Preventing Catheter-Associated UTIs: A Complex Review
Assessment of the Complexity of Preventing Catheter-Associated Urinary Tract Infection and Bacterial Biofilms
Jose Luis Amorone Leiva¹, Claudia Caballero Gonzalez¹, Jose Gabriel Alvarez Rodriguez¹, Andres Patricio Vera Macias², Maria Del Pilar Vera²
- Hospital Mariano y Luciano de la Vega, Moreno, Buenos Aires, Argentina. Universidad de Morón.
- Hospital Mariano y Luciano de la Vega, Moreno, Buenos Aires, Argentina.
OPEN ACCESS
PUBLISHED: 30 September 2025
CITATION Amorone Leiva, JL., Caballero Gonzalez, C., et al., 2025. Assessment of the Complexity of Preventing Catheter-Associated Urinary Tract Infection and Bacterial Biofilms. Medical Research Archives, [online] 13(9). https://doi.org/10.18103/mra.v13i9.6951
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v13i9.6951
ISSN 2375-1924
Abstract
Catheter-associated urinary tract infections (CAUTI) are among the most common healthcare-associated infections, arising from biofilm-forming bacteria and cause increased morbidity, mortality, length of stay and hospital costs. The linear model of the microbial theory of disease has failed to solve CAUTI. It was hoped that, knowing the nosological diagnosis (urinary tract infection), the cause (bacteria) and its treatment (antibiotic), the cure would be simple. However, the prevention and treatment of CAUTI imposes challenges, which can only be addressed, if it is understood that CAUTI is a complex entity where different actors and multiple factors interact, in various settings. The objective of this article is to review the intervention of different actors and elements in the prevention of CAUTI, focusing on the assessment of the complexity of preventing CAUTI and bacterial biofilm. The unnecessary use of indwelling urethral catheter is the primary risk factor for CAUTI, so the use of the catheter with appropriate indications, its proper insertion and maintenance, and its rapid removal are recommended. In selected patients, alternatives to indwelling urethral catheterization, such as the male external catheter and the prostatic stent, may be considered, although they are not without complications. The health care setting should be considered in the design of prevention strategies. CAUTIs in intensive care units are associated with increased lengths of stay, healthcare costs and antibiotic resistance development. In the twenty-first century, the proportion of older people tends to increase globally, which adds value to the fact that elder age is a significant risk factor for CAUTI. Biofilm is the central factor in the pathogenesis of CAUTI. Various antibiofilm strategies have been designed because once biofilms are formed, they are difficult to treat. Biomedical technology continuously introduces new antifouling and antibacterial materials for urethral catheter coating. The efficiency of the different coatings is proven in vitro, in animal models, and clinical trials. However, its application in clinical settings has not been satisfactory. Education should include both patients and their families and/or caregivers as well as health personnel. Patient and family participation is necessary in the patient’s care. It is important to consider the values, attitudes, and beliefs of health personnel, which will impact clinical practice. The overload of work of health personnel and inadequate and/or insufficient infrastructure are added as barriers to adherence to prevention recommendations. Therefore, it is necessary to address and optimize the complexity of CAUTI care, through a multimodal and multidisciplinary intervention, which minimizes its incidence and complications.
Keywords
- Catheter-associated urinary tract infections
- Bacterial biofilms
- Healthcare-associated infections
- Indwelling urethral catheter
- Antibiofilm strategies
Introduction
In 1500 B.C. an ancient Egyptian papyrus described the use of a urethral catheter (UC) to relieve urinary retention. Currently, the use of long-term indwelling urethral catheters is a basic need in inpatient care. Approximately 20% of hospitalized patients have a urethral catheter at any given time. The global urethral catheter market size was estimated at USD 5,229.6 million in 2022 and is projected to reach USD 8,060.9 million by 2030, growing at a compound annual growth rate of 5.4% from 2023 to 2030. In terms of region, North America was the highest revenue-generating market in 2023.
Urinary Tract Infections are the most common type of healthcare-associated infection, accounting for more than 30% of infections reported by acute care hospitals. The insertion of a urethral catheter allows bacteria to colonize the urinary tract, promoting infection. Catheter-associated urinary tract infections (CAUTI) are among the most common healthcare-associated infections, arising from biofilm-forming bacteria. They cause increased morbidity, mortality, length of stay, and hospital costs. CAUTIs are the main cause of bacteremia associated with secondary health care.
In USA, 1.3 CAUTI per 1000 UC-days were reported. The length of stay was 6.57 days for patients without health care–associated infections and 24.41 days for those with CAUTI. The mortality rates were 14.06% for patients without urethral catheter and 31.14% for those with CAUTI. The costs associated with each CAUTI were USD$1006 per case.
Although research activities dedicated to CAUTI are advancing, a permanent solution to this problem has not yet been found. The evaluation and management of CAUTI presents significant complexity due to the intervention of different actors, such as the patient and health professionals and administrators, and multiple factors from urethral catheters and antibiotics to available resources. Each of these elements has different particularities. Adding to this complexity is the fact that CAUTI can occur in a variety of settings, including intensive care, long-term care, outpatient and trauma settings, each with unique risk factors and contributing factors.
Despite the publication of several studies and guidelines on CAUTI and bacterial biofilms, many challenges remain regarding their prevention and treatment. This article aims to review every element involved in the production of CAUTI and biofilms to equip healthcare providers with the most effective and innovative approaches on how to address and optimize the complexity of CAUTI care, leading to minimizing its incidence and complications.
CAUTI: Definition
Different definitions of CAUTI have been used in research, which makes meta-analyses difficult and prevents us from assessing the quality of the evidence in the literature. Some of them include asymptomatic bacteriuria, which is not a clinical infection. This can lead to an overestimation of CAUTI rates and possible misallocation of resources. The National Health Care Safety Network (NHSN) revised the criteria for defining urinary tract infection (UTI) surveillance. It considers CAUTI to be a symptomatic urinary tract infection in a patient who has had permanent urethral catheter for more than two consecutive days at a hospitalization site on the date of the event.
Along these lines, the diagnosis of CAUTI requires:
- Positive urine culture with no more than two species of organism, at least one of which is a bacterium of ≥ 105 CFU/ml. All elements of the UTI criterion must occur during the Infection Window Period. If none of the organisms present at ≥ 105 CFU/ml are bacteria, it’s excluded.
- Had an indwelling UC that had been in place for > two days and was either:
- a) still present for any portion of the calendar day on date of event or
- b) removed day before date of event.
- At least one of the following signs of symptoms:
- a) Any age patient: fever (>38.0°C), hypothermia (<36.0°C); or suprapubic tenderness, or costovertebral angle pain, with no other recognized cause; or urgency, dysuria, frequency (these symptoms cannot be used when UC is in place).
- b) Patients ≤1 year of age: fever (>38.0°C), hypothermia (<36.0°C); or suprapubic tenderness, costovertebral pain, apnea, bradycardia, lethargy, or vomiting, with no other recognized cause.
Urethral Catheter & CAUTI
The urethral catheter plays a leading role in the prevention of CAUTI. Its placement should be justified by an appropriate clinical indication: acute urinary retention, chronic urinary retention without bladder outlet obstruction, accurate measurement of urinary output in critically ill patients, perioperative use for selected surgical procedures, assistance in the healing of stage III and IV perineal and sacral wounds in incontinent patients, palliative care. The urethral catheter should be removed quickly once it is no longer needed.
In routine clinical practice, the professional should select the catheter and drainage system that fits the indication. Consider the material, design, caliber, length, and the ball’s capacity. Care should be taken with the techniques for insertion and removal of the catheter with adequate hand hygiene. All procedures must be recorded in writing.
The evidence-based European Association of Urology recommends: treating symptomatic CAUTI according to recommendations for complicated Urinary Tract Infections; perform a urine culture before starting antibiotic treatment in catheterized patients who have had their catheter removed; not medicating asymptomatic catheter-associated bacteriuria; treat catheter-associated asymptomatic bacteriuria prior to traumatic urinary tract interventions (e.g., transurethral resection of the prostate); replace or remove the catheter before starting antibiotic treatment; do not apply topical antiseptics or antibiotics to the catheter, the urethra or meatus; not using prophylactic antibiotics to prevent CAUTI; not routinely using antibiotic prophylaxis to prevent clinical urinary tract infections after catheter removal. The use of silicone catheters is associated with a lower risk of incrustation in indwelling catheterization.
Urethral catheter: Alternatives
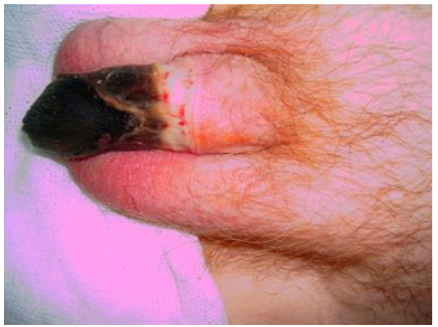
In selected patients, alternatives to indwelling urethral catheterization, such as the male external catheter, may be considered. This external catheter is not a true catheter, it is a simple sheath that is placed over the penis, and it has a drainage tube that allows urine to pass into a storage bag fastened around the leg. The male external catheter is also known as a: condom catheter. It may be appropriate for cooperative men without urinary retention or bladder outlet obstruction who are not expected to frequently manipulate the urinary catheter because of behavioural problems such as delirium. External catheters are especially useful for the treatment of incontinence in cooperative elderly male patients with dementia. They are better tolerated than the urethral catheter. However, irritative, allergic, and compressive complications related to the use of a male external catheter have been reported. Routine care of condom catheters is important to prevent complications, which can be serious such as penile necrosis.
The urethral stent is a self-expanding steel mesh that opens the urethral lumen. It is a procedure under vision, outpatient, with little or no bleeding and does not require a urethral catheter, the patient urinates immediately after stent implantation. It can be considered as an alternative to the indwelling urethral catheter in selected patients with bladder outlet obstruction. It’s a very good option in elderly patients with benign prostatic hyperplasia, chronic retentionists and with high surgical risk with multiple pathologies and polypharmacy or who refuse surgery.

CAUTI & Health Care Settings
The health care setting should be considered in the design of CAUTI prevention strategies. This can be an acute hospital, a long-term care facility, a post-acute care facility, or an outpatient setting. In the same hospital there are different healthcare settings, e.g. intensive care units, acute care wards.
A meta-analysis has been published concluding that CAUTIs in intensive care units are associated with increased lengths of stay, healthcare costs and antibiotic resistance development. Another meta-analysis reports that, in the intensive care unit, the infection rates are 3–5 times higher than other hospital patient care areas and the incidence of CAUTI is 7.78 per 1000 catheter days. In a US study, a mean of 3.2 urinary tract infections per 1000 catheter days was reported in long-term care facilities.
CAUTI & Age
A retrospective cohort and case-control study with 3598 eligible patients reported that CAUTI usually occurs in hospitalized pediatric patients. Pediatric outpatient CAUTI occur in 18% of patients with catheters; are associated with morbidity and health care utilization. The twenty-first century is characterized by a global and unprecedented phenomenon of population aging, driven by the decline in the birth rate and the increase in life expectancy. Elder age is a significant risk factor for CAUTI. In a case-control study, CAUTI occurred in 182 of 7295 elderly hospitalized patients, and the infection rate was 3.4/per 1000 catheter days. Urine pH ≥ 6.5, moderate dependence or severe dependence in the classification of self-care ability, age ≥ 74 years, male sex, hospitalization ≥ 14 days, indwelling urinary catheter ≥ 10 days, diabetes and malnutrition were independent risk factors for CAUTI.
CAUTI & Biofilms
Biofilm formation on the urethral catheter is a key aspect in the pathogenesis of CAUTI. Biofilms were first described by Antonie van Leeuwenhoek. Using his groundbreaking microscopes, in the 17th century, he observed the presence of communities of microbes that he called the “animalcules” in the plaque of his own teeth. In the 20th century, numerous papers were written about microbial films. The first use of “biofilm” in a publication is from the journal Microbial Ecology in 1975. The importance of biofilms was recognized by the American Society for Microbiology in 1993. In 1999, Costerton et al. described biofilm as an organized population of microbes encased in a polymeric matrix produced by the microbe that attaches to a surface. His scientific contributions were joined by those of Dr. Characklis, which led to the opening of the Center for Biofilm Engineering at Montana State University in 1990. Since then, the field of biofilm research has experienced a huge boom. Numerous research laboratories around the world study biofilms. In recent decades, the definition of biofilm has been modified.
At the beginning of the twenty-first century, Dolan and Coleman propose a new definition of a biofilm: it is a microbially derived sessile community characterized by cells that are irreversibly attached to a substratum or interface or to each other, are embedded in a matrix of extracellular polymeric substances that they have produced and exhibit an altered phenotype with respect to growth rate and gene transcription.
Biofilm Formation
The formation of biofilms is a complex multifactorial phenomenon that generally develops in stages. First, organic, inorganic and cellular components, which are found in the environment surrounding the urethral catheter, are deposited on it. Bacteria migrate to the catheter and interact with this surface through appendages, attractive forces, or adhesins. Reversible fixation occurs through non-specific physical interactions. This initial attachment of bacteria to the catheter allows for subsequent irreversible adhesion to the catheter surface. Bacteria proliferate rapidly and form microcolonies. Simultaneously, gene expression is activated for components needed for a biofilm matrix, such as polysaccharides, proteins, eDNA, and lipids. The cells continue to proliferate, and additional surrounding planktonic cells are also incorporated into the biofilm. The biofilm increases in thickness and transforms into a mature biofilm with a three-dimensional arrangement, called a “mushroom” structure. Water-filled channels are formed in the biofilm that allow for the transport of nutrients, signalling molecules, and waste removal. Once the biofilm is mature, planktonic bacteria break away from the biomaterial, migrate and colonize new surfaces, spreading the infection.
Biofilm & Antibiotic Resistance
Biofilm is responsible for antibiotic resistance, one of the top ten threats to global public health. The bacteria resident in the biofilm are protected by the matrix of extracellular polymeric substances. They have greater tolerance to antibiotics and greater resistance to the immune system compared to free-floating bacteria. They tolerate up to 1000 times more concentration of antibiotics than planktonic ones.
Numerous studies are devoted to the study of biofilm formation. An in vitro model of the catheterized urinary tract has been presented, which replicates the closed drainage system of the catheter as used in clinical practice, whose purpose is to facilitate the study of CAUTI and the catheter biofilm under representative conditions. Microfluidics-based research is being applied to understand the dynamic mechanism of bacterial adhesion and biofilm development. Some authors criticize the concept that the indwelling catheter assumes a passive role as a conduit between the outside world and the sterile bladder environment. They suggest that a patient’s basal urogenital microbiota may predispose them to the development of CAUTI.
Antibiofilm Strategies
Various antibiofilm strategies have been designed because once biofilms are formed, they are difficult to treat. Biomedical technology continuously introduces new antifouling and antibacterial materials for the urethral catheter coating, to prevent microbial attachment, or antimicrobial effect, respectively. The efficiency of the different coatings is proven in vitro, in animal models of CAUTI, and clinical trials. Unfortunately, Andersen and Flores-Mireles’ question remain unanswered: why do such promising coatings that come out of research fall short when translated to the bedside?
Antimicrobial Coatings
The mechanism of action of antimicrobial coatings would be the inhibition of the synthesis of nucleic acids and proteins involved in the synthesis of the cell wall of the bacterium. Various materials have been used, e.g. metals, antibiotics, nitric oxide, antimicrobial peptides, bacteriophages and natural bioactive molecules. Nanoparticles have been used to deliver metals, such as silver, gold, copper, and zinc. Recently, an antibacterial and antibiofilm coating with nanogel has been designed that encapsulates a mimetic ceragenin of the synthetic broad-spectrum antimicrobial peptide (CSA-131). In vitro and in vivo assays showed that the coating provided antimicrobial and antibiofilm activity for up to 7 days of catheterization, along with catheter lubricity, without toxic effects. Another study reports the synthesis of biocompatible silver nanoparticles immobilized in carboxymethyl chitosan. In vivo experiments revealed its superior antibiofilm performance in urinary catheters implanted in rat bladders.
Antifouling Coatings
Antifouling coatings prevent bacteria from adhering to the surface of the urethral catheter, which prevents the formation of biofilms. Unlike antimicrobial coatings, antifouling coatings do not harm bacteria but repel them. These antifouling properties of the material are achieved with physicochemical modifications of the polymers. The most mentioned are those coated with hydrogels, whose hydrophilic structure reduces bacterial growth. Other antifouling materials that can coat UC are poly(tetrafluoroethylene), polyzwitterions, and poly(ethylene glycol) and enzymatic coatings.
CAUTI & Education
Education should include both patients and their families and/or caregivers as well as health personnel. Communication with the patient and/or their relatives or caregivers is very important. The patient should know why a catheter is placed, what the procedure is like, and how to care for it. His privacy must be respected, in an appropriate environment and all their concerns must be listened to.
With the right scaffolding, professionals can prepare themselves, both in content and skills, to handle the complexity of CAUTI, in a learning environment. Lectures can be organized on indications for urethral catheter, strategies to reduce its use, information about CAUTI and alternatives to indwelling catheter. Staff should be trained in both urethral catheter insertion, maintenance, and removal procedures. Simulation training and case-based education are useful tools. Likewise, daily patient monitoring, electronic reminders, and urinary retention protocol are resources that require adequate training.
It is important to consider the values, attitudes, and beliefs of health personnel, which will impact clinical practice. The overload of work of health personnel and inadequate and/or insufficient infrastructure are added as barriers to adherence to prevention recommendations. Different types of evaluations can be carried out, including design, institutional, process, product, outcome and impact evaluations. The evaluation is an opportunity to obtain useful information to reorient the management of CAUTI prevention. It can be inserted at the beginning, in the course or at the end of the process. The intention is always to contribute to the success of prevention programs.
The alternatives for evaluating performance are diverse. Critical incident assessment, Objective Structured Clinical Examination and check list can be used.
CAUTI & Prevention Programs
Prevention programs are multidimensional approach and typically include bundles with sets of evidence-based practices, education, CAUTI surveillance, monitoring compliance with recommendations to prevent CAUTI, internal reporting of CAUTI rates, and performance feedback.
Conclusions
CAUTI are among the most common healthcare-associated infections. The linear model of the microbial theory of disease has failed to solve CAUTI. It was hoped that, knowing the nosological diagnosis (urinary tract infection), the cause (bacteria) and its treatment (antibiotic), the cure would be simple. However, the prevention and treatment of CAUTI imposes challenges, which can only be addressed, if it is understood that CAUTI is a complex entity where different actors and multiple factors interact, in various settings. The unnecessary use of indwelling urethral catheter is the primary risk factor for CAUTI, so the use of the catheter with appropriate indications, its proper insertion and maintenance, and its rapid removal are recommended. In selected patients, alternatives to indwelling urethral catheterization, such as the male external catheter and the prostatic stent, may be considered, although they are not without complications. CAUTIs in intensive care units are associated with increased lengths of stay, healthcare costs and antibiotic resistance development. The elder age is a significant risk factor for CAUTI. New antifouling and antibacterial materials for urethral catheter coating are reported. However, its application in clinical settings has not been satisfactory. Education should include both patients and their families and/or caregivers as well as health personnel. Therefore, it is necessary to address and optimize the complexity of CAUTI care, through a multimodal and multidisciplinary intervention, which minimizes its incidence and complications.
References
- anafy HM, Saad SM, Al-Ghorab MM. Ancient Egyptian medicine: contribution to urology. Urology. 1974 Jul;4(1):114-20.
- Saint S, Trautner BW, Fowler KE, Colozzi J, Ratz D, Lescinskas E, Hollingsworth JM, Krein SL. A Multicenter Study of Patient-Reported Infectious and Noninfectious Complications Associated With Indwelling Urethral Catheters. JAMA Intern Med. 2018 Aug 1;178(8):1078-1085.
- Saint S. Clinical and economic consequences of nosocomial catheter-related bacteriuria. Am J Infect Control. 2000 Feb;28(1):68-75.
- Grand View Research. Medical Devices. Urinary Catheters Market Size. Share & Trends Report, 2030. Report ID: GVR-1-68038-381-2. Available at: https://www.grandviewresearch.com/industry-analysis/urinary-catheters-market
- Klevens RM, Edwards JR, Richards CL,Jr, et al. Estimating health care-associated infections and deaths in U.S. hospitals, 2002. Public Health Rep. 2007;122(2):160-166.
- Centers for Disease Control and Prevention. Guideline for prevention of catheter-associated urinary tract infections (2009). January 2025. Available at: https://www.cdc.gov/infectioncontrol/hcp/cauti/background.html?CDC_AAref_Val=https://www.cdc.gov/infectioncontrol/guidelines/cauti/background.html
- European Association of Urology Guidelines on Urological Infections: Summary of the 2024 Guidelines. Kranz, Jennifer et al. European Urology, Volume 86, Issue 1, 27 – 41.
- Rosenthal VD, Memish ZA, Nicastri E, Leone S, Bearman G. Preventing catheter-associated urinary tract infections: A position paper of the International Society for Infectious Diseases, 2024 update. Int J Infect Dis. 2025 Feb;151:107304.
- Stone PW. Economic burden of healthcare-associated infections: an American perspective. Expert Rev Pharmacoecon Outcomes Res 2009; 9 :417–22. doi: 10. 1586/erp.09.53.
- Patel PK, Advani SD, Kofman AD, et al. Strategies to prevent catheter-associated urinary tract infections in acute-care hospitals: 2022 Update. Infection Control & Hospital Epidemiology. 2023;44(8):1209-1231.
- National Institute for Health and Care Excellence (NICE). Infection prevention and control. QS61. April 2014. Available at: https://www.nice.org.uk/guidance/qs61
- Reid, S., Brocksom, J., Hamid, R., Ali, A., Thiruchelvam, N., Sahai, A., Harding, C., Biers, S., Belal, M., Barrett, R., Taylor, J. and Parkinson, R. (2021), British Association of Urological Surgeons (BAUS) and Nurses (BAUN) consensus document: management of the complications of long-term indwelling catheters. BJU Int, 128: 667-677.
- Kranz J, Bartoletti R, Bruyère F, Cai T, Geerlings S, Köves B, Schubert S, Pilatz A, Veeratterapillay R, Wagenlehner FME, Bausch K, Devlies W, Horváth J, Leitner L, Mantica G, Mezei T, Smith EJ, Bonkat G. European Association of Urology Guidelines on Urological Infections: Summary of the 2024 Guidelines. Eur Urol. 2024 Jul;86(1):27-41.
- The European Association of Urology Nurses (EAUN). V. Geng (Chair), H. Lurvink, I. Pearce, S. Vahr Lauridsen. Guideline Indwelling catheterisation in adults – Urethral and suprapubic. 2024 (online) Available at: https://nurses.uroweb.org/guideline/indwelling-catheterisation-in-adults-urethral-and-suprapubic/
- Amorone Leiva JL, María Agüero D, Caballero González C, Cecilia Postiglione M. Necrosis total de pene por cuidado inapropiado del colector urinario externo [Total penile necrosis due to inappropriate care of condom catheter]. Rev Esp Geriatr Gerontol. 2019 Jan-Feb;54(1):57-58.
- Amorone Leiva JL, Puscinski AJ. Recomendaciones para el tratamiento de LUTS por HPB. 2012.Rev. Arg. de Urol. Vol. 77 (Suplemento 1).
- Puscinski AJ, Amorone JL. Implante de Prótesis y Biomateriales en Urología. 2013. ISBN 9789872731991. NLM ID: 101660531.
- Werneburg GT. Catheter-Associated Urinary Tract Infections: Current Challenges and Future Prospects. Res Rep Urol. 2022 Apr 4;14:109-133.
- Chant C, Smith OM, Marshall JC, Friedrich JO. Relationship of catheter-associated urinary tract infection to mortality and length of stay in critically ill patients: a systematic review and meta-analysis of observational studies. Crit Care Med. 2011;39(5):1167–1173.
- Peng D, Li X, Liu P, et al. Epidemiology of pathogens and antimicrobial resistance of catheter-associated urinary tract infections in intensive care units: a systematic review and meta-analysis. Am J Infect Control. 2018;46(12):e81–e90.
- Stevenson KB, Moore J, Colwell H, Sleeper B. Standardized infection surveillance in long-term care interfacility comparisons from a regional cohort of facilities. Infect Cont Hosp Epidemiol. 2005;26(3):231–238.
- Rinke ML, Oyeku SO, Heo M, Saiman L, Zachariah P, Rosenberg RE, DeLaMora P, Rabin B, Mirhaji P, Klein E, Ford WJH, Obaro-Best O, Drasher M, Peshansky A, Balem KA, Bundy DG. Pediatric ambulatory catheter-associated urinary tract infections (CAUTIs): Incidence, risk factors, and patient outcomes. Infect Control Hosp Epidemiol. 2020 Aug;41(8):891-899.
- Tandogdu Z, Wagenlehner FM. Global epidemiology of urinary tract Infections. Curr Opin Infect Dis. 2016;29(1):73–9.
- Zilberberg MD, Nathanson BH, Sulham K, Shorr AF. Descriptive epidemiology and outcomes of hospitalizations with complicated urinary tract Infections in the United States, 2018. Open Forum Infect Dis. 2022;9(1):ofab591.
- Redder JD, Leth RA, Moller JK. Analysing risk factors for urinary tract Infection based on automated monitoring of hospital-acquired Infection. J Hosp Infect. 2016;92(4):397–400.
- Detweiler K, Mayers D, Fletcher SG. Bacteruria and urinary tract Infections in the Elderly. Urol Clin North Am. 2015;42(4):561–8.
- Shen L, Fu T, Huang L, Sun H, Wang Y, Sun L, Lu X, Zhang J, Yang Z, Ni C. 7295 elderly hospitalized patients with catheter-associated urinary tract infection: a case-control study. BMC Infect Dis. 2023 Nov 24;23(1):825.
- Mack WN, Mack JP, Ackerson AO. Microbial film development in a trickling filter. 1975, Volume 2, Issue 3, pp 215–226.
- Costerton, J.W.; Geesey, G.G.; Cheng, K.J. How bacteria stick. Sci. Am. 1978, 238, 86–95.
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322.
- Center for Biofilm Engineering. Biofilm Basics. Brief history of biofilm. Available at: https://biofilm.montana.edu/biofilm-basics/brief-history-biofilm.html
- Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002 Apr;15(2):167-93. doi: 10.1128/CMR.15.2.167-193.2002.
- Bouhrour N, Nibbering PH, Bendali F. Medical Device-Associated Biofilm Infections and Multidrug-Resistant Pathogens. Pathogens. 2024 May 8;13(5):393.
- EClinicalMedicine. Antimicrobial resistance: a top ten global public health threat. EClinicalMedicine. 2021 Nov 24;41:101221.
- Yuan L, Straub H, Shishaeva L, Ren Q. Microfluidics for Biofilm Studies. Annu Rev Anal Chem (Palo Alto Calif). 2023 Jun 14;16(1):139-159.
- Clarke OE, Bennett V, Jones BV. An In Vitro Bladder Model of Catheter-Associated Urinary Tract Infection. J Vis Exp. 2025 Jun 24;(220).
- Muraveva, V., Lomadze, N., Gordievskaya, Y.D. et al. Manipulation of artificial and living small objects by light driven diffusioosmotic flow. Sci Rep 14, 18342 (2024). https://doi.org/10.1038/s41598-024-69001-6
- Stewart E, Hochstedler-Kramer BR, Khemmani M, Clark NM, Parada JP, Farooq A, Doshi C, Wolfe AJ, Albarillo FS. Characterizing the urobiome in geriatric males with chronic indwelling urinary catheters: an exploratory longitudinal study. Microbiol Spectr. 2024 Nov 5;12(11):e0094124.
- Francolini I, Vuotto C, Piozzi A, Donelli G. Antifouling and antimicrobial biomaterials: an overview. APMIS. 2017 Apr;125(4):392-417.
- Li C, Gao D, Li C, Cheng G, Zhang L. Fighting against biofilm: The antifouling and antimicrobial material. Biointerphases. 2024 Jul 1;19(4):040802.
- Zhang Q, Zong Q, Feng X, Luo M, Sun W, Zhai Y. Antibacterial and antifouling materials for urinary catheter coatings. Acta Biomater. 2025 Jan 15;192:28-47.
- Andersen MJ, Flores-Mireles AL. Urinary Catheter Coating Modifications: The Race against Catheter-Associated Infections. Coatings. 2020; 10(1):23.
- Sharma S, Mohler J, Mahajan SD, Schwartz SA, Bruggemann L, Aalinkeel R. Microbial Biofilm: A Review on Formation, Infection, Antibiotic Resistance, Control Measures, and Innovative Treatment. Microorganisms. 2023 Jun 19;11(6):1614. doi: 10.3390/microorganisms11061614. Erratum in: Microorganisms. 2024 Sep 27;12(10):1961.
- Wang L, Zhang S, Keatch R, Corner G, Nabi G, Murdoch S, Davidson F, Zhao Q. In-vitro antibacterial and anti-encrustation performance of silver-polytetrafluoroethylene nanocomposite coated urinary catheters. J Hosp Infect. 2019 Sep;103(1):55-63.
- Arunachalam KD, Annamalai SK, Hari S. One-step green synthesis and characterization of leaf extract-mediated biocompatible silver and gold nanoparticles from Memecylon umbellatum. Int J Nanomedicine. 2013;8:1307-15.
- Malka E, Perelshtein I, Lipovsky A, Shalom Y, Naparstek L, Perkas N, Patick T, Lubart R, Nitzan Y, Banin E, Gedanken A. Eradication of multi-drug resistant bacteria by a novel Zn-doped CuO nanocomposite. Small. 2013 Dec 9;9(23):4069-76.
- Folawewo AD, Bala MD. Nanocomposite Zinc Oxide-Based Photocatalysts: Recent Developments in Their Use for the Treatment of Dye-Polluted Wastewater. Water. 2022; 14(23):3899. https://doi.org/10.3390/w14233899
- Yan P, Chen D, Muhammad S, Guo Y, Gao Q, Niu B, Liu Y, Liu C. Carboxymethyl chitosan-immobilized silver nanoparticles for intravesical instillation: A strategy to prevent catheter-associated urinary tract infections. Int J Biol Macromol. 2025 Jul 14;320(Pt 4):146001.
- Mosayyebi A, Manes C, Carugo D, Somani BK. Advances in Ureteral Stent Design and Materials. Curr Urol Rep. 2018 Apr 10;19(5):35.
- Diaz Blanco C, Ortner A, Dimitrov R, Navarro A, Mendoza E, Tzanov T. Building an antifouling zwitterionic coating on urinary catheters using an enzymatically triggered bottom-up approach. ACS Appl Mater Interfaces. 2014 Jul 23;6(14):11385-93.
- Lipovsky A, Thallinger B, Perelshtein I, Ludwig R, Sygmund C, Nyanhongo G, et al. Ultrasound coating of polydimethylsiloxanes with antimicrobial enzymes. Journal of Materials Chemistry B. 2015;3(35):7014-9.
- Snarr BD, Baker P, Bamford NC, Sato Y, Liu H, Lehoux M, Gravelat FN, Ostapska H, Baistrocchi SR, Cerone RP, Filler EE, Parsek MR, Filler SG, Howell PL, Sheppard DC. Microbial glycoside hydrolases as antibiofilm agents with cross-kingdom activity. Proc Natl Acad Sci U S A. 2017 Jul 3;114(27):7124-7129.
- Bell MM, Alaestante G, Finch C. A multidisciplinary approach to preventing catheter-associated urinary tract infections through education, care continuity, and system-wide support. 2016. Ochsner Journal 16:96–100.
- Lifshitz Guinzberg A, Pomposo García-Cohen ASF. Las ciencias de la complejidad y la educación médica. Inv Ed Med. 2017;6(24):267-271.
- Bell MM, Alaestante G, Finch C. A multidisciplinary approach to preventing catheter-associated urinary tract infections through education, care continuity, and system-wide support. 2016. Ochsner Journal 16:96–100.
- Atkins L, Sallis A, Chadborn T, Shaw K, Schneider A, Hopkins S, Bunten A, Michie S, Lorencatto F. Reducing catheter-associated urinary tract infections: a systematic review of barriers and facilitators and strategic behavioural analysis of interventions. Implement Sci. 2020 Jul 6;15(1):44. doi: 10.1186/s13012-020-01001-2. PMID: 32624002; PMCID: PMC7336619.
- Asociación Española de Urología. Posicionamiento basado en la evidencia sobre la Prevención de las Infecciones del Tracto Urinario (ITU) relacionadas con el uso del Sondaje Vesical (SV).2020. Available at: https://www.aeu.es/UserFiles/files/PosicionamientoAEUPrevencionITU-SV.pdf
- Tenutto M, Sabelli, MJ. Evaluación de desempeño: evaluación por estaciones. In Ferreyra, Horacio y Tenutto, Marta (comp.) Planificar, enseñar, aprender y evaluar en educación superior. 202. Novedades Educativas. ISBN 978-987-538-792-8.
- Rosenthal VD, Memish ZA, Nicastri E, Leone S, Bearman G. Preventing catheter-associated urinary tract infections: A position paper of the International Society for Infectious Diseases, 2024 update. Int J Infect Dis. 2025 Feb;151:107304. doi: 10.1016/j.ijid.2024.107304. Epub 2024 Nov 17. PMID: 39551089.




