Premature Adrenarche and Obesity in Preterm Children
PREMATURE ADRENARCHE AND OBESITY IN CHILDREN BORN PRETERM AND/OR SMALL FOR GESTATIONAL AGE: A CASE SERIES AND LITERATURE REVIEW
Farwa Khurshid1, Elizabeth Littlejohn2
- Michigan State University, Department of Pediatrics, University of Michigan Health-Sparrow, Lansing, Michigan, USA
- Michigan State University, Department of Pediatrics, University of Michigan Health-Sparrow, Lansing, Michigan, USA
OPEN ACCESS
PUBLISHED 31 August 2025
CITATION Khurshid, F., Littlejohn, E., Premature Adrenarche and Obesity in Children Born Preterm or Small for Gestational Age: A Case Series and Literature Review. Medical Research Archives, [online] 13(8). https://doi.org/10.18103/mra.v13i8.6782
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i8.6782
ISSN 2375-1924
ABSTRACT
Premature adrenarche is increasingly recognized among children born preterm and/or small for gestational age (SGA), groups at elevated risk for metabolic complications. This retrospective case series describes three prepubertal girls, all born preterm, who presented with premature adrenarche and varying degrees of overweight or obesity. Clinical findings included early development of pubic or axillary hair and elevated levels of dehydroepiandrosterone sulfate. One patient born preterm and SGA also exhibited early metabolic abnormalities, including high triglyceride levels and elevated insulin-like growth factor 1. These cases highlight the importance of early identification and cardiometabolic monitoring in children with premature adrenarche, particularly those with a history of prematurity and/or SGA. This report contributes to growing evidence linking early-life growth patterns, androgen excess, and future metabolic risk.
Keywords
Premature adrenarche, obesity, preterm birth, small for gestational age, metabolic risk
INTRODUCTION
Premature adrenarche (PA), which refers to the early onset of adrenal androgen production, is a frequently encountered phenomenon in pediatric endocrinology. It presents with signs of androgen action such as pubic and/or axillary hair, adult-type body odor, oily skin, or acne before the age of 8 in girls and 9 in boys. This is typically accompanied by elevated levels of adrenal androgen precursors, primarily dehydroepiandrosterone (DHEA) and its sulfate (DHEAS), beyond what is expected for chronological age.
The process reflects early maturation of the adrenal zona reticularis. Recent studies have highlighted the role of 11-oxygenated androgens, particularly 11-ketotestosterone, as dominant bioactive androgens during both normal and PA. The underlying mechanisms remain incompletely understood, but both intrinsic factors (e.g., adrenal enzyme activity) and extrinsic factors, including low birth weight, rapid catch-up growth, and obesity, are thought to influence the timing and degree of adrenal androgen secretion.
Although traditionally considered a benign and self-limited variant of normal development, PA is increasingly linked to later adverse outcomes. Epidemiologic and cohort studies have associated it with increased risk of central obesity, insulin resistance, dyslipidemia, early menarche, and polycystic ovary syndrome (PCOS), especially among girls born SGA and/or preterm. Despite these associations, clinical guidelines for risk stratification remain limited, and reliable predictors of progression to metabolic disease are lacking.
The prevalence of PA varies considerably, ranging from 9% to 23% in girls and 2% to 10% in boys, depending on the population studied. Diagnosis remains clinical and biochemical, requiring exclusion of other causes of hyperandrogenism such as central precocious puberty, congenital adrenal hyperplasia, androgen-secreting tumors, and exogenous androgen exposure.
This retrospective case series highlights three cases of PA in preterm girls with early signs of metabolic dysregulation. By compiling clinical, biochemical, and imaging findings, we aim to emphasize the potential link between early life growth restriction and the development of androgen excess and metabolic risk. This report contributes to growing efforts to better characterize this population and inform early, tailored monitoring strategies.
CASE SERIES
In 2024, three prepubertal girls presented to our clinic with clinical signs consistent with PA. Each met the diagnostic criteria for PA: the development of androgenic features (e.g., pubic or axillary hair, body odor, acne) before 8 years of age, elevated DHEAS levels for age, and exclusion of other causes including central precocious puberty, congenital adrenal hyperplasia, androgen-secreting tumors, and exogenous androgen exposure.
DEMOGRAPHIC AND PERINATAL CHARACTERISTICS
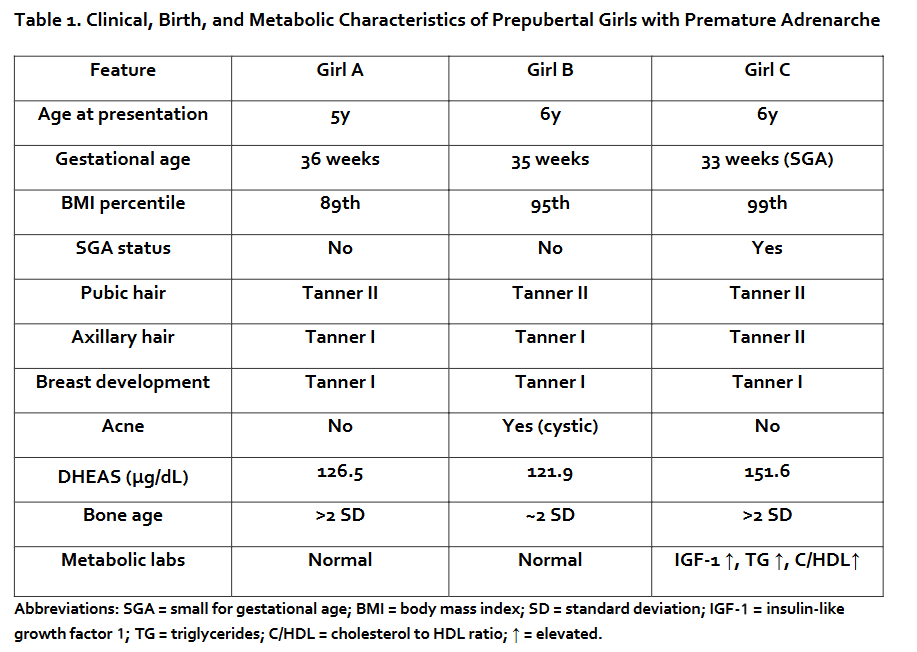
All three patients were female and aged 5 to 6 years at presentation. Each was born preterm (gestational age 33–36 weeks), and one was preterm and small for gestational age (SGA).
CLINICAL FEATURES
Common presenting signs included early pubic and/or axillary hair in all patients, with body odor and cystic acne observed in one patient with each. BMI percentiles ranged from the percentile, indicating that all were either overweight or obese. All patients were Tanner stage I for breast development and Tanner II for pubic hair. Axillary hair ranged from Tanner I to II. None had exogenous androgen exposure per report.
BIOCHEMICAL FINDINGS
All three patients had elevated DHEAS levels for age (121.9–151.6 μg/dL), consistent with PA. In the patient born SGA, additional laboratory findings included elevated insulin-like growth factor 1 (IGF-1) (347 μg/mL), hypertriglyceridemia (186 mg/dL), and a total cholesterol to high density lipoprotein (HDL) ratio of , findings suggestive of early metabolic risk. 17-hydroxyprogesterone (17-OHP) was within normal range in all cases, essentially ruling out non-classic congenital adrenal hyperplasia.
IMAGING
Bone age was advanced by more than 2 standard deviations (SD) in two cases and at the upper limit of normal in the third, reflecting the known association between PA, adiposity, and accelerated skeletal maturation. To facilitate comparison, the key clinical, birth, and metabolic characteristics of all three patients are summarized in Table 1.
DISCUSSION
The interplay between prematurity, SGA status, PA, and obesity represents a critical area of investigation in pediatric endocrinology, with mounting evidence supporting their combined contribution to increased cardiometabolic risk in childhood and beyond.
INDEPENDENT EFFECTS OF PREMATURITY AND SGA
Prematurity, defined as birth before 37 weeks of gestation, is associated with a spectrum of metabolic derangements. Large-scale cohort and meta-analytic data indicate that children born preterm, including those born late preterm (34–36 weeks), exhibit higher rates of hypertension, impaired glucose metabolism, and dyslipidemia in childhood and adulthood compared to term born peers. Mechanistically, prematurity disrupts critical periods of organogenesis, leading to permanent alterations in vascular structure (e.g., increased arterial stiffness), reduced nephron endowment, and altered metabolic programming, all of which contribute to long-term cardiometabolic risk.
SGA status, typically defined as birth weight below the 10th percentile for gestational age, is independently associated with increased risk of insulin resistance, type 2 diabetes, and adverse lipid profiles from childhood through adulthood. Meta-analyses demonstrate that SGA individuals have a more than twofold increased risk of type 2 diabetes and higher Homeostasis Model Assessment for Insulin Resistance (HOMA-IR) values in childhood and adolescence compared to those born appropriate for gestational age (AGA). The pathophysiology is multifactorial, involving intrauterine growth restriction (IUGR), epigenetic modifications, and a propensity for rapid postnatal catch-up growth, which further amplifies metabolic risk.
SYNERGISTIC AND INTERACTIVE EFFECTS
Emerging evidence suggests that prematurity and SGA may interact to further elevate metabolic risk. In large pediatric obesity cohorts, SGA status was associated with higher odds of hypertensive blood pressure, elevated HbA1c, and adverse lipid profiles, and these associations were accentuated in the context of preterm birth. Notably, preterm SGA infants may experience compounded risk due to the convergence of disrupted organ development and suboptimal intrauterine nutrition, leading to more pronounced alterations in metabolic homeostasis.
Longitudinal studies indicate that both SGA and preterm-born children exhibit a progression of adverse metabolic profiles from childhood into adolescence, with worsening insulin resistance and clustering of cardiometabolic risk factors over time. The risk is further modulated by postnatal growth patterns: accelerated catch-up growth in SGA and/or preterm infants is strongly linked to increased adiposity, earlier pubertal timing, and heightened risk of metabolic syndrome. Conversely, optimal growth trajectories, characterized by adequate but not excessive weight gain, may mitigate some of these risks.
MECHANISTIC INSIGHTS AND EPIGENETIC PROGRAMMING
The mechanisms that connect prematurity, SGA status, and PA to subsequent metabolic diseases are intricate, likely involving both genetic and epigenetic factors. Intrauterine growth restriction and preterm birth can trigger adaptive changes in neuroendocrine and metabolic pathways, including modifications in appetite regulation, insulin sensitivity, and hypothalamic-pituitary adrenal (HPA) axis function. While these adaptations may be advantageous in the context of early undernutrition, they could predispose individuals to excessive energy storage, obesity, and early pubertal development when exposed to postnatal energy abundance. Epigenetic modifications during critical developmental windows may further perpetuate these risks throughout life.
CLINICAL IMPLICATIONS AND PREVENTIVE STRATEGIES
The clinical implications of the strong association between prematurity, SGA status, and PA with an increased risk of obesity and metabolic syndrome are significant. This underscores the necessity for early identification and targeted preventive strategies in pediatric practice. Children born preterm and/or SGA, particularly those exhibiting rapid catch-up growth or early adrenarche, constitute a vulnerable subgroup with a markedly elevated risk for adverse cardiometabolic outcomes, including hypertension, insulin resistance, dyslipidemia, and type 2 diabetes, as demonstrated in large cohort studies and meta-analyses.
Early multidisciplinary lifestyle interventions, such as nutritional counseling and the promotion of physical activity, are most effective when initiated in early childhood and tailored to the unique risk profiles of these children. Evidence supports modest but clinically meaningful improvements in metabolic parameters and BMI, especially with high-intensity, longitudinal approaches. Despite these advances, gaps remain in the ability to precisely stratify risk and personalize interventions, highlighting the need for ongoing research to refine predictive models, elucidate underlying mechanisms, and evaluate the long-term impact of targeted preventive strategies in diverse populations (low vs. high socioeconomic status).
CONCLUSION
PA is increasingly recognized as a condition with potential long-term metabolic and reproductive consequences, particularly in children with a history of prematurity and/or SGA status. The pathophysiology is multifactorial, involving altered HPA axis programming, rapid postnatal catch-up growth, and increased adiposity, all of which contribute to early androgen excess and heightened cardiometabolic risk.
By systematically documenting these cases, this report advances current understanding by underscoring the critical importance of early recognition, longitudinal follow-up, and tailored intervention strategies in children with prematurity and/or SGA with PA. The heterogeneity of clinical outcomes in this population necessitates a nuanced approach to risk assessment, as most children with PA experience a benign course, while a distinct subset, particularly those born SGA or exhibiting rapid postnatal catch-up growth, demonstrate a heightened propensity for adverse metabolic and reproductive sequelae, including insulin resistance, metabolic syndrome, and polycystic ovary syndrome (PA as a risk factor).
This case series is limited by its small sample size and retrospective nature. Nevertheless, the clinical patterns observed raise important considerations for risk stratification in similar pediatric populations and reinforce the need for early metabolic screening in children with PA, particularly those born preterm and/or SGA, while supporting the inclusion of birth history in pediatric endocrine assessments.
CONFLICT OF INTEREST:
The authors have no conflicts of interest to declare.
FUNDING STATEMENT:
No financial disclosure.
ACKNOWLEDGEMENTS:
No acknowledgements.
REFERENCES:
- Rosenfield RL. Normal and premature adrenarche. Endocr Rev. 2021;42(6):783-814. doi:10.1210/endrev/bnab009.
- Augsburger P, Liimatta J, Fluck CE. Update on adrenarche—still a mystery. J Clin Endocrinol Metab. 2024;109(6):1403-1422. doi:10.1210/clinem/dgae008.
- Voutilainen R, Jaaskelainen J. Premature adrenarche: etiology, clinical findings, and consequences. J Steroid Biochem Mol Biol. 2015;145:226-236. doi:10.1016/j.jsbmb.2014.06.004.
- Utriainen P, Laakso S, Liimatta J, Jaaskelainen J, Voutilainen R. Premature adrenarche— a common condition with variable presentation. Horm Res Paediatr. 2015;83(4):221-231. doi:10.1159/000369458.
- Rege J, Turcu AF, Kasa-Vubu JZ, et al. 11-Ketotestosterone is the dominant circulating bioactive androgen during normal and premature adrenarche. J Clin Endocrinol Metab. 2018;103(12):4589-4598. doi:10.1210/jc.2018-00736.
- Jee YH, Jumani S, Mericq V. The association of accelerated early growth, timing of puberty, and metabolic consequences in children. J Clin Endocrinol Metab. 2023;108(9):e663-e670. doi:10.1210/clinem/dgad202.
- Idkowiak J, Lavery GG, Dhir V, et al. Premature adrenarche: novel lessons from early onset androgen excess. Eur J Endocrinol. 2011;165(2):189-207. doi:10.1530/EJE-11-0223.
- Williams RM, Ward CE, Hughes IA. Premature adrenarche. Arch Dis Child. 2012;97(3):250-254. doi:10.1136/archdischild-2011-300011.
- Janner M, Sommer G, Groessl M, Fluck CE. Premature adrenarche in girls characterized by enhanced 17,20-lyase and 17β-hydroxysteroid dehydrogenase activities. J Clin Endocrinol Metab. 2020;105(12):dgaa598. doi:10.1210/clinem/dgaa598.
- Oberfield SE, Sopher AB, Gerken AT. Approach to the girl with early onset of pubic hair. J Clin Endocrinol Metab. 2011;96(6):1610-1622. doi:10.1210/jc.2011-0225.
- Ibanez L, Dimartino-Nardi J, Potau N, Saenger P. Premature adrenarche—normal variant or forerunner of adult disease? Endocr Rev. 2000;21(6):671-696. doi:10.1210/edrv.21.6.0416.
- Witchel SF, Pinto B, Burghard AC, Oberfield SE. Update on adrenarche. Curr Opin Pediatr. 2020;32(4):574-581. doi:10.1097/MOP.0000000000000928.
- Martin-Calvo N, Goni L, Tur JA, Martinez JA. Low birth weight and small for gestational age are associated with complications of childhood and adolescence obesity: systematic review and meta-analysis. Obes Rev. 2022;23(Suppl 1):e13380. doi:10.1111/obr.13380.
- Prinz N, Putri RR, Reinehr T, et al. The association between perinatal factors and cardiometabolic risk factors in children and adolescents with overweight or obesity: a retrospective two-cohort study. PLoS Med. 2023;20(1):e1004165. doi:10.1371/journal.pmed.1004165.
- Yoshida-Montezuma Y, Sivapathasundaram B, Brown HK, et al. Association of late preterm birth and size for gestational age with cardiometabolic risk in childhood. JAMA Netw Open. 2022;5(5):e2214379. doi:10.1001/jamanetworkopen.2022.14379.
- Mericq V, Martinez-Aguayo A, Uauy R, et al. Long-term metabolic risk among children born premature or small for gestational age. Nat Rev Endocrinol. 2017;13(1):50-62. doi:10.1038/nrendo.2016.127.
- Chiavaroli V, Marcovecchio ML, de Giorgis T, et al. Progression of cardio-metabolic risk factors in subjects born small and large for gestational age. PLoS One. 2014;9(8):e104278. doi:10.1371/journal.pone.0104278.
- Cauzzo C, Chiavaroli V, Di Valerio S, Chiarelli F. Birth size, growth trajectory and later cardio-metabolic risk. Front Endocrinol (Lausanne). 2023;14:1187261. doi:10.3389/fendo.2023.1187261.
- Chernausek SD. Update: consequences of abnormal fetal growth. J Clin Endocrinol Metab. 2012;97(3):689-695. doi:10.1210/jc.2011-2741.
- Kim YJ, Shin SH, Lee ES, et al. Impact of size at birth and postnatal growth on metabolic and neurocognitive outcomes in prematurely born school-age children. Sci Rep. 2021;11(1):6836. doi:10.1038/s41598-021-86292-1.
- de Jong M, Cranendonk A, van Weissenbruch MM. Components of the metabolic syndrome in early childhood in very-low-birth-weight infants and term small and appropriate for gestational age infants. Pediatr Res. 2015;78(4):457-461. doi:10.1038/pr.2015.118.
- Maguolo A, Olivieri F, Zusi C, et al. The risk of metabolic derangements is higher in children and adolescents with overweight or obesity born small for gestational age. Nutr Metab Cardiovasc Dis. 2021;31(6):1903-1910. doi:10.1016/j.numecd.2021.02.025.
- Roth CL, Sathyanarayana S. Mechanisms affecting neuroendocrine and epigenetic regulation of body weight and onset of puberty: potential implications in the child born small for gestational age (SGA). Rev Endocr Metab Disord. 2012;13(2):129-140. doi:10.1007/s11154-.

