Precocious Puberty Linked to Endocrine Disruptors
Precocious Puberty in Association to Exposure to Endocrine Disrupting Chemicals: A Case Series
Julianne M. Hall¹ and Lee Ann Serrano²
- Department of Medical Sciences, Frank H. Netter MD School of Medicine, Quinnipiac University, NH-MED, 300 Bassett Road, North Haven, CT 06473, USA
- Frank H. Netter MD School of Medicine, Quinnipiac University, NH-MED, 300 Bassett Road, North Haven, CT 06473, USA
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Hall, JM., and Serrano, LA., 2026. Precocious Puberty in Association to Exposure to Endocrine Disrupting Chemicals: A Case Series. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i1.7133
ISSN 2375-1924
ABSTRACT
Endocrine disrupting chemicals are ubiquitously present in our environment, and it has become apparent that these agents pose significant health risks. A number of endocrine disrupting chemicals have been detected in humans at levels capable of interfering with endogenous hormone action. Notably, there are well-established effects on male and female sexual development as a consequence of either prenatal or childhood exposure. Given the observed accelerated rate of puberty in the past few decades, it has been postulated that contact with endocrine disrupting chemicals during critical development periods may be a causative factor in promoting precocious puberty in children. This case series presents patient evidence correlating endocrine disrupting chemicals exposure with clinical evidence of premature sexual development. The identification of numerous cases of precocious puberty related to exposures highlights the need for physicians to take an active role in educating parents about the risks posed during pregnancy and child development.
Keywords: Endocrine disrupting chemicals; EDCs; precocious sexual development; puberty; Sexual Maturity Rating; hypothalamic-pituitary-gonadal axis; HPG axis
1. Introduction
Endocrine disrupting chemicals (EDCs) are natural or synthetic agents in the environment that alter physiological hormone functions. Some of the most commonly encountered EDCs include bisphenol A, phytoestrogens, phthalate esters, pesticides, dioxins, polychlorinated biphenyls, and flame retardants. EDCs are highly present globally, as they are found in many household and environmental products, including plastic toys and bottles, lining of food cans, personal care products, medical devices and tubing, household furnishings, and contaminated food, water, and soil. Importantly, EDCs have been detected in human blood, urine, hair, sweat, breast milk, placental tissue, and amniotic fluid at levels sufficient to disrupt endocrine function. Exposure to EDCs at various stages of life has been associated with the abnormal development and function of numerous organ systems, including the nervous, endocrine, cardiovascular, renal, immune, and reproductive systems. Specifically, EDC actions on adipose tissue, the adrenal glands, and the hypothalamic-pituitary-gonadal (HPG) axis may affect reproductive function at multiple points during fetal and neonatal life, early childhood, puberty, and adulthood.
2. Puberty
Puberty is defined as the development of secondary sexual features and the ability to reproduce. Driven by activation of the HPG axis and a balance of hormones, on average puberty begins between the ages of eight to thirteen in females and nine to fourteen in males. Typical puberty begins with the release of gonadotropin-releasing hormone (GnRH) from the hypothalamus. GnRH stimulates release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the anterior pituitary in a pulsatile fashion. LH acts on the interstitial cells of the gonads, to initiate steroidogenesis. Specifically, ovarian thecal cells and testicular Leydig cells synthesize androgens in response to LH. FSH acts on ovarian follicular cells to promote conversion of androgens to estrogens, and on ovarian oocytes to stimulate development. In testes, FSH stimulates spermatogenesis and enables maturation of spermatozoa.
During pubertal development, estrogen accelerates breast growth, stimulates development of pubic hair, as well as increases growth of the female genital tract. In males, testosterone expands testes length and stimulates development of pubic hair. In females and males, both hormones act to increase bone length, while testosterone also enlarges bone width, whereas estrogen stimulates closure of the epiphyseal plates. In females, increasing concentrations of estrogen also enable the female neuroendocrine system to secrete LH. LH then acts on the ovary to prepare for ovulation and menarche.
In addition, the adrenal glands also play a contributory role in puberty as a source of sex steroid hormones. Adrenocorticotropin releasing hormone (ACTH) release from the anterior pituitary stimulates adrenal production of dehydroepiandrosterone (DHEA), which is then converted to testosterone and other forms of estrogen in peripheral tissues. While the gonads are the primary source of sex hormones in both males and females, the adrenal glands contribute a small amount of sex hormones through this pathway.
In the United States, puberty is staged by the Tanner scale which is also known as Sexual Maturity Rating. Sexual Maturity Rating (SMR) is more widely used in literature and will therefore be used in this case series. (Table 1). SMR stage 1 refers to the prepubertal stage. SMR stage 2 is defined as onset of thelarche in females and testicular enlargement in males. Ongoing puberty is classified as SMR stages 3-4, and completion of puberty is classified as SMR stage 5. In females, puberty typically starts with development of thelarche, followed by pubarche and menarche. In males, puberty typically starts with lengthening of the testes and pubarche.
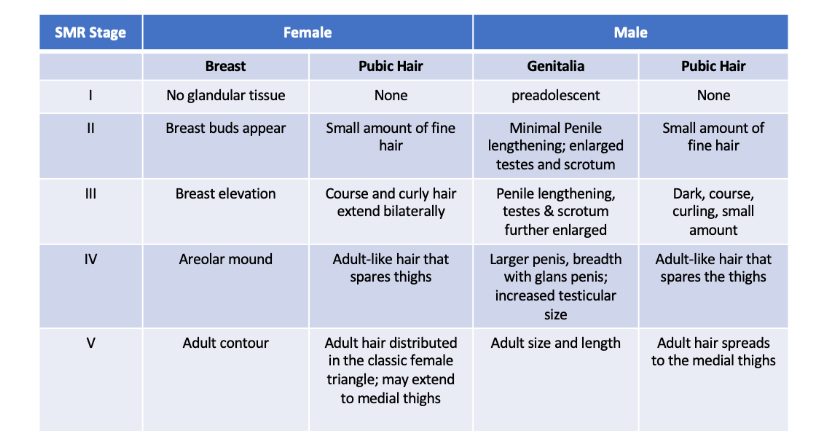
3. Case Presentations: Precocious Puberty
3.1. SEXUAL MATURITY RATING 2
Multiple case studies were found in which children were experiencing precocious puberty, classified as SMR stage 2. At the University of California Irvine Pediatric Endocrinology Division and Children’s Hospital of Orange County’s clinic, four patients were evaluated. The first was a 7 year 6-month-old female who presented after concern for development of a left breast bud at 6 years of age in the context of normal prepubertal FSH, LH, and estradiol levels. At the time of evaluation, breast development was SMR stage 2. The second patient was a 3 year 11-month-old female who had right sided breast development starting at age one; no data was reported on prepubertal gonadotropin and gonadal hormone levels. The third patient was a 7 year 9-month-old female who developed a left breast bud at 6 years of age; no data was provided on hormones. The fourth patient was a 7 year 11-month-old male who developed a left breast bud at four years of age. At the time of evaluation, he had bilateral breast enlargement and bilateral testicular volume, with normal gonadotropin and sex steroid hormones, and normal prolactin levels. Thorough histories of the 4 patients were obtained, which revealed that each child had exposure to lavender, in the forms of cologne, soap, oil in a diffuser, and again cologne, respectively. After discontinuation of the exposure, breast development had resolved in patients 1, 2, and 4, and regressed in patient 3.
Similarly, a 14-month-old female was referred to an endocrinology clinic for concern of breast development starting at six months of age. She had no accelerated growth development and otherwise normal developmental milestones. Her history revealed frequent exposure to high levels of lavender oil in a wide variety of formulations. The patient’s parents reported routine use of lavender wipes, lavender shampoo, lavender body wash, lavender moisturizing cream, and lavender skin soothing lotion. The child’s gonadotropin and estradiol levels were normal. However, elevated prolactin was observed as well as marked increase of the tumor marker alpha-fetoprotein. After the parents were counseled on discontinuing these products, the patient’s breast tissue receded.
Precocious puberty was also seen with exposure to phytosterol, an EDC with established actions on the HPG axis. A 20-month-old male was evaluated for concern of pubic hair and increased penile length. Pubarche was classified as SMR stage 2. After a thorough history was obtained, it was discovered that this patient was consuming large amounts of chicken essence seasoning, which contained phytosterol. No gonadotropin or sex steroid hormone levels were reported. After discontinuing the exposure, the patient’s pubarche and penile lengthening halted.
3.2. SEXUAL MATURITY RATING 3
Multiple case studies of patients with SMR stage 3 thelarche were documented in Turkey. A five-year-old patient presented to the clinic for evaluation of thelarche. The patient was also found to be experiencing vaginal discharge as well. Similarly, three additional female pediatric patients, ages 5 months, three and five years, were identified who were also experiencing thelarche, SMR stage 3. All 4 pediatric patients presented with estradiol levels 15-20 times higher than appropriate for their ages. Parent interviews revealed that the pediatric patients had all been heavy consumers (2-3 times daily) of herbal tea, containing the phytoestrogen Foeniculum vulgare, to alleviate gas pain. Coincidentally, at the same time, a 65-year-old postmenopausal patient was evaluated for postmenopausal bleeding, presenting as regular menstrual cycles. It was discovered that the 65-year-old patient had a long-term history of drinking Foeniculum vulgare tea on a daily basis. The parents were instructed to stop administering tea to their children, after which thelarche resolved and estradiol levels normalized within three to six months. No follow up data was provided for the adult patient.
3.3. SEXUAL MATURITY RATING 4
Cases of precocious puberty, classified as SMR stage 4, were also documented in the literature. In France, a four-month-old female was evaluated for SMR stage 4 breasts as well as vaginal bleeding. It is widely understood that newborns can have breast buds because of maternal hormonal transfer in utero, however it was inexplicable why the patient was also experiencing menstruation. Endocrine investigation revealed highly elevated estradiol, and Gonadotropin stimulation resulted in marked increases in FSH, and moderate elevation of LH. The physicians concluded that the child was experiencing peripheral precocious puberty. The patient history mentioned no excessive soy/phytoestrogen intake, however, the family resided on a farm that stored more than 20 tons of several pesticides with known endocrine disrupting activities (i.e. DDT, endosulfan). It was determined that exposure to several pesticides was the cause of the patient’s precocious puberty, after increased plasma levels were detected in that of the patient and the parents.
Similarly, in Morocco, a five-year-old female presented to a pediatric endocrine clinic for breast development, SMR stage 4, as well as SMR stage 1 pubarche (downy pubic hair). Breast enlargement had been observed beginning at 4 months of age. Family history was unremarkable, and the child had no history of neurological, or behavioral deficits and an otherwise normal perinatal developmental experience. On pelvic ultrasound, the child displayed a uterine axis twice the normal length, increased ovarian follicles, and 9 mm diameter follicles, the size associated with ovulation. Endocrine testing revealed highly elevated estradiol, FSH, and LH. Gonadotropin stimulation testing resulted in marked increases in FSH, and moderate elevation of LH, and an LH/FSH ratio > 1. Based on these findings, the child was diagnosed with peripheral precocious puberty. Though the parents reported no excessive phytoestrogen intake, it was noted that the patient lived on a farm with widespread exposure to endocrine-disrupting pesticides. Per recommendations from the medical providers, the family moved to a pesticide-free residence. A causal link between EDC exposure and pubertal symptoms was established when the patient’s hormonal balance, and uterine and ovarian ultrasounds normalized after a year living in the new environment.
4. Discussion
This case series highlights multiple deviations from typical pubertal onset and maturation in response to exposure to EDCs. Precocious puberty is defined as onset of puberty before eight years of age in females and nine years of age in males. Precocious puberty may be the consequence of various factors including improved public health measures, which may result in improved nutrition among children. However, exposure to EDCs may account for earlier onset of puberty in females and males, given the established actions of these agents in adipose tissue and on the HPG axis.
Essential oils, such as lavender, are known to display estrogenic and antiandrogenic effects in vitro and possibly in vivo effects, as evidenced by the multiple case reports demonstrating prepubertal-aged children with SMR stage 2 development. In addition, components of herbal teas can also cause individuals to undergo precocious puberty. Pesticides have also been described to be estrogen receptor agonists as well as androgen receptor antagonists. In addition to having estrogenic and antiandrogenic effects, EDCs can also predispose individuals to obesity, another risk factor for precocious puberty.
Rates of exposure to EDCs and other exogenous chemicals can vary by location, occupation, socioeconomic status, race, and other factors that determine quality of living. This is evidenced in this series, as cases of precocious puberty were described in the United States, China, Turkey, France, and Morocco. However, there are global efforts in place by the World Health Organization to develop policies regarding the use of EDCs and to decrease exposures. For example, some chemicals in pesticides have been banned by the European Union.
5. Conclusion
Exposure to EDCs over a lifetime can lead to dysfunction of the HPG axis and may disproportionately affect those exposed during infancy and childhood. This study aimed to highlight case reports of precocious puberty seen in association to EDCs. Continued research to further establish the relationship between EDCs and precocious puberty is necessary to prevent further health disparities between people of different backgrounds, to improve public health outcomes, and to promote patient education and health literacy.
Author Contributions: Conceptualization, L.A.S. and J.M.H.; methodology, L.A.S.; investigation, L.A.S. and J.M.H.; writing—original draft preparation, L.A.S. and J.M.H.; writing—review and editing, J.M.H.; supervision, J.M.H. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement: Not applicable.
Informed Consent Statement: Patient consent was waived due to the fact that this manuscript summarized previous publications of patient data for which informed consent was granted.
Acknowledgments: The authors thank the Scholarly Reflection and Concentration Capstone Program at the Frank H. Netter MD School of Medicine for supporting scholarly projects for medical students and faculty.
Conflicts of Interest: The authors declare no conflicts of interest.
References
- Gore, AC, Chappell VA, Fenton SE, Flaws JA., Nadal A, Prins GS, Toppari J, Zoeller RT. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr Rev 2015;36(6):E1-E150.
- Verga JU, Huff M, Owens D, Wolf, B.J, Hardiman, G. Integrated Genomic and Bioinformatics Approaches to Identify Molecular Links between Endocrine Disruptors and Adverse Outcomes. Int J Environ Res Public Health 2022;19:574.
- Lee JE, Jung HW, Lee YJ, Lee YA. Early-life exposure to endocrine-disrupting chemicals and pubertal development in girls. Ann Pediatr Endocrinol Metab. 2019;24(2):78-91.
- Bordini B, Rosenfield RL. Normal pubertal development: part II: clinical aspects of puberty. Pediatr Rev. 2011;32(7):281-292.
- Bordini B, Rosenfield RL. Normal pubertal development: Part I: The endocrine basis of puberty. Pediatr Rev. 2011;32(6):223-229.
- Wolf RM, Long D. Pubertal Development. Pediatr Rev. 2016;37(7):292-300.
- Ramsey JT, Li Y, Arao Y, Naidu A, Coons LA, Diaz A, Korach KS. Lavender Products Associated With Premature Thelarche and Prepubertal Gynecomastia: Case Reports and Endocrine-Disrupting Chemical Activities. J Clin Endocrinol Metab. 2019;104(11):5393-5405.
- Linklater A, Hewitt JK. Premature thelarche in the setting of high lavender oil exposure. J Paediatr Child Health 2015;51(2):235.
- Xue P, Wang Y, Chen Y, Wu X, Tang Y, Wang X, Ding Y, Wang X, Liu SA. Case of Peripheral Precocious Puberty May Be Caused by a Diet Containing Phytosterols in a 20-Month-Old Boy. Horm Res Paediatr. 2022;95(5):484-491.
- Türkyilmaz Z, Karabulut R, Sönmez K, Can Başaklar A. A striking and frequent cause of premature thelarche in children: Foeniculum vulgare. J Pediatr Surg. 2008;43(11):2109-2111.
- Gaspari L, Paris F, Jeandel C, Sultan C. Peripheral precocious puberty in a 4-month-old girl: role of pesticides. Gynecol Endocrinol. 2011;27(9):721-724.
- Helyaich A, Elqadiry R, El Fakiri K, Rada N, Draiss G, Bouskraoui M. Case Report of Central Precocious Puberty in a Female: Role of Environmental Pollutants. Clin Med Insights Case Rep. 2020;13:1179547620933585.
- Ruiz D, Becerra M, Jagai JS, Ard K, Sargis RM. Disparities in Environmental Exposures to Endocrine-Disrupting Chemicals and Diabetes Risk in Vulnerable Populations. Diabetes Care 2018;41(1):193-205.
- World Health Organization. Report by the World Health Organization on the Engagement of the Health Sector in the Strategic Approach to International Chemicals Management. Geneva, Switzerland: World Health Organization; 2015.




