Primate Perirhinal Cortex: Memory and Emotion Insights
Role of the Primate Perirhinal Cortex in Memory and Emotional Regulation: Ontogeny and Early Insults
Jocelyne Bachevalier 1 and Alison R. Weiss 2
- Emory National Primate Research Center, Atlanta, GA, USA
- Oregon National Primate Research Center, Beaverton, OR, USA
OPEN ACCESS
PUBLISHED: 31 January 2026
CITATION: Bachevalier, J., and Weiss, A.R., 2026. Role of the Primate Perirhinal Cortex in Memory and Emotional Regulation: Ontogeny and Early Insults. Medical Research Archives, [online] 14(1).
COPYRIGHT: © 2026 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v14i2.7272
ISSN 2375-1924
Abstract
The perirhinal cortex, a small strip of the anterior medial temporal cortex, first came into prominence through studies of memory. While examining patients with damage to the medial temporal lobe as well as animals with similar regional damage, findings showed that combined damage to the hippocampus, amygdala, and adjacent cortical areas, including the perirhinal cortex, were responsible for the profound memory loss observed. Later, however, the evidence demonstrated that the accompanying damage to the underlying medial temporal cortical areas were largely responsible for the memory deficit that had been attributed to the combined hippocampal and amygdala lesions. The perirhinal cortex has become appreciated as a critical structure supporting familiarity judgement, recognition memory, flexible executive control and behavioral regulation. The objective of this article is first to review the anatomy of the perirhinal cortex and its interactions with other medial temporal structures as well as the neocortex. A series of neurosurgical ablation studies in nonhuman primates will provide evidence for its role in memory and behavioral regulation in adulthood. The next section will highlight the functional maturation of the perirhinal from infancy through adulthood and will show that its role in support of recognition memory emerges in early infancy. The findings will also show that the neonatal perirhinal dysfunction results in functional compensation of recognition and working memory in adulthood but impacts higher-order executive processes such as cognitive control and flexibility. Interestingly, the discovery of the role of perirhinal cortex in familiarity judgements instead of recollection, which is mediated by the hippocampus, is now well documented in several clinical neurodevelopmental disorders (epilepsy, Alzheimer’s disease, schizophrenia), providing valuable markers in the prodromal phase of the diseases for early diagnosis and treatment.
Keywords
Medial temporal lobe, recognition memory, working memory, executive functions, epilepsy, Alzheimer’s disease, schizophrenia
Introduction
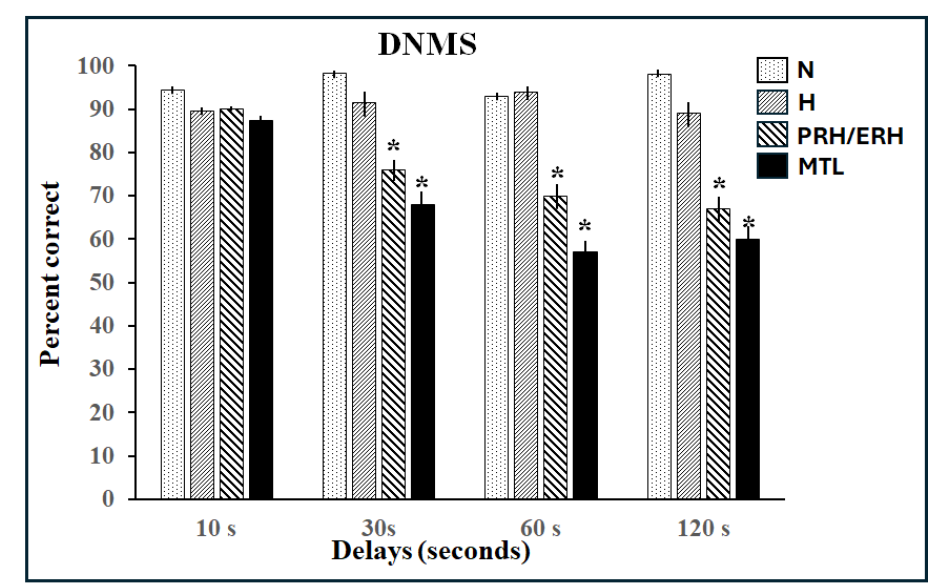
Declarative memory enables access to information previously encoded and stored in long-term memory and involves processes such as recognition and recall. This memory system is distinguished – functionally and anatomically – from procedural, implicit, habit memory systems, which expresses previously acquired skills through performance. Substantial progress was made in the last half century to further our understanding of the neural substrates involved in each of these memory systems. The discovery of the crucial role of the medial temporal lobe in human declarative memory, and the numerous subsequent monkey studies that this discovery prompted, has been the main driving force of research on recognition memory in primates. Hence, we chose to keep the present review focused on the medial temporal lobe. Human patients with large extensive damage to this region suffer global anterograde amnesia in that they are unable to recognize or recall events even after a few minutes. Yet, this profound memory deficit left patients’ skill learning and memory abilities intact, leading to the view that the medial temporal lobe supports declarative memory. The modular organization of memory processes was replicated in nonhuman primates following bilateral medial temporal damage originally reported in the amnesic patients, which included two deep structures – the amygdala (AMY) and hippocampus (HIP)- wrapped rostrally by two medial temporal cortical areas, the entorhinal (ERH) and perirhinal (PRH) shown below. Monkeys with medial temporal lobe lesions failed to recognize objects a minute or so after they were presented when tested with the delayed nonmatching-to-sample task (DNMS, Figures 1A and 2), compared to normal intact control groups.
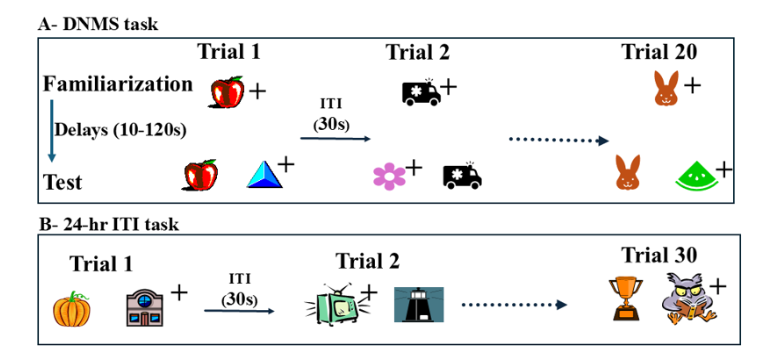
Despite the profound recognition deficit, they were still able to learn long lists of 30 pairs of objects (in which only one of the objects of the pair was rewarded) as rapidly as normal animals even though this list was presented only once every 24 hours (24-hr ITI task, see Figure 1B). Although damage to the medial temporal lobe region in monkeys replicated some of the memory loss seen in the amnesic patients, the results from DNMS task were soon called into question when more selective lesion techniques made it possible to parse out the individual contributions of specific medial temporal lobe structures to recognition memory. Although aspiration amygdala lesions in adult monkeys had no effects on recognition memory at any delays, restricted damage to the subjacent PRH/ERH cortex devastated animals’ performance even at the shortest delay. Hippocampal damage resulted at best in a mild impairment only at longest delays of 10 minutes and beyond. Taken together, these data indicated that the impaired recognition memory observed after the large medial temporal lobe lesions likely resulted from damage to the medial temporal cortical areas and not from damage to the amygdala or hippocampus. Given the anatomical organization of the region, it is currently being argued that the PRH may be important for visual perception of complex items as well as memory (see review for a more detailed discussion). Given the prominent impacts these new findings had on the neural bases of memory processes and on the neural underpinning of clinical neurodevelopmental disorders, this manuscript reviews the discoveries made in the last decade. Following a brief summary of the anatomical organization of the PRH cortex, we describe its functional organization in adult primates demonstrating the modulation of its interactions with the hippocampus to support recognition memory as well as its interactions with the AMY to reduce anxiety in presence of familiar positive stimuli. Next, the postnatal morphological maturation of the PRH cortex as well as the developmental trajectory of PRH recognition memory functions will illustrate that the PRH cortex functionally matures earlier after birth than the hippocampus in monkeys. The long-lasting effects of neonatal PRH damage on memory functions will then be briefly described. The review will end with an account of the implication of PRH dysfunction to clinical neurodevelopmental disorders, including epilepsy, schizophrenia, and Alzheimer’s Disease (AD).
Primate Perirhinal Cortex Anatomy and Functional Organization
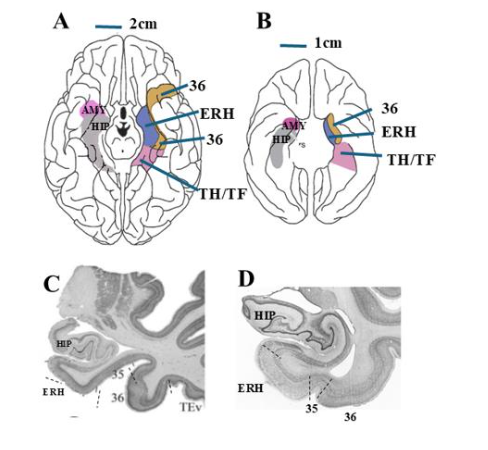
In both humans and monkeys, the PRH is a narrow band of cortex lying lateral to the collateral and rhinal sulcus, respectively, in the anterior portion of the medial temporal lobe (Figure 3A-D). The PRH, defined by Brodman areas (BA) 35 and 36, receives prominent and convergent inputs from sensory areas (visual, somatosensory, and auditory), polymodal areas (cingulate cortex, superior temporal sulcus, and parahippocampal gyrus), reward-related cortical areas (orbitofrontal cortex) as well as interconnections with other MTL structures (hippocampus and amygdala), but its densest afferent inputs originate in ventral visual cortical region (temporal areas TE and TEO). In turn, the PRH projects to the anterior and lateral portions of the ERH, with lighter projections to the distal section of CA1 and the proximal section of the subiculum of the hippocampus. It also projects to the lateral amygdala nuclei and has bi-directional monosynaptic projections with the lateral prefrontal fields (BA 45, 47/12, 46, 9). Multi-synaptic pathways through the thalamus connect the PRH to the medial prefrontal areas (Brodmann’s areas 24, 25, 32, and 14). In sum, its convergent inputs from sensory, polymodal, and reward-related cortical areas together with its prominent interconnections with other medial temporal lobe and prefrontal cortex structures place it in a unique position to coordinate neural representations of non-spatial visual information generated in the ventral visual stream with brain structures known to be important for memory (hippocampus), executive functions (prefrontal cortex) and emotional regulation (amygdala). Given these widespread connections with other brain structures, the PRH cortex has been implicated in diverse functions that are summarized next.
Perirhinal Cortex and Visual Perception
Neurocognitive processes underlying visual perception begin in peripheral sensory organs (eyes), where visual stimuli are transduced into neural signals. Basic information about color, movement and form are coded and assembled into progressively complex representations, first in primary visual cortex (areas V1-V4) and then in the higher-order visual cortical areas of the dorsal and ventral visual streams. Seminal work demonstrated that dorsal visual areas in inferior parietal cortex allowed integration of visuospatial features to represent locations, whereas the ventral visual areas in inferior temporal cortex allowed integration of perceptual features to represent objects. It is in the PRH, the last station of the object visual pathway, that a complete representation of a perceived object is realized. In this way, the PRH may generate complex representations of objects from a compilation of perceptual features represented at lower levels of the visual hierarchy.
Most of the knowledge on the PRH role in perception comes from a body of work that has used neuroimaging and electrophysiological recording techniques to monitor PRH activity during perceptual tasks or tested visual discrimination abilities after PRH damage. Neuroimaging data in healthy human adults indicate that the PRH is highly active during perceptual tasks requiring the integration of visual features into configurally-based representations. This pattern of activation corresponds to the activity in ventral temporal visual areas that have been historically linked to visual processes. Additionally, when subjects performed difficult figure-ground discrimination problems, the activity recorded in area V2 mimicked the activity usually recorded in PRH. Taken together, these findings suggest that the PRH may interact with other cortical areas important for feature-based visual processing to construct visual representations from configurations of familiar features. Thus, it was not surprising to observe that PRH lesions resulted in visual discrimination impairment when stimulus complexity was high or perceptual overlap between stimuli was extensive (i.e. feature ambiguity), although they spared the ability to discriminate two highly dissimilar stimuli. The specificity of the visual deficits following PRH lesions gave credibility to the neuroimaging data suggesting the importance of this cortical area in mechanisms of complex visual perception.
Perirhinal Cortex and Familiarity Judgments
The PRh may not only be critical for object perception, as its strong anatomical link with the ERH and hippocampus has led scientists to believe that it may also be critical for memory processes such as recognition. Recognition of a previously encountered item can be accomplished by two processes: either recollecting the specific episode in which it was previously encountered, or by assessing the degree of its familiarity. In this way, mechanisms of recollection and familiarity are both capable of supporting recognition memory. Lesion studies in rodents and monkeys, as well as work with human neuropsychiatric populations have all demonstrated the importance of the PRH for recognition memory. Yet, newer studies have begun to provide evidence for the presence of a functional dissociation in the contribution of the PRH and hippocampus to recognition memory, with the PRH being critical for familiarity judgments and the hippocampus associated with recollection. For instance, patients with extensive medial temporal lobe damage encompassing both hippocampus and PRH show deficits in recollection and familiarity judgments. However, selective damage to the hippocampus, sparing the PRH, impairs recollection but spares familiarity. Furthermore, electrophysiological recordings have demonstrated that neuronal firing in the PRH precedes cell firing in the hippocampus, suggesting that there is a rapid familiarity signal mediated by the PRH, which is then followed by a later-onset recollection signal mediated by the hippocampus. Additional neuroimaging studies in healthy adults have shown that hippocampal activity increases in response to retrieval of information but not in response to judgments of familiarity. In sum, the PRH appears to be important to support familiarity processes involved in recognition memory.
Perirhinal Cortex and Working Memory
The PRH is also involved in the integration of multiple sensory features, across multiple domains, into an abstract view-invariant representation of a stimuli and is critical for familiarity recognition of these stimuli; in this fashion, the strong projection of the PRH with the lateral prefrontal cortex could enable memory processes that have been linked to this area, such as working memory. Working memory involves the maintenance of a limited set of cognitive representations of objects, places, ideas, goals, or rules. Furthermore, these cognitive representations are kept active in a manner flexible enough to cooperate with simultaneous/parallel working memory process that monitor or manipulate the representations being kept ‘in mind. In the last 50 years, overwhelming evidence has accumulated from human functional imaging and electrophysiological and lesion studies in monkeys to indicate the importance of the ventrolateral prefrontal cortex for maintenance processes and the dorsolateral prefrontal cortex for higher-order monitoring/manipulation processes. However, more recent studies suggest that the prefrontal cortex is part of a broader network of interconnected brain areas involved in working memory. Specifically, medial temporal lobe structures are also recruited during working memory tasks, such as the PRH cortex that is well positioned to play a role in working memory, mainly because of its direct reciprocal connections with lateral and orbital prefrontal cortex fields.
Electrophysiological and functional imaging studies indicate increased activity in PRH during object-based working memory tasks, suggesting that it supports object representations used by the prefrontal cortex during working memory. Specifically, cells in the macaque PRH are highly activated during working tasks requiring the temporary maintenance of object representations, but this activity is not observed in other temporal visual areas such as area TE. Likewise, 2-Deoxyglucose imaging studies demonstrate increased PRH activity during a delayed object alternation task requiring the maintenance and monitoring of information, whereas the same increase was not seen in the ERH. Taken together, these results point to a unique contribution of the PRH to performance on tasks that require the active/flexible representation of familiar items, that is strengthened by the concurrent lack-of-contribution of both its primary afferent inputs (area TE/TEO) as well as its primary efferent projections to ERH.
Perirhinal Cortex and Proactive Interference
Proactive interference occurs when previously acquired information impedes the ability to learn or apply new information and may result in behavior that is dominated by rules no longer appropriate to the current situation. To resolve proactive interference, the influence of formerly active, and now competing, response sets must be suppressed. This requires the inhibition of behavioral responses based on “old” information, and a flexible shift of cognitive resources towards learning/remembering “new” information. These cognitive mechanisms are critical for performance on tasks requiring participants to flexibly update cognitive representations or shift response strategies, such as the Wisconsin card sort task, the self-ordered pointing task, and attentional set-shifting task. Lesion studies in monkeys have already demonstrated that behavioral inhibition is supported by the orbital frontal cortex, whereas cognitive flexibility is supported by the ventrolateral and medial prefrontal cortex. Given that the PRH has robust interconnections with these prefrontal areas, its contribution to mechanisms underlying cognitive flexibility and/or behavioral inhibition is expected. There is already evidence that combined damage to PRH and ERH in adulthood impairs performance on reversal-learning tasks, suggesting that the PRH plays a role in mechanisms of behavioral inhibition. In contrast, preliminary results on the effects of extended medial temporal lobe damage in adulthood indicates that the medial temporal lobe structures are not important to support performance on attentional set shifting tasks. However, few studies to date have directly addressed the role of the PRH in mechanisms of cognitive flexibility in adulthood, or during development and additional information on this topic will be valuable.
Perirhinal Cortex and Emotional Regulation
As reviewed above, recent efforts to define the functions of the primate ERH and PRH areas have focused on their interactions with the hippocampus in the mediation of normal memory. Its implication in affective functions through, for example, its dense connections with the amygdala, has received little attention despite supporting experimental and theoretical evidence from the rodent literature. In an electrophysiological study simultaneously recording neurons in PRH, ERH, and the basolateral amygdala nucleus (BLA) during a trace-conditioning task, Paz, Pelletier, Bauer and Pare reported that early in the learning process, BLA activity was associated with increased signal transmission from PRH to ERH, and this activity was increased markedly after reward delivery. In this way, the PRH appears to serve as an active gateway of information flow from neocortical areas toward the hippocampus, which can be modulated by the emotional salience of the present situation provided by the amygdala. To provide additional evidence for this proposal, we first explored the role of the primate PRH, by comparing the behavioral responses of monkeys with combined lesions of the PRH and ERH with those of monkeys with neurotoxic or aspiration amygdala lesions. Four stimuli probed affective functions (construed in a broad sense to encompass social behavior, emotion, and motivation). Two of these stimuli had a social component (an unfamiliar human and a conspecific stimulus) and two were nonsocial items, one affectively positive (a generally rewarded object) and one affectively negative (a toy snake). A detailed ethogram was built to quantify behaviors during three weekly presentations of each stimulus. In this paradigm, combined ERH/PRH ablations yielded none of the symptoms recorded after lesions involving the amygdala; i.e. hyperorality (a tendency to explore any items by mouth), and hypermetamorphosis (compulsive eating, hypersexuality, excessive affiliation, and diminished fear). Rather, they led to more subtle behavioral changes that were opposite in direction, namely, the ERH/PRH lesions reduced affiliative responses and heightened defensiveness, indicating that ERH and PRH damage can interfere with responses to affectively salient stimuli in a way radically opposite from those following amygdala damage. A follow up study investigating the effects of separate lesions of the ERH and PRH in the same paradigm showed similar emotional changes to those of the combined lesions (i.e., attenuated affiliation and enhanced defense). Although failure to modulate responses based on previous experience (i.e., memory difficulties) may explain these affective changes, it does not account, however, for the sparing of some memory-dependent modulations of defense, nor for the lack of correlation between the animals’ affective changes and their own recognition memory performance. Alternatively, we had proposed that damage to ERH and PRH introduces a negative bias in the risk assessment of affectively salient stimuli; a proposal more compatible with Gray and McNaughton’s anxiety-centered view of medial temporal functions, than with prominent mnemonic/perceptual functional models of the hippocampus-PRH duo. To sum, the PRH could modulate the interactions between the hippocampus and the amygdala to reduce anxiety in presence of familiar positive stimuli.
Early Perirhinal Cortex Functions and Dysfunction
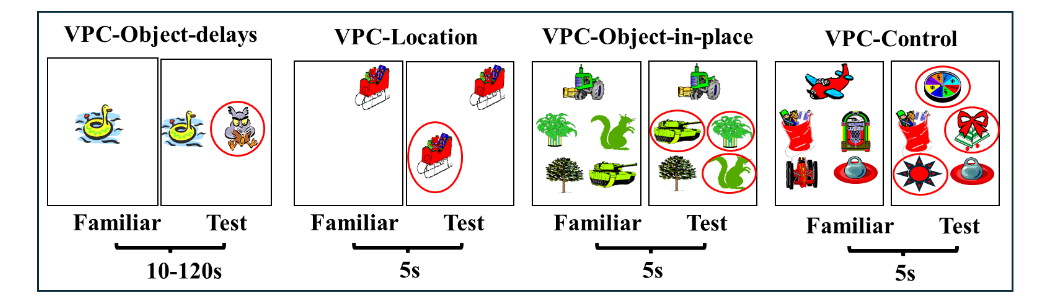
As reviewed above for adult monkeys, in addition to its role in building representations of objects, the role of PRH in object recognition memory has received growing support from studies in several species including rodents, monkeys and humans. Yet, its role in the maturation of early memory functions had not been explored in human infants or infant monkeys. However, this was made possible with the exploitation of another paradigm, the visual paired comparison task (VPC) that enabled evaluation of incidental recognition memory skills in nonverbal infant primates of both species. Thus, we began a series of prospective developmental studies to assess the role of the hippocampus and PRH in the maturation of recognition memory using the VPC-Object-delays task. In this task, monkeys are first familiarized to view an object at the center of a monitor and after a delay their recognition memory was indexed by longer looking time to novel stimuli as compared to familiar ones. Earlier studies had already shown that incidental recognition-memory ability is present as early as 3–4 days of age with either no delays or 2 min delays and become stronger in 3-month-old human infants. Similarly, by 4 weeks of age, infant macaques show novelty preference that becomes stronger by 13 weeks of age.
Neonatal Perirhinal Lesions and Object and Spatial Memory Development
To gain additional knowledge on the developmental trajectory of recognition memory abilities from birth to adolescence and its neural substrate, we longitudinally tested sham-operated controls (Neo-C) and those with neonatal lesions to either the PRH cortex (Neo-PRH) or the hippocampus (Neo-HIP, for comparison) from 1 to 48 months, using the 4 VPC tasks shown in Figure 4. All lesions were performed between 10-12 days of age. The pattern of performance in the age-matched controls as well as the memory impairment following the two types of lesions was surprising, but at the same time very revealing.
Visual recognition memory: The development of object recognition abilities was assessed in infant monkeys from 1.5 to 18 months with the VPC-Object-delays task. Normally developing monkeys showed robust recognition memory across short and long delays, with a delay-dependent forgetting emerging only at 18 months of age that was similar to that shown in adult monkeys. Interestingly, infants with Neo-HIP lesions performed as well as controls at the 2 youngest ages, but, at 18 months, showed a significant forgetting that became evident and reliable only at the longest delay of 120s, suggesting that, with maturation, animals with neonatal hippocampal lesions grew into a recognition memory loss that remained present even when reaching adulthood.
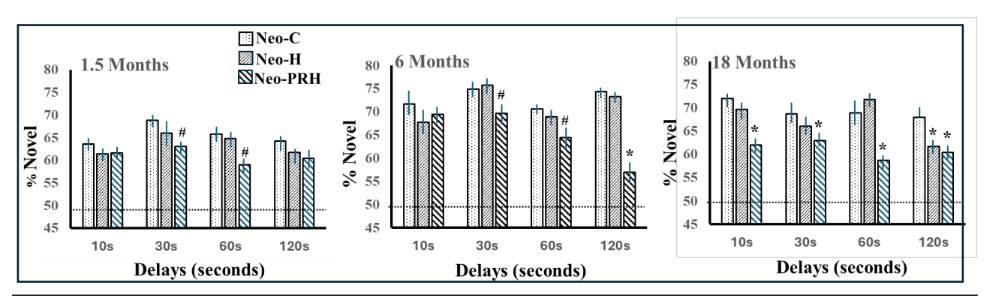
Both the emergence of delay-dependent recognition memory performance at 18 months of age in the control animals together with the recognition memory loss observed after neonatal hippocampal lesions at that same age suggest that important maturational changes in the neural substrate supporting incidental recognition memory occurred after six months of age in monkeys. This interpretation was supported by anatomical findings indicating that the primate hippocampus is not fully developed at birth and go through significant anatomical remodeling until at least 1-2 years of age in monkeys. By contrast, these object recognition memory abilities were severely compromised in infant monkeys with neonatal PRH damage early after birth. This memory loss emerged after 1.5 months of age but became more pronounced in juvenile (6 months) and adolescent monkeys (18 months). Interestingly, the magnitude of the recognition memory deficit after neonatal PRH lesions was milder than following adult-onset PRH lesions, suggesting functional compensation may have occurred following the early perturbation. It is possible that other brain structures compensate for the loss of PRH-supported familiarity processes in the event of an early PRH malfunction. There is evidence of increased recovery of sensorimotor and visual function following early injury, but comparably fewer studies in cognitive systems. Taken together, this work has enhanced our understanding of the development of recognition memory and of early brain plasticity.
Spatial memory
Contrary to its significant contribution to object recognition memory, the PRh plays a limited role in memory for spatial locations, especially when delays are kept short. Thus, we assessed the effects of neonatal PRH lesions on spatial location memory using the same controls and animals with neonatal PRH lesions as they reached adulthood using a Spatial Memory Span task (SMS), measuring recognition of spatial locations, and the Delayed Nonmatching-to-Sample (DNMS), measuring object recognition for comparison. The neonatal PRH lesions had no impact on spatial memory, though they did impair performance on the DNMS task when the delays were extended from 30 s to 600 s. The dissociation between the sparing of spatial memory together with the loss of object recognition memory after neonatal PRH lesions is similar to that described when the PRH damage occurs in adulthood, suggesting that the PRH is a cortical structure critical for the normal development of processes supporting object memory but not spatial memory.

Neonatal Perirhinal Lesions and Perceptual Ability versus Familiarity Judgment
Previous studies in adult monkeys with PRH lesions report a mild perceptual impairment when test stimuli were black and white (B&W), or had overlapping/similar features, suggesting that this cortical area may also contribute to higher-order visual processes. To test the impact of the neonatal-PRH lesions on perceptual abilities, and to confirm a specific impairment in familiarity judgement, we conducted a set of studies in the same monkeys with neonatal PRH lesions discussed above. First, we re-tested the animals on a version of the VPC-object-delays task using highly similar black and white (B&W) stimuli. Both Control animals and those with neonatal PRH lesions performed well on the VPC task with similar B&W objects at short (10s) delays, but neonatal PRH animals had significantly lower novelty preferences than controls when the delays were extended beyond 30s. Given the normal levels of novelty preference after the shorter delay, coupled with delay-dependent reduced novelty preference, these data suggest that the neonatal PRH animals had perceptual abilities within the normal range, indicating normal visual perception but displayed recognition memory impairment.
In parallel, the same animals were also tested on a task measuring the progressive accumulation of familiarity signals, the Constant Negative task. This experiment was inspired by the observation that, compared with adult-onset PRH lesions, monkeys with neonatal PRH lesions exhibited a partial sparing of recognition memory when measured using VPC, but not when measured using DNMS. A possible explanation for this difference was that the DNMS task used a shorter familiarization time (usually 3-7s) than the VPC task (30s), and therefore that the recognition memory sparing observed with the VPC task could be due to a longer opportunity to become familiar with the sample stimuli. To confirm this, we administered an object discrimination task that measured the rate at which neonatal PRH-operated animals became familiar with objects. The results revealed clear group differences in learning curves, with control animals having significantly steeper slopes than the neonatal PRH-operated animals, suggesting a slower ability to familiarize. Importantly, an alternative interpretation is that, rather than completing the Constant Negative task using novelty-guided strategies, the neonatal PRH-operated monkeys learned to avoid incorrect, unrewarded objects using “habit” based strategies. Habit learning was tested in the same animals using a Concurrent Discrimination task, and comparisons of errors on both tasks indicated that control animals made fewer numbers of errors on the Constant Negative task than the Concurrent Discrimination task, whereas neonatal PRH-operated animals made similar numbers of errors on both. These data suggest that the neonatal PRH-operated animals may have developed a behavioral strategy that relied more heavily on habit systems to compensate for their impaired recognition memory.
Neonatal Perirhinal Lesions and Working Memory Tasks with High-proactive Interference
In adulthood, the same control animals and those with neonatal PRH lesions were evaluated on three object-based working memory tasks that targeted distinct cognitive demands. One task emphasized simple maintenance (Session-Unique Delayed Nonmatching-to-Sample; SU-DNMS), which is like the DNMS shown in Figure 1A, but only two stimuli are used on each trial and serve either as the familiar or the novel stimulus. The other two WM tasks required maintenance plus monitoring of temporal item order: the Object Self-Order (S-OBJ) and the Serial Order Memory Task (SOMT).
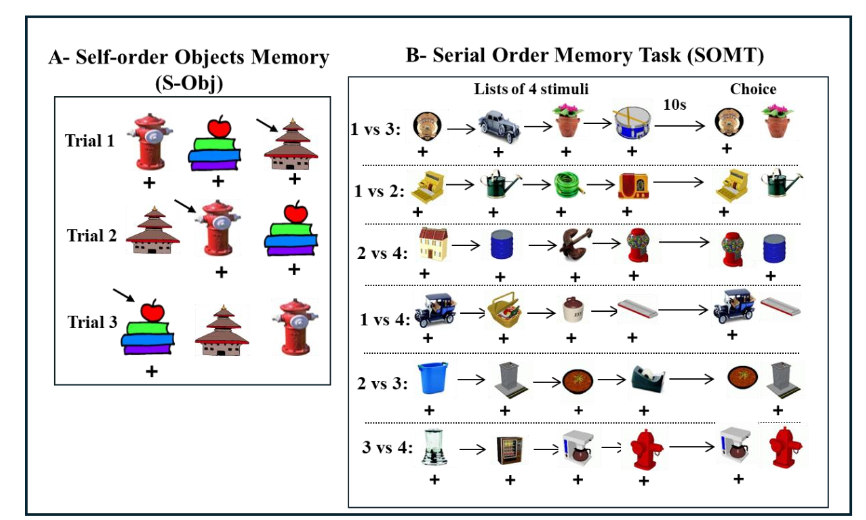
Monkeys with neonatal PRH damage showed a deficit on SU-DNMS when they were initially tested with a short delay of 5 seconds, but they performed normally when re-tested and the delay extended to 30 seconds, indicating that this impairment was transient and that they were able to perform this task at a level comparable to controls with extended opportunities for practice. Similarly, performance on the SOMT was unaffected, indicating preserved ability to track the temporal sequence of items in working memory following these early lesions. In contrast, the neonatal PRH-operated animals made significantly more errors on the S-OBJ task, most notably reflected in a marked rise in perseverative responding. Together, the evidence from the SU-DNMS, S-OBJ, and SOMT tasks indicated that early damage to the PRH impacted the development of mechanisms that help resolve proactive interference and/or control perseverative responding, but spared mechanisms supporting object representations in working memory. Due to increased brain plasticity during development, it is possible that other brain areas, such as the hippocampus or temporal visual cortical areas could be recruited to support object representations in working memory in the absence of a functional PRH.
Neonatal Perirhinal Lesions and Cognitive Flexibility
As described above, the performance patterns of neonatal PRH-operated animals on object-based working memory tasks suggested that the early PRH damage disrupted the development of mechanisms that resolve proactive interference. To further test whether this vulnerability reflected broader impairments in inhibitory control or difficulty shifting behavioral strategies, we evaluated the same neonatal PRH-operated monkeys and Control animals on an Intradimensional–Extradimensional (ID–ED) set-shifting task in adulthood. This paradigm has proved valuable for distinguishing between two components of executive function: (1) reversal learning, which depends heavily on inhibiting previously reinforced responses, and (2) cognitive flexibility, which requires shifting attention away from a previously relevant stimulus dimension toward a newly relevant one. In the early phases of the task (simple discriminations, compound discriminations, and reversals), the neonatal PRH-operated monkeys performed comparably to controls, indicating intact basic stimulus–reward learning and relatively preserved behavioral inhibitory control. However, when required to shift attention across stimulus dimensions, their performance declined sharply. Unlike control animals that rapidly adapted to the new rule, neonatal PRH-operated monkeys showed persistent perseverative responding, continuing to rely on the previously learned dimension despite repeated negative feedback. This selective impairment at the extradimensional shift stage pointed to a deficit in cognitive flexibility rather than a generalized learning or memory problem.
Together with the working memory findings, these results indicate that early PRH removal alters the maturation of neural systems that support flexible, interference-resistant behavior. The sparing of reversal learning suggests that the basic ability to suppress previously rewarded responses remains largely intact, whereas the capacity to reconfigure behavior in response to changing task demands is compromised. Importantly, the deficit in cognitive flexibility following neonatal PRH damage contrasts with the minimal cognitive flexibility effects reported after broad medial temporal lobe lesions in adulthood, including damage to the PRH. Given the dense reciprocal connections between the PRH and ventrolateral prefrontal cortex, an area known to undergo protracted postnatal maturation, one interpretation of these results is that the neonatal PRH lesions disrupted the developmental scaffolding that normally supports the emergence of flexible executive control. These observations are consistent with the broader pattern in this cohort, in which recognition and working memory show partial functional compensation, but higher-order control processes remain particularly vulnerable to early PRH dysfunction. Taken together, these findings indicate that the absence of normal PRH projections to ventrolateral and medial PFC during early development may interfere with the establishment of prefrontal networks necessary for adaptive behavioral control, leading to long-lasting impairments in cognitive flexibility even when working processes remain relatively preserved.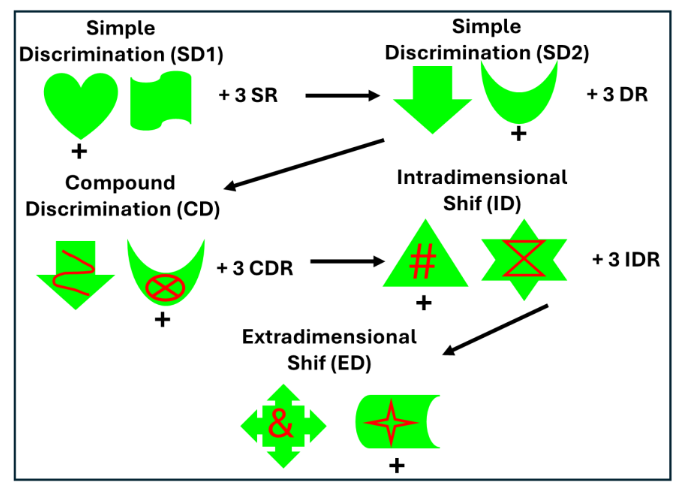
Figure 8: IntraDimensional/ExtraDimensional (ID/ED) task measures behavioral inhibition and cognitive flexibility. The animal first learns two simple discrimination problems (SD1, SD2) followed by 3 reversals of the second problem (SR). The animal learns a compound discrimination problem (SC) with red lines occurring on top of the green shapes of the last discrimination but continues to respond to the shape, followed by 3 reversals (CDR). In the intradimensional shift (ID), new shapes and lines form a new discrimination problem during which the animal continues responding to the shape, followed again by 3 reversals (IDR). Finally, in the extradimensional shift (ED), a new discrimination problem is presented with new shapes and lines, but now the animal needs to respond to the lines. Described in details in Weiss et al. (2019).Given the dense reciprocal connections between the PRH and ventrolateral prefrontal cortex¹⁹,²²,²⁴,¹³²,¹³³, an area known to undergo protracted postnatal maturation, one interpretation of these results is that the neonatal PRH lesions disrupted the developmental scaffolding that normally supports the emergence of flexible executive control. These observations are consistent with the broader pattern in this cohort, in which recognition and working memory show partial functional compensation, but higher-order control processes remain particularly vulnerable to early PRH dysfunction. Taken together, these findings indicate that the absence of normal PRH projections to ventrolateral and medial PFC during early development may interfere with the establishment of prefrontal networks necessary for adaptive behavioral control, leading to long-lasting impairments in cognitive flexibility even when working processes remain relatively preserved.
Perirhinal Cortex and Human Clinical Disorders
Finally, the PRH has now been shown to be a prominent neural marker of the prodromal phase in at least three human clinical neurodevelopmental disorders, epilepsy, Alzheimer’s disease, and schizophrenia, briefly reviewed below.
Temporal Lobe Epilepsy
Recurrent unprovoked seizures are the hallmark of epilepsy, and medial temporal lobe epilepsy (TLE) is the most common type of medically intractable focal epilepsy in adolescents and adults that necessitates surgical evaluation. Ninety years of studies on TLE patients with either medial temporal lobe lesions or electrical stimulations have strengthened the theoretical view of the respective role of PRH, ERH and hippocampus in memory processes reviewed above. Neuroimaging studies have documented volume loss of the anterior temporal lobe, temporal neocortical gray and white matter, and more refined analyses have also revealed volumetric changes of the hippocampus, PRH, and ERH. Interestingly, a small subset of individuals with TLE consistently experience “déjà-vu” phenomenon during the aura of their seizures. This experiential phenomenon was recognized very early by the pioneering work of Jackson and subsequently followed by Penfield and Gloor. Déjà-vu phenomenon is defined as an alteration of consciousness characterized by memory-like hallucinations, and/or a feeling of familiarity, with an epileptiform activity localized to the anterior parahippocampal region. The link between déjà-vu and medial temporal region in TLE is of particular interest in the context of the role of that brain region in recognition memory.
As reviewed above, the flow of information about items from PRH to ERH, through the hippocampal fields, and back to ERH and PRH provides a reverberatory circuit that, in normal conditions, plays a crucial role in the processing of declarative memory. Under pathological conditions, however, this neural loop tends toward excessive propagation and loop-gain amplification, which is a hallmark of TLE. Concurrently, many current models of recognition memory offer a critical distinction between familiarity assessment and recollection. Familiarity assessment involves evaluating memory strength of an item independent from recovery of contextual detail about a specific past encounter and has been proposed to depend specifically on computations performed in PRH, and not in the hippocampus. Recollection, by contrast, involves direct recovery of contextual details about a specific prior encounter with an item and has been linked specifically to hippocampal functioning. When considered within this model, part of the typical déjà-vu experience in the aura of TLE has been characterized as a static sense of familiarity. Accordingly, the phenomenon may reflect, at its core, an erroneous familiarity signal that is generated by abnormal activity in PRH, or perhaps combined PRH-ERH. Preserved recollection and metacognitive inferences may produce the subjective sense of inappropriateness for this familiarity signal. Considered together, the evidence reviewed hints that TLE patients who experience déjà-vu in the aura of their seizures may present with lasting interictal behavioral impairments that are specific to familiarity assessment.
Yet, there is still very little direct evidence for this proposal, but provocative results exist. Bowles and colleagues reported a single TLE case study presenting with a surgical removal of the MTL including the PRH but sparing the hippocampus showing impaired familiarity with preserved recollection. Furthermore, Martin and colleagues demonstrated selective familiarity deficits sparing recollection in patients with unilateral TLE experiencing déjà vu that contrasted with the broader pattern of recognition-memory impairments present in a control group of unilateral TLE patients without déjà-vu. Interestingly, medial temporal lobe structures were less broadly affected in TLE patients with déjà-vu than in TLE patients without déjà-vu, with a trend for more focal volume reductions in the PRH+ERH cortices of those patients with déjà-vu. Additional evidence for the distinctive contribution of the PRH vs hippocampus in the familiarity feeling experienced during déjà-vu episodes was provided by numerous brain stimulation studies of TLE patients. The results indicate that electrical stimulation of the amygdala and hippocampus induced reminiscences of episodic memories, whereas stimulations of the PRH-ERH lead mostly to familiarity and semantic memories, in other words to memories devoid of contextual information. Fernández and Tendolkar hypothesized that the PRH-ERH could be a gatekeeper to the declarative memory system and acts as an adaptable interface between the neocortex and the hippocampus. For example, an early PRH familiarity signal could trigger a source retrieval process for recollection in the hippocampus, which could in turn recruit PRH representations during episodic memory processes.
Alzheimer’s Disease
Alzheimer’s disease (AD) is one of the most prominent degenerative diseases and the major cause of elderly disability. It typically progresses from a long preclinical asymptomatic phase to mild cognitive impairment (MCI) that becomes incurable cognitive impairment in the transition to AD stage. Thus, identifying the earliest signs of AD has sparked a critical interest in the field as this will allow the initiation of treatments as early in the disease progression as possible. Neuropathological brain changes associated with AD, namely β-amyloid plaques and neurofibrillary tangles (NFT), are thought to begin years before clinical symptoms become evident. In contrast to β-amyloid plaques, NFT are more strongly correlated with cognitive deficits and progress in a hierarchical manner throughout the brain in typical AD. This continuous accumulation of NFT is strongly related to loss of neurons and was shown to be causally associated to cerebral atrophy in affected regions.
The most obvious clinical symptom of AD is the progressive loss of declarative memory. Therefore, a large emphasis has been placed to identify the integrity of hippocampus and related medial temporal cortical areas in AD, due to their critical role in declarative memory. With the advent of MRI tools and the growing use of imaging in clinical settings, the earliest AD-related changes on structural MRI are usually seen in the medial temporal lobe structures and notably the hippocampus. Hence, the hippocampal volume is reduced by 10-15% in MCI patients and by 40% in clinical AD patients compared to elderly controls. Rate of hippocampal atrophy has been shown not only via automated and manual segmentation on structural MRI, but with additional neuroimaging tools, such as 3-D dimensional surface shape measures and metabolic neuroimaging, and is associated with changes in cognitive performance. In both, MCI and AD, surface inward-deformation of the hippocampus is more prominent in its anterior portion as well as its lateral border (likely associated with CA1 atrophy).
Thus, hippocampal alteration was viewed as major topographical marker linking molecular pathology of AD to the clinical and cognitive decline. Nevertheless, in addition to hippocampal atrophy, Pennanen and colleagues showed that ERH volume loss was the dominant structural feature at the stage of MCI, followed by profound macroscopic hippocampal atrophy in patient with clinical AD. Positron Emission Tomography (PET) studies have also revealed that hypometabolism in the posteromedial association cortex is among the earliest brain areas containing Aβ plaques and may reflect a disconnection of the medial temporal cortical areas from the hippocampus. Thus, the ERH and PRH are some of the earliest cortical changes, prior to the hippocampal atrophy and preceding the cognitive impairment. Utilizing the U-Net framework, Henzen and colleagues recently confirmed that the medial PRH is one of the earliest regions affected by neurofibrillary tau pathology, which approximately corresponds to Brodmann area 35. This neurofibrillary tau pathology spreads to the medially located ERH and eventually to the hippocampus, and throughout the brain. Both automated MRI segmentation, optical imaging techniques to illuminate large-scale structures in postmortem tissue at a micron-scale resolution, as well as transcriptional regulation of the gene for synaptic protein, are becoming promising tools to establish early markers of PRH pathology. The importance of future longitudinal studies using these methodologies together with neuropsychological testing using tasks specifically assessing recognition memory processes (familiarity versus recollection), as well as working memory, and cognitive flexibility will enhance diagnostic precision and pave the way for early, targeted intervention strategies. Furthermore, as for epilepsy reviewed above, the clinical data may strengthen the proposal for the distinctive role of PRH and hippocampus in recognition memory processes.
Schizophrenia
Schizophrenia is a chronic neurodegenerative psychiatric disorder with an early onset in young adulthood reflecting the interplay of genetics and the environment. The disease characteristically evolves from a premorbid phase in which the clinical phenotype is not, or only partially, expressed through a series of stages culminating in the syndromal manifestation (hallucination, delusion, speech and behavior disorder, apathy, social withdrawal, and cognitive impairment) meeting diagnostic criteria and constituting a first episode of a psychotic disorder. The subsequent course varies markedly based on illness severity, adequacy of treatment and environmental, including social, factors. As with other progressive disorders of the brain, such as Alzheimer’s disease, early detection and treatment during prodromal stages, when the disease is restricted to relatively confined areas of the brain, has emerged as an important therapeutic strategy in alleviating symptoms and disease modification. Previous research indicates that people with schizophrenia exhibit impairments in declarative memory and their severity correlates with the magnitude of structural abnormalities within the medial temporal lobe structures, including the hippocampus together with its neighboring cortical areas, ERH and PRH cortex. Consistent with this idea, structural changes in the hippocampus (reduced volume, presence of cellular and molecular abnormalities, and abnormal activity; see for a review) and abnormal volumes of the parahippocampal gyrus (ERH and PRH) have been found in schizophrenia patients. Furthermore, a resting state functional connectivity study showed alterations characterized by increases and decreases in the strength of the positive connectivity between the PRH and ERH and the hippocampal subregions when comparing patients with schizophrenia with healthy subjects. Concurrently, schizophrenia patients experience difficulties in recognition tasks with both familiarity and recollection deficits reported in some studies, but only recollection deficits in others, suggesting multi-focal medial temporal lobe dysfunction. Thus, both the medial temporal lobe neuropathological findings as well as the memory deficits in schizophrenia offer significant evidence to implicate a role of the hippocampus and adjacent cortical areas in schizophrenia. However, additional research is necessary. The integration of anatomical and functional neuroimaging tools coupled with direct assessment of recognition memory processes to further characterize specific recollection versus familiarity deficits in the schizophrenia prodrome phase will likely provide critical neuroanatomical markers for better targeted treatment of memory impairments.
Conclusions
Significant advances have been made in the last decade on the specific role played by the medial temporal lobe structures in memory processes. Although extensive literature exists on the prominent function of the hippocampus in recollection, more recent emphasis was placed onto one of the medial temporal cortical areas surrounding the hippocampus, namely the PRH. In this review we showed that the PRH plays a critical role in recognition memory, particularly familiarity judgements rather than recollection, and in object memory rather than spatial memory. In addition, the PRH modulates anxiety as well as impulse control and behavioral flexibility. A series of developmental studies in nonhuman primates also indicated that anatomically and functionally the perirhinal cortex matures early in infancy, unlike the more protracted maturation of the hippocampus in adolescence, and PRH damage in infancy impacts object recognition memory at an early age. Further findings indicated that as animals with neonatal perirhinal damage reached adulthood, they displayed some functional compensation of object recognition and working memory but were severely impaired in higher-order executive processes such as cognitive control and flexibility. Interestingly, the discovery of the role of PRH in familiarity judgements instead of recollection is now well documented in several clinical neurodevelopmental disorders associated with memory disorders and cognitive impairments and is providing critical markers for early diagnoses and treatment.
Ethical Approval
All protocols for the nonhuman primate studies reviewed in this paper were approved by Emory University Institutional Animal Care and Use Committee (IACUC) in accordance with the Animal Welfare Act and the U.S. Department of Health and Human Services “Guide for Care and Use of Laboratory Animals”.
Conflicts of Interest Statement
The authors disclose no conflicts of interest.
Funding Statement
Preparation of this review was funded in part by NIH grants MH058846, HD090925, and AG070704, the NIH’s Office of the Director, Office of Research Infrastructure Programs P51OD011132 (ENPRC Base Grant) and P51OD011092 (ONPRC Base Grant), and the EPC Fund for Excellence. The ENPRC and ONPRC are fully accredited by AAALAC, International.
Acknowledgments
This review is dedicated to the memory of Martine Meunier whose pioneering research on the perirhinal cortex has transformed the study of memory processes mediated by structures within the medial temporal lobe. Her enlightening research impacted our understanding of not only the nature of memory but also how we modulate our emotions. Her immense contributions to neuroscience using nonhuman primate models have had a tremendous impact to a better understanding of the neuropathology of human clinical disorders and will continue to reverberate throughout the field. Her scientific impact will be felt for generations to come. We thank the JB laboratory trainees, students and research assistants that have participated in the execution of the series of nonhuman primate experiments described in this review as well as the veterinary and animal husbandry staff at ENPRC for expert care and handling of the animals, and the image core facility for their support during the MR imaging.
References
1. Milner B. The medial temporal-lobe amnesic syndrome. Psychiatr Clin North Am. 2005;28(3):599-611, 09. doi: 10.1016/j.psc.2005.06.002. PubMed PMID: 16122569.
2. Warrington EK, Weiskrantz L. Organisational aspects of memory in amnesic patients. Neuropsychologia. 1971;9(1):67-73. doi: 10.1016/0028-3932(71)90063-7. PubMed PMID: 5146487.
3. Squire LR. Declarative and Nondeclarative Memory: Multiple Brain Systems Supporting Learning and Memory. Memory Systems 1994: The MIT Press; 1994. p. 203-31.
4. Malamut BL, Saunders RC, Mishkin M. Monkeys with combined amygdalo-hippocampal lesions succeed in object discrimination learning despite 24-hour intertrial intervals. Behav Neurosci. 1984;98(5):759-69. doi: 10.1037//0735-7044.98.5.759. PubMed PMID: 6487412.
5. Meunier M, Bachevalier J, Mishkin M, Murray EA. Effects on visual recognition of combined and separate ablations of the entorhinal and perirhinal cortex in rhesus monkeys. J Neurosci. 1993;13(12):5418-32. doi: 10.1523/JNEUROSCI.13-12-05418.1993. PubMed PMID: 8254384; PMCID: PMC6576426.
6. Meunier M, Hadfield W, Bachevalier J, Murray EA. Effects of rhinal cortex lesions combined with hippocampal excision on visual recognition memory in rhesus monkeys. J Neurophysiol. 1996;75(3):1190-205. doi: 10.1152/jn.1996.75.3.1190. PubMed PMID: 8867128.
7. Baxter MG, Murray EA. Opposite relationship of hippocampal and rhinal cortex damage to delayed nonmatching-to-sample deficits in monkeys. Hippocampus. 2001;11(1):61-71. doi: 10.1002/1098-1063(2001)11:1<61::AID-HIPO1021>3.0.CO;2-Z. PubMed PMID: 11261774.
8. Heuer E, Bachevalier J. Effects of selective neonatal hippocampal lesions on tests of object and spatial recognition memory in monkeys. Behav Neurosci. 2011;125(2):137-49. doi: 10.1037/a0022539. PubMed PMID: 21341885; PMCID: PMC3072249.
9. Murray EA, Mishkin M. Object recognition and location memory in monkeys with excitotoxic lesions of the amygdala and hippocampus. J Neurosci. 1998;18(16):6568-82. doi: 10.1523/JNEUROSCI.18-16-06568.1998. PubMed PMID: 9698344; PMCID: PMC6793180.
10. Beason-Held LL, Rosene DL, Killiany RJ, Moss MB. Hippocampal formation lesions produce memory impairment in the rhesus monkey. Hippocampus. 1999;9(5):562-74. doi: 10.1002/(SICI)1098-1063(1999)9:5<562::AID-HIPO10>3.0.CO;2-X. PubMed PMID: 10560927.
11. Zola SM, Squire LR, Teng E, Stefanacci L, Buffalo EA, Clark RE. Impaired recognition memory in monkeys after damage limited to the hippocampal region. J Neurosci. 2000;20(1):451-63. doi: 10.1523/JNEUROSCI.20-01-00451.2000. PubMed PMID: 10627621; PMCID: PMC6774137.
12. Murray EA. Quality Controls: The Role of Self-Corrective Science in Explorations of Primate Memory Systems. Hippocampus. 2024;35(1). doi: 10.1002/hipo.23667.
13. Brodmann K. Vergleichende lokalisationslehre der grobhirnrinde. Vergleichende lokalisationslehre der grobhirnrinde 1909. p. 324-.
14. Suzuki WA, Amaral DG. Topographic organization of the reciprocal connections between the monkey entorhinal cortex and the perirhinal and parahippocampal cortices. The Journal of Neuroscience. 1994;14(3):1856-77. doi: 10.1523/jneurosci.14-03-01856.1994.
15. Suzuki WA, Amaral DG. Perirhinal and parahippocampal cortices of the macaque monkey: cortical afferents. J Comp Neurol. 1994;350(4):497-533. doi: 10.1002/cne.903500402. PubMed PMID: 7890828.
16. Suzuki WA. The anatomy, physiology and functions of the perirhinal cortex. Curr Opin Neurobiol. 1996;6(2):179-86. doi: 10.1016/s0959-4388(96)80071-7. PubMed PMID: 8725959.
17. Ding SL, Van Hoesen GW. Borders, extent, and topography of human perirhinal cortex as revealed using multiple modern neuroanatomical and pathological markers. Human Brain Mapping. 2010;31(9):1359-79. doi: 10.1002/hbm.20940.
18. Miyashita Y. Perirhinal circuits for memory processing. Nat Rev Neurosci. 2019;20(10):577-92. Epub 20190904. doi: 10.1038/s41583-019-0213-6. PubMed PMID: 31485007.
19. Lavenex P, Suzuki WA, Amaral DG. Perirhinal and parahippocampal cortices of the macaque monkey: projections to the neocortex. J Comp Neurol. 2002;447(4):394-420. doi: 10.1002/cne.10243. PubMed PMID: 11992524.
20. Insausti R, Amaral DG, Cowan WM. The entorhinal cortex of the monkey: II. Cortical afferents. J Comp Neurol. 1987;264(3):356-95. doi: 10.1002/cne.902640306. PubMed PMID: 2445796.
21. Amaral DG, Price JL. Amygdalo-cortical projections in the monkey (Macaca fascicularis). Journal of Comparative Neurology. 2004;230(4):465-96. doi: 10.1002/cne.902300402.
22. Kondo H, Saleem KS, Price JL. Differential connections of the perirhinal and parahippocampal cortex with the orbital and medial prefrontal networks in macaque monkeys. J Comp Neurol. 2005;493(4):479-509. doi: 10.1002/cne.20796. PubMed PMID: 16304624.
23. Aggleton JP. Multiple anatomical systems embedded within the primate medial temporal lobe: Implications for hippocampal function. Neuroscience & Biobehavioral Reviews. 2012;36(7):1579-96. doi: 10.1016/j.neubiorev.2011.09.005.
24. Saunders RC, Mishkin M, Aggleton JP. Projections from the entorhinal cortex, perirhinal cortex, presubiculum, and parasubiculum to the medial thalamus in macaque monkeys: identifying different pathways using disconnection techniques. Exp Brain Res. 2005;167(1):1-16. Epub 20051029. doi: 10.1007/s00221-005-2361-3. PubMed PMID: 16143859.
25. Baizer JS, Ungerleider LG, Desimone R. Organization of visual inputs to the inferior temporal and posterior parietal cortex in macaques. J Neurosci. 1991;11(1):168-90. doi: 10.1523/JNEUROSCI.11-01-00168.1991. PubMed PMID: 1702462; PMCID: PMC6575184.
26. Desimone R, Schein SJ, Moran J, Ungerleider LG. Contour, color and shape analysis beyond the striate cortex. Vision Res. 1985;25(3):441-52. doi: 10.1016/0042-6989(85)90069-0. PubMed PMID: 4024463.
27. Haxby JV, Grady CL, Horwitz B, Ungerleider LG, Mishkin M, Carson RE, Herscovitch P, Schapiro MB, Rapoport SI. Dissociation of object and spatial visual processing pathways in human extrastriate cortex. Proc Natl Acad Sci U S A. 1991;88(5):1621-5. doi: 10.1073/pnas.88.5.1621. PubMed PMID: 2000370; PMCID: PMC51076.
28. Mishkin M, Ungerleider LG. Contribution of striate inputs to the visuospatial functions of parieto-preoccipital cortex in monkeys. Behav Brain Res. 1982;6(1):57-77. doi: 10.1016/0166-4328(82)90081-x. PubMed PMID: 7126325.
29. Ungerleider LG, Haxby JV. ‘What’ and ‘where’ in the human brain. Current Opinion in Neurobiology. 1994;4(2):157-65. doi: 10.1016/0959-4388(94)90066-3.
30. Wilson HR, Wilkinson F. From orientations to objects: Configural processing in the ventral stream. J Vis. 2015;15(7):4. doi: 10.1167/15.7.4. PubMed PMID: 26024513.
31. Murray EA, Wise SP. Why is there a special issue on perirhinal cortex in a journal called hippocampus? The perirhinal cortex in historical perspective. Hippocampus. 2012;22(10):1941-51. doi: 10.1002/hipo.22055.
32. Murray EA, Richmond BJ. Role of perirhinal cortex in object perception, memory, and associations. Curr Opin Neurobiol. 2001;11(2):188-93. doi: 10.1016/s0959-4388(00)00195-1. PubMed PMID: 11301238.
33. Devlin JT, Price CJ. Perirhinal contributions to human visual perception. Curr Biol. 2007;17(17):1484-8. doi: 10.1016/j.cub.2007.07.066. PubMed PMID: 17764947; PMCID: PMC1971135.
34. Peterson MA, Cacciamani L, Barense MD, Scalf PE. The perirhinal cortex modulates V2 activity in response to the agreement between part familiarity and configuration familiarity. Hippocampus. 2012;22(10):1965-77. doi: 10.1002/hipo.22065.
35. Barense MD, Ngo JKW, Hung LHT, Peterson MA. Interactions of Memory and Perception in Amnesia: The Figure–Ground Perspective. Cerebral Cortex. 2012;22(11):2680-91. doi: 10.1093/cercor/bhr347.
36. Bussey TJ, Saksida LM, Murray EA. Perirhinal cortex resolves feature ambiguity in complex visual discriminations. Eur J Neurosci. 2002;15(2):365-74. doi: 10.1046/j.0953-816x.2001.01851.x. PubMed PMID: 11849302.
37. Bussey TJ, Saksida LM, Murray EA. Impairments in visual discrimination after perirhinal cortex lesions: testing ‘declarative’ vs. ‘perceptual‐mnemonic’ views of perirhinal cortex function. European Journal of Neuroscience. 2003;17(3):649-60. doi: 10.1046/j.1460-9568.2003.02475.x.
38. Bussey TJ, Saksida LM, Murray EA. The Perceptual-Mnemonic/Feature Conjunction Model of Perirhinal Cortex Function. The Quarterly Journal of Experimental Psychology Section B. 2005;58(3-4b):269-82. doi: 10.1080/02724990544000004.
39. Bussey TJ, Saksida LM, Murray EA. Perirhinal cortex and feature-ambiguous discriminations. Learn Mem. 2006;13(2):103-5; author reply 6-7. doi: 10.1101/lm.163606. PubMed PMID: 16585785.
40. Hales JB, Broadbent NJ, Velu PD, Squire LR, Clark RE. Hippocampus, perirhinal cortex, and complex visual discriminations in rats and humans. Learn Mem. 2015;22(2):83-91. Epub 20150115. doi: 10.1101/lm.035840.114. PubMed PMID: 25593294; PMCID: PMC4341362.
41. Malkova L, Bachevalier J, Webster M, Mishkin M. Effects of neonatal inferior prefrontal and medial temporal lesions on learning the rule for delayed nonmatching-to-sample. Dev Neuropsychol. 2000;18(3):399-421. doi: 10.1207/S1532694207Malkova. PubMed PMID: 11385832.
42. Mumby DG, Pinel JP. Rhinal cortex lesions and object recognition in rats. Behav Neurosci. 1994;108(1):11-8. doi: 10.1037//0735-7044.108.1.11. PubMed PMID: 8192836.
43. Nemanic S, Alvarado MC, Bachevalier J. The Hippocampal/Parahippocampal Regions and Recognition Memory: Insights from Visual Paired Comparison versus Object-Delayed Nonmatching in Monkeys. The Journal of Neuroscience. 2004;24(8):2013-26. doi: 10.1523/jneurosci.3763-03.2004.
44. Wan H, Aggleton JP, Brown MW. Different contributions of the hippocampus and perirhinal cortex to recognition memory. J Neurosci. 1999;19(3):1142-8. doi: 10.1523/JNEUROSCI.19-03-01142.1999. PubMed PMID: 9920675; PMCID: PMC6782155.
45. Bowles B, Crupi C, Mirsattari SM, Pigott SE, Parrent AG, Pruessner JC, Yonelinas AP, Köhler S. Impaired familiarity with preserved recollection after anterior temporal-lobe resection that spares the hippocampus. Proceedings of the National Academy of Sciences. 2007;104(41):16382-7. doi: 10.1073/pnas.0705273104.
46. Schoemaker D, Gauthier S, Pruessner JC. Recollection and Familiarity in Aging Individuals with Mild Cognitive Impairment and Alzheimer’s Disease: A Literature Review. Neuropsychology Review. 2014;24(3):313-31. doi: 10.1007/s11065-014-9265-6.
47. Yonelinas AP, Kroll NE, Quamme JR, Lazzara MM, Sauve MJ, Widaman KF, Knight RT. Effects of extensive temporal lobe damage or mild hypoxia on recollection and familiarity. Nat Neurosci. 2002;5(11):1236-41. doi: 10.1038/nn961. PubMed PMID: 12379865.
48. Mayes AR, Holdstock JS, Isaac CL, Montaldi D, Grigor J, Gummer A, Cariga P, Downes JJ, Tsivilis D, Gaffan D, Gong Q, Norman KA. Associative recognition in a patient with selective hippocampal lesions and relatively normal item recognition. Hippocampus. 2004;14(6):763-84. doi: 10.1002/hipo.10211. PubMed PMID: 15318334.
49. Aggleton J, Kyd R, Bilkey D. When is the perirhinal cortex necessary for the performance of spatial memory tasks? Neuroscience & Biobehavioral Reviews. 2004;28(6):611-24. doi: 10.1016/j.neubiorev.2004.08.007.
50. Staresina BP, Fell J, Do Lam AT, Axmacher N, Henson RN. Memory signals are temporally dissociated in and across human hippocampus and perirhinal cortex. Nat Neurosci. 2012;15(8):1167-73. Epub 20120701. doi: 10.1038/nn.3154. PubMed PMID: 22751037; PMCID: PMC3428860.
51. Staresina BP, Fell J, Dunn JC, Axmacher N, Henson RN. Using state-trace analysis to dissociate the functions of the human hippocampus and perirhinal cortex in recognition memory. Proc Natl Acad Sci U S A. 2013;110(8):3119-24. Epub 20130204. doi: 10.1073/pnas.1215710110. PubMed PMID: 23382181; PMCID: PMC3581882.
52. Eldridge LL, Knowlton BJ, Furmanski CS, Bookheimer SY, Engel SA. Remembering episodes: a selective role for the hippocampus during retrieval. Nat Neurosci. 2000;3(11):1149-52. doi: 10.1038/80671. PubMed PMID: 11036273.
53. Vilberg KL, Rugg MD. Dissociation of the neural correlates of recognition memory according to familiarity, recollection, and amount of recollected information. Neuropsychologia. 2007;45(10):2216-25. Epub 20070312. doi: 10.1016/j.neuropsychologia.2007.02.027. PubMed PMID: 17449068; PMCID: PMC1933497.
54. Yonelinas AP. Unpacking the Medial Temporal Lobe: Separating Recollection and Familiarity. Hippocampus. 2025;35(5):e70033. doi: 10.1002/hipo.70033. PubMed PMID: 40919742; PMCID: PMC12416122.
55. Cannon TD, Glahn DC, Kim J, Van Erp TG, Karlsgodt K, Cohen MS, Nuechterlein KH, Bava S, Shirinyan D. Dorsolateral prefrontal cortex activity during maintenance and manipulation of information in working memory in patients with schizophrenia. Arch Gen Psychiatry. 2005;62(10):1071-80. doi: 10.1001/archpsyc.62.10.1071. PubMed PMID: 16203952.
56. D’Esposito M, Postle BR, Ballard D, Lease J. Maintenance versus Manipulation of Information Held in Working Memory: An Event-Related fMRI Study. Brain and Cognition. 1999;41(1):66-86. doi: 10.1006/brcg.1999.1096.
57. Glahn DC, Kim J, Cohen MS, Poutanen VP, Therman S, Bava S, Van Erp TG, Manninen M, Huttunen M, Lonnqvist J, Standertskjold-Nordenstam CG, Cannon TD. Maintenance and manipulation in spatial working memory: dissociations in the prefrontal cortex. Neuroimage. 2002;17(1):201-13. doi: 10.1006/nimg.2002.1161. PubMed

