128. doi:10.1038/leu.2013.313
- Sonneveld P. Management of multiple myeloma in the relapsed/refractory patient. Hematology Am Soc Hematol Educ Program. 2017;2017(1):508-517. doi:10.1182/asheducation-2017.1.508
- Laubach J, Garderet L, Mahindra A, et al. Management of relapsed multiple myeloma: recommendations of the International Myeloma Working Group. Leukemia. 2016;30(5):1005-1017. doi:10.1038/leu.2015.356
- Anderson KC, Kyle RA, Rajkumar SV, et al. Clinically relevant end points and new drug approvals for myeloma. Leukemia. 2008;22(2):231-239. doi:10.1038/sj.leu.2405016
- Sonneveld P, Broijl A. Treatment of relapsed and refractory multiple myeloma. Haematologica. 2016;101(4):396-406. doi:10.3324/haematol.2015.129189
- Rajkumar SV. Multiple myeloma: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97(8):1086-1107. doi:10.1002/ajh.26590
- Atrash S, Thompson-Leduc P, Tai MH, et al. Treatment patterns and effectiveness of patients with multiple myeloma initiating Daratumumab across different lines of therapy: a real-world chart review study. BMC Cancer. 2021;21(1):1207. Published 2021 Nov 12. doi:10.1186/s12885-021-08881-7
- US Food and Drug Administration. FDA approves daratumumab for multiple myeloma ineligible for autologous stem cell transplant [Internet]. Silver Spring (MD): FDA; 2019 [cited 2024 Jun 14].
- Sonneveld P, Dimopoulos MA, Boccadoro M, et al. Daratumumab, Bortezomib, Lenalidomide, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2024;390(4):301-313. doi:10.1056/NEJMoa2312054
- Spicka I, Lapidot I, Merz M, et al. P944: Real-world patient characteristics and survival outcomes of lenalidomide refractory vs. Lenalidomide exposed rrmm patients in the honeur federated data network. Hemasphere. 2023 Aug 8;7(Suppl): e258126d. doi: 10.1097/01.HS9.0000970680.25812.6d.
- Usmani SZ, Quach H, Mateos MV, et al. Final analysis of carfilzomib, dexamethasone, and daratumumab vs carfilzomib and dexamethasone in the CANDOR study. Blood Advances. 2023;7(14):3739-3748.
- Delimpasi S, Dimopoulos MA, Straub J, et al. Ixazomib plus daratumumab and dexamethasone: Final analysis of a phase 2 study among patients with relapsed/refractory multiple myeloma. American Journal of Hematology. 2024; 99(9):1746–1756.
- Antonio Palumbo A, Chanan-Khan A, Weisel K, et al. Daratumumab, Bortezomib, and Dexamethasone for Multiple Myeloma. The New England Journal of Medicine. 2016;375(8):754-766.
- Mateos MV, Sonneveld P, Hungria V, et al. Daratumumab, bortezomib, and dexamethasone versus bortezomib and dexamethasone in patients with previously treated multiple myeloma: Three-year follow-up of CASTOR. Clinical Lymphoma, Myeloma and Leukemia. 2020;20(8):509-518.
- Martin T, Dimopoulos MA, MikhaelJ, et al. Isatuximab, carfilzomib, and dexamethasone in patients with relapsed multiple myeloma: updated results from IKEMA, a randomized Phase 3 study. Blood Cancer Journal. 2023;13(1):72-79.
- Dimopoulos M, Terpos E, Boccadoro M, et al. Subcutaneous daratumumab plus pomalidomide and dexamethasone versus pomalidomide and dexamethasone in patients with relapsed or refractory multiple myeloma (APOLLO): extended follow up of an open-label, randomised, multicentre, phase 3 trial. The Lancet Haematology. 2023;10(10):813-824.
- Attal M, Richardson PG, Rajkumar SV, et al. Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): a randomised, multicentre, open-label, phase 3 study. The Lancet. 2019; 394(10214):2096-2107.
- Ailawadhi S, Spicka I, Spencer A, et al. Isatuximab subcutaneous by on-body injector versus isatuximab intravenous plus pomalidomide and dexamethasone in relapsed/refractory multiple myeloma: Phase III IRAKLIA study. Journal of Clinical Oncology. 2025;43(22):2527-2537.
- Zayad A, Magee G, Mansour R, et al. Efficacy and safety of daratumumab, pomalidomide and dexamethasone compared to daratumumab, carfilzomib and dexamethasone in relapsed multiple myeloma: Multicenter real-world experience. Clinical Lymphoma, Myeloma and Leukemia. 2025.doi: 10.1016/j.clml.2025.08.017. Online ahead of print.
- Dimopoulos MA, Dytfeld D, Grosicki S, et al. Elotuzumab plus Pomalidomide and Dexamethasone for Multiple Myeloma. The New England Journal of Medicine. 2018; 379 (19):1811-1822.
- Jakubowiak A, Offidani M, Pégourie B, et al. Randomized phase 2 study: elotuzumab plus bortezomib/dexamethasone vs bortezomib/dexamethasone for relapsed/refractory MM. Blood. 2016;127 (23): 2833–2840.
- Silvennoinen RH, Nahi H, Anttila P, et al. Carfilzomib, elotuzumab and dexamethasone for relapsed or refractory myeloma patients. Blood. 2020; 136 (Suppl. 1): 20.
- Hungria V, Pawel Robak P, Hus M, et al. Belantamab mafodotin, bortezomib, and dexamethasone for multiple myeloma. The New England Journal of Medicine. 2024;391(5):393-407.
- Dimopoulos MA, Beksac M, Pour L, et al. Belantamab mafodotin, pomalidomide, and dexamethasone in multiple myeloma. The New England Journal of Medicine. 2024;391(5):408-421.
- Moreau P, Garfall AL, van de Donk N, et al. Teclistamab in relapsed or refractory multiple myeloma. The New England Journal of Medicine. 2022;387(6):495-505.
- Lesokhin AM, Tomasson MH, Arnulf B, et al. Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results. Nature Medicine. 2023;29 (9): 2259–2267.
- Bumma N, Richter J, Jagannath S, et al. Linvoseltamab for treatment of relapsed/refractory multiple myeloma. Journal of Clinical Oncology. 2024;42(22):2702-2712.
- Chari A, Minnema MC, Berdeja JG, et al. Talquetamab, a T-cell–redirecting GPRC5D bispecific antibody for multiple myeloma. The New England Journal of Medicine. 2022;387(24):2232-2244.
- Parrondo RD, LaPlant BR, Elliott J, et al. Phase II trial of elotuzumab with pomalidomide and dexamethasone for daratumumab-refractory multiple myeloma. Blood Cancer Journal.2024;14 (1):152. doi: 10.1038/s41408-024-01134-3.
- Mian HS, Riley CH, Popat R, et al. Cevostamab plus pomalidomide (pom) and dexamethasone (dex) in relapsed/refractory multiple myeloma (RRMM): Phase I dose expansion results from the CAMMA 1 study. Presented at: 22nd Annual International Myeloma Society Meeting and Exposition; September 17-20, 2025; Toronto, Canada. Abstract OA-14.
- Matous J, Biran N, Perrot A, et al. Talquetamab + pomalidomide in patients with relapsed/refractory multiple myeloma: Safety and preliminary efficacy results from the phase 1b MonumenTAL-2 Study. Blood. 2023; 142 (Suppl.1): 1014.
- Tomasson MH, Gabayan E, Ali SA, et al. Efficacy of elranatamab (ELRA) in combination with carfilzomib (CFZ) and dexamethasone (DEX) in the phase 1b MagnetisMM-20 Trial in relapsed or refractory multiple myeloma (RRMM). Blood. 2024;144 (Suppl. 1):1024.
- Rodríguez-Otero P, Delimpasi S, Oriol A, et al. Linvoseltamab (LINVO) + bortezomib (BTZ) in patients (pts) with relapsed/refractory multiple myeloma (RRMM): First results from the LINKER-MM2 trial. Journal of Clinical Oncology. 2025;43 (Suppl.16):7510.
- Manier S, Ocio EM, et al. Linvoseltamab (LINVO) + carfilzomib (CFZ) in patients (pts) with relapsed/refractory multiple myeloma (RRMM): Initial results from the LINKER-MM2 trial. Journal of Clinical Oncology. 2025;43 (Suppl 16);7513.

