Precision Thyroid Hormone Therapy for Hypothyroidism
Precision Thyroid Hormone Therapy: Integrating Physiology, Symptoms, and Clinical Context
Angela D Mazza1
- Metabolic Center for Wellness
OPEN ACCESS
PUBLISHED: 31 March 2026
CITATION: Mazza, AD., 2026. Precision Thyroid Hormone Therapy: Integrating Physiology, Symptoms, and Clinical Context. Medical Research Archives, [online] 14(3).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ISSN 2375-1924
ABSTRACT
Thyroid hormone replacement remains the cornerstone of hypothyroidism management, yet a substantial subset of patients continue to experience persistent symptoms despite achieving biochemical euthyroidism with standard levothyroxine therapy. This clinical reality highlights the need for a more individualized approach to thyroid hormone replacement that accounts for interindividual variability in thyroid hormone metabolism, tissue sensitivity, and systemic physiologic context. This review proposes a clinician-focused framework for personalized thyroid hormone replacement, integrating current evidence with real-world clinical experience. This review will examine the physiologic determinants of variability in thyroid hormone action, including deiodinase activity, hormone transport, inflammatory and metabolic influences, and the impact of age, sex hormones, and comorbid conditions. Distinct clinical phenotypes of hypothyroid patients are described to guide patient stratification and therapeutic decision-making. Therapeutic strategies are reviewed, including levothyroxine monotherapy, combination T4/T3 therapy, and alternative formulations, with emphasis on patient selection, dosing principles, and safety considerations. A practical clinical algorithm is presented to support individualized treatment adjustments based on both biochemical markers and symptom patterns. In addition, the importance of integrating lifestyle and systemic factors—such as metabolic health, inflammation, nutrient status, and stress physiology—into comprehensive thyroid care will be discussed. Case-based examples illustrate the application of a personalized approach in common clinical scenarios. By moving beyond a one-size-fits-all model and embracing a precision framework, clinicians can better tailor thyroid hormone replacement to optimize patient outcomes. Future research priorities include phenotype-driven clinical trials and the development of biomarkers that more accurately reflect tissue-level thyroid hormone activity.
Keywords
- Thyroid hormone therapy
- Hypothyroidism
- Levothyroxine
- Personalized medicine
- Clinical phenotypes
1. Introduction
Hypothyroidism remains one of the most common endocrine disorders worldwide, and levothyroxine (LT4) monotherapy continues to serve as the standard first-line treatment. For the majority of patients, LT4 effectively restores serum thyroid-stimulating hormone (TSH) concentrations to within the reference range. Yet in routine clinical practice, normalization of TSH does not invariably translate into restoration of well-being. A meaningful subset of patients report persistent symptoms—including fatigue, cognitive slowing, mood disturbance, weight changes, and reduced quality of life—despite biochemical euthyroidism. Patient-reported outcome studies suggest that dissatisfaction with therapy remains clinically relevant and may influence long-term adherence and trust in care. This discordance has prompted renewed examination of thyroid hormone physiology beyond circulating TSH values. Thyroid hormone action is regulated at multiple levels, including cellular transport, intracellular deiodinase activity, and receptor-mediated transcriptional signaling. Local conversion of thyroxine (T4) to triiodothyronine (T3) by deiodinase enzymes determines tissue-specific T3 availability, which may not always parallel serum measurements. Polymorphisms in deiodinase genes and thyroid hormone transporters have been associated with altered intracellular T3 generation and variable symptom response in some patients receiving LT4 monotherapy. These findings support the concept that thyroid hormone replacement requirements may differ across individuals due to biologic variability in hormone activation and tissue responsiveness.
In addition to intrinsic enzymatic differences, clinical context matters. Aging, menopausal transition, chronic inflammatory states, metabolic dysfunction, and coexisting autoimmune disease can influence thyroid hormone requirements and symptom expression. Such factors may complicate interpretation of laboratory values and contribute to persistent symptoms even when TSH is within range. Recognition of these modifying influences underscores the limitation of a purely laboratory-driven approach.
Within the broader movement toward precision medicine, there is increasing recognition that hypothyroidism management may benefit from phenotype-driven personalization. Although LT4 remains appropriate for many patients, selected individuals with persistent symptoms may warrant reassessment of dosing strategies, formulation, or—after careful evaluation—consideration of combination T4/T3 therapy. Importantly, the presence of ongoing symptoms should prompt thoughtful clinical evaluation rather than reflexive reassurance.
This review focuses specifically on personalized thyroid hormone replacement in patients with persistent or discordant symptoms despite standard therapy. We propose a clinician-oriented framework that integrates physiologic variability, recognition of reproducible clinical phenotypes, and practical therapeutic strategies. The goal is not to supplant established guidelines, but to extend them by incorporating tissue-level biology and patient-centered outcomes into decision-making.
2. Physiologic Foundations of Individual Variability
2.1 THYROID HORMONE METABOLISM AND TISSUE-LEVEL REGULATION
T4→T3 Conversion Thyroxine (T4) serves predominantly as a prohormone that requires intracellular conversion to T3, the active ligand for nuclear thyroid hormone receptors. This conversion is catalyzed by the iodothyronine deiodinases: type 1 (D1) and type 2 (D2), which generate T3, and type 3 (D3), which inactivates T4 and T3. Importantly, deiodinase activity is tissue-specific and dynamically regulated, allowing individual organs to fine-tune intracellular T3 concentrations independently of circulating hormone levels. D2, expressed prominently in the brain, pituitary, brown adipose tissue, and skeletal muscle, plays a particularly critical role in maintaining local T3 availability. In contrast, D1 contributes more substantially to circulating T3 levels through hepatic and renal conversion. D3 functions as a counter-regulatory enzyme, reducing thyroid hormone signaling under conditions such as critical illness or inflammatory stress. The coordinated balance of these enzymes determines tissue-specific thyroid hormone exposure and may create physiologic discordance between serum markers and intracellular thyroid hormone signaling.
Emerging evidence suggests that serum TSH normalization does not necessarily ensure restoration of euthyroidism at the tissue level in all individuals. Experimental models demonstrate that different tissues achieve euthyroidism at distinct circulating T4 concentrations, reflecting variability in local deiodinase expression and sensitivity to thyroid hormone signaling. These findings provide a biologic explanation for why some patients report persistent symptoms despite biochemically adequate replacement.
Physiologic and pathologic states—including systemic inflammation, caloric restriction, chronic illness, and glucocorticoid exposure—can alter deiodinase expression and intracellular T3 generation. Such adaptive changes may initially serve protective roles but could also contribute to reduced tissue T3 availability in selected patients receiving LT4 monotherapy. Recognition of this complexity is central to understanding heterogeneity in treatment response.
Deiodinase Polymorphisms
Genetic variation introduces additional heterogeneity in thyroid hormone activation. The Thr92Ala polymorphism in the DIO2 gene has been among the most studied variants, with some investigations suggesting associations with altered intracellular T3 signaling, differences in neurocognitive outcomes, and variable response to LT4 monotherapy. While the magnitude and clinical reproducibility of these findings remain under active investigation, translational studies indicate that altered D2 localization and function may affect tissue-specific thyroid hormone action. Not all carriers of DIO2 polymorphisms exhibit clinically meaningful differences, and routine genetic screening is not currently recommended. Nevertheless, these observations reinforce the broader principle that interindividual biologic variability may influence treatment experience. In selected patients with persistent symptoms despite adequate biochemical replacement, such variability may partially explain discordant clinical responses.
Cellular Transporters
Thyroid hormone entry into cells is an active, transporter-mediated process. Monocarboxylate transporters (MCT8 and MCT10) and organic anion transporting polypeptides (OATPs) facilitate cellular uptake of T4 and T3. Pathogenic mutations in MCT8, as observed in Allan–Herndon–Dudley syndrome, illustrate the profound physiologic consequences of impaired intracellular transport, underscoring the importance of these mechanisms. Even in the absence of overt transporter mutations, variability in transporter expression and function may influence tissue responsiveness to circulating hormone levels. This layer of regulation further supports the concept that serum measurements provide only a partial representation of intracellular thyroid hormone action.
Tissue-Specific Thyroid Hormone Signaling
Thyroid hormone action is ultimately mediated by nuclear thyroid hormone receptors, primarily TRα and TRβ isoforms, which exhibit tissue-specific distribution and differential gene regulatory effects. TRα predominates in cardiac and skeletal muscle, whereas TRβ is more prominent in liver and hypothalamic-pituitary tissues. This receptor heterogeneity contributes to organ-specific physiologic responses and may partly explain why cardiovascular, metabolic, and neurocognitive symptoms can diverge among patients with similar laboratory values. Beyond receptor expression, co-regulatory proteins and epigenetic modifications influence transcriptional responsiveness to T3. These modulatory mechanisms further contribute to variability in clinical presentation and underscore the limitation of relying solely on circulating hormone measurements to define therapeutic adequacy. Recognition of tissue-level regulation provides a mechanistic foundation for phenotype-driven personalization of thyroid hormone replacement.
2.2 FACTORS INFLUENCING THYROID HORMONE REQUIREMENTS
Age and Sex Hormones
Aging is associated with shifts in thyroid axis regulation, altered TSH reference ranges, and changes in tissue sensitivity to thyroid hormone. Sex steroids also interact closely with thyroid physiology. Estrogen influences thyroid-binding globulin concentrations and peripheral hormone dynamics, while androgen status modulates metabolic responses. Recent reviews emphasize that transitional stages such as perimenopause and menopause can alter thyroid hormone requirements and symptom expression, supporting the need for periodic reassessment of therapy.
Inflammation and Chronic Illness
Inflammation and chronic systemic disease disrupt thyroid homeostasis through cytokine-mediated effects on deiodinase activity, hypothalamic–pituitary regulation, and peripheral metabolism. Contemporary models of nonthyroidal illness highlight complex adaptive changes in thyroid hormone signaling during chronic disease states. These mechanisms may contribute to persistent symptoms in patients with coexisting inflammatory or metabolic disorders.
Stress and the Hypothalamic–Pituitary–Adrenal Axis
Activation of the hypothalamic–pituitary–adrenal (HPA) axis during chronic stress influences TSH secretion and peripheral thyroid hormone metabolism. Bidirectional interactions between glucocorticoids and thyroid signaling may modify tissue responsiveness and symptom burden. Emerging research in neuroendocrine integration underscores the importance of considering stress physiology in thyroid management.
Nutrient Status
Micronutrients such as selenium, iron, zinc, and iodine are essential for thyroid hormone synthesis and deiodinase function. Updated reviews highlight the role of nutrient status in modulating thyroid hormone metabolism and autoimmune thyroid disease risk. Subclinical deficiencies may contribute to variability in therapeutic response and represent modifiable targets in personalized care.
Gut–Liver Axis and Medication Interactions
The gut–liver axis influences levothyroxine absorption, enterohepatic circulation, and peripheral hormone metabolism. Recent literature emphasizes the impact of gastrointestinal disorders, microbiome alterations, and polypharmacy on thyroid hormone bioavailability. Medications such as proton pump inhibitors, calcium and iron supplements, and bile acid sequestrants can significantly interfere with absorption. Recognition of these factors is essential for optimizing individualized replacement strategies.
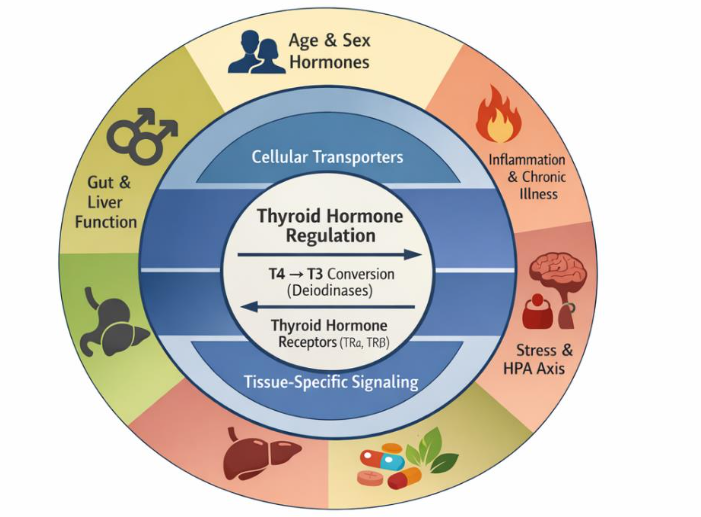
Thyroid hormone signaling is regulated through multiple interconnected layers that extend beyond circulating serum concentrations. At the core, intracellular conversion of thyroxine (T4) to triiodothyronine (T3) by deiodinase enzymes and receptor-mediated transcriptional activity determine tissue-level hormone effects. Cellular transporters and tissue-specific regulatory mechanisms further modulate intracellular hormone availability. Surrounding systemic influences—including age and sex hormones, inflammation and chronic illness, stress and hypothalamic–pituitary–adrenal (HPA) axis activity, nutrient status, and gut–liver interactions—shape thyroid hormone metabolism and clinical responsiveness. Together, these multilevel factors contribute to interindividual variability and provide a biologic rationale for personalized thyroid hormone replacement strategies.
3. Clinical Phenotypes of Hypothyroid Patients
Biochemical indices alone incompletely capture the heterogeneity of hypothyroid presentations encountered in clinical practice. Patients with similar laboratory values may exhibit markedly different symptom profiles, metabolic responses, and therapeutic needs. Recognition of recurring clinical phenotypes can assist clinicians in tailoring thyroid hormone replacement strategies beyond a uniform laboratory-driven approach. The following phenotypes represent common patterns observed in clinical practice and are supported by emerging physiologic and outcomes-based literature.
3.1 PERSISTENT SYMPTOMATIC EUTHYROID PATIENTS
A subset of patients treated with LT4 achieve serum TSH values within the reference range yet continue to report symptoms such as fatigue, cognitive slowing, mood disturbance, and impaired quality of life. Population surveys and longitudinal studies confirm that persistent dissatisfaction with therapy remains common despite biochemical euthyroidism. These patients often present with nonspecific but functionally significant symptoms that overlap with metabolic, neurocognitive, and psychosocial domains.
Laboratory evaluation typically demonstrates normal TSH and free T4 levels, with free T3 values that may reside in the lower portion of the reference range in some individuals. While laboratory findings alone do not establish causality, they may suggest differences in peripheral hormone activation or tissue sensitivity. Treatment considerations include careful reassessment of diagnosis, evaluation for comorbid contributors (e.g., anemia, sleep disorders, mood disorders, inflammatory conditions), and shared decision-making regarding therapeutic adjustments. In selected patients, a monitored trial of combination T4/T3 therapy may be considered, consistent with contemporary expert discussions emphasizing individualized care. Symptom tracking and functional outcomes should guide ongoing management.
3.2 IMPAIRED T4→T3 CONVERTERS
Some patients exhibit clinical and biochemical features suggestive of reduced peripheral conversion of T4 to T3. These individuals may report persistent hypothyroid symptoms despite adequate levothyroxine dosing and may demonstrate relatively low serum free T3 levels compared with free T4. Experimental and translational studies implicate variability in deiodinase activity and genetic polymorphisms in contributing to altered intracellular T3 availability. Clinically, this phenotype may present with disproportionate fatigue, impaired thermoregulation, and metabolic slowing. Although the concept of impaired conversion remains an area of ongoing investigation, recognition of this pattern supports consideration of individualized dosing strategies. Carefully titrated combination T4/T3 therapy, initiated at low doses and monitored closely, may improve symptom burden in selected patients. Ongoing assessment of cardiovascular risk and symptom response is essential.
3.3 PATIENTS WITH METABOLIC SYNDROME AND INFLAMMATORY PHENOTYPES
Hypothyroidism frequently coexists with metabolic syndrome, insulin resistance, and low-grade systemic inflammation. These patients often present with central adiposity, dyslipidemia, impaired glucose tolerance, and fatigue. Inflammatory mediators can influence deiodinase activity and thyroid hormone signaling, potentially modifying tissue responsiveness. Laboratory patterns may include subtle alterations in lipid profiles and inflammatory markers in addition to thyroid indices. Management extends beyond hormone replacement to include aggressive attention to metabolic health, anti-inflammatory lifestyle strategies, and optimization of comorbid conditions. Integrating thyroid therapy with broader metabolic interventions may enhance overall outcomes.
3.4 PERIMENOPAUSAL AND MENOPAUSAL WOMEN
Hormonal transitions during perimenopause and menopause introduce dynamic changes in thyroid physiology and symptom expression. Fluctuations in estrogen levels influence thyroid-binding globulin concentrations and may alter peripheral hormone availability. Symptom overlap between hypothyroidism and menopausal transition—including fatigue, mood changes, and cognitive complaints—can complicate clinical assessment. Laboratory interpretation may require contextualization within age- and sex-specific reference ranges. Treatment considerations include periodic reassessment of thyroid hormone requirements and attention to concurrent hormonal and metabolic factors. A holistic approach that integrates thyroid management with broader endocrine transitions is particularly important in this population.
3.5 PATIENTS WITH AUTOIMMUNE THYROID DISEASE
Autoimmune thyroiditis represents the most common etiology of hypothyroidism in many populations. These patients may exhibit fluctuating thyroid function, systemic inflammatory features, and an increased prevalence of comorbid autoimmune conditions. Symptom patterns may extend beyond classic hypothyroid features to include fatigue, musculoskeletal discomfort, and neurocognitive complaints. Laboratory evaluation often demonstrates thyroid autoantibodies alongside variable thyroid hormone indices. Management involves not only appropriate hormone replacement but also monitoring for disease progression and associated autoimmune conditions. Emerging research highlights potential interactions between immune regulation, micronutrient status, and thyroid autoimmunity. Addressing modifiable contributors to immune dysregulation may complement pharmacologic therapy.
3.6 HIGHLY SENSITIVE OR MEDICATION-INTOLERANT PATIENTS
A minority of patients exhibit heightened sensitivity to thyroid hormone adjustments or intolerance to standard formulations. These individuals may report palpitations, anxiety, or fluctuating symptoms with small dosing changes. Variability in absorption, excipient sensitivity, and individual pharmacodynamic responses may contribute to this phenotype. Management strategies include gradual dose titration, consideration of alternative formulations (e.g., liquid or soft-gel levothyroxine), and close clinical follow-up. Emphasis on patient education and shared decision-making is essential to maintain adherence and therapeutic trust.
These recurring clinical patterns can be conceptualized as distinct phenotypes of hypothyroid patients, each with unique therapeutic considerations.
| Clinical Phenotype | Typical Clinical Features | Common Laboratory Patterns |
|---|---|---|
| Persistent symptomatic euthyroid | Fatigue, cognitive slowing, mood disturbance despite normalized TSH | TSH within range; free T4 normal; free T3 low–normal in some cases |
| Impaired T4→T3 conversion phenotype | Disproportionate fatigue, cold intolerance, metabolic slowing | Normal/high-normal free T4 with relatively lower free T3 |
| Metabolic/inflammatory phenotype | Central adiposity, insulin resistance, low exercise tolerance | Thyroid indices within range; metabolic abnormalities present |
| Perimenopausal/menopausal transition | Fatigue, sleep disturbance, mood variability overlapping with menopausal symptoms | Fluctuating thyroid indices within reference range |
| Autoimmune thyroid disease | Fluctuating symptoms; possible systemic autoimmune features | Positive thyroid antibodies; variable thyroid function over time |
| Highly sensitive/intolerant phenotype | Palpitations or anxiety with small dose adjustments | Biochemically euthyroid with exaggerated symptom response |
| Absorption challenges | Variable symptom control despite adherence | Lab variability with medication timing sensitivity |
Representative clinical phenotypes commonly encountered in hypothyroid patients are shown with their characteristic symptom patterns, laboratory features, and individualized management strategies. These phenotypes illustrate how patients with similar biochemical profiles may exhibit distinct clinical presentations and therapeutic needs. Recognition of phenotype-specific patterns supports a precision-oriented approach to thyroid hormone replacement that integrates symptoms, physiology, and comorbid context alongside standard laboratory assessment.
4. Therapeutic Options in Personalized Thyroid Replacement
Personalized thyroid hormone replacement requires thoughtful selection among available therapeutic options, guided by patient phenotype, physiologic context, and clinical response. While levothyroxine monotherapy remains the standard initial treatment for most patients, contemporary literature increasingly supports a nuanced, patient-centered framework that allows for individualized therapeutic trials in selected populations. The goal of personalized therapy is not to replace evidence-based standards, but to optimize outcomes through careful patient selection, dosing, and monitoring.
4.1 LEVOTHYROXINE MONOTHERAPY: WHEN IT WORKS BEST
Ideal Patient Profiles
Levothyroxine monotherapy remains highly effective for the majority of patients with primary hypothyroidism and continues to be endorsed as first-line therapy by major professional societies. It is particularly appropriate for patients with uncomplicated autoimmune hypothyroidism, post-thyroidectomy states, and individuals without significant metabolic or inflammatory comorbidities. Recent outcome-focused reviews emphasize that most patients achieve stable symptom control and biochemical normalization with LT4 alone when dosing and absorption factors are optimized. In these individuals, intact peripheral conversion mechanisms likely support adequate tissue T3 availability.
Dosing Strategies
Weight-based initial dosing remains standard in younger adults, with conservative titration recommended in older patients or those with cardiovascular disease. Recent clinical guidance highlights the importance of individualized titration intervals, attention to gastrointestinal absorption, and consistency in formulation and administration timing. Emerging data underscore that variability in absorption remains a common contributor to unstable control.
Monitoring Nuances
Serum TSH remains the primary monitoring tool, with reassessment typically 6–8 weeks after dose adjustment. However, recent expert discussions stress the importance of integrating patient-reported outcomes with biochemical targets. Persistent symptoms should prompt evaluation for nonthyroidal contributors rather than reflexive dose escalation. Overtreatment is associated with increased risks of atrial fibrillation and bone loss, particularly in older populations.
4.2 COMBINATION T4/T3 THERAPY
Rationale and Evidence
Combination therapy with LT4 and LT3 is grounded in the physiologic observation that the healthy thyroid gland secretes both T4 and T3. Approximately 20% of circulating T3 is derived directly from thyroidal secretion, with the remainder generated through peripheral conversion. LT4 monotherapy, while effective in normalizing TSH in most patients, results in reliance on peripheral deiodination to generate adequate intracellular T3. Over the past two decades, randomized controlled trials comparing LT4 monotherapy with combination therapy have produced heterogeneous results. Most studies demonstrate no consistent population-level superiority of combination therapy with respect to quality-of-life measures, mood, or cognitive outcomes. However, several trials report patient preference for combination therapy despite minimal differences in standardized outcome metrics. Meta-analyses suggest that while average treatment effects may be neutral, subgroup variability is substantial. Interpretation of the literature is complicated by methodological limitations. Many trials enrolled unselected populations, used fixed T4:T3 ratios without phenotype stratification, and had relatively short follow-up periods. Additionally, conventional LT3 preparations produce transient serum T3 peaks that may not replicate physiologic secretion patterns, potentially influencing both symptom perception and adverse event profiles. These factors may obscure benefits in selected patients with impaired peripheral conversion or tissue-level T3 availability.
Patient Selection
Appropriate candidates for consideration of combination therapy include patients with persistent symptoms despite stable biochemical euthyroidism on optimized LT4 dosing. Prior to initiation, clinicians should reassess adherence, medication timing, absorption factors, comorbid conditions (including depression, sleep disorders, anemia, and cardiometabolic disease), and psychosocial contributors. Emerging frameworks emphasize phenotype-driven selection rather than empirical escalation. Patients with clinical features suggestive of impaired T4→T3 conversion, disproportionate neurocognitive symptoms, or strong preference for shared therapeutic experimentation may warrant discussion of risks and benefits. Shared decision-making is central, with explicit acknowledgment that evidence remains mixed and therapeutic goals should be clearly defined.
Practical Dosing Approaches
When implemented, combination therapy typically involves reducing the LT4 dose and substituting a small physiologic dose of LT3. Common starting strategies approximate a T4:T3 ratio between 13:1 and 20:1, reflecting estimated endogenous secretion patterns, though optimal ratios remain debated. Because immediate-release LT3 produces rapid serum peaks, divided dosing (e.g., twice daily) is often employed to mitigate fluctuations. Gradual titration is advisable, with attention to both biochemical markers and symptom response. Monitoring should extend beyond TSH alone and include free T4, free T3, heart rate, blood pressure, and patient-reported outcomes. If no meaningful symptomatic improvement is observed after an adequate trial period, reversion to LT4 monotherapy should be considered.
Risk Mitigation
Potential risks of combination therapy include symptoms of thyrotoxicosis, palpitations, anxiety, and theoretical long-term cardiovascular or skeletal consequences in the setting of overtreatment. Older adults, individuals with established cardiovascular disease, and those with osteoporosis require particular caution. Recent expert guidance underscores that combination therapy should be viewed as a monitored therapeutic trial rather than a default escalation strategy. Periodic reassessment of risk–benefit balance is essential, and therapy should be discontinued if adverse effects outweigh symptomatic gains.
4.3 ALTERNATIVE FORMULATIONS
Liquid and Soft-Gel Levothyroxine
Liquid and soft-gel LT4 formulations demonstrate improved absorption profiles and reduced sensitivity to gastric pH and interfering medications. Recent comparative studies confirm more stable TSH control in patients with malabsorption or polypharmacy. These formulations are increasingly recognized as valuable tools in personalized therapy.
Compounded Therapies
Compounded preparations permit individualized dosing but carry concerns regarding potency variability and regulatory oversight. Recent reviews recommend reserving compounded therapy for select cases with documented excipient intolerance or special clinical circumstances.
Desiccated Thyroid Extract
Desiccated thyroid extract (DTE) was the first widely available form of thyroid hormone replacement and has a long history of clinical use. Derived from porcine thyroid tissue, it contains both T4 and T3 in a fixed ratio and therefore appeals to some patients seeking a therapy that more closely resembles natural thyroid secretion. A subset of patients reports subjective symptomatic improvement with DTE, and patient preference remains an important consideration in shared decision-making. At the same time, the fixed T4:T3 ratio in DTE differs from physiologic human secretion and may result in relatively higher circulating T3 levels in some individuals. Variability in hormone content and pharmacokinetics can also complicate dose titration. Contemporary expert guidance therefore recommends thoughtful, individualized use of DTE when chosen, with careful clinical and biochemical monitoring and clear discussion of potential benefits and limitations.
Therapeutic strategies can be aligned with specific hypothyroid clinical phenotypes to support individualized care.
| Clinical Phenotype | Primary Strategy | Selected Adjuncts / Alternatives | Key Implementation Considerations |
|---|---|---|---|
| Persistent symptomatic euthyroid patients | Optimize LT4 dosing; reassess comorbid conditions | Monitored trial of T4/T3 combination in selected patients | Exclude alternative diagnoses; use shared decision-making; track symptom and quality-of-life outcomes |
| Impaired T4→T3 conversion phenotype | Physiologic T4/T3 combination therapy | Individualized LT4 adjustment | Start low-dose LT3; divide dosing; monitor cardiovascular tolerance |
| Metabolic/inflammatory phenotype | LT4 with concurrent metabolic optimization | Lifestyle and insulin resistance management | Address cardiometabolic risk; coordinate endocrine and metabolic care |
| Perimenopausal/menopausal women | LT4 with periodic reassessment | Integration with sex hormone therapy when appropriate | Reevaluate dosing during hormonal transitions; distinguish menopausal from thyroid symptoms |
| Autoimmune thyroid disease | LT4 with monitoring for progression | Targeted nutritional or immune-supportive measures | Screen for comorbid autoimmune disease; monitor antibodies and clinical trajectory |
| Highly sensitive/intolerant patients | Gradual low-dose LT4 titration | Liquid or soft-gel LT4 formulations | Use small dose increments; ensure formulation consistency; close follow-up |
| Absorption challenges | Liquid or soft-gel LT4 | Medication timing adjustments | Review interfering medications; optimize administration timing |
Therapeutic strategies aligned with common hypothyroid clinical phenotypes. Management approaches are matched to symptom patterns and physiologic context to illustrate phenotype-driven personalization. Combination therapy should be implemented cautiously and reassessed periodically based on clinical response and safety.
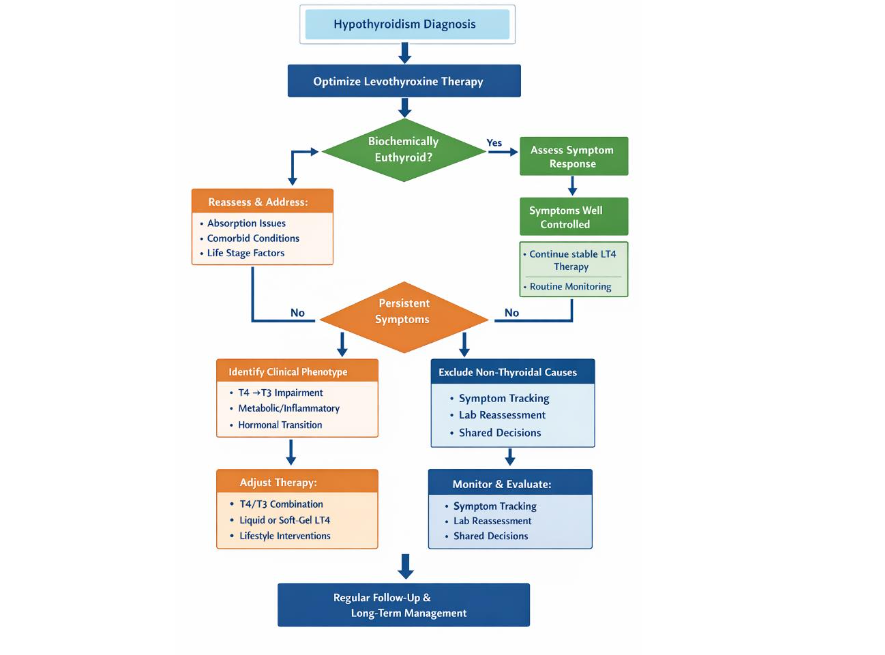
5. A Clinical Algorithm for Individualized Treatment
The integration of physiologic principles, clinical phenotyping, and therapeutic options supports a structured algorithm for individualized thyroid hormone replacement. Rather than relying exclusively on biochemical targets, a stepwise approach that incorporates symptom assessment, laboratory interpretation, and patient-specific context allows clinicians to tailor therapy more precisely. The following framework outlines a practical decision pathway for personalized management.
5.1 STEPWISE DECISION FRAMEWORK
The initial step in hypothyroidism management is confirmation of diagnosis and optimization of levothyroxine therapy according to established guidelines. Once biochemical euthyroidism is achieved, clinicians should systematically evaluate symptom response and functional outcomes. Patients who report satisfactory symptom resolution may continue stable therapy with routine monitoring. For patients with persistent symptoms, the algorithm shifts toward phenotype-based reassessment. This includes evaluation for comorbid conditions, absorption factors, inflammatory or metabolic contributors, and life-stage transitions such as menopause. Identification of a dominant clinical phenotype can guide targeted therapeutic adjustments, including optimization of LT4 dosing, consideration of alternative formulations, or a carefully monitored trial of combination therapy in selected cases.
5.2 SYMPTOM-GUIDED THERAPEUTIC ADJUSTMENTS
Symptom patterns provide essential information beyond laboratory values. Structured symptom assessment tools or patient-reported outcome measures can help quantify functional impairment and track response over time. Importantly, symptom persistence should prompt a broad differential diagnosis rather than reflexive escalation of thyroid hormone dose. When thyroid-related contributors remain plausible after exclusion of alternative causes, incremental therapeutic adjustments may be considered. These adjustments should be conservative, individualized, and accompanied by clear goals and predefined evaluation intervals. Shared decision-making is central to this process, ensuring that patients understand both potential benefits and risks.
5.3 INTEGRATION OF LABORATORY AND CLINICAL DATA
Effective personalization requires balanced interpretation of biochemical and clinical information. Serum TSH remains the primary biochemical anchor, but free hormone levels and clinical context may inform nuanced decision-making in selected cases. Laboratory trends should be interpreted alongside symptom trajectories rather than in isolation. Overemphasis on laboratory normalization without attention to patient experience risks undertreatment of persistent symptoms, while excessive dose escalation increases the likelihood of iatrogenic thyrotoxicosis. A calibrated approach that integrates both domains supports safer and more effective individualized care.
5.4 MONITORING AND FOLLOW-UP STRATEGY
Ongoing monitoring is essential to maintain therapeutic stability and detect emerging risks. After any dose adjustment, reassessment of thyroid function tests is typically performed after 6–8 weeks. Once stability is achieved, periodic monitoring intervals may be extended based on clinical context. Follow-up visits should include structured reassessment of symptoms, cardiovascular status, bone health risk, and relevant comorbid conditions. In patients undergoing combination therapy or alternative treatment strategies, closer surveillance is warranted. Documentation of symptom response and shared decision-making discussions enhances continuity of care and supports long-term therapeutic alignment.
This clinical framework lends itself naturally to a visual decision tree summarizing the stepwise pathway from diagnosis to phenotype-guided treatment adjustments and longitudinal monitoring. A schematic algorithm can serve as a practical reference tool for clinicians implementing personalized thyroid care.
6. Contextual Modifiers of Persistent Symptoms
Thyroid hormone replacement does not occur in physiologic isolation. In selected patients with persistent symptoms despite biochemical euthyroidism, systemic factors may modify tissue responsiveness to thyroid hormone and influence clinical presentation. These contextual modifiers should be considered adjunctive contributors rather than primary explanations.
Low-grade inflammation and metabolic dysfunction have been shown to influence deiodinase activity, receptor signaling, and mitochondrial energy metabolism. Insulin resistance, adiposity, and dyslipidemia frequently coexist with hypothyroidism and may amplify fatigue and metabolic complaints independent of thyroid hormone dosing. Addressing cardiometabolic risk through structured physical activity, dietary quality, and weight optimization may therefore improve symptom burden in appropriate patients.
Micronutrient status also warrants consideration in selected cases. Selenium, iron, iodine, zinc, and vitamin D play established roles in thyroid physiology and immune regulation. Clinically meaningful deficiencies should be corrected using evidence-informed strategies, while avoiding excessive supplementation. Routine empirical supplementation without documented need is not supported.
Chronic stress and sustained activation of the hypothalamic–pituitary–adrenal axis may further influence thyroid hormone metabolism and perceived energy levels. While behavioral interventions such as sleep optimization and stress management are not substitutes for pharmacologic therapy, they may support symptom stability in selected individuals.
These contextual factors should be evaluated as part of a comprehensive reassessment when symptoms persist. However, they should not be invoked reflexively to dismiss ongoing patient concerns. Rather, they serve as potential contributors within a phenotype-driven framework that integrates physiologic variability, clinical presentation, and individualized therapeutic strategy.
7. Case-Based Clinical Applications
Case-based examples illustrate how a phenotype-guided framework can inform individualized thyroid hormone replacement. The following vignettes demonstrate practical application of symptom assessment, laboratory interpretation, and targeted therapeutic adjustments.
7.1 PERSISTENT SYMPTOMATIC PATIENT ON LEVOTHYROXINE
A 42-year-old woman with primary hypothyroidism treated with levothyroxine for three years presents with persistent fatigue, cognitive slowing, and reduced exercise tolerance. Her TSH has remained within the reference range (1.8 mIU/L) with stable dosing. Free T4 is mid-normal, and free T3 is at the lower end of the reference range. Evaluation excludes anemia, sleep apnea, depression, and other systemic contributors. Using a phenotype-guided approach, the patient is categorized as a persistent symptomatic euthyroid phenotype with possible relative T4→T3 conversion inefficiency. After shared decision-making and discussion of risks and benefits, a carefully monitored trial of low-dose combination T4/T3 therapy is initiated, replacing a small portion of her LT4 dose. Over 12 weeks, she reports measurable improvement in energy and cognitive clarity without cardiovascular side effects. Laboratory monitoring confirms maintenance of biochemical euthyroidism. Ongoing follow-up emphasizes symptom tracking and periodic reassessment of risk–benefit balance. This case illustrates how selected patients with persistent symptoms may benefit from individualized therapeutic trials when alternative causes have been excluded and monitoring is structured.
7.2 PERIMENOPAUSAL WOMAN WITH FLUCTUATING THYROID NEEDS
A 49-year-old woman with autoimmune hypothyroidism reports new fatigue, sleep disturbance, and mood variability during the perimenopausal transition. Her TSH has fluctuated between 2.5 and 4.0 mIU/L over the past year despite adherence to therapy. She also reports vasomotor symptoms and irregular menstrual cycles. Clinical reassessment recognizes the interaction between hormonal transition and thyroid physiology. Rather than escalating thyroid hormone dosing reflexively, the care plan integrates evaluation of menopausal status and lifestyle contributors such as sleep quality and stress. After modest LT4 dose adjustment and coordinated management of menopausal symptoms, the patient experiences improved stability in both thyroid indices and overall well-being. This vignette highlights the importance of contextualizing thyroid management within broader endocrine transitions and avoiding isolated laboratory-driven decision-making.
7.3 AUTOIMMUNE PHENOTYPE WITH SYSTEMIC CONTRIBUTORS
A 35-year-old woman with Hashimoto thyroiditis presents with fatigue, myalgias, and intermittent cognitive fog despite adequate levothyroxine therapy and TSH of 1.5 mIU/L. She has positive thyroid peroxidase antibodies and a history of other autoimmune symptoms. Further evaluation reveals vitamin D insufficiency and markers of low-grade inflammation. Management extends beyond dose adjustment to include correction of nutrient deficiencies, anti-inflammatory lifestyle strategies, and screening for associated autoimmune conditions. Thyroid hormone dosing remains stable, but systemic interventions lead to gradual improvement in symptom burden. This case demonstrates how an autoimmune phenotype may require a systems-oriented approach in which thyroid replacement is integrated with immune and metabolic optimization.
8. Future Directions and Research Gaps
Despite major advances in thyroid hormone replacement over recent decades, important gaps remain in understanding interindividual variability in treatment response. Emerging developments in precision medicine, biomarker discovery, and clinical trial design offer opportunities to refine personalized approaches to hypothyroidism. Addressing these gaps will require integration of molecular science with clinically meaningful outcome measures and patient-centered endpoints.
8.1 PRECISION MEDICINE AND GENOMIC INSIGHTS
Advances in genomics and systems biology are reshaping the understanding of endocrine variability. Individual differences in genes regulating deiodinase activity, thyroid hormone transport, receptor signaling, and immune function may contribute to heterogeneous responses to therapy. Contemporary reviews emphasize that although routine genomic testing is not yet standard in hypothyroidism care, genotype-informed stratification represents a promising future direction. Emerging precision frameworks extend beyond single-gene polymorphisms toward multi-omic profiling, including transcriptomic and metabolomic signatures that more directly reflect tissue-level thyroid hormone activity. Integration of genomic and phenotypic data may eventually support predictive models that guide individualized therapy selection and dosing strategies.
8.2 BIOMARKERS BEYOND SERUM TSH
Serum TSH remains the primary biomarker guiding hypothyroidism management, yet it functions as an indirect surrogate for tissue-level thyroid hormone action. Increasing recognition of discordance between biochemical euthyroidism and patient-reported outcomes has stimulated investigation into complementary biomarkers that better capture intracellular thyroid signaling and functional status. Recent research highlights the potential role of metabolomic and physiologic markers in characterizing thyroid hormone effects on energy metabolism and systemic homeostasis. Advances in digital health technologies and wearable physiologic monitoring may further expand opportunities to assess functional outcomes in real-world settings. Validation of clinically practical biomarkers that correlate with symptom improvement remains a key research priority.
8.3 NEED FOR PHENOTYPE-DRIVEN CLINICAL TRIALS
Most randomized trials of thyroid hormone replacement have evaluated heterogeneous populations without stratification by biologic or clinical phenotype, potentially obscuring meaningful subgroup effects. Contemporary commentaries increasingly call for phenotype-driven trial designs that incorporate genetic, metabolic, and clinical stratification. Future studies would benefit from greater emphasis on patient-reported outcomes, functional endpoints, and longer follow-up intervals that reflect real-world therapeutic goals. Adaptive trial methodologies and pragmatic clinical designs may accelerate translation into clinical practice. A shift toward phenotype-informed investigation aligns with broader trends in precision endocrinology and may clarify which patient subgroups derive the greatest benefit from alternative therapeutic strategies.
9. Conclusion
Hypothyroidism management is evolving from a uniform, laboratory-centered model toward a more nuanced framework that recognizes the biologic and clinical diversity of patients. While levothyroxine monotherapy remains an effective foundation for most individuals, persistent symptoms in a meaningful subset of patients highlight the limitations of a one-size-fits-all approach. Variability in thyroid hormone metabolism, tissue responsiveness, systemic physiology, and patient context underscores the need for individualized therapeutic strategies. A phenotype-guided model that integrates physiologic understanding with careful symptom assessment, laboratory interpretation, and shared decision-making provides a practical pathway toward personalized care. Incorporating lifestyle and systems-level factors—including metabolic health, inflammation, nutrient status, and stress regulation—further expands the therapeutic landscape beyond pharmacologic replacement alone. Case-based applications demonstrate how structured clinical reasoning can translate these principles into real-world decision-making. Future advances in precision medicine, biomarker development, and phenotype-driven clinical trials hold promise for refining individualized thyroid therapy. As research continues to clarify the determinants of treatment response, clinicians can adopt a flexible, patient-centered framework that balances evidence-based standards with thoughtful personalization. Such an approach aims not only to normalize laboratory values but to optimize functional outcomes and quality of life for patients living with hypothyroidism.
Key Takeaways
- Hypothyroidism is biologically heterogeneous. Interindividual variability in thyroid hormone metabolism, tissue signaling, and systemic physiology contributes to differing treatment responses among patients.
- Biochemical euthyroidism does not always equal clinical euthyroidism. Persistent symptoms in some patients warrant thoughtful reassessment rather than automatic dose escalation.
- Clinical phenotyping supports personalized care. Recognizable patient patterns—including persistent symptomatic euthyroid states, impaired T4→T3 conversion, metabolic/inflammatory phenotypes, hormonal transitions, and autoimmune presentations—can guide targeted therapeutic strategies.
- Levothyroxine remains first-line therapy for most patients. However, carefully selected individuals may benefit from alternative approaches, including monitored T4/T3 combination therapy or formulation adjustments.
- Lifestyle and systems factors influence therapeutic outcomes. Metabolic health, inflammation, nutrient status, and stress regulation interact with thyroid hormone action and should be integrated into comprehensive management.
- Structured clinical algorithms improve decision-making. A stepwise framework that combines symptom assessment, laboratory data, and phenotype-guided adjustments supports safer and more effective individualized treatment.
- Future progress depends on precision approaches. Advances in genomics, biomarker development, and phenotype-driven clinical trials are needed to refine personalized thyroid hormone replacement.
Conflict of Interest Statement: None.
Funding Statement: None.
Acknowledgements: None.
References:
- Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid. 2014 Dec;24(12):1670–751. doi: 10.1089/thy.2014.0028. PMID: 25266247; PMCID: PMC4267409.
- Peterson SJ, Cappola AR, Castro MR, et al. An online survey of hypothyroid patients demonstrates prominent dissatisfaction. Thyroid. 2018;28(6):707–721. DOI: 10.1089/thy.2017.0681.
- McAninch EA, Bianco AC. The history and future of treatment of hypothyroidism. Ann Intern Med. 2016;164(1):50–56. DOI: 10.7326/M15-1799
- Wiersinga WM. Paradigm shifts in thyroid hormone replacement therapies for hypothyroidism. Nat Rev Endocrinol. 2014;10:164–174. DOI: 10.1038/nrendo.2013.258
- Taylor PN, Albrecht D, Scholz A, et al. Global epidemiology of hyperthyroidism and hypothyroidism. Nat Rev Endocrinol. 2018;14:301–316. DOI: 10.1038/nrendo.2018.18
- Bianco AC, Kim BW. Deiodinases and local thyroid hormone action. J Clin Invest. 2006;116(10):2571–2579. DOI: 10.1172/JCI29812
- Gereben B, Zavacki AM, Ribich S, et al. Cellular and molecular basis of deiodinase-regulated thyroid hormone signaling. Endocr Rev. 2008;29(7):898–938. DOI: 10.1210/er.2008-0019
- Panicker V. Genetics of thyroid function and disease. Clin Biochem Rev. 2011 Nov;32(4):165-75. PMID: 22147956; PMCID: PMC3219766.
- Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. Lancet. 2017;390(10101):1550–1562. DOI: 10.1016/S0140-6736(17)30703-1
- Biondi B, Cappola AR, Cooper DS. Subclinical hypothyroidism: a review. JAMA. 2019;322(2):153–160. DOI: 10.1001/jama.2019.9052
- Wiersinga WM. T4 + T3 combination therapy: an unsolved problem of increasing Magnitude and Complexity. Eur Thyroid J. 2021;10(1):7–15. DOI: 10.3803/EnM.2021.501
- Hoermann R, Midgley JEM, Larisch R, Dietrich JW. Recent advances in thyroid hormone regulation: toward a new paradigm for optimal diagnosis and treatment. Front Endocrinol. 2017;8:364. DOI: 10.3389/fendo.2017.00364
- Luongo C, Dentice M, Salvatore D. Deiodinases and their intricate role in thyroid hormone homeostasis. Nat Rev Endocrinol. 2019 Aug;15(8):479–488. doi: 10.1038/s41574-019-0218-2. PMID: 31160732.
- Brent GA. Mechanisms of thyroid hormone action. J Clin Invest. 2012 Sep;122(9):3035-43. doi: 10.1172/JCI60047. Epub 2012 Sep 4. PMID: 22945636; PMCID: PMC3433956.
- Fliers E, Boelen A. An update on nonthyroidal illness syndrome. J Endocrinol Invest. 2021. DOI: 10.1007/s40618-020-01482-4
- Panicker V, Saravanan P, Vaidya B, et al. Common variation in the DIO2 gene predicts baseline psychological well-being and response to combination thyroxine plus triiodothyronine therapy in hypothyroid patients. J Clin Endocrinol Metab. 2009 May;94(5):1623-9. doi: 10.1210/jc.2008-1301. Epub 2009 Feb 3. PMID: 19190113.
- Bianco AC, Kim BS. Pathophysiological relevance of deiodinase polymorphism. Curr Opin Endocrinol Diabetes Obes. 2018 Oct;25(5):341-346. doi: 10.1097/MED.0000000000000428. PMID: 30063552; PMCID: PMC6571023.
- Köhrle J. Thyroid hormone transporters in health and disease: advances in thyroid hormone deiodination. Best Pract Res Clin Endocrinol Metab. 2007 Jun;21(2):173-91. doi: 10.1016/j.beem.2007.04.001. PMID: 17574002.
- Mullur R, Liu YY, Brent GA. Thyroid hormone regulation of metabolism. Physiol Rev. 2014; updated concepts reviewed in Nat Rev Endocrinol. 2020. doi: 10.1152/physrev.00030.2013
- Biondi B, Cappola AR. Subclinical hypothyroidism in older individuals. Lancet Diabetes Endocrinol. 2022 Feb;10(2):129-141. doi: 10.1016/S2213-8587(21)00285-0. Epub 2021 Dec 22. PMID: 34953533.
- Strelow B, O’Loughlin D, Anderson T, et al. Menopause decoded: what’s happening and how to manage it. J Prim Care Community Health. 2024 Jan-Dec;15:2150131924307460. doi: 10.1177/2150131924307460. PMID: 39707880; PMCID: PMC11663262.
- Warner MH, Beckett GJ. Mechanisms behind the non-thyroidal illness syndrome: an update. J Endocrinol. 2010 Apr;205(1):1–13. doi: 10.1677/JOE-09-0412. Epub 2009 Dec 16. PMID: 20016054.
- Feldt-Rasmussen U, Klose M, Benvenega S. Interactions between hypothalamic pituitary thyroid axis.Santos LR, Vasconcelos Bessa I, Gaspar da Rocha A, et al. Survey on selenium clinical supplementation in autoimmune thyroid disease. Eur Thyroid J. 2023 Feb;8(12):e220090. doi: 10.1530/ETJ-22-0090. PMID: 36622057; PMCID: PMC9986388.
- Virili C, Brusca N, Capriello S, et al. Levothyroxine therapy in gastric malabsorptive disorders. Front Endocrinol (Lausanne). 2021 Jan 28;11:621616. doi: 10.3389/fendo.2020.621616. PMID: 33584549; PMCID: PMC7876372.
- Hoang TD, Olsen CH, Mai VQ, et al. Desiccated thyroid extract compared with levothyroxine in the treatment of hypothyroidism: a randomized, double-blind, crossover study. J Clin Endocrinol Metab. 2013 May;98(5):1982-90. doi: 10.1210/jc.2012-4107. Epub 2013 Mar 28. PMID: 23539727.
- Taylor S, Davis SR. Is it time to revisit recommendations for initiation of menopausal hormone therapy? Lancet Diabetes Endocrinol. 2025 Jan;13(1):69-74. doi: 10.1016/S2213-8587(24)00270-5. Epub 2024 Oct 14. PMID: 39419045.
- Mazur M, Szymańska M, Malik A, et al. Nutrition and micronutrient interactions in autoimmune thyroid disorders: implications for cardiovascular health. Pathophysiology. 2025 Aug 1;32(3):37. doi: 10.3390/pathophysiology32030037. PMID: 40843807; PMCID: PMC12372124.
- Ettleson MD, Bianco AC. Individualized therapy for hypothyroidism: is T4 enough for everyone? J Clin Endocrinol Metab. 2020 Sep 1;105(9):e3090-104. doi: 10.1210/clinem/dgaa430. PMID: 32614450; PMCID: PMC7382053.
- Liu H, Li W, Zhang W, et al. Levothyroxine: conventional and novel drug delivery formulations. Endocr Rev. 2023 May 8;44(3):393-416. doi: 10.1210/endrev/bnac030. PMID: 36412275; PMCID: PMC10166268.
- Kahaly GJ, editor. 70 Years of Levothyroxine [Internet]. Cham (CH): Springer; 2021. Available from: https://www.ncbi.nlm.nih.gov/books/NBK585638/. doi: 10.1007/978-3-030-63277-9
- Jonklaas J, Bianco AC, Cappola AR, et al. Evidence-based use of levothyroxine/liothyronine combinations in treating Hypothyroidism: a consensus document. Eur Thyroid J. 2021 Mar;10(1):10-38. doi: 10.1159/000512970. Epub 2021 Feb 16. PMID: 33777817; PMCID: PMC7983670.
- Biondi B, Celi FS, McAninch EA. Critical approach to hypothyroid patients with persistent symptoms. J Clin Endocrinol Metab. 2023 Sep 18;108(10):2708-2716. doi: 10.1210/clinem/dgad224.
- Hoermann R, Midgley JEM, Larisch R, et al. Individualised requirements for optimum treatment of hypothyroidism: complex needs, limited options. Drugs Context. 2019 Aug 13;8:212597. doi: 10.7573/dic.212597. PMID: 31516533; PMCID: PMC6726361.
- Gatta E, Bambini F, Buoso C, et al. Liquid levothyroxine formulations in patients taking drugs interfering with L-T4 absorption. Front Endocrinol (Lausanne). 2022 Dec 6;13:1080108. doi: 10.3389/fendo.2022.1080108. PMID: 36561558; PMCID: PMC9764388.
- Xu Y, Derakhshan A, Hysaj O, et al. The optimal healthy ranges of thyroid function defined by the risk of cardiovascular disease and mortality: systematic review and individual participant data meta-analysis. Lancet Diabetes Endocrinol. 2023 Oct;11(10):743-754. doi: 10.1016/S2213-8587(23)00227-9. Epub 2023 Sep 8. PMID: 37696273; PMCID: PMC10866328.
- Pedersen BK, Saltin B. Exercise as medicine – evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand J Med Sci Sports. 2015 Dec;25 Suppl 3:1-72. doi: 10.1111/sms.12581. PMID: 26606383.
- Souza LSL, Campos RO, Braga JS Filho, et al. Selenium nutritional status and thyroid dysfunction. Arch Endocrinol Metab. 2025 Feb 24;69(1):e230348. doi: 10.20945/2359-4292-2023-0348. PMID: 39992731; PMCID: PMPedersen BK, Saltin B. Exercise as medicine – evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand J Med Sci Sports. 2015 Dec;25 Suppl 3:1-72. doi: 10.1111/sms.12581. PMID: 26606383.
- Souza LSL, Campos RO, Braga JS Filho, et al. Selenium nutritional status and thyroid dysfunction. Arch Endocrinol Metab. 2025 Feb 24;69(1):e230348. doi: 10.20945/2359-4292-2023-0348. PMID: 39992731; PMCID: PMC11849045.
- Arczewska KD, Piekielko-Witkowska A. The Influence of Micronutrients and Environmental Factors on Thyroid DNA Integrity. Nutrients. 2025 Jun 21;17(13):2065. doi: 10.3390/nu17132065. PMID: 40647171; PMCID: PMC12251246.
- Ortiga-Carvalho TM, Chiamolera MI, et al. Hypothalamus-Pituitary-Thyroid Axis. Compr Physiol. 2016 Jun 13;6(3):1387-428. doi: 10.1002/cphy.c150027. PMID: 27347897.
- Irwin MR. Sleep and inflammation: partners in sickness and in health. Nat Rev Immunol. 2019 Nov;19(11):702-715. doi: 10.1038/s41577-019-0190-z. PMID: 31289370.Mazza AD. Personalized approaches to hypothyroidism: The Role of triiodothyronine (T3) in thyroid hormone replacement. Cureus. 2025 Aug 21;17(8):e90685. doi: 10.7759/cureus.90685. PMID: 40851876; PMCID: PMC12370163.
- Hoermann R, Pekker MJ, Midgley JEM, et al. Principles of endocrine regulation: reconciling tensions between robustness in performance and adaptation to change. Front Endocrinol (Lausanne). 2022 Jun 9;13:825107. doi: 10.3389/fendo.2022.825107. PMID: 35757421; PMCID: PMC9219553.
- Fitzgerald SP, Falhammar H. Redefinition of successful treatment of patients with hypothyroidism. Is TSH the best biomarker of euthyroidism? Front Endocrinol (Lausanne). 2022 Jun 16;13:920854. doi: 10.3389/fendo.2022.920854. PMID: 35784560; PMCID: PMC9243528.

