Prenatal Arbovirus Infection: ASD and ADHD Risks
Prenatal Arbovirus Infection, Autism Spectrum Disorder, and Attention Deficit and Hyperactivity Disorder: A Scoping Review
Tomás A.M. Gomes1, Lavínia Schuler-Faccini1,2
- Post-Graduate Program in Children and Adolescent Health, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil
- Post-Graduate Program in Children and Adolescent Health, Universidade Federal do Rio Grande do Sul, Porto Alegre, Brazil ; Medical Genetics Service, Hospital de Clínicas de Porto Alegre, Porto Alegre, Brazil
OPEN ACCESS
PUBLISHED: 31 October 2025
CITATION: Gomes, TAM., and Schuler-Faccini, L., 2025. Prenatal Arbovirus Infection, Autism Spectrum Disorder, and Attention Deficit and Hyperactivity Disorder: A Scoping Review. Medical Research Archives, [online] 13(10).
https://doi.org/10.18103/mra.v13i10.6998
COPYRIGHT © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i10.6998
ISSN 2375-1924
Abstract
Many arbovirus infections have well-known risks of vertical transmission, causing congenital anomalies, therefore being a concern for women who are either planning a pregnancy or are already pregnant. Research on the effects of prenatal exposure to arbovirus infection on long-term neurodevelopment is limited. Of particular concern is the potential association of prenatal infections as risk factors for Autism Spectrum Disorder (ASD) and Attention Deficit and Hyperactivity Disorder (ADHD). In this scoping review, we’ve researched studies that investigate the impact of arbovirus infection on ASD and ADHD through published case-control, cohort, case series, cross-sectional, and experimental study designs. Sixteen articles met our inclusion criteria, five experimental in vitro, one experimental in vivo, one both in vitro and in vivo, and nine in humans. Evidence of a possible association between arboviral infection and ASD was observed in seven articles, and almost all had prenatal exposure to ZIKV. No specific research on prenatal arboviral infection and ADHD was located. However, the similarity of the neurological pathways shared by these two disorders suggests the need for specific investigations in this field.
Keywords
arbovirus, prenatal infection, Autism Spectrum Disorder, Attention Deficit Hyperactivity Disorder, Zika virus
Introduction
Zika virus (ZIKV) is an arbovirus transmitted by the Aedes aegypti mosquito. When a pregnant woman is infected with the ZIKV, the newborn can be affected by the Congenital Zika syndrome (CZS). Between 2015 and 2016, a sudden increase in ZIKV cases occurred in the northeastern states of Brazil, with the outbreak quickly evolving into an epidemic and a serious public health concern. CZS includes not only a variety of symptoms, but also extremely debilitating ones, such as severe motor disability, cerebral paralysis, neurodevelopmental delays, dystonia, oropharyngeal dysphagia. 1–4 In exposed children born without microcephaly (normocephalic), there is evidence that neurodevelopmental delays are more frequent than in non-exposed children. Utilizing the Bayley-III neurodevelopmental assessment scale, children’s cognitive, linguistic, and motor skills were assessed, with language development being the most significantly affected neurological domain.5,6 In a case report, a normocephalic child exposed to ZIKV during gestation was diagnosed with Autism Spectrum Disorder (ASD) at 2 years of age,7 bringing the possibility that infection by ZIKV during gestation could lead to a broader spectrum of neurodevelopmental disorders, such as ASD and Attention Deficit Hyperactivity Disorder (ADHD). Common manifestations of ASD and ADHD include difficulties with motor responses, behavioral reactions, underdeveloped social skills, the lack of ability to evaluate risks, planning, and prioritizing tasks.8
Besides ZIKV, other relevant arboviral infections in Brazil are dengue, chikungunya, yellow fever, and oropouche (sloth) fever. The dengue (DENV) and chikungunya viruses (CHIKV) are also transmitted by the Aedes aegypti mosquito, while the oropouche and yellow fever vectors are the insects Culicoides paraensis and Aedes spp., respectively.9–12 Perinatal exposure to CHIKV is associated with meningoencephalitis, fever, and exanthema. A cohort study found reduced neurodevelopment in children born from such gestations.13 Dengue virus infection affects gestations by increasing abortions, fetal death, and premature births. A single case of perinatal transmission of yellow fever was registered with severe symptoms, including cyanosis, hemorrhage, and liver complications, leading to death on the 12th day after birth.14 Finally, exposure to the Oropouche virus has been recently linked to causing microcephaly, arthrogryposis, and possibly polyhydramnios and generalized edema.15
Therefore, our objective is to perform a scoping review of the current literature to map the existing evidence of the risks related to arboviral prenatal infections by arbovirus and the risk of neurodevelopmental disorders, specifically ASD and ADHD, and establish the need for further elaborate investigations.
Methodology
In this scoping review, we employed the standard reporting guidelines outlined by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for scoping reviews.16 We defined ASD and ADHD as disorders that affect the neurodevelopment of children and young adolescents. We defined arboviral prenatal infections as infections from an arbovirus that affect the gestation at any given time during said gestation.
The inclusion criteria were as follows: (1) articles that analyzed how gestational arbovirus affects the immunological system or neurodevelopment in normocephalic children; (2) studies with humans or experimental (in vivo or in vitro); (3) studies published between January 1st, 2015, and December 31st, 2024. The date of January 2015 was chosen because it was the year when the teratogenicity of ZIKV was described. The exclusion criteria were (1) reviews, (2) articles that did not relate to arbovirus infections with neurodevelopmental disorders, and (3) when the infection is postnatal.
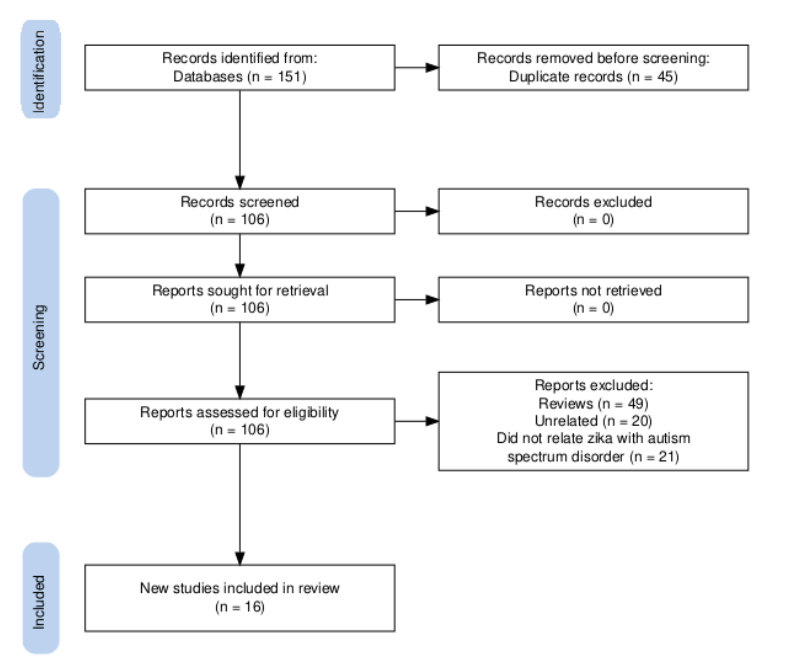
We searched publications indexed in PubMed and Scopus databases using the following descriptors: (Zika) AND (Autism) or (Zika) AND (ADHD); (Dengue) AND (Autism) or (Dengue) AND (ADHD); (Arbovirus) AND (Autism) or (Arbovirus) AND (ADHD). The resulting articles were then exported to Excel®, where duplicates were manually removed. Themes were identified through careful reading and discussion of the abstracts between both authors and, when applicable, the results. We then extracted the following data from included studies: author, year of publication, country, type of study, summary and main outcome of said study. The PRISMA flowchart is detailed in Figure 1.
Results
Table 1 contains all 16 articles included in this review, with a summary of the subject discussed in each article.
| Author/ Country* | Type of study | Summary and main outcomes |
|---|---|---|
| Watanabe et al., 201718 | Experimental in vitro | Brain organoids study: ZIKV increases interferon activity during fetal development linked to ASD. |
| Beys-da-Silva et al., 201919 | Experimental in vitro | Stem cells: Proteins associated with ASD are highly expressed in ZIKV infection. |
| Lottini et al., 202220 | Experimental in vitro | FOXG1 gene with altered expression during ZIKV infection; related to ASD. |
| Benazzato et al., 202421 | Experimental in vitro | CSZ infants’ fibroblasts to form neural networks have decreased synapses and deficits similar to those described in ASD patients. |
| Ohki et al., 202422 | Experimental in vitro and in vivo | ZIKV infection affects astrocytes’ functionality; an in vivo mouse model presented autistic-like behavior. |
| Nani et al., 202523 | Experimental in vitro | Brain organoids: expression of proteins NDE1 and NDEL1 is associated with both autism and prenatal ZIKV infection. |
| Ma et al., 202124 | Experimental in vivo | Mice with prenatal exposure to ZIKV: slight behavioral changes (autistic-like behavior) when compared to control mice. |
| Santi et al., 2021 / Brazil7 | Case-report | Child with prenatal ZIKV infection later diagnosed with ASD. |
| Abtibol-Bernardino et al., 2020 / Brazil25 | Case-control | Children with prenatal ZIKV infection (n=26) with language impairment, neurological disorders, and two cases of ASD. |
| Mulkey et al., 2024 / USA26 | Case-control | Neurodevelopment of 12 children exposed prenatally to ZIKV. |
| Roth et al., 2023 / Puerto Rico27 | Cross-sectional | 109 children diagnosed with ASD: 60 with laboratory evidence of prenatal zika exposure. |
| Nielsen-Saines et al., 2019 / Brazil28 | Cohort | Follow-up of 216 children (8 microcephalic) with prenatal exposure to ZIKV: three previously healthy children diagnosed with ASD in the second year of life. |
| Gazeta et al., 2021 / Brazil29 | Cohort | 57 confirmed cases of ZIKV during pregnancy: two cases of ASD. |
| Grant et al., 2021, Guadeloupe, Martinique and French Guiana30 | Cohort | 156 toddlers with prenatal exposure to ZIKV: no major differences observed in behavior disorder screening risk (M-CHAT) between ZIKV-exposed and ZIKV-unexposed (P = 0.15). |
| Abtibol-Bernardino et al., 2022 / Brazil31 | Cohort | Children with prenatal exposure to ZIKV: four cases of ASD. ** |
| Toizumi et al., 2023, Vietnam32 | Cohort | Follow-up study of 11 children with prenatal zika infection: 10 resulted in low risk for autism according to M-CHAT testing. |
ASD: autism spectrum disorder; ASQ: ages and stages questionnaire; CSZ: congenital zika syndrome; M-CHAT: modified checklist for autism in toddlers; ZIKV: zika virus;
* Country is mentioned only for studies with humans.
** Possibly two children were the same from Abtibol-Bernardino et al, 202023
Although we searched for arbovirus infections, the research regarding neurodevelopment primarily investigated ZIKV, with no results on the development of ADHD. Most available evidence was based on cohort and case-control studies (n=7). Four were conducted in Brazil; one was in the USA, one in Vietnam, and one included multiple countries (Guadeloupe, Martinique, and French Guiana). There was also one case report from Brazil and one cross-sectional study from Puerto Rico. The other six studies were in vitro, using organoids, stem cells, or astrocytes. Two in vivo mouse models were used to observe behavioral patterns derived from prenatal exposure to ZIKV. One of the in vivo papers is a double study, conducting an in vitro ZIKV infection analysis using astrocytes and an in vivo mouse model for behavior analysis.
In the epidemiological studies in humans, three were follow-up studies of children’s cohorts at different moments of life.26,28,32 While two of the studies only assessed prenatal exposure to zika virus and physical examination,33,34 one investigated the neurological impact of prenatal exposure through neuroimaging and neurological examinations, with confirmed and possible ZIKV exposure being the groups with the highest proportion of abnormal imaging and examinations.35
Most (7/9) of the studies involving humans have some cases of formally diagnosed ASD, one case being macrocephalic.7,25–29,31 Furthermore, the results of applied Bayley-III show slightly below average neurodevelopment in linguistic criteria for children exposed prenatally to ZIKV compared to non-exposed.25,28,29,31 One case control study used different tests, known as Pediatric Evaluation of Disability Inventory (PEDI-CAT), Behavior Rating Inventory of Executive Function (BRIEF-P), and the Bracken School Readiness Assessment (BSRA), with similar results.26
Finally, tests applied specifically for ASD screening, M-CHAT and ASQ-3, indicated overall a low risk for this condition.27,30,32
Discussion
Neurodevelopmental disorders (NDD) affect nearly 10-13% of children. Its etiology is complex (multifactorial) with gene/environment interactions.36,37 On the other hand, only a minority of children whose mothers had ZIKV infection during pregnancy were born with microcephaly or obvious neurological impairments.38
Neurodevelopmental disorders resulting from prenatal arbovirus infection present novel challenges. Initial evidence suggests that there is more to it than meets the eye, specifically beyond the structural defects characteristic of the Congenital Zika Syndrome. The main question is whether there is an elevated risk for neurodevelopmental disorders in individuals exposed to ZIKV born without apparent brain abnormalities.
Febrile illness and inflammation during pregnancy are correlated with risk for autism, attention deficit/hyperactivity disorder, and developmental delay in the offspring in human and animal models. Different congenital viral syndromes have been associated with autism spectrum disorders, such as rubella and cytomegalovirus.39 Although there is no evidence for arbovirus infection influencing the development of ADHD in the studies we reviewed here, the similar molecular and neuronal pathways of both conditions make ADHD a plausible outcome in children with pre-natal ZIKV infection.8,40
The six in vitro experimental studies included in this scoping review highlighted different molecular pathways or pathogenic mechanisms that could lead to neurodevelopmental disorders. Inflammatory response is one of the mechanisms that perpass different investigations. It is known that immune cells and immunity-related signaling molecules participate in the development of the nervous system and associations between autoimmune diseases and ADHD and autism have already been investigated.41–43 In the paper by Watanabe et al.18, one-third of the 50 most upregulated genes following ZIKV infection were associated with interferon signaling and innate immune responses. Elevated interferon levels during fetal and postnatal development were implicated in NDD, including autism and ADHD, in independent studies not related to congenital infections.44 Other in vitro studies have reported altered pathways in the inflammatory response after ZIKV-infected cell cultures, as seen in Beys-da-Silva et al.19. They identified 145 differentially expressed proteins, comprising 27 upregulated and 118 downregulated proteins, compared to control cells. Interestingly, in their study, they observed that some proteins previously described in several brain pathologies, including autism spectrum disorder, showed altered expression due to ZIKV. The study by Benazzato et al.21 also showed that reprogrammed iPSCs (induced pluripotent stem cells) from children with Congenital Zika Syndrome, when reprogrammed into neuroprogenitor cells, exhibited elevated levels of cytokine production, including IL-6, already associated with the ASD phenotype.42
Another pathway involved in the pathogenesis of ASD is the GABAergic inhibitory circuit. One study included in our review20 reported that ZIKV infection altered the nuclear localization of FOXG1 (transcription factor Forkhead box G1), leading to its downregulation and consequently impairing the expression of genes involved in cell replication and apoptosis in human neural progenitor cells, which could be one of the mechanisms leading to microcephaly. Moreover, the altered expression of the gene FOXG1 in both excitatory and inhibitory neurons results in ASD-related circuit and social behavior deficits in our mouse models.45,46
The in vivo experimental studies have yielded interesting results regarding behavioral elements observed in mice with prenatal exposure to ZIKV, known as “autistic-like behavior”. These mice exhibited stereotyped behavior (repetition of a given task), impaired social interaction, impaired social memory, and repetitive self-grooming when compared to the control group.22,24 Furthermore, these experiments complement the in vitro models, allowing observation of the degree to which the neuronal pathways are affected by the ZIKV. Activation of the immune response, as indicated by cytokines such as IL-6 and interferon, is associated with neurodevelopmental disturbances resulting from impaired synaptogenesis and improper astrocyte function.18,22,47
Considering the amount of evidence pointing to ASD and ADHD being potential outcomes after congenital ZIKV infection, it is surprising that there are so few epidemiological studies in our search (n=8). As expected, most of the studies were performed in Brazil, the country that identified ZIKV as a teratogen implicated in a surge in microcephalic children born in 2015.48
Age at follow-up is an essential factor to consider in these investigations. Proper ASD diagnosis is generally made at 3 to 4 years of age.49,50 Late diagnosis makes it harder to associate ASD/ADHD with prenatal ZIKV exposure, either by loss of data, addition of confounding factors during the patient’s life, or health professionals ruling out the possibility of ZIKV as a potential risk. These components illustrate how we currently occupy a window of opportunity to properly investigate and determine the association between pre-natal ZIKV infection and neurodevelopmental issues/disorders.
The observations in these studies should consider the baseline of ASD estimated as 1:54 in the general population.49 The study of Nielsen-Saines et al.28, detected a ratio of 1:72 in a series of 216 children exposed to ZIKV in pregnancy, which is not different from the expected in the general population. Other studies had limited sample sizes, which made it difficult to detect significant increases in ASD.25,26,29,31,32 Also, the epidemiological studies in humans have contradictory results. Although most studies indicated both a risk for neurodevelopmental delay and proper ASD diagnosis of some of the patients25,26,27,28,29,31, two studies found no significant association between neurodevelopmental delay and ZIKV exposure during pregnancy.30,32
Conclusion
Prenatal exposure to ZIKV is known to have teratogenic effects, with physical symptoms being the most predominant. With this review, we highlight the effects on fetal neurodevelopment caused by prenatal ZIKV. The global climate change and the spread of arboviral infections underscore the importance of epidemiological and longitudinal studies to follow up pregnancies and offspring neurodevelopment.
Conflict of Interests
The authors declare no conflicts of interest.
Funding Statements
The authors received no funding for production of this research.
References
- Freitas DA, Souza-Santos R, Carvalho LMA, et al. Congenital Zika syndrome: A systematic review. PLoS One. 2020;15(12 December). doi:10.1371/journal.pone.0242367
- Leal MC, van der Linden V, Bezerra TP, et al. Characteristics of dysphagia in infants with microcephaly caused by congenital zika virus infection, Brazil, 2015. Emerg Infect Dis. 2017;23(8):1253-1259. doi:10.3201/eid2308.170354
- Pereira HVFS, dos Santos SP, Amâncio APRL, et al. Neurological outcomes of congenital Zika syndrome in toddlers and preschoolers: a case series. Lancet Child Adolesc Health. 2020;4(5):378-387. doi:10.1016/S2352-4642(20)30041-9
- Ribeiro MFM, Prudente COM, de Queiróz KBP. Motor development of children exposed to the zika virus: systematic review. Revista Brasileira de Saude Materno Infantil. 2022;22(4):739-751. doi:10.1590/1806-9304202200040002
- Gerzson LR, de Almeida CS, da Silva JH, Feitosa MMA, de Oliveira LN, Schuler-Faccini L. Neurodevelopment of Nonmicrocephalic Children, After 18 Months of Life, Exposed Prenatally to Zika Virus. J Child Neurol. 2020;35(4):278-282. doi:10.1177/0883073819892128
- Moreira MEL, Nielsen-Saines K, Brasil P, et al. Neurodevelopment in Infants Exposed to Zika Virus In Utero. New England Journal of Medicine. 2018;379(24):2377-2379. doi:10.1056/nejmc1800098
- Santi L, Riesgo RS, Quincozes-Santos A, et al. Zika Virus Infection Associated with Autism Spectrum Disorder: A Case Report. Neuroimmunomodulation. 2021;28(4):229-232. doi:10.1159/000516560
- Miyake A, Friedman NP. The nature and organization of individual differences in executive functions: Four general conclusions. Curr Dir Psychol Sci. 2012;21(1):8-14. doi:10.1177/0963721411429458
- Halstead SB. Seminar Dengue. Vol 370.; 2007. www.thelancet.com
- Weaver SC, Lecuit M. Chikungunya Virus and the Global Spread of a Mosquito-Borne Disease. New England Journal of Medicine. 2015;372(13):1231-1239. doi:10.1056/nejmra1406035
- Zhang Y, Liu X, Wu Z, et al. Oropouche virus: A neglected global arboviral threat. Virus Res. 2024;341. doi:10.1016/j.virusres.2024.199318
- Wilder-Smith A. Yellow Fever in Travelers. Curr Infect Dis Rep. 2019;21(11). doi:10.1007/s11908-019-0701-x
- Gérardin P, Sampériz S, Ramful D, et al. Neurocognitive Outcome of Children Exposed to Perinatal Mother-to-Child Chikungunya Virus Infection: The CHIMERE Cohort Study on Reunion Island. PLoS Negl Trop Dis. 2014;8(7). doi:10.1371/journal.pntd.0002996
- Bentlin MR, de Barros Almeida RAM, Coelho KIR, et al. Perinatal Transmission of Yellow Fever, Brazil, 2009. Emerging Infections Diseases. 2011;17(9):1389-1393. doi:10.1099/jmm.0.2008/003632-0
- das Neves Martins FE, Chiang JO, Nunes BTD, et al. Newborns with microcephaly in Brazil and potential vertical transmission of Oropouche virus: a case series. Lancet Infect Dis. Published online February 1, 2024. doi:10.1016/S1473-3099(24)00617-0
- Tricco AC, Lillie E, Zarin W, et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann Intern Med. 2018;169(7):467-473. doi:10.7326/M18-0850
- Haddaway NR, Page MJ, Pritchard CC, McGuinness LA. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Systematic Reviews. 2022;18(2). doi:10.1002/cl2.1230
- Watanabe M, Buth JE, Vishlaghi N, et al. Self-Organized Cerebral Organoids with Human-Specific Features Predict Effective Drugs to Combat Zika Virus Infection. Cell Rep. 2017;21(2):517-532. doi:10.1016/j.celrep.2017.09.047
- Beys-da-Silva WO, Rosa RL, Santi L, et al. Zika Virus Infection of Human Mesenchymal Stem Cells Promotes Differential Expression of Proteins Linked to Several Neurological Diseases. Mol Neurobiol. 2019;56(7):4708-4717. doi:10.1007/s12035-018-1417-x
- Lottini G, Baggiani M, Chesi G, et al. Zika virus induces FOXG1 nuclear displacement and downregulation in human neural progenitors. Stem Cell Reports. 2022;17(7):1683-1698. doi:10.1016/j.stemcr.2022.05.008
- Benazzato C, Lojudice F, Pöehlchen F, et al. Zika virus vertical transmission induces neuroinflammation and synapse impairment in brain cells derived from children born with Congenital Zika Syndrome. Sci Rep. 2024;14(1). doi:10.1038/s41598-024-65392-8
- Ohki CMY, Benazzato C, van der Linden V, et al. Zika virus infection impairs synaptogenesis, induces neuroinflammation, and could be an environmental risk factor for autism spectrum disorder outcome. Biochim Biophys Acta Mol Basis Dis. 2024;1870(5). doi:10.1016/j.bbadis.2024.167097
- Nani J V., Krenn V, Christoff RR, Rabello T, Garcez PP, Hayashi MAF. RNA-seq transcriptome of ZIKV-infected brain organoids reanalysis implicates Ndel1 oligopeptidase and its cytoskeleton protein binding partners in the infection pathogenesis and recovery following interferon treatment. Brain Res. 2025;1850. doi:10.1016/j.brainres.2024.149371
- Ma L, Wang J, Ge J, et al. Reversing neural circuit and behavior deficit in mice exposed to maternal inflammation by Zika virus. EMBO Rep. 2021;22(8). doi:10.15252/embr.202051978
- Abtibol-Bernardino MR, de Almeida Peixoto L de FA, de Oliveira GA, et al. Neurological findings in children without congenital microcephaly exposed to zika virus in utero: A case series study. Viruses. 2020;12(11). doi:10.3390/v12111335
- Mulkey SB, Corn E, Williams ME, et al. Neurodevelopmental Outcomes of Preschoolers with Antenatal Zika Virus Exposure Born in the United States. Pathogens. 2024;13(7). doi:10.3390/pathogens13070542
- Roth NM, Delgado-López C, Wiggins LD, et al. Notes from the Field: Autism Spectrum Disorder Among Children with Laboratory Evidence of Prenatal Zika Virus Exposure — Puerto Rico, 2023. MMWR Morb Mortal Wkly Rep. 2023;72(29):802-804. doi:10.15585/mmwr.mm7229a5
- Nielsen-Saines K, Brasil P, Kerin T, et al. Delayed childhood neurodevelopment and neurosensory alterations in the second year of life in a prospective cohort of ZIKV-exposed children. Nat Med. 2019;25(8):1213-1217. doi:10.1038/s41591-019-0496-1
- Gazeta RE, Bertozzi APAP, Dezena R de C de AB, et al. Three-year clinical follow-up of children intrauterine exposed to zika virus. Viruses. 2021;13(3). doi:10.3390/v13030523
- Grant R, Fléchelles O, Tressières B, et al. In utero Zika virus exposure and neurodevelopment at 24 months in toddlers normocephalic at birth: a cohort study. BMC Med. 2021;19(1). doi:10.1186/s12916-020-01888-0
- Abtibol-Bernardino MR, Peixoto L de FA de A, Castilho M da C, et al. Would Zika virus Infection in Pregnancy Be a Sentence of Poor Neurological Prognosis for Exposed Children? Neurodevelopmental Outcomes in a Cohort from Brazilian Amazon. Viruses. 2022;14(12). doi:10.3390/v14122659
- Toizumi M, Vu CN, Huynh HT, et al. A Birth Cohort Follow-Up Study on Congenital Zika Virus Infection in Vietnam. Viruses. 2023;15(9). doi:10.3390/v15091928
- Tun MMN, Moriuchi M, Toizumi M, et al. Congenital zika virus infection in a birth cohort in Vietnam, 2017-2018. American Journal of Tropical Medicine and Hygiene. 2020;103(5):2059-2064. doi:10.4269/ajtmh.20-0286
- Brasil P, Pereira JP, Moreira ME, et al. Zika Virus Infection in Pregnant Women in Rio de Janeiro. New England Journal of Medicine. 2016;375(24):2321-2334. doi:10.1056/nejmoa1602412
- Mulkey SB, Ansusinha E, Cristante C, et al. Complexities of zika diagnosis and evaluation in a u.s. congenital zika program. American Journal of Tropical Medicine and Hygiene. 2021;104(6):2210-2219. doi:10.4269/ajtmh.20-1256
- Halfon N, Houtrow A, Larson K, Newacheck PW. The Changing Landscape of Disability in Childhood. Vol 22.; 2014. http://www.jstor.orgURL:http://www.jstor.org/stable/41475645
- Boyle CA, Boulet S, Schieve LA, et al. Trends in the prevalence of developmental disabilities in US children, 1997-2008. Pediatrics. 2011;127(6):1034-1042. doi:10.1542/peds.2010-2989
- Schuler-Faccini L, del Campo M, García-Alix A, et al. Neurodevelopment in Children Exposed to Zika in utero: Clinical and Molecular Aspects. Front Genet. 2022;13. doi:10.3389/fgene.2022.758715
- Yates EF, Mulkey SB. Viral infections in pregnancy and impact on offspring neurodevelopment: mechanisms and lessons learned. Pediatr Res. 2024;96(1):64-72. doi:10.1038/s41390-024-03145-z
- Reiersen AM, Constantino JN, Todd RD. Co-occurrence of motor problems and autistic symptoms in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2008;47(6):662-672. doi:10.1097/CHI.0b013e31816bff88
- Daniel B, Acquaviva E, Bader-Meunier B, et al. Predictive factors of comorbid attention-deficit/hyperactivity disorder in early systemic autoimmune and auto-inflammatory disorders. Pediatric Rheumatology. 2025;23(1). doi:10.1186/s12969-025-01103-5
- Upthegrove R, Corsi-Zuelli F, Couch A, Barnes N, Vernon A. Current Position and Future Direction of Inflammation in Neuropsychiatric Disorders: a Review. JAMA Psychiatry. Published online July 9, 2025. doi:10.1001/jamapsychiatry.2025.1369
- Nielsen T, Nassar N, Shand A, et al. Association of Maternal Autoimmune Disease with Attention-Deficit/Hyperactivity Disorder in Children. JAMA Pediatr. 2021;175(3). doi:10.1001/jamapediatrics.2020.5487
- Zengeler KE, Lukens JR. Innate immunity at the crossroads of healthy brain maturation and neurodevelopmental disorders. Nat Rev Immunol. 2021;21(7):454-468. doi:10.1038/s41577-020-00487-7
- Younger S, Boutros S, Cargnin F, et al. Behavioral Phenotypes of Foxg1 Heterozygous Mice. Front Pharmacol. 2022;13. doi:10.3389/fphar.2022.927296
- Miyoshi G, Ueta Y, Natsubori A, et al. FoxG1 regulates the formation of cortical GABAergic circuit during an early postnatal critical period resulting in autism spectrum disorder-like phenotypes. Nat Commun. 2021;12(1). doi:10.1038/s41467-021-23987-z
- Rosa-Fernandes L, Cugola FR, Russo FB, et al. Zika virus impairs neurogenesis and synaptogenesis pathways in human neural stem cells and neurons. Front Cell Neurosci. 2019;13. doi:10.3389/fncel.2019.00064
- Schuler-Faccini L, Ribeiro E, Feitosa I, et al. Possible Association between Zika Virus Infection and Microcephaly – Brazil, 2015. Vol 65. Centers for Disease Control and Prevention (CDC); 2016. doi:10.3201/eid2110.150847
- Redfield RR, Kent CK, Leahy MA, et al. Prevalence of Autism Spectrum Disorder Among Children Aged 8 Years-Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2014. MMWR Morb Mortal Wkly Rep. Published online April 27, 2018.
- Arlington V. American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. 5th ed.; 2013.

