Prevalence of Cirrhotic Cardiomyopathy in Kenya
Prevalence of Cirrhotic Cardiomyopathy among Patients with Liver Cirrhosis at Kenyatta National Hospital
Isaak M. Osman 1, Prof. Elly O. Ogutu 1, Prof. E. N. Ogola 1, Dr. Adam Sheikh 1, Dr. E.M. Karari 1
- Department of Clinical Medicine & Therapeutics, University of Nairobi, Nairobi, Kenya
OPEN ACCESS
PUBLISHED: 30 November 2025
CITATION: Osman, IM., Ogutu, EO., et al., 2025. Prevalence of Cirrhotic Cardiomyopathy among Patients with Liver Cirrhosis at Kenyatta National Hospital. Medical Research Archives, [online] 13(11). https://doi.org/10.18103/mra.v13i11.7095
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i11.7095
ISSN 2375-1924
ABSTRACT
Background – Cardiac dysfunction is not uncommon in liver cirrhosis. Cirrhotic Cardiomyopathy is an impaired contractile responsiveness to stress and/or abnormal diastolic function with associated electrophysiological abnormalities in lack of other known cardiac diseases. This form of cardiac disorder bears significant morbidity and mortality particularly after invasive procedures.
Objective – Determine the Prevalence of Cirrhotic Cardiomyopathy among patients with liver Cirrhosis at Kenyatta National Hospital.
Design: This was hospital based descriptive cross-sectional study
Setting: Kenyatta National Hospital out-patient liver clinic and medical wards. Nairobi, Kenya.
Subjects: Forty-four patients with liver cirrhosis presenting to Kenyatta National Hospital.
Interventions: All patients underwent resting echocardiography and 12-lead electrocardiogram
Results: Forty-four patients were recruited. The mean age of the participants was 44 years. 27 (61.4%) were males. Hepatitis B infection (38.6%) and chronic alcohol consumption (38.6%) were the two leading etiology of liver cirrhosis. Out of 44 patients, 17 (38.6%) belonged to class A, 16 (36.4%) to class B, and 11 (25.0%) to class C. Three (6.8%) patients were found to have systolic dysfunctions. 19 (43.2%) diastolic dysfunction; more than three quarter (94.7%) of these had grade1 diastolic dysfunction. Prolonged (>440ms) QTc interval was present in 25 (56.8%) patients. Cirrhotic cardiomyopathy was recorded in 43.2% of the study population. Liver disease severity did not show correlation with the cardiomyopathy.
Conclusion: The prevalence of cirrhotic cardiomyopathy as mainly driven by diastolic and systolic dysfunction was high in this black African population of liver cirrhosis. QTc interval prolongation was common in our study. The severity of liver disease did not show association with the presence of cirrhotic cardiomyopathy.
Keywords: Liver Cirrhosis, Cirrhotic cardiomyopathy, Echo, ECG, Systolic dysfunction, Diastolic dysfunction, QTc interval
1. Introduction
Cirrhosis causes significant morbidity and mortality worldwide; it is associated with wide-ranging cardiovascular abnormalities which are now described as cirrhotic cardiomyopathy. According to the World Congress of Gastroenterology of 2005, Cirrhotic cardiomyopathy is a chronic cardiac dysfunction with impaired contractile responsiveness to stress stimuli and/or impaired diastolic relaxation with electrophysiological abnormalities particularly QT interval prolongation in the absence of other known cardiac disease. Cirrhotic Cardiomyopathy occurs in variable degrees in patients with liver cirrhosis with an estimated prevalence of 40% to 50%.
In patients with liver cirrhosis, portal hypertension leads to a hyperdynamic circulatory state characterized by reduced systemic vascular resistance, resulting in decreased afterload, increased preload, and consequently elevated cardiac output. These hemodynamic alterations may obscure early manifestations of systolic dysfunction and subtle reductions in myocardial contractility.
Cirrhotic cardiomyopathy is often under-diagnosed as it is not widely recognized because most of the patients are asymptomatic at rest with overt heart failure being uncommon. This latent cardiomyopathy is recognized when patients with liver cirrhosis experience pathological, physiological, or pharmacological stress. Cirrhotic cardiomyopathy assumes clinical importance in the setting of events that challenge the heart such as liver transplant surgery and the insertion of transjugular intrahepatic porto-systemic shunts (TIPS). These stressful procedures can precipitate acute cardiac failure due to hemodynamic instability. Currently, there is a lack of data on the prevalence of cirrhotic cardiomyopathy in Kenya. Therefore, this study aims to determine the prevalence of cirrhotic cardiomyopathy among patients with liver cirrhosis at Kenyatta National Hospital.
2. Materials and Methods
Area of study: The study was carried out at KNH outpatient liver clinic and inpatient medical wards from March to June 2018. KNH is public tertiary teaching and referral hospital. The hospital runs weekly based liver clinic.
Study Subjects: The study participants consisted of patients with diagnosis of liver cirrhosis that were on follow-up at KNH liver clinic or admitted to the medical wards.
Study design: Descriptive cross-sectional study.
Inclusion criteria: These included patients who were ≥13 years diagnosed with liver cirrhosis irrespective of aetiology.
Ethical considerations: Approval to perform the study was sought from Kenyatta National Hospital-University of Nairobi Ethical and Research committee (KNH-UoN ERC).
Data collection: The study subjects were recruited from Kenyatta National Hospital liver clinic and inpatient medical wards by the principal investigator (PI). Patients meeting the inclusion criteria were given information about the study and asked to participate in the study. Eligible patients agreeable to take part in the study signed a certificate of consent. All study subjects underwent echocardiography by trained echocardiography technician and recorded videos were reviewed by two cardiologists. Philips iE33 machine equipped with S5-1 Sector Array Transducer was used. Modified Simpson’s technique in apical 4 chamber view was used to calculate Left Ventricular (LV) ejection fraction (EF). Pulsed Wave Doppler echocardiography in the setting of apical four chamber view and the sample volume placed at the tips of the mitral leaflets was used to obtain trans-mitral LV filling velocity measurements from three successive cardiac cycles to measure the peak early diastolic flow velocity (E), the peak of atrial flow velocity (A) and E/A ration.
Data analysis: The data collected was analyzed by means of SPSS and presented in tables and graphs. The accepted level of significance was 5%.
3. Results
The mean age of the study participants was 44 years, 27 (61.4%) were males. Liver disease severity was characterized according to child pugh score. Out of 44 patients, 17 (38.6%) belonged to class A, 16 (36.4%) belonged to class B, and 11 (25.0%) to belonged to class C.
| Characteristic | n= 44 | |
|---|---|---|
| Age, (Mean in yrs, ± SD) | 44.0 (12.2) | |
| Gender (n, %) | • Male • Female |
27 (61.4) 17 (38.6) |
| Marital status (n, %) | • Single • Married • Widowed |
14 (31.8) 27 (61.4) 3 (6.8) |
| Education level (n, %) | • None • Primary • Secondary • Tertiary |
2 (4.5) 22 (50.0) 18 (40.9) 2 (4.5) |
| Occupation (n, %) | • Self employed • Employed • Unemployed • Retired |
21 (47.7) 4 (9.1) 17 (38.6) 2 (4.5) |
| Child Pugh Score (n, %) | • A • B • C |
17 (38.6) 16 (36.4) 11 (25.0) |
| Jaundice (n, %) | • Yes • No |
26 (59.1) 18 (40.9) |
| Ascites (n, %) | • Yes • No |
26 (59.1) 18 (40.9) |
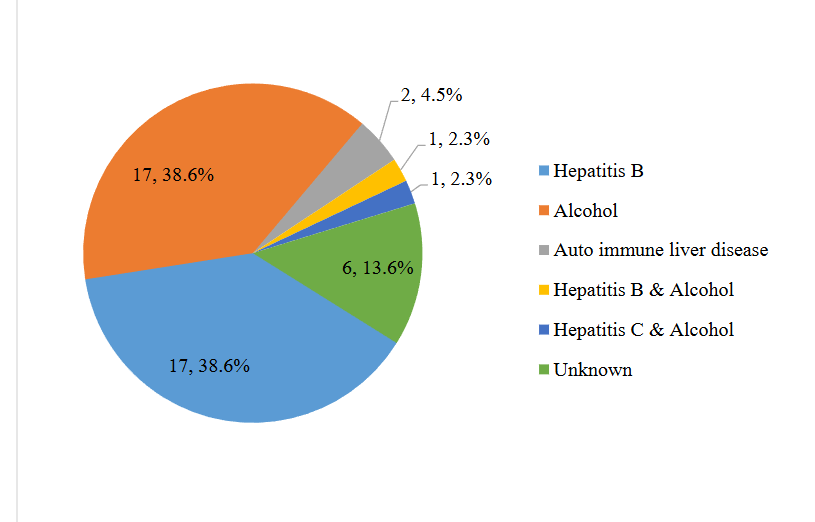
Regarding the etiology of liver cirrhosis, 17 (38.6%) patients had hepatitis B infection and another 17 had history of chronic alcohol consumption. Two patients had both viral hepatitis and history of chronic alcohol intake. 2 (4.5%) patients had auto-immune liver disease and 6 (13.6%) patients had unknown etiology of liver cirrhosis.
Only 3 (6.8%) patients were found to have systolic dysfunction. 18 (40.9%) participants had age-adjusted E/A ratio <1. One (2.3%) patient had E/A ration >1.5, deceleration time (DT) <160ms, and isovolumetric relaxation time (IVRT) <70ms. Overall, 19 (43.2%) patients were found to have diastolic dysfunction based on the above parameters. 18 (94.7%) had grade1 diastolic dysfunction, only 1 (5.3%) patient had grade 3 diastolic dysfunction. Resting 12-lead ECG showed normal QTc interval in 19 (43.2%) patients while 25 (56.8%) patients had prolonged (>440ms) QTc interval.
| Echocardiographic & ECG Findings | All Study Participants | N | % |
|---|---|---|---|
| Systolic function (LVEF): | • Abnormal (<55 %) • Normal (>55 %) |
3 41 |
6.8 93.2 |
| Diastolic dysfunction E/A ratio | • <1 • >1.5 • >1 |
19 18 1 |
43.2 40.9 2.3 |
| Deceleration time (DT) | • >200 ms • <200 ms |
18 26 |
40.9 59.1 |
| IVRT (ms) | • >80 • <80 |
18 26 |
40.9 59.1 |
| Diastolic dysfunction Grades | • Grade I • Grade II • Grade III |
18 0 1 |
94.7 5.3 |
| QTc Interval | • Prolonged (>440 ms) • Normal (<440 ms) |
25 19 |
56.8 43.2 |
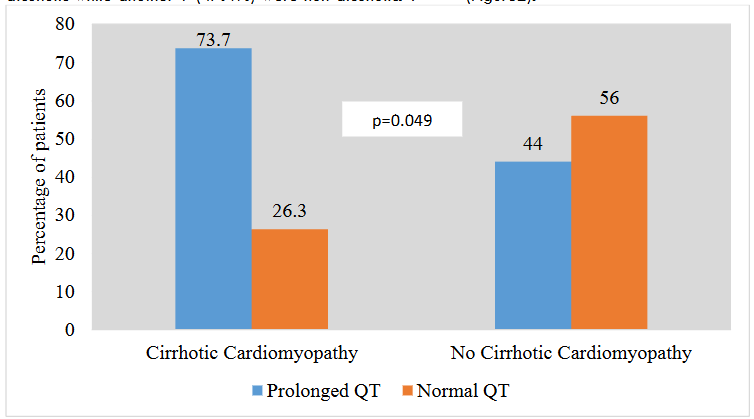
Out of 44 study participants, 19 (43.2%) patients had evidence of cirrhotic cardiomyopathy indicated by systolic and/or diastolic dysfunction. 3 (6.8%) patients had both systolic and diastolic dysfunctions while 16(36.4%) had only diastolic dysfunction. 9 (47.4%) of the patients who had cirrhotic cardiomyopathy were alcoholic while another 9 (47.4%) were non-alcoholic. 1 (5.3%) patients had both hepatitis C infection and history of chronic alcohol intake. Out of 19 patients with cirrhotic cardiomyopathy, 14 (73.7%) of them had prolonged (>440 ms) QTc interval. Chi square statistical test applied to this have showed significant (P=0.049) association between QTc prolongation and cirrhotic cardiomyopathy.
Out of 19 patients with cirrhotic cardiomyopathy, 5 (26.4%) were child pugh class A, 7(36.8%) were class B, and 7(36.8%) class C. The severity of liver disease as assessed by child Pugh score did not show significant association with cirrhotic cardiomyopathy (p=0.117). During study procedures, other echocardiographic and ECG abnormalities were observed.
| Echo and ECG Findings | All study participants | N | % |
|---|---|---|---|
| Pulmonary Hypertension | • Mild • Moderate |
11 10 |
90.9 9.1 |
| Tricuspid Regurgitation | • Mild • Moderate |
16 14 |
36.4 87.5 12.5 |
| Mitral Regurgitation | • Mild • Moderate • Severe |
6 3 2 |
13.6 50.0 33.3 16.7 |
| Aortic Regurgitation | 1 | 2.3 | |
| LVH | 5 | 11.4 | |
| Pleural Effusion | 6 | 13.6 | |
| Pericardial Effusion | 3 | 6.8 | |
| Premature ventricular contractions (PVCs) | 4 | 9.1 | |
| Sinus tachycardia | 4 | 9.1 | |
| Sinus bradycardia | 2 | 4.5 | |
| 1st degree AV block | 2 | 4.5 | |
| Low voltage | 3 | 6.8 | |
| RBBB | 1 | 2.3 | |
| Left axis deviation | 1 | 2.3 | |
| Right axis deviation | 1 | 2.3 | |
| Atrial Premature complex | 1 | 2.3 |
4. Discussion
The study was designed to find out the prevalence of cirrhotic cardiomyopathy among liver cirrhosis patients at Kenyatta National Hospital. Regarding the stage of liver disease, majority (75%) of the patients were in child Pugh class A & B. Alcohol and hepatitis B infection were the main causes of liver cirrhosis in this study. In this study, 6.8% of the participants had systolic dysfunction on resting echocardiography. In Portugal, Barbosa M et al reported systolic dysfunction in 38.5% of cirrhotic patients in whom, alcohol was the major aetiology of cirrhosis. As in our study, most of the patients were in child Pugh class A & B. Barbosa et al used stress echocardiography with intravenous infusion of dobutamine instead of resting echocardiography to evaluate left ventricular systolic function. Another study by Moon young et al in Korea showed left ventricular blunted response defined by <10% increase of ejection fraction after dobutamine infusion in 25.4% of liver cirrhosis who had normal systolic function at rest. Systolic dysfunction is commonly manifested during physiologic, pharmacologic or pathologic stressful conditions in cirrhosis patients. The higher numbers of systolic dysfunction in these two studies compared to our study could be due to the different echocardiographic modality used in these studies. This supports the concept of subtle systolic dysfunction in liver cirrhosis. Stress echocardiography is an ideal tool to unmask this latent contractile dysfunction.
In this study, diastolic dysfunction was present in 43.2% of the cases, mainly driven by reduced E/A ratio. This is consistent with what is reported in other several studies. In India, Shweta Patil et al reported diastolic dysfunction in 48.3% of the study cases. Shweta Patil et al used Model for End stage liver disease (MELD) Score to assess liver disease severity and most of the patients were in MELD stage 1. This is comparable to child Pugh class B in present study; so, liver disease severity was almost same with our patients. In Egypt, Manal Eldeeb et al found 66.6% of diastolic dysfunction in non-alcoholic Liver Cirrhosis Patients. Majority of the cases were in child Pugh class B & C compared to our study where most of the cases were in classes A & B. Another study in Egypt by Mashahit et al reported diastolic dysfunction in 60% of liver cirrhosis patients. Studies have shown that diastolic dysfunction is well correlated with liver disease severity. Some of the differences observed in prevalence of diastolic dysfunction across different populations are attributable to liver disease severity difference in different study populations.
Most of the studies on cirrhotic cardiomyopathy used mitral E/A ratio for diastolic dysfunction assessment. In our study we similarly applied age-adjusted E/A ratio to detect diastolic dysfunction.
In this study we recorded 43.2% prevalence of cirrhotic cardiomyopathy determined by presence of left ventricular systolic or diastolic dysfunction irrespective of QTc interval prolongation. The high burden of cirrhotic cardiomyopathy in this study is mostly driven by diastolic dysfunction. Our findings are consistent with what other studies reported in populations of Europe, America, Asia and Africa. Barbosa et al in Portugal reported prevalence of 61.5% of cirrhotic cardiomyopathy. Barbosa et al used dobutamine stress echocardiography to detect systolic dysfunction which commonly not revealed by resting echocardiography that we used in our study. This may explain the higher prevalence of cardiomyopathy in this study compared to our findings. In USA, T. Belay et al found 51.1% prevalence of cirrhotic cardiomyopathy. T. Belay et al used both systolic and diastolic dysfunction, and prolonged QTc interval to define cirrhotic cardiomyopathy. Use of prolonged QTc interval as defining criteria for cirrhotic cardiomyopathy and the larger sample size may explain the slightly higher figures of cirrhotic cardiomyopathy in this study compared to our findings. Studies on similar populations in India and Pakistan had reported results closer to our findings. S. Sakthi et al in India studied the prevalence of cirrhotic cardiomyopathy by using resting echocardiographic findings of systolic and diastolic dysfunction. He found 44.6% prevalence of cirrhotic cardiomyopathy. In Pakistan Sajid et al reported 49.0% prevalence of cirrhotic cardiomyopathy. In Egypt, study conducted by Abdel-bary et al showed 64% prevalence of cirrhotic cardiomyopathy in 66 HCV-related liver cirrhosis patients. This Egyptian study had higher sample size and higher child Pugh class C indicating that majority of the participants got advanced liver cirrhosis compared with ours.
The present study showed high prevalence of cirrhotic cardiomyopathy in this African black population. Published studies have revealed that cirrhotic cardiomyopathy is linked to poor survival and significant mortality due to heart failure and arrhythmias flowing invasive interventions such as hepatic transplantation and TIPS. Cirrhotic cardiomyopathy has been also related with the development of hepato-renal syndrome as it may cause alterations in the effective circulatory blood volume. Sepsis, such as spontaneous bacterial peritonitis generates metabolic stress due to increased production of inflammatory cytokines. This may result in an overt heart failure in cirrhotic patients. Therefore; perioperative and post-operative cardiac evaluation by echocardiography and ECG, and careful monitoring of cardiac decompensation in all stressful conditions is a reasonable recommendation. Hepatic transplantation is the sole definitive therapy for advanced liver cirrhosis and it is reported to resolve liver cirrhosis related cardiovascular disorders.
In our study, we recorded 56.8% prevalence of QTc interval prolongation that was highly present in subjects with evidence of cirrhotic cardiomyopathy. Prolongation of QTc interval is not uncommon in liver cirrhosis as several previous studies revealed. In Portugal, Borbosa M et al documented QTc prolongation in 68.8% of liver cirrhosis patients. Om Parkash in Pakistan reported 35% prevalence of QTc prolongation among liver cirrhosis patients, 33.8% of the cases in their study were on beta-blocker therapy which shortens QTc interval; this may explain the difference in findings with the present study. Leili et al in Iran reported QTc prolongation in 63.5% of cirrhotic cases. The bigger sample size of this study may explain the difference with our study. Long QTc interval is shown to be associated with lower survival rate and risk of arrhythmias in patients with liver cirrhosis. Therefore, it is worth to monitor QTc interval in subjects with advanced liver cirrhosis with attention to any modifiable cause of QTc prolongation such as medications and electrolyte imbalance.
This study did not show correlation between cirrhotic cardiomyopathy and child pugh score (p =0.117). Previous studies reported positive correlation while others found negative correlation between cirrhotic cardiomyopathy and child pugh score. Study by T. Perumal et al in India showed association of cirrhotic cardiomyopathy and the stage liver dysfunction as determined by child pugh score. In this study, majority (66%) of the subjects were in child pugh class C. Another Indian study by P. Thirumal et al also established association between child pugh score and the occurrence of cirrhotic cardiomyopathy. This study also had higher proportion (70.7%) of child pugh class C cases compared to the present study. Lack of association between cirrhotic cardiomyopathy and child pugh severity score in our study may be due to inadequate sample size. Our study had limitation that resting echocardiography was used to evaluate systolic function. This may have underestimated the burden of systolic dysfunction.
Conclusion
Our study concluded that the prevalence of cirrhotic cardiomyopathy as mainly driven by diastolic dysfunction was high in this black African population of liver cirrhosis. Long QTc interval was common in this study. This was significantly higher in patients with cirrhotic cardiomyopathy. Therefore; we recommend cardiac evaluation by 2-D echocardiography and 12-lead ECG for patients with liver cirrhosis.
Acknowledgements
I am very grateful to my supervisors for their contribution in ideas, feedback and advice in this research project. I also thank the hospital administrations in Kenyatta National Hospital for their permission and co-operation, during the period of this work.
Conflict of Interest
There is no conflict of interest in this study.
References
- Henriksen JH, Møller S. Cardiac and systemic haemodynamic complications of liver cirrhosis. Scandinavian cardiovascular journal: SCJ. 2009;43(4): 218-225.
- Rahman S, Mallett S V. Cirrhotic cardiomyopathy: Implications for the perioperative management of liver transplant patients. World Journal of Hepatology. 2015;7(3):507-520.
- Izzy MJ, VanWagner LB. Current Concepts of Cirrhotic Cardiomyopathy. Clin Liver Dis.W.B. Saunders. 2021;25(2):471-481.
- Liu H, Naser JA, Lin G, Lee SS. Cardiomyopathy in cirrhosis: From pathophysiology to clinical care. JHEP Reports. 2024;6(1):100911.
- Rahman S, Mallett S V. Cirrhotic cardiomyopathy: Implications for the perioperative management of liver transplant patients. World Journal of Hepatology. 2015;7(3):507-520.
- Barbosa M, Guardado J, Marinho C, et al. Cirrhotic cardiomyopathy: Isn’t stress evaluation always required for the diagnosis? World Journal of Hepatology. 2016;8(3):200-206.
- Kim MY, Baik SK, Won CS, et al. Dobutamine stress echocardiography for evaluating cirrhotic cardiomyopathy in liver cirrhosis. Published online 2010:376-382.
- Patil S, Lal B, Pandey M, Haldia SS, Rishi JP. A Clinical Study of Cardiovascular Dysfunction in Patients of Cirrhosis of Liver . (2):212-215.
- Eldeeb M, Fouda R, Hammady M, Rashed L. Echocardiographic Evaluation of Cardiac Structural and Functional Changes in Hepatitis C Positive Non-Alcoholic Liver Cirrhosis Patients and Their Plasma NT-ProBNP Levels Evaluation of Cardiac Structural and Functional Changes in Hepatitis C Positive Non. Vol 9.; 2012.
- Mashahit M, Younan H, El-amir M, Mohamed A. Cirrhotic Cardiomyopathy in Egyptian Patients. 2016;3(3):2336.
- Salari A, Shafaghi A, Ofoghi M, Saeidinia A, Mansour-Ghanaei F. Diastolic dysfunction and severity of cirrhosis in nonalcoholic cirrhotic patients. International journal of hepatology. 2013;2013:892876.
- Belay T, Gress T, Sayyed R. Cirrhotic cardiomyopathy among patients with liver cirrhosis. American Journal of Gastroenterology. 2013;108(December):S129.
- S Sakthi Selva Kumar. Prevalence of cirrhotic cardiomyopathy in patients with cirrhosis of liver. Japi. 2016;64(january):1-29.
- Nisar S, Qazi S, Ashraf Y. To assess the frequency of cirrhotic cardiomyopathy among patients with cirrhosis of liver. Pakistan Journal of Medical and Health Sciences. 2013;7(3):851-854.
- Abdel-Bary SA, Safwat E, Galal H. Correlation of echocardiographic and electrocardiographic changes with liver disease severity in HCV-related liver cirrhosis. Egyptian Liver Journal. 2013;3(2):35-40.
- Ruíz-del-Árbol L, Achécar L, Serradilla R, et al. Diastolic dysfunction is a predictor of poor outcomes in patients with cirrhosis, portal hypertension, and a normal creatinine. Hepatology. 2013;58(5):1732-1741.
- Cazzaniga M, Salerno F, Pagnozzi G, et al. Diastolic dysfunction is associated with poor survival in patients with cirrhosis with transjugular intrahepatic portosystemic shunt. Gut. 2007;56(6):869-875.
- Liu H, Jayakumar S, Traboulsi M, Lee SS. Cirrhotic cardiomyopathy: Implications for liver transplantation. Liver Transplantation. 2017;23(6):826-835.
- Ih N, Mahmood K, Talib A, Nz R. Looking Beyond Liver ! Cirrhotic Cardiomyopathy: Pathophysiology, Clinical Presentation and Management Strategies. 2017;2(1):1-11.
- Torregrosa M, Aguadé S, Dos L, et al. Cardiac alterations in cirrhosis: reversibility after liver transplantation. Journal of Hepatology. 2005;42(1):68-74.
- Parkash O, Alishah H, Mohyuddin GR, Ayub A, Jafri W, Hamid S. Electrophysiological changes in patients with liver cirrhosis in tertiary care hospital Karachi Pakistan. Hepatology International. 2013;7(4):S529.
- Pourafkari L, Ghaffari S, Nazeri L, et al. Electrocardiographic findings in hepatic cirrhosis and their association with the severity of disease. Cor et Vasa. 2017;59(2):e105-e113.
- Mozos I. Arrhythmia risk in liver cirrhosis. World Journal of Hepatology. 2015;7(4):662-672.
- Josefsson A, Fu M, Allayhari P, et al. Impact of peri-transplant heart failure & left-ventricular diastolic dysfunction on outcomes following liver transplantation. Liver International. 2012;32(8):1262-1269.

