Potent Antimicrobial Device for Diabetic Foot Treatment
Potent Antimicrobial properties of a Biodegradable Medical Device in the treatment of Diabetic Foot
Jorge Cueto García, MD 1; Gerardo Aparicio Ozores 2; Mario Adán Moreno Eutimio, PhD 3; José Moreno Rodríguez PhD 4
- American College of Surgeons Department of Surgery, American British Cowdray Hospital of Mexico & Research Department, Anahuac University of Mexico
- Chief of Department of Microbiology in National School of Biological Sciences, National Polytechnic Institute of Mexico.
- PhD Chemistry, Faculty of Chemistry, National Autonomous University of Mexico.
- National System of Research.
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Cueto García, J., Aparicio Ozores, G., et al., 2025. Potent Antimicrobial properties of a Biodegradable Medical Device in the treatment of Diabetic Foot. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7020
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i11.7020
ISSN 2375-1924
Abstract
There is an urgent need for effective antimicrobial agents to treat infected diabetic foot ulcers (DFUs), which are associated with debilitating symptoms, high rates of amputation, long-term disability, elevated mortality, and significant healthcare costs. This need is even more critical in the era of increasing antimicrobial resistance (AMR). Recent IWGDF/IDSA international guidelines recommend “avoiding antibiotics unless there is confirmed bacterial infection and demonstrated susceptibility to the prescribed agents.”
Keywords
Diabetic Foot, Antimicrobial Resistance, CPZO, Wound Healing, Biodegradable Medical Device
Introduction:
According to the U.S. Centers for Disease Control and Prevention (CDC), over 12% of U.S. adults are affected by type 2 diabetes (T2DM), and more than one-third of adults are obese or prediabetic. Treatment outcomes for diabetic foot (DF) are frequently poor due to neuropathy, impaired healing, immune dysfunction, vascular insufficiency, and renal complications. In low- and middle-income countries, delays in accessing specialized care further compromise multidisciplinary management.
Methods:
Over the past several years, the antimicrobial efficacy of a non-antibiotic, non-toxic topical medical device — CPZO (Carbohydrate Polymer with Zinc Oxide, Pebisut®) — has been demonstrated through in vitro and in vivo preclinical studies in rodent models at the Experimental Laboratory of Anahuac University. Clinical trials in adult patients with chronic venous ulcers at Juarez Hospital of Mexico have also shown promising outcomes. The formulation demonstrated potent antimicrobial and anti-inflammatory effects, even against multidrug-resistant bacterial strains.
Further microbiological evaluation was performed at the National School of Biological Sciences (ENCB-IPN), assessing CPZO’s bactericidal activity against three biofilm-forming bacteria: methicillin-resistant Staphylococcus aureus (MRSA), Escherichia coli, and Pseudomonas aeruginosa, using bacterial suspension, biofilm disruption, and an in vitro wound biofilm model. Separate studies at Juarez Hospital also evaluated the product’s efficacy against MRSA and P. aeruginosa.
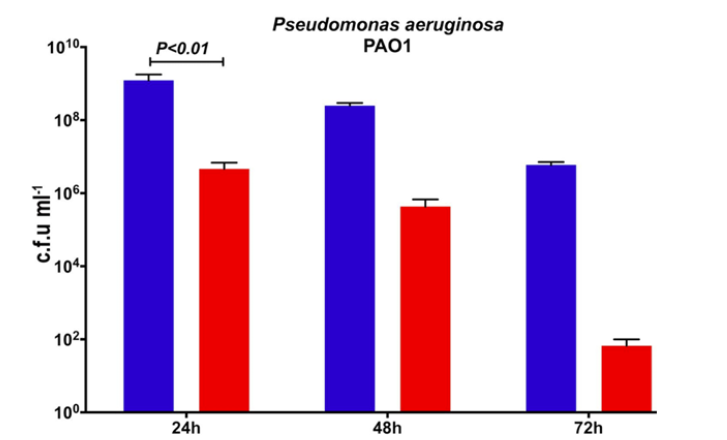
Results:
In rodent studies, both acute and chronic models showed that CPZO was well tolerated and fully resorbed within 6 days. In clinical trials involving patients with venous ulcers, diabetes, obesity, and hypertension, no adverse effects were observed. Within 3–5 days of topical CPZO application, patients reported resolution of pain, burning, exudate, and insomnia.
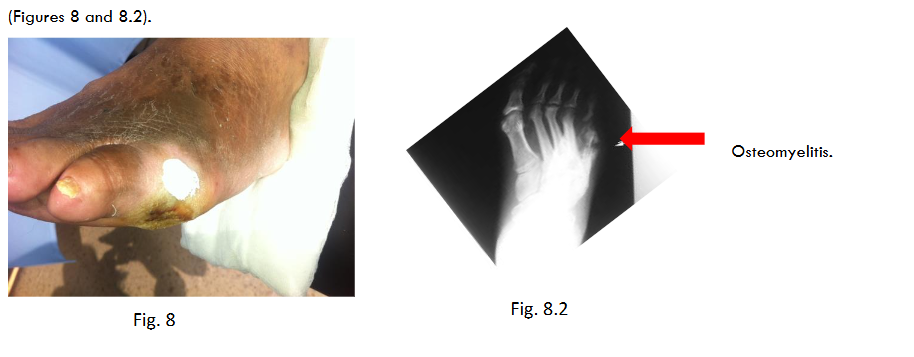
Subsequently, 102 patients with advanced DFUs were treated across four hospitals, including emergency admissions. Most patients showed significant improvement or complete healing. Surgical intervention was required in a limited number of cases: two above-the-knee, four below-the-knee, four transmetatarsal amputations, and 25 toe excisions. Importantly, no patient mortality was recorded.
Discussion:
CPZO is environmentally friendly, requires no refrigeration, and has shown no evidence of toxicity or inflammation in animal studies. Histological analysis revealed no signs of acute or chronic inflammation or infection. In clinical trials, no adverse effects were noted, and CPZO significantly accelerated healing in chronic venous ulcers compared to control groups.
Since its approval for clinical use over six years ago, CPZO has been adopted by public and private healthcare institutions in Mexico, including the ISSSTE, with no reports of toxicity, allergy, or adverse reactions. The morbidity and mortality associated with diabetes and DFUs continue to rise, possibly due to increased life expectancy and childhood obesity. Global obesity rates have more than doubled in adults and quadrupled in adolescents since 1990.
Extensive research by Moreno et al. confirmed CPZO’s anti-inflammatory activity in rodent models, including reduced cytokine release and histological evidence of healing. These findings correlate with the rapid clinical improvement observed in patients, typically within 3–4 days of application.
Although other studies have shown that maltodextrins and zinc possess antimicrobial effects, the most comprehensive evidence comes from the work of Bello-López et al. and Aparicio et al., demonstrating CPZO’s potent bactericidal action against MRSA and P. aeruginosa in both experimental models and clinical settings, without toxicity.
These results are especially relevant given the global threat of AMR. According to the WHO, antimicrobial resistance contributed to 4.95 million deaths in 2019. The 2023 IWGDF/IDSA guidelines emphasized prudent antibiotic use, reinforcing the importance of alternative therapies such as CPZO.
Conclusion
CPZO demonstrates potent bactericidal and anti-inflammatory properties without toxicity, offering a promising first-line treatment for DFUs, venous ulcers, and other complex chronic wounds. Its ability to prevent and control infections while accelerating healing makes it a valuable asset in managing difficult-to-treat lesions.
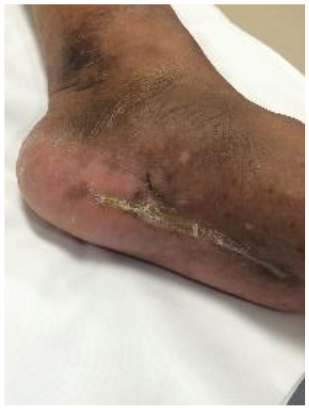
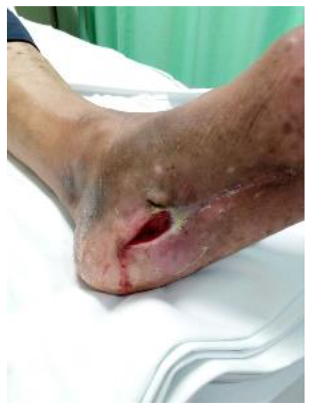
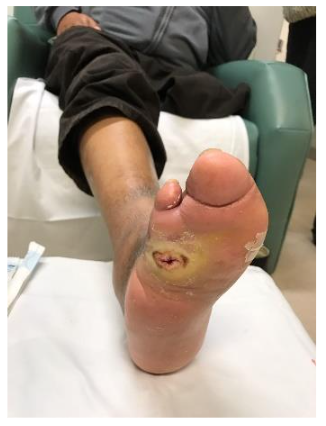
Case Report (Juarez Hospital).
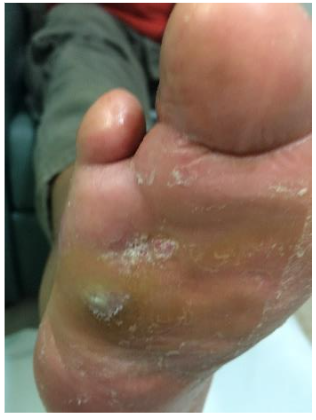
M, 58 y.o. with T2DM, cellulitis, and a fistula extending from the dorsum to the sole of the foot. After 8 months of unsuccessful treatment, the patient was scheduled for infra-condylar amputation.
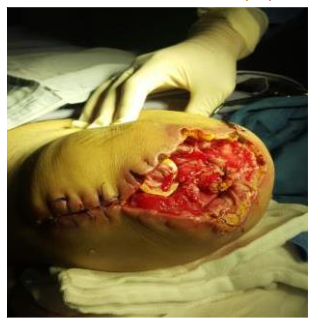
Our Group received an emergency call requesting immediate delivery of 12 jars of CPZO to be sent to a mountain clinic in Jalisco area.
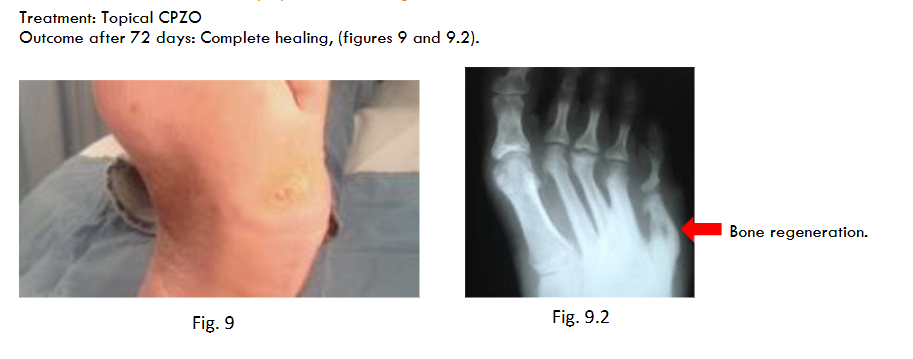
Treatment:
Topical CPZO
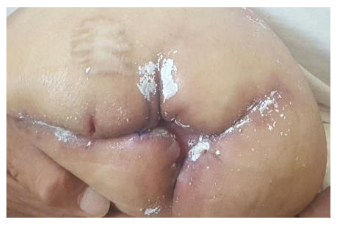
Outcome after 72 days:
Complete healing.
Conflict of Interest:
Author J. Cueto holds patents related to the external application of CPZO described in this study. The remaining authors declare no conflicts of interest.
Patents: USPTO #8,252,333 and #9,808,484; Canadian #2,661,686 and #2,926,115; European #2,062,602; Mexican #280,754 and #350080.
References:
- Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of diabetes and its burden in the United States. Published November 29, 2023. Accessed June 17, 2025. https://www.cdc.gov/diabetes/data/statisticsreport/index.html
- Swaminathan N, Awuah WA, Bharadwaj HR, et al. Early intervention and care for diabetic foot ulcers in low and middle income countries: addressing challenges and exploring future strategies—a narrative review. Health Sci Rep. 2024;7(5):e2075. doi:10.1002/hsr2.2075
- Cueto J, Barrientos T, Rodríguez E, Del Moral P. A new biodegradable adhesive for protection of intestinal anastomoses: preliminary communication. Arch Med Res. 2011;42(6):475-481. doi:10.1016/j.arcmed.2011.09.004
- Cueto J, Moreno M, Bahena Z, et al. Tratamiento de las úlceras venosas varicosas complicadas y refractarias con polímero de maltodextrina y óxido de zinc: reporte inicial. Rev Mex Angiol. 2015;43(3):102-108.
- Moreno MA, Espinosa-Monroy L, Torres Y, et al. Potent anti-inflammatory activity of carbohydrate polymer with zinc oxide. Biomed Res Int. 2014;2014:712312. doi:10.1155/2014/712312
- Moreno-Eutimio MA, Espinosa-Monroy L, Orozco-Amaro T, et al. Enhanced healing and anti-inflammatory effects of a carbohydrate polymer with zinc oxide in patients with chronic venous leg ulcers: preliminary results. Arch Med Sci. 2018;14(2):336-344. doi:10.5114/aoms.2016.59851
- Bello-López JM, López-Ornelas A, Vilchis-Rangel RE, et al. In vitro bactericidal activity of a carbohydrate polymer with zinc oxide for the treatment of chronic wounds. J Med Microbiol. 2020;69(5):874-880. doi:10.1099/jmm.0.001204
- Cueto J, Ochoa R, Bert E, Tron S, Moreno MA. Chronic venous ulceration in obese patients with diabetes mellitus: a therapeutic challenge. J Diabetol. 2019;3(1):1-6.
- Cueto J, Ochoa R, Bert E. Multi-traumatized patient with a dehiscent fistulized abdomen and a central venous line-associated infection successfully treated with a new medical device II. Ann Clin Med Case Rep. 2022;9(1):1-3.
- Suárez D, Franco D, Rey R, Mena T, Cueto J. Successful healing of two very large pelvic-perineal chronic fistulized ulcers in a patient with congenital myelomeningocele treated with a carbohydrate polymer with zinc oxide: case report. J Dermatol Res Rev Rep. 2023;4(2):1-3. doi:10.47363/JDMRS/2023(4)146
- World Bank. Obesity prevalence, adults (% of adults ages 18+). Gender Data Portal. Accessed June 17, 2025. https://genderdata.worldbank.org/en/indicator/shsta-ob18-zs
- National Institute of Diabetes and Digestive and Kidney Diseases. Overweight & obesity statistics. U.S. Department of Health and Human Services. Accessed June 17, 2025. https://www.niddk.nih.gov/health-information/health-statistics/overweight-obesity
- Jones N, Ray B, Ranjit KT, Manna AC. Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of microorganisms. FEMS Microbiol Lett. 2008;279(1):71-76. doi:10.1111/j.1574-6968.2007.01056.x
- Korresteijn J, Driegsman D, Assendelft W, et al. Patient education for preventing diabetic foot ulceration. Cochrane Database Syst Rev. 2010;12:CD001488. doi:10.1002/14651858.CD001488.pub2
- Infectious Diseases Society of America. Diabetic foot infections. Published October 2, 2023. Accessed June 17, 2025. https://www.idsociety.org/practice-guideline/diabetic-foot-infections/
- World Health Organization. Antimicrobial resistance. Published November 17, 2023. Accessed June 17, 2025. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance

