Preclinical Study of CLX-155A in Triple-Negative Breast Cancer
Preclinical Evaluation of CLX-155A: A Novel 5-FU and Valproic Acid Prodrug in Nude Mouse Model for Activity in Triple-Negative Breast Cancer
John M. York, PharmD, MBA¹˒²˒³, Michika Maeda, MD¹, Giovanni Lara, PharmD¹, Mahesh Kandula, M.Tech, MBA⁴˒⁵, Subbu Apparsundaram, PhD⁴˒⁵
- Ernest Mario School of Pharmacy, Rutgers University, Newark, NJ
- Institute for the Global Entrepreneur, University of California, San Diego, CA
- Burnett School of Medicine, Texas Christian University, Ft. Worth, TX
- Celix Bio Private Limited, Hyderabad, India
- Celix Biosciences, Inc., Newark, NJ
OPEN ACCESS
PUBLISHED: 30 April 2025
CITATION: York, J.M., Maeda, M., et al., 2025. Preclinical Evaluation of CLX-155A: A Novel 5-FU and Valproic Acid Prodrug in Nude Mouse Model for Activity in Triple-Negative Breast Cancer. Medical Research Archives, [online] 13(4).
https://doi.org/10.18103/mra.v13i4.6470
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i4.6470
ISSN 2375-1924
Abstract
New therapeutic approaches are needed to improve patient outcomes. Traditional pyrimidine antimetabolic chemotherapy agents like 5-FU and capecitabine face challenges of resistance, toxicity, and variability in patient response. CLX-155A, a novel oral prodrug, aims to enhance chemotherapy efficacy by combining 5-fluorouracil (5-FU) and valproic acid (VPA) to utilize synergistic mechanisms. This preclinical study addresses the research question: What is CLX-155A’s preclinical activity in nude mouse models of triple-negative breast cancer (TNBC)? This study assesses the anticancer efficacy of CLX-155A in TNBC nude xenograft mouse models, focusing on tumor growth inhibition and its potential effects when combined with paclitaxel. Mice inoculated with cancer cells were treated with CLX-155A at 1000 mg/kg/D either as monotherapy or in combination with paclitaxel at 15mg/kg daily. The study utilized comparisons with vehicle and capecitabine at 1000 mg/kg/D. The primary endpoint was tumor growth rate (%). Secondary assessments included survival and weight loss. CLX-155A displayed significant antitumor activity in a TNBC model. The two CLX-155A groups (CLX-155A at 1000 mg/kg and CLX-155A at 1000 mg/kg + paclitaxel at 15 mg/kg) showed a significant effect (p<0.001) versus the vehicle control. At equal dosages to capecitabine, CLX-155A showed similar effects. It was numerically superior compared to capecitabine at equal dosages, both alone and in combination with paclitaxel. Further, CLX-155A as monotherapy showed similar effects to capecitabine with paclitaxel at 15mg/kg daily. Overall, CLX-155A exhibits promising preclinical efficacy in CRC and TNBC models. Due to its dual-action mechanism, it may offer potential advantages over existing therapies. Additional studies are warranted to explore its clinical potential further and optimize dosing strategies.
Keywords
Capecitabine, CLX-155A, 5-FU, Nude mouse model, Triple-negative breast cancer, Valproic acid
Introduction
The interference of DNA and RNA synthesis, vital for cell replication and survival, are key mechanisms for pyrimidine analogs in cancer treatment. 5-fluorouracil (5-FU) is a commonly utilized intravenous (IV) chemotherapeutic drug, demonstrating effectiveness in the treatment of solid tumors, such as colorectal and breast cancers. By mimicking the pyrimidine nucleotide uracil, 5-FU inhibits thymidylate synthase and incorporates itself into DNA and RNA, disrupting their normal function and inducing cell death. While effective, continuous infusion of 5-FU is required to maintain therapeutic levels due to the occurrence of severe side effects, such as myelosuppression, mucositis, and cardiotoxicity. Resistance issues and drug availability caused by supply, demand, and manufacturing issues further restrict the effectiveness of 5-FU.
Overcoming these limitations, researchers created capecitabine, an oral prodrug of 5-FU. The development offers a more convenient and potentially less toxic alternative to the intravenous administration of 5-FU since capecitabine undergoes enzymatic conversion to 5-FU in both the liver and tumor tissues. Oral administration offers increased flexibility in dosing schedules and has the potential to enhance patient compliance. However, capecitabine possesses challenges, including patient metabolism variability, reliance on liver function for activation, and notable toxicities in the gastrointestinal and dermatological systems, such as diarrhea and hand-foot syndrome. The occurrence of these side effects often requires reducing the dosage and temporarily halting treatment, which can potentially compromise the effectiveness of the therapy.
Unmet needs in cancer therapy, specifically triple-negative breast cancer (TNBC), involve addressing drug resistance, minimizing toxicity, and enhancing patient compliance. Drug resistance development in cancer treatment poses a significant obstacle, as treatment failure can arise from cancer cells employing mechanisms to evade chemotherapy. Furthermore, the use of specific chemotherapeutic agents can be restricted due to their associated toxicity, especially in patients with comorbidities or advanced age. Enhancing patient compliance is equally as complicated as inconvenient dosing regimens may result in suboptimal adherence to treatment protocols.
Within the current TNBC landscape, a variety of pharmaceutical agents are used, including alkylating agents, antimetabolites, taxanes, and biologics. This variety of treatment options is due to the poor prognosis of this type of cancer, with multiple agents being quite common. Within the NCCN guidelines for TNBC to date, capecitabine is part of the standard regimen, alongside pembrolizumab and olaparib, despite its side effect profile. The existing gaps emphasize the need for innovative therapeutic strategies to improve effectiveness and minimize adverse effects. For 5-FU, unmet needs include ensuring a consistent drug supply, addressing resistance concerns, and exploring alternative delivery methods to simulate continuous infusion. The needs for capecitabine include consistent drug levels and the limiting of gastrointestinal and liver effects, hand and foot syndrome issues (50% of patients), and dose adjustments in patients with hepatic or renal disease as well as in older patients.
CLX-155 offers a groundbreaking solution to meet these unmet needs by introducing a novel prodrug that combines 5-FU with caprylic acid. Rather than metabolize in the liver, it is specifically designed to undergo metabolism in the intestinal wall. The addition of caprylic acid is rationalized by its antibacterial, antifungal, anti-inflammatory, and digestive health capabilities and enhances the effects of 5-FU through various established anti-cancer mechanisms, which include inducing inhibiting cell proliferation, reducing the viability of cancer cells, modulating gene expression, and apoptosis. The combination offers the potential for achieving a more consistent pharmacokinetic profile and minimizing toxicity.
CLX-155A integrates valproic acid (Figure 1), an inhibitor of histone deacetylase (HDAC), to further augment the anticancer activity through the modulation of epigenetics. By showcasing its capability to trigger apoptosis, inhibit cell proliferation, and modulate gene expression, Valproic acid (VPA) proves to be a valuable addition to cancer therapy. The role of HDAC inhibitors, such as VPA, in cancer therapy is of great significance as they influence the acetylation status of histones, leading to changes in gene expression and the promotion of cancer cell death. CLX-155A, the combination of 5-FU and VPA, aims to improve treatment outcomes in TNBC by harnessing these synergistic effects.
Accordingly, this study addresses the following research questions: 1) What is the anticancer activity of CLX-155A in preclinical models of TNBC? and 2) How do the effects of CLX-155A compare to existing treatments, such as capecitabine? Through a thorough assessment, this study seeks to establish the preclinical basis for the potential clinical utilization of CLX-155A in the TNBC setting. The paper structure first presents the methods used in the preclinical evaluation of CLX-155A, including the design of xenograft models in nude mice, schedule, dosing regimens, assessments, and analysis. Next, the results section details tumor growth inhibition and toxicity assessment findings. Finally, the discussion considers the implications of these results, comparing them with relevant literature and exploring the potential mechanisms underlying the observed efficacy of CLX-155A and the role of VPA.
Methods
STUDY DESIGN
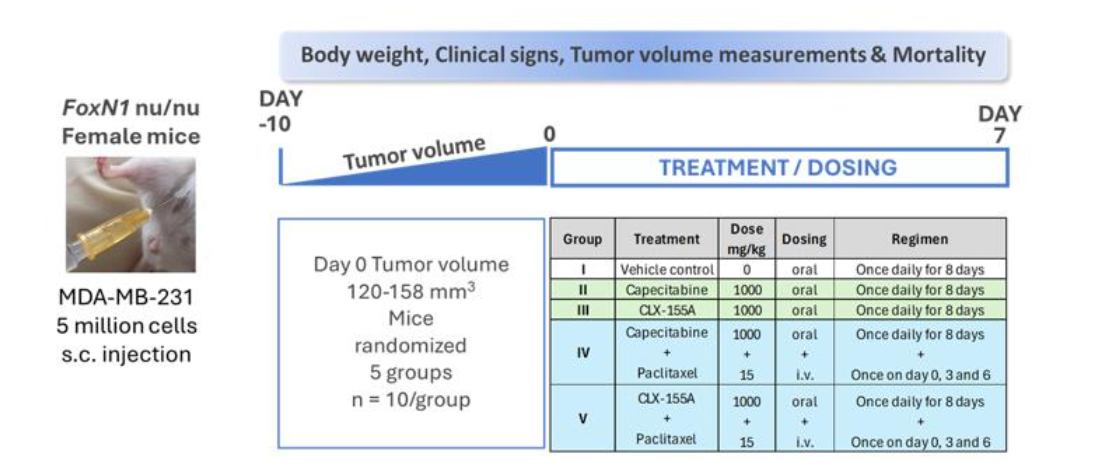
The study design drew guidance from works by Pulaski and Ostrand-Rosenberg (2001) and Contreras-Zárate and collaborators. It utilized female FoxN1 athymic nude mice obtained from Vivo Bio Tech (Hyderabad, India). The evaluation involved five groups (Figure 2), with ten mice per group (n=10/group). All animal care and use procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IAEC/JDC/2017-120) prior to the start of the study. The study followed the guidelines outlined in the 8th Edition, 2010, of the Guide for the Care and Use of Laboratory Animals, ensuring proper animal care and use. The experimentation facility is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC).
ANIMALS AND HANDLING
For one week, all animals resided within individually ventilated cages in groups of five in a dedicated rodent quarantine room within an immunocompromised facility. Investigators monitored the animals daily during this time for any clinical signs of disease. To acclimate to the experimental condition, healthy animals transitioned to an experimental room for seven days following the completion of the quarantine period. The conditions in which the animals resided included: 12-hour light/dark cycle, barrier (quarantine) conditions, and continuously monitored temperature and humidity-regulated aseptic and access-controlled environment (target ranges: temperature 22 ± 2°C; relative humidity 60 ± 4%; and 60 air changes per hour). Investigators routinely monitored the entire facility to detect any airborne infections. Animals had an autoclaved commercial diet (Nutrilab Rodent Feed, cylindrical-shaped pellets) and free access to autoclaved water.
CANCER CELL LINES AND INOCULATION
The TNBC study utilized the human cell line MDA-MB-231, obtained from the American Type Culture Collection (ATCC), Manassas, Virginia, USA. The culture media used to grow the MDA-MB-231 cell line consisted of McCoy’s 5a medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin. Investigators harvested cells by trypsinization at 70-80% confluence and then re-suspended cells in a serum-free medium prior to animal inoculation. Investigators implanted the MDA-MB-231 cells (5 million cells/site) subcutaneously in the dorsal right flank. Injections contained viable MDA-MB-231 cells in serum-free medium at a concentration of 5 × 106/100 μL mixed with an equal volume of Matrigel (1:1 ratio) for implanting at the subcutaneous site per mouse. Each injection consisted of 200 μL per site using a 1 mL BD syringe attached to a 23-gauge needle. Investigators measured the size of the xenografts approximately ten days after cell injection and once the xenografts became palpable. Investigators randomized animals into five groups (N = 10 per group). On Day 0, the tumor volume ranged from 120 to 158 mm3.
PREPARATION OF EXPERIMENTAL TREATMENTS
The administration of all compound formulations occurred within one hour of preparation. CLX-155A formulations consisted of CLX-155A at 100 mg/mL in 0.5% w/v hydroxypropyl methylcellulose (HPMC E15) in water containing 0.5% v/v Polysorbate 80. Investigators prepared the capecitabine 1000 mg/kg dose in 0.5% w/v hydroxypropyl methylcellulose (HPMC E15) in 40 mM citrate buffer, pH 6.0 in 0.2 μm filtered water vehicle for a capecitabine dose concentration of 100 mg/mL and a dose volume of 10 mL/kg. For the TNBC study, investigators diluted freshly on the day of dosing with 0.9% sodium chloride injection pre-formulated solution for injection 6 mg/mL paclitaxel (Intaxel®, Intas, India) to achieve the final concentration of 1.5 mg/mL to deliver a dose of 15 mg/kg/mouse intravenously at a dose volume of 10 mL/kg via an in-line filter (0.22 microns) unit.
TREATMENT GROUPS AND EXPERIMENTAL PROCEDURES
For the triple-negative study, treatments occurred over eight days (Days 0-7). Group 1 was the vehicle control. Groups 2 and 3 were capecitabine and CLX-155A, both dosed at 1000 mg/kg daily, respectively, on Days 0-7. Groups 4 and 5 were capecitabine and CLX-155A, both dosed at 1000 mg/kg QD and with paclitaxel at 15 mg/kg daily, respectively. Paclitaxel dosing occurred on Days 1, 3, and 6.
MEASUREMENTS AND ASSESSMENTS
The study team conducted mortality checks daily. Investigators inspected animals daily for tumors for necrosis, ulceration, wounds, and scars, as well as visible clinical signs (e.g., illness and behavioral changes) throughout the study. On the first day of treatment and three times weekly, the study team recorded animal body weights. The presence of body weight loss aided in the evaluation of treatment toxicity. The study team recorded MDA-MB-231 tumor growth on Days 0, 3, 6, 9, 12, 15, 18, 21, and 24. The study team measured tumor length and width with a digital Vernier caliper. The following calculation was used to calculate tumor volume: tumor length × (tumor width)2 × 0.52. To calculate tumor growth inhibition, tumor volume on a given day was compared to the initial measurement. If animals exhibited clinical signs of severe toxicity (e.g., >20% decrease in body weight from pre-test level, >15% decrease in body weight in a day, or tumor volumes exceeding 2000 mm3), investigators stopped treatment and humanely sacrificed the affected animal.
ANALYSIS AND STATISTICS
Investigators used Prism 5.0 for all statistical calculations. The study team evaluated the primary endpoint, tumor volume, utilizing a two-way ANOVA followed by Bonferroni’s multiple comparison tests, with a p-value <0.05 compared to sham considered significant. These investigators calculated the percent of tumor growth inhibition utilizing the equation below. The study team used the ratio between the tumor volume on the day of measurements and tumor volume on the first day of drug treatment to calculate tumor growth rate. The definition for a complete response was a tumor with a volume less than 25 mm3 for three consecutive measurements. In contrast, a partial response indicates a tumor decreased to less than 50% of its initial volume for three consecutive measurements. The study investigators reported the results as standard deviation. Where TGI is Tumor Growth Inhibition and TV is tumor volume.
Results
The study evaluated CLX-155A’s activity in a TNBC xenograft model using MDA-MB-231 cells in nude mice. It involved fifty mice divided over five groups, each receiving different treatments, including vehicle control, Capecitabine, CLX-155A, and combinations with paclitaxel. Fifty-four animals completed the study treatment: six expired in the CLX-155A and paclitaxel group. Only twelve remained over the twenty-five days involving treatment and follow-up, with five in the capecitabine group, three in the CLX-155A group, and two in each combination group. CLX-155A demonstrated a significant effect versus sham (p<0.001) on % TGI as both monotherapy and in combination with paclitaxel (Figure 3). All treatment groups showed statistically significant tumor reduction compared to the vehicle control (p<0.001, Days 3 and 6), indicating that all treatments effectively inhibit tumor growth in TNBC models. The combination of CLX-155A with paclitaxel exhibited enhanced anticancer activity compared to CLX-155A alone when compared to the vehicle, suggesting a synergistic effect.
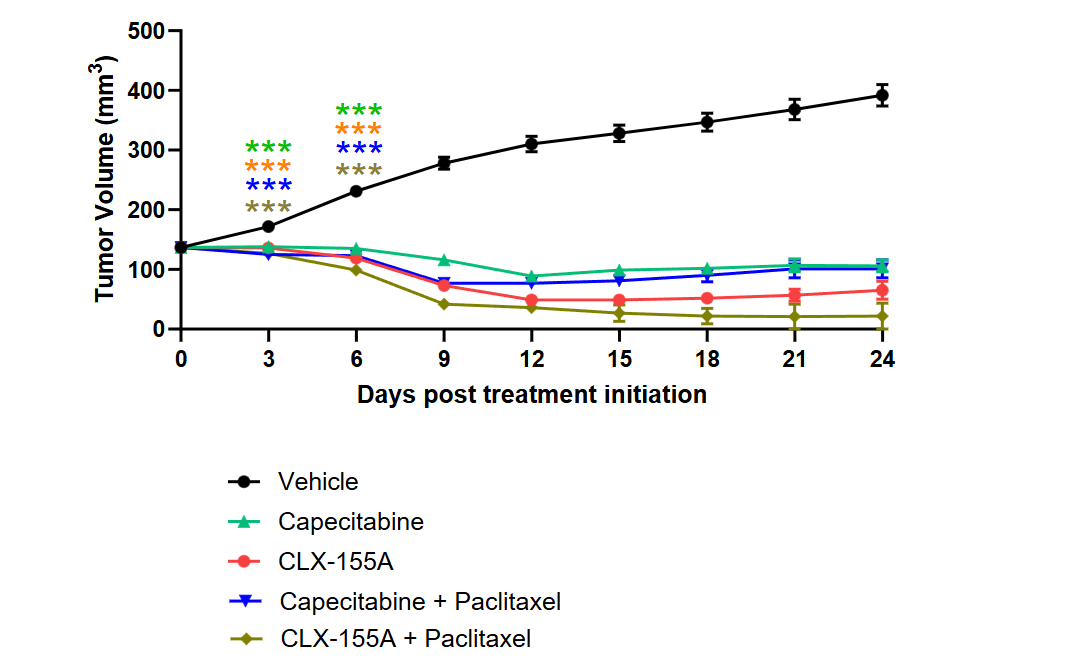
When compared with capecitabine, CLX-155A showed numerically higher % TGI activity versus sham on Day 9, both as monotherapy and in combination with paclitaxel (Figure 3). At this time point, CLX-155A monotherapy % TGI appeared equivalent to that of capecitabine and paclitaxel. The study also observed changes in body weight across different treatment groups (Figure 4). All treatment groups showed reduced weights at Days 6 (range 3%-8%) and 9 (range 13% to 22%) (p<0.001). The CLX-155A groups presented with -5 ± 1% and -22 ± 2% change in body weight on Days 6 and 9, respectively, then capecitabine with -3 ± 1% and -15 ± 2% on Days 6 and 9 (NS).
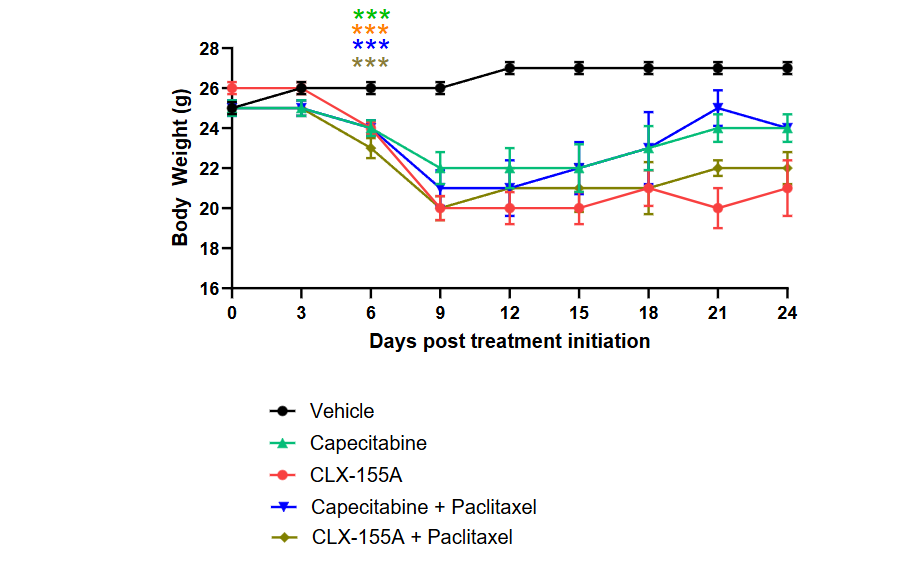
A secondary area of interest was the effect on weight changes. The percentage change findings in body weight during and after treatment reflected insights into the animals’ general health and treatments’ potential toxicities. Treatments showed significant effects on weight versus vehicle (p<0.001) as soon as Day 6 (Figure 4) with CLX-155A monotherapy at 24 ± 0.3 grams vs vehicle control at 26 ± 0.3 grams. The change in body weight in the TNBC study patterned treatment over the nine days. These effects either leveled off or reverted toward baseline during the follow-up period, reinforcing the effect of dose and schedule on this effect. CLX-155A (mono and combination therapy) showed the most pronounced effect numerically compared with those with capecitabine (NS), which was consistent with the study design since it set statistical power versus vehicle.
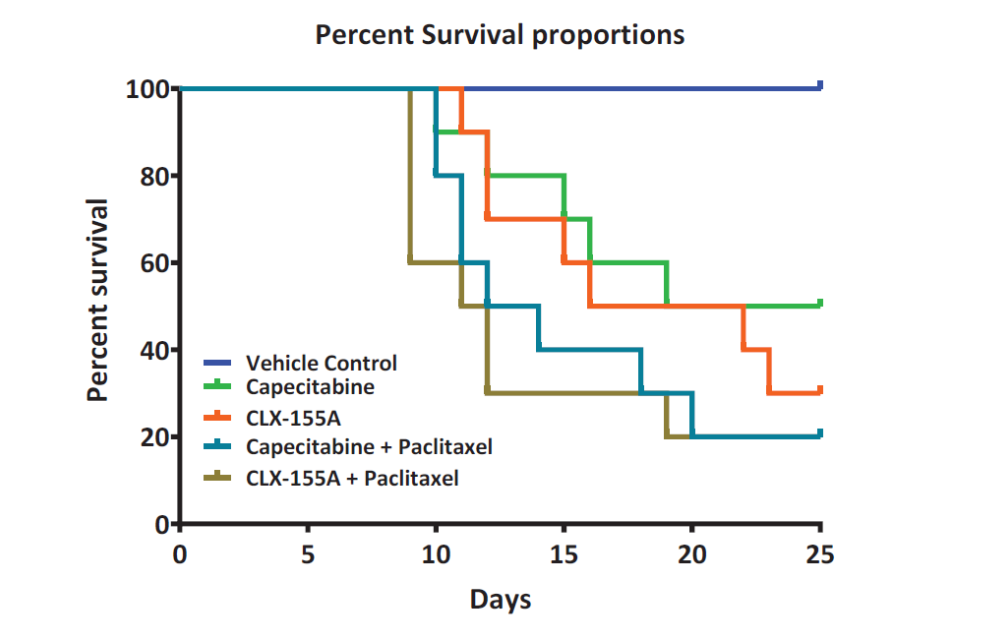
Another observation involved mortality. Again, while this finding was not a primary assessment of this study, notable differences existed. During the 25-day experiment, mortality occurred in all experimental groups, ranging from 50% to 80% mortality (Figure 5). Interestingly, all treatment groups in the TNBC study displayed similar weight loss reductions and mortality as well as significant mortality, where the drug doses were 1000 mg/kg/day. The highest mortality occurred with CLX-155A and capecitabine in combination with paclitaxel, with eight animals in each group expiring. Monotherapy with CLX-155A fared slightly better, with seven deaths. Capecitabine showed less mortality than CLX-155A; however, five animals treated with this agent expired during the study.
Discussion
In this current research, the TNBC study addressed activity based on seven days of treatment at 1000 mg/kg/day as monotherapy or combined with paclitaxel at 15 mg/kg/day. This study’s findings addressed this research question of CLX-155A’s antitumor activity in the nude mouse xenograft model of TNBC. It found differences versus vehicle in % TGI at Day 6 (101% TGI for CLX-155A, 140% TGI for CLX-155A + paclitaxel) and 9 (143% TGI for CLX-155A, 115% TGI for CLX-155A + paclitaxel) during treatment (p<0.001) and no tumor regrowth out to Day 25 in follow-up. It also observed numerically improved % TGI changes versus capecitabine (alone and in combination), with monotherapy appearing similar to that of capecitabine with paclitaxel. Further, the twice daily administration and at higher doses led to more consistent %TGI effects across animals tested. This work’s findings appear consistent with preclinical studies that have further supported capecitabine’s use in TNBC. For example, a study investigating the combination of Wee1 inhibitor AZD1775 and capecitabine in TNBC patient-derived xenograft (PDX) models found that the combination significantly inhibited tumor growth compared to either agent alone, suggesting a synergistic effect. Another study evaluated the effects of anti-PD-1 antibody therapy in TNBC PDX models, with some models also receiving capecitabine. It found varying responses, with some PDX models showing significant tumor reduction and increased survival when treated with the combination.
This study found comparable levels of mortality between groups given capecitabine and paclitaxel versus groups given CLX-155A and paclitaxel. Studies have tied capecitabine’s impact on mortality to higher doses due to toxicity. Additionally, Liu et al. highlighted that capecitabine maintenance therapy at doses of 1,000 mg/m² twice daily (approximately 54.05 mg/kg/day) in nasopharyngeal carcinoma models resulted in manageable toxic effects but also noted instances of progression to death. Midgley and Kerr (2008) discussed the challenges of determining the optimal dose of capecitabine to balance efficacy and safety, noting that high doses, such as 1,250 mg/m² twice daily (approximately 67.57 mg/kg/day), can lead to significant toxicity and mortality. Reichardt et al. also reported increased mortality at doses of 1,250 mg/m² twice daily (approximately 67.57 mg/kg/day) in metastatic breast cancer models. Zielinski et al. further confirmed that doses of 1,000-1,250 mg/m² twice daily (approximately 54.05-67.57 mg/kg/day) could result in significant toxicity and increased mortality. Furthermore, studies involving combination therapy with paclitaxel have shown that while the combination can enhance antitumor efficacy, it can also contribute to increased toxicity and mortality at higher doses, such as 1,250 mg/m² of capecitabine combined with standard doses of paclitaxel.
As seen in this study, body weight reductions occurred and are not uncommon in preclinical animal cancer studies. Numerous preclinical studies report capecitabine to cause weight loss. The drug’s gastrointestinal toxicity can reduce food intake, thus causing weight loss. In a study by Ishikawa et al., it was observed that animals treated with capecitabine experienced notable weight loss, particularly at higher doses. Researchers noted that higher doses lead to more pronounced weight loss. Notably, these findings on weight loss and mortality highlight the need to minimize adverse effects while maximizing the therapeutic benefits by employing precise dose management in preclinical studies.
Differences between capecitabine and CLX-155A may be due to the presence of VPA in CLX-155A, which provides additional insight into its activity in these models. Intestinal enzymes cleave the prodrug, leading to the formation of the active compound VPA and the 5-FU precursor, 5′-DFCR (Figure 1). VPA, primarily recognized as an anticonvulsant and mood stabilizer, inhibits voltage-gated sodium and calcium channels and enhances GABAergic neurotransmission. It also exhibits other properties, such as histone deacetylase inhibition, which can induce changes in gene expression to promote cell cycle arrest, differentiation, and apoptosis. Additionally, VPA can cause cell cycle arrest, induce apoptosis, and exert antiangiogenic effects. VPA enhances the efficacy of other anticancer treatments, including chemo and radiotherapy, through its ability to sensitize cancer cells to these treatments. Additionally, VPA shows activity in multiple animal cancer models, including bladder, pancreatic, glioma, and breast cancer. VPA potentiates capecitabine’s anticancer activity via thymidine phosphorylase expression induction in the breast cancer model by Terranova-Barberio et al.
Due to the 1000 mg/kg/day dose, weight loss appears to be an issue that aligns with the findings in a CRC study the investigators also conducted. Comparing the two studies, the TNBC evaluation appeared to have higher animal mortality rates than the CRC study, with the highest mortality rate in the combination groups. Dose, combination therapy, disease, and schedule may contribute to these observations. To date, vorinostat, romidepsin, belinostat, and panobinostat are the only US Food and Drug Administration-approved HDAC inhibitors for the treatment of hematological malignancies. Studies show limited success with HDAC-inhibitor in treating solid tumors despite their positive experience in hematological malignancies. Conversely, several studies suggest that HDAC inhibitors, when combined with another agent, can produce additional antitumor activity. For example, in a mouse tumor model using syngeneic mice, the co-administration of a low dose of trichostatin-A, a HDAC inhibitor, along with anti-PD-L1, resulted in enhanced tumor reduction and extended survival in mice with tumors, surpassing the effects observed with either treatment alone. In a separate study involving xenograft models utilizing cell lines from various solid tumor lineages, the combination of paclitaxel with either clinostat or ACY-241, both HDAC inhibitors, exhibited enhanced cell proliferation inhibition and increased cell death in comparison to the individual agents alone.
HDAC inhibitors have demonstrated the potential for clinical applications in recent research in gliomas, pancreatic cancer, and breast cancer. Investigators have initiated multiple clinical studies in solid tumors, advanced solid tumors including glioblastoma, head and neck cancer, and acute myeloid leukemia or myelodysplastic syndromes.
Like all research, while this study provides valuable insights, it is important to recognize its limitations. Notable is the use of the MDA-MB-231 TNBC xenograft model in FoxN1 athymic nude mice. While xenograft models provide valuable insights into antitumor efficacy, they have limitations due to the absence of an intact immune system. The lack of immune responses in these models may not fully capture the intricate interactions between the immune system and the tumor microenvironment observed in human subjects. The model employed in this study addressed a specific question as an initial activity indicator. Still, its inability to replicate tumor metastasis does not entirely represent advanced disease. The MDA-MB-231 TNBC model provides additional insights into the drug’s efficacy across different cancer types. Still, additional models for breast cancer and other solid tumors would enhance CLX-155A’s activity profile. Moreover, introducing the unique CLX-155A in this study brings a novel aspect, but it requires a cautious interpretation of the results. The specific metabolic conversions and subsequent release of active compounds (e.g., valproic acid) in vivo warrant further clarification, particularly considering potential variations across different tumor types or patient populations.
Additionally, it is crucial to acknowledge the limited translatability of preclinical oncology studies to successful clinical treatments, as only approximately 15% of preclinical findings have been effective in clinical applications. This limited translatability emphasizes the necessity for thorough and diverse preclinical testing to enhance the probability of achieving clinical success. Hence, further research in more diverse models to confirm and broaden the activity profile of CLX-155A while defining the dose and safety profile will enhance the package for regulatory submission and first-in-human testing.
Conclusion
Overall, the results address the overarching research questions. The findings indicate that CLX-155A is a promising candidate within the TNBC treatment model. CLX-155A promotes a significant %TGI reduction at all doses (p<0.01) versus vehicle as early as Day 8 of treatment. This investigation in TNBC reflects a significant favorable %TGI activity versus vehicle (p<0.001) at 1000 mg/kg/day dose as monotherapy or combined with paclitaxel. It also displays encouraging numerical trends when compared with capecitabine at the same dose and combination, with CLX-155A monotherapy appearing similar to capecitabine with paclitaxel in %TGI. This study sets the stage for further preclinical investigation to elucidate activity, safety, and pharmacokinetics so this compound can move into clinical testing.
Conflicts of Interest Statement
Subbu Apparsundaram, PhD, and Mahesh Kandula, MTech, MBA are Directors in Cellix Biosciences, Inc and Cellix Bio Private Limited. Michika Maeda, MD and Giovanni Lara, PharmD are Post-Doctoral Fellows at Novartis. John York, PharmD, MBA is a Consultant to Cellix Biosciences, Inc, COASTAR Therapeutics, Crestec Therapeutics, HRA Rare Disease, JD Biosciences, Reviva Pharmaceuticals and Teikoku Pharma USA.
Funding Statement
None
Acknowledgments
CLX-155A Patents US20210171564 and WO2020/026054
References
- Longley DB, Harkin DP, Johnston PG. 5-Fluorouracil: Mechanisms of action and clinical strategies. Nature Reviews Cancer. 2003;3(5):330-338. doi:10.1038/nrc1074
- Grem JL. 5-Fluorouracil: Forty-plus and still ticking. A Cancer Journal for Clinicians. 2000;50(6):345-358. doi:10.3322/canjclin.50.6.345
- Rosen F, Muggia F, Jeffers S, Waugh W. Biological modification of protracted infusion of 5-fluorouracil with weekly leucovorin: A dose-seeking clinical trial for patients with disseminated gastrointestinal cancers. Cancer Chemotherapy. Springer; 1985:25-35.
- Saif MW, Choma A, Salamone SJ, Chu E. Pharmacokinetically guided dose adjustment of 5-fluorouracil: A rational approach to improving therapeutic outcomes. JNCI: Journal of the National Cancer Institute. 2009;101(22):1543-1552. doi:10.1093/jnci/djp328
- Amorim LC, Peixoto RD. Should we still be using bolus 5-FU prior to infusional regimens in gastrointestinal cancers? Curr Colorectal Cancer Rep. 2021;17(5):49-54. doi:10.1007/s11888-021-00505-4
- Barathan M, Kumar R, Singh A, Patel S. Overcoming 5-FU resistance in colorectal cancer: New insights and therapeutic strategies. Cancer Treatment Reviews. 2024;104:102345. doi:10.1016/j.ctrv.2024.102345
- Cassidy J, Twelves C, Cutsem E, et al. Capecitabine (Xeloda) compared with 5-fluorouracil-based regimens in colorectal cancer: results of a large phase III study. Journal of Clinical Oncology. 2002;20(11):2282-2292. doi:10.1200/JCO.2002.09.005
- Cassidy J, Twelves C, Van Cutsem E, et al. First-line oral capecitabine therapy in metastatic colorectal cancer: a favorable safety profile compared with intravenous 5-fluorouracil/leucovorin. Ann Oncol. Apr 2002;13(4):566-75. doi:10.1093/annonc/mdf089
- Blum JL, Jones SE, Buzdar AU, et al. Multicenter phase II study of capecitabine in paclitaxel-refractory metastatic breast cancer. Journal of Clinical Oncology. 2001;17(2):485-493. doi:10.1200/JCO.2001.172.485
- Sikora K, Zhang L, Li Y. Effect of capecitabine maintenance therapy using lower dosage and higher frequency vs observation on disease-free survival among patients with early-stage who had received standard treatment: The SYSUCC-001 randomized clinical trial. JAMA. 2015;314(24):2641-2652. doi:10.1001/jama.2015.16017
- Garg P, Malhotra J, Kulkarni P, Horne D, Salgia R, Singhal SS. Emerging therapeutic strategies to overcome drug resistance in cancer cells. Cancers. 2024;16(13):2478. doi:10.3390/cancers16132478
- Tian Y, Wang X, Wu C, Qiao J, Jin H, Li H. A protracted war against cancer drug resistance. Cancer Cell International. 2024;24, Article 326 doi:10.1186/s12935-024-03510-2
- Feliu J, Heredia-Soto V, Gironés R, et al. Management of the toxicity of chemotherapy and targeted therapies in elderly cancer patients. Clin Transl Oncol. Apr 2020;22(4):457-467. doi:10.1007/s12094-019-02167-y
- Baranova A, Krasnoselskyi M, Starikov V, et al. Triple-negative breast cancer: current treatment strategies and factors of negative prognosis. J Med Life. Feb 2022;15(2):153-161. doi:10.25122/jml-2021-0108
- National Comprehensive Cancer Network. Breast Cancer (Version 3.2025). NCCN Clinical Practice Guidelines in Oncology. Published March 2025. Accessed April 4, 2025. https://www.nccn.org/guidelines/guidelines-detail?id=1419
- Suresh D, Kumar R, Singh A. Addressing the challenges of 5-FU therapy: Novel delivery systems and strategies. Drug Delivery. 2020;27(1):123-134. doi:10.1080/10717544.2020.1717523
- Williams ML, Smith JR, Johnson ME, Brown CS, Davis LK. Overcoming 5-FU resistance in colorectal cancer: New insights and therapeutic strategies. Cancer Treatment Reviews. 2018:104,-102345. doi:10.1016/j.ctrv.2018.102345
- Bryson HM, Sorkin EM, McTavish D. Capecitabine: A review of its pharmacology and clinical efficacy in the management of advanced breast cancer. Drugs. 2023;56(1):37-65. doi:10.2165/00003495-2023560100-00004
- Visacri MB, Lima TM, Mazzola PG. Capecitabine-induced hand-foot syndrome: A review of current management and preventive strategies. Journal of Oncology Pharmacy Practice. 2022;28(1):123-134. doi:10.1177/1078155221101234
- Xoloda PI. Capecitabine prescribing information. 2024.
- Boyette N, Dalton A, Tak Y, et al. CLX-155: A Novel, Oral 5-FU Prodrug Displaying Antitumor Activity in Human Colon Cancer Xenograft Model in Nude Mice. Medical Research Archives. 2024;12(6) doi:https://doi.org/10.18103/mra.v12i6.5219.
- Anderson RC, Salyers AA. Antibacterial effects of caprylic acid on Escherichia coli and Salmonella enterica. Journal of Food Protection. 2006;69(7):1636-1641.
- Bergsson G, Arnfinnsson J, Steingrímsson Ó, Thormar H. In vitro killing of Candida albicans by fatty acids and monoglycerides. Antimicrobial Agents and Chemotherapy. 2001;45(11):3209-3212.
- Isaacs CE, Litov RE, Thormar H. Antimicrobial activity of lipids added to human milk, infant formula, and bovine milk. Journal of Nutrition and Biochemistry. 1995;6(7):362-366.
- Liu Y, Wang X. Caprylic acid suppresses inflammation through modulation of the TLR4/NF-κB signaling pathway in atherosclerosis. Journal of Nutritional Biochemistry. 2018;57:56-64.
- Yoon BK, Jackman JA, Valle-González ER, Cho NJ. Antibacterial free fatty acids and monoglycerides: biological activities, experimental testing, and therapeutic applications. International Journal of Molecular Sciences. 2018;19(4):1114.
- Zhao J, Hu J, Ma X. Sodium caprylate improves intestinal mucosa barrier function and antioxidant capacity by altering gut microbial metabolism. Journal of Animal Science and Biotechnology. 2021;12(1):1-14.
- Narayanan NK, Narayanan BA, Nixon DW. Caprylic acid in cancer therapy: A review. Journal of Cancer Research and Therapeutics. 2015;11(3):543-548. doi:10.4103/0973-1482.157334
- York JM, Kang S, Dalton A, Tak Y, Boyette N, Kandula M, Apparsundaram S. Pharmacokinetics of Single-dose CLX-155 and Metabolites in Female Balb/C Mice. Med Res Arch. 2024;12(9). doi:10.181-03/mra.v12i9.5709.
- Göttlicher M, Minucci S, Zhu P, et al. Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. The EMBO Journal. 2001;20(24):6969-6978. doi:10.1093/emboj/20.24.6969
- Blaheta RA, Cinatl J. Anti-tumor mechanisms of valproate: A novel role for an old drug. Medical Research Reviews. 2002;22(5):492-511. doi:10.1002/med.10023
- Michaelis M, Michaelis UR, Fleming I, et al. Valproic acid is an anticancer drug. Current Pharmaceutical Design. 2004;10(21):2619-2635. doi:10.2174/1381612043383792
- Munster PN, Marchion D, Bicaku E, et al. A phase II study of valproic acid in patients with advanced cancer. Cancer Chemotherapy and Pharmacology. 2009;64(4):733-740. doi:10.1007/s00280-009-0923-0
- Phiel CJ, Zhang F, Huang EY, Guenther MG, Lazar MA, Klein PS. Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. The Journal of Biological Chemistry. 2001;276(39):36734-36741. doi:10.1074/jbc.M101287200
- Marks PA, Richon VM, Breslow R, Rifkind RA. Histone deacetylase inhibitors as new cancer drugs. Curr Opin Oncol. Nov 2001;13(6):477-83. doi:10.1097/00001622-200111000-00010
- Pulaski BA, Ostrand-Rosenberg S. Mouse 4T1 Breast tumor model. Curr Prot Immunology. 2001;(Chap 20 Unit 20.2) doi:https://doi.org/10.1002/0471142735.im2002s39
- Contreras-Zárate MJ, Ormond DR, Gillen AE, et al. Development of Novel Patient-Derived Xenografts from Breast Cancer Brain Metastases. Front Oncol. 2017;7:252. doi:10.3389/fonc.2017.00252
- Pitts TM, Simmons DM, Bagby SM, et al. Wee1 Inhibition Enhances the Anti-Tumor Effects of Capecitabine in Preclinical Models of Triple-Negative Breast Cancer. Cancers (Basel). Mar 19 2020;12(3) doi:10.3390/cancers12030719
- Rosato RR, Dávila-González D, Choi DS, et al. Evaluation of anti-PD-1-based therapy against triple-negative breast cancer patient-derived xenograft tumors engrafted in humanized mouse models. Breast Cancer Res. Sep 5 2018;20(1):108. doi:10.1186/s13058-018-1037-4
- Shah AN, Flaum L, Helenowski I, et al. Phase II study of pembrolizumab and capecitabine for triple negative and hormone receptor-positive, HER2-negative endocrine-refractory metastatic breast cancer. J Immunother Cancer. Feb 2020;8(1) doi:10.1136/jitc-2019-000173
- Midgley R, Kerr DJ. Capecitabine: have we got the dose right? Nature Clinical Practice Oncology. 2008;5(12):682-692. doi:10.1038/ncponc1240
- Reichardt P, Minckwitz G, Thuss-Patience PC, et al. Multicenter phase II study of oral capecitabine (Xeloda) in patients with metastatic breast cancer relapsing after treatment with a taxane-containing therapy. Annals of Oncology. 2003;14(8):1227-1233. doi:10.1093/annonc/mdg328
- Zielinski C, Gralow J, Martin M. Optimizing the dose of capecitabine in metastatic breast cancer: confused, clarified or confirmed? Annals of Oncology. 2010;21(11):2145-2152. doi:10.1093/annonc/mdq258
- Liu GY, Li WZ, Wang DS, et al. Effect of Capecitabine Maintenance Therapy Plus Best Supportive Care vs Best Supportive Care Alone on Progression-Free Survival Among Patients with Newly Diagnosed Metastatic Nasopharyngeal Carcinoma Who Had Received Induction Chemotherapy: A Phase 3 Randomized Clinical Trial. JAMA Oncology. 2022;8(2):234-243. doi:10.1001/jamaoncol.2021.6163
- Gradishar WJ, Meza LA, Amin B, et al. Capecitabine plus paclitaxel as front-line combination therapy for metastatic breast cancer: A multicenter phase II study. Journal of Clinical Oncology. 2004;22(12):2321-2327. doi:10.1200/JCO.2004.12.128
- Ishikawa T, Sekiguchi F, Fukase Y, Sawada N, Ishitsuka H. Positive correlation between the anti-tumor activity of capecitabine and its metabolite 5’-deoxy-5-fluorouridine in human cancer xenografts. Clinical Cancer Research. 1998;4(4):1013-1019.
- Brodie SA, Brandes JC. Could valproic acid be an effective anticancer agent? The evidence so far. Expert Review of Anticancer Therapy. 2014;14(10):1097-1100. doi:10.1586/14737140.2014.940329
- Han W, Guan W. Valproic Acid: A Promising Therapeutic Agent in Glioma Treatment. Frontiers in Oncology. 2021;11:687362.
- Jonaid A. The evidence for repurposing anti-epileptic drugs to target cancer. Molecular Biology Reports. 2023;50:7667-7680.
- Nakatsuji T, Kao MC, Fang JY, et al. Antimicrobial property of lauric acid against Propionibacterium acnes: its therapeutic potential for inflammatory acne vulgaris. Journal of Investigative Dermatology. 2009;129(10):2480-2488.
- Tsai HC. Valproic Acid Enhanced Temozolomide-Induced Anticancer Activity in Human Glioma Through the p53–PUMA Apoptosis Pathway. Frontiers in Oncology. 2021;11:722754. doi:10.3389/fonc.2021.722754
- Wang LL, Johnson EA, Ray B. Inhibition of Listeria monocytogenes by fatty acids and monoglycerides. Applied and Environmental Microbiology. 1994;60(11):4172-4177.
- Wang D, Ning W, Zhao L, Chen S, Li X. Inhibitory effect of valproic acid on bladder cancer in combination with chemotherapeutic agents in vitro and in vivo. This study demonstrates the potential of valproic acid as a promising component in the treatment of bladder cancer. Cancer Letters. 2013;335(2):201-209. doi:10.1016/j.canlet.2013.02.037
- Sun G, Kashiwakura G, Komatsu N, Aoki Y, Matsumoto K, Saito Y. The histone deacetylase inhibitor valproic acid induces cell growth arrest in hepatocellular carcinoma cells via suppressing Notch signaling. Journal of Experimental & Clinical Cancer Research. 2015;34:125. doi:10.1186/s13046-015-0231-3
- Terranova-Barberio MS. Valproic acid potentiates the anticancer activity of capecitabine in vitro and in vivo in breast cancer models via induction of thymidine phosphorylase expression. Breast Cancer Research and Treatment. 2016;155(3):425-435.
- York JM, Maeda M, Lara G, Kandula M, Apparsundaram S. Preclinical Evaluation of CLX-155A: A Novel 5-FU and Valproic Acid Prodrug in Nude Mouse Model for Activity in Colon Cancer. Medical Research Archives. 2025.
- Yoon S, Eom G. HDAC and HDAC Inhibitor: From Cancer to Cardiovascular Diseases. Chonnam Med J. 2016;52(1):1-11. doi:10.4068/cmj.2016.52.1.1
- Hu Z, Wei F, Su Y, et al. Histone deacetylase inhibitors promote breast cancer metastasis by elevating NEDD9 expression. Signal transduction and Targeted Therapy. 2023;8:11. doi:10.1038/s41392-022-01221-6
- West A, Johnstone R. New and emerging HDAC inhibitors for cancer treatment. The Journal of Clinical Investigation. 2014;124(1):30-39. doi:10.1172/JCI69738
- Li X, Su Z, Liu R, et al. HDAC inhibition potentiates anti-tumor activity of macrophages and enhances anti-PD-L1-mediated tumor suppression. Oncogene. Mar 2021;40(10):1836-1850. doi:10.1038/s41388-020-01636-x
- Huang P, Almeciga-Pinto I, Jarpe M, et al. Selective HDAC inhibition by ACY-241 enhances the activity of paclitaxel in solid tumor models. Oncotarget. 2017;8(2):2694-2707.
- Lipska K, Gumieniczek A, Filip AA. Anticonvulsant valproic acid and other short-chain fatty acids as novel anticancer therapeutics: Possibilities and challenges. Acta Pharmaceutica. 2020;70(3):291-301. doi:10.2478/acph-2020-0021
- Budillon A, Leone A, Passaro E, et al. Randomized phase 2 study of valproic acid combined with simvastatin and gemcitabine/nab-paclitaxel-based regimens in untreated metastatic pancreatic adenocarcinoma patients: the VESPA trial study protocol. BMC Cancer. 2024;24(1) doi:10.1186/s12885-024-12936.
- M.D. Anderson Cancer Center. Bevacizumab and Temsirolimus Alone or in Combination with Valproic Acid or Cetuximab in Treating Patients with Advanced or Metastatic Malignancy or Other Benign Disease. ClinicalTrials.gov. Updated October 23, 2024. Accessed October 25, 2024, https://clinicaltrials.gov/ct2/show/NCT01552434
- Camphausen K. Valproic Acid With Temozolomide and Radiation Therapy to Treat Brain Tumors. ClinicalTrials.gov. Updated August 18, 2016. Accessed October 1, 2024, https://clinicaltrials.gov/study/NCT00302159
- Barretos Cancer Hospital. Chemoprevention of Head and Neck Squamous Cell Carcinoma (HNSCC) With Valproic Acid (GAMA. Updated February 1, 2018. Accessed October 1, 2024, https://clinicaltrials.gov/study/NCT02608736
- University of Kansas. Valproic Acid and Decitabine in Treating Patients With Acute Myeloid Leukemia or Myelodysplastic Syndromes. ClinicalTrials.gov. Updated March 26, 2013. Accessed October 1, 2024, https://clinicaltrials.gov/study/NCT01130662
- Brown P, Smith L, Jones M. The role of MDA-MB-231 in triple-negative breast cancer research. Breast Cancer Research and Treatment. 2018;170(2):345-356. doi:10.1007/s10549-018-4756-7
- Seyhan AA. Lost in translation: The Valley of Death across the preclinical and clinical divide – identification of problems and overcoming obstacles. Translational Medicine Communications. 2019;4, Article 18 doi:10.1186/s41231-019-0050-7

