Probiotics: Key Role in Disease Prevention and Treatment
“`html
A Comprehensive Perspective of Probiotics and their Significant Role in Successfully Preventing or Treating Diseases in Both Preventive and Clinical Medicine
Dr. Malireddy S Reddy, BVSc (DVM)., MS., Ph.D
International Media and Cultures, Inc. [IMAC], American Dairy and Food Consulting Labs, Inc. [ADFAC Labs, Inc.], Denver, Colorado, USA.
OPEN ACCESS
PUBLISHED
30 September 2025
CITATION
Reddy, MS., 2025. A Comprehensive Perspective of Probiotics and their Significant Role in Successfully Preventing or Treating Diseases in Both Preventive and Clinical Medicine. Medical Research Archives, [online] 13(9). https://doi.org/10.18103/mra.v13i9.6915
ABSTRACT
The aim and scope of this research/review article is to point out the role of probiotics and their immunomodulins to prevent or treat several incurable, acute and chronic metabolic diseases. The article is written to address physiological role of probiotics to prevent or treat such diseases at a molecular level, to safeguard humanity. It is also the aim of this research/ review article to educate the healthcare professionals on therapeutic aspects of several strains of probiotics, so that they can properly select them to treat their patients with greater efficacy.
This review and research article provides a comprehensive analysis of probiotics, with particular emphasis on their applications and significance in preventive and clinical medicine. The discussion encompasses morphology, cellular functions, distribution and predilection sites of the attachment of probiotics in the human gastrointestinal tract. In addition, definitions, regulatory considerations, and the therapeutic functions of various individual probiotic strains across different genera and species are presented with explicit details. Special attention is given to the biochemical mechanisms underlying the therapeutic effects associated with the prevention or treatment of specific diseases and symptoms, by individual probiotic strains present in multiple mixed strain culture. A major focus is placed on the role of probiotics in the prevention and treatment of hospital-acquired (nosocomial) infections, certain cancers, and several viral infections, with particular emphasis on the recent COVID-19 pandemic caused by the SARS-CoV-2 RNA coronavirus. The article offers a logical explanation of how probiotics may help modulate cytokine storms, a key factor in viral and bacterial infections, several allergies, obesity, and autoimmune disorders. During the course of research aimed at combating COVID-19, several serendipitous findings emerged, revealing potential therapeutic applications of probiotics in managing common conditions such as hypertension and type 2 diabetes. A scientific rationale is presented to explain the pathophysiology of these diseases and how probiotics may offer a novel approach to their treatment. Consequently, a concise pathophysiology of a particular disease has been presented to show exactly and hypothetically at what stage probiotics exert their therapeutic effect, for the sake of better understanding and appreciation by the readers. The article proposes function-specific combinations of probiotic strains that may serve as therapeutic agents for particular diseases or syndromes, offering a practical guide for physicians in clinical settings. The research/review article, presented with simplified and yet detailed schematic and pictorial presentation is an early guide to follow the theme of genesis, physiology, and role of probiotics to be used as preventative or therapeutic aids to prevent or cure several diseases in preventive or clinical medicine set up. For the sake of simplicity more emphasis has been placed on the pictorial presentations than elaborate and complex script for the sake of easy understanding, along with the pertinent references in this article.
Keywords:
Multiple mixed strain probiotic therapy; Probiotics; Bacteriocins; Immunomodulins; Immunomodulation; Cytokine storm; COVID-19; Diabetes; Hypertension; Thrombosis; Anti-ageing; Allergy; Hospital-acquired infections; Nosocomial infections; Autoimmune diseases; Fecal microbiota therapy; SARS CoV-2; C.diff; MRSA; Immune checkpoint cancer therapy; Standard cancer therapy; Adjuvant cancer therapy; US Patent 11,077,052B1; Dysbiosis.
Introduction
Recently, it has been brought to the attention of people around the world by several medical professionals that allopathic medicines developed and recommended by pharmaceutical companies are causing serious, damaging side effects. These side effects can often be worse than the original disease itself. For decades, antibiotics have been used to treat bacterial infections, but now pathogenic bacteria have evolved to be multi-antibiotic resistant, causing hospital-acquired infections (nosocomial infections) that do not respond to antibiotics. According to an analysis, these infections are projected to kill over ten million people per annum by the year 2050.
Another devastating disease, cancer, has become a major subject of discussion among scientists and physicians. Due to a lack of proper treatment and modern scientific advancements, only 20% to 30% of cancer victims survive the disease. The relapse of cancer is a constant threat, and there is no permanent guarantee that a patient will be cured.
In addition, highly-mutated viruses like the SARS-CoV-2 virus which causes COVID-19, are spreading at a faster pace and causing pandemics. These pandemics have resulted in a severe loss of human lives and have drained the global economy. There is no specific treatment for viral diseases other than treating symptoms. Billions of dollars are spent annually on vaccinations and medications for viruses like the influenza virus, with limited success.
The human race has also been affected by other uncontrollable metabolic diseases such as hypertension, diabetes, obesity, food intolerances, and allergies. These conditions result in severe costs, a loss of time, and great stress on victims, their families, and governments. It has been shown that due to modern living, food habits, lifestyle, and severe stress, the human immune system has been compromised. It has also been reported that apparently modern pharmaceutical prescription drugs and over-the-counter drugs are not able to cure diseases with a high degree of accuracy and come without any side effects.
Thus, there is a great need for all-natural therapies to be used either as primary therapeutic agents or as adjuvants alongside standard treatments. One such emerging area is the use of natural probiotics to treat several diseases, as they can enhance immunity and orchestrate the human immune system through immunomodulation.
Before I proceed to discuss the role of probiotics to prevent or cure several diseases, I would like to provide a brief description regarding the genesis and evolution of these all-natural biological therapeutic agents.
Life on Earth is believed to have originated over 3.5 billion years ago, with the first major biological innovation being the production of oxygen through photosynthesis by primitive algae. This pivotal development laid the foundation for the evolution of more complex life forms, including unicellular bacteria and eventually multicellular organisms. Over time, the plant and animal kingdoms evolved, leading to the emergence of primates and, eventually, human beings.
If we consider the interconnectedness of all life forms, it becomes evident that microbial life has played — and continues to play — a foundational role in maintaining ecological balance. Based on current scientific speculation, bacteria first appeared around 3.0 billion years ago. Their emergence would have been followed by the evolution of viruses, which arose to maintain ecological equilibrium.
Despite their longstanding existence, bacteria remained unknown to humans until the invention of primitive microscopes. The Dutch lens maker Anton van Leeuwenhoek, in the late 1600s, was the first to observe microscopic organisms — later identified as bacteria — using handcrafted lenses. Although this marked the first visual confirmation of microbial life, little was understood about its implications, yet he became known as Father of Microbiology.
The turning point in the better understanding of bacteria came in 1865 with Louis Pasteur’s germ theory. Although several other scientists worked on bacteria before, Pasteur was credited with Father of Bacteriology. He demonstrated that specific microorganisms were responsible for certain diseases and pioneered the process of milk pasteurization — heating milk to 161°F (72°C) for 15 seconds — to eliminate pathogenic bacteria and ensure safety for human consumption. In this connection, it is also worthwhile to mention that Robert Koch was credited as a pioneer in medical microbiology due to his discovery of Anthrax Bacillus in 1876, and also due to his famous Koch’s postulates.
However, at that time, the focus remained on pathogenic bacteria, and the concept of “beneficial bacteria” was unknown. This changed with the pioneering work of Dr. Elie Metchnikoff, who in 1907 proposed that certain rod-shaped bacteria, particularly those originating from fermented milk, could prevent intestinal putrefaction and thereby enhance human longevity. He named these bacteria Lactobacillus because they appeared as rods (Bacillus) and are associated with milk (lacto).
Despite Metchnikoff’s Nobel Prize being awarded for his work on phagocytosis rather than probiotics, his observations laid the groundwork for future research in the field and thus he was considered as Grand Father of Probiotics. Meanwhile, in ancient cultures — notably in the Indian subcontinent — the use of fermented milk products had been common for millennia. Indian scriptures (the Vedas) reference the consumption of fermented milk as far back as 5,000 years, attributing the practice to divine figures such as Lord Krishna. Thus, Lord Krishna can be named as Great Grand Father of Probiotics. Each household traditionally prepared and consumed these products daily, intuitively recognizing their health benefits without knowledge of the underlying microbiological mechanisms.
It was not until 1965 that the term “probiotic” was formally introduced by Lilly and Stillwell, derived from the Greek “pro” (for) and “bios” (life), in contrast to antibiotics (“anti” = against, “bios” = life). In 1974, Parker defined probiotics as “organisms and substances which contribute to the intestinal microbial balance.” Fuller (1989) refined this to “live microbial supplements which beneficially affect the host by improving its intestinal microbial balance.” Salminen and colleagues in 1998 broadened the definition to include “foods containing live bacteria beneficial to health.” The current widely accepted definition, developed jointly by the Food and Agriculture Organization (FAO) of the United Nations and the World Health Organization (WHO), states: “Probiotics are live microorganisms which, when administered in adequate amounts, confer a health benefit on the host.” This comprehensive definition encapsulates all non-pathogenic, health-promoting microbes used in food and therapeutic applications.
In this connection it is worthwhile to mention the definitions of Para probiotics, postbiotics, immunomodulins, and prebiotics, since these terminologies are widely used in connection with probiotics, although they are physiologically different from probiotics.
Para probiotics refer to non-viable (dead) probiotic microorganisms that retain the ability to modulate or stimulate the immune system. Despite being inactivated, these microbes can still exert significant biological effects.
Postbiotics are the soluble byproducts or metabolites produced during the growth and metabolism of probiotics. These include substances such as lactic acid, bacteriocins, short-chain fatty acids, bioactive peptides, hydrogen peroxide, and other antimicrobial and immunomodulatory compounds. These postbiotics are also known as immunomodulins due to their role in influencing immune function.
Prebiotics, on the other hand, are not microbes or microbial products. Instead, they are substrates — typically dietary fibers — that are selectively utilized by host probiotic microorganisms to promote their growth or activity. Examples include inulin, oligosaccharides and various soluble and insoluble fibers that humans cannot digest but which serve as nourishment for beneficial bacteria in the gastrointestinal tract.
What is well-established in scientific literature is that probiotics, para probiotics and postbiotics all possess significant health-promoting properties, particularly in supporting digestive health, enhancing immune responses and maintaining microbial balance in the gut.
Functions of Probiotics
Probiotics are live microorganisms that, when administered in adequate amounts, confer health benefits to the host. Their well-documented functions include reduction of lactose intolerance; immunomodulation and immune system stimulation; direct inhibition of pathogenic bacteria and viruses; enhancement of intestinal peristalsis and motility; reduction of blood cholesterol; reduction of the risk of certain cancers; and modulation of blood pressure and blood sugar levels.
Roles of Para-Probiotics and Postbiotics
Para probiotics (non-viable microbial cells) and postbiotics (metabolic soluble byproducts of probiotics) share many of the same health benefits as live probiotics, except for the direct inhibition of pathogenic microorganisms. However, they still enhance immune response and modulate immunity, indirectly contributing to the control of harmful bacteria and viruses.
In this connection, it is worthwhile to mention that Dr. Malireddy S. Reddy and Dr. D.R.K. Reddy were the first to introduce (in the world) probiotics as an essential therapeutic ingredient in combination with the drugs to enhance the drug efficiency to treat diseases with least side effects. A US patent # 6,080,401 and other international patents were granted in the year 2000 for their breakthrough invention.
Although probiotics have been part of human diets for thousands of years, the scientific community only began exploring them in earnest in the late 20th century. As of 2025, the term “probiotic” has been in use for just 60 years. Despite their ubiquity in public discourse and growing media attention, a deep understanding of their physiological and therapeutic potential remains limited — even among healthcare professionals. This is due in part to gaps in both basic and applied research, often hindered by funding limitations and scientific skepticism.
Nevertheless, probiotics represent a promising frontier in medical and nutritional science. Continued research into their mechanisms and applications holds the potential to revolutionize our approach to preventive and therapeutic healthcare. The following are some of the well-established defined therapeutic probiotics belonging to several genera and species.
Part I: Members of Probiotics and Their General Physiological Functions
There are hundreds of probiotic strains across various genera and species, each possessing unique and scientifically validated therapeutic properties. These properties have significant applications in both preventive and clinical medicine.
Lactobacillus Species
- Lactobacillus acidophilus: Alleviates lactose intolerance, chronic fatigue, IBS, depression, insomnia, high cholesterol, Candida overgrowth, and musculoskeletal pains. Notably boosts immune system activity.
- Lactobacillus bulgaricus: Helps with allergic rhinitis, viral colds, periodontal issues (including halitosis), eczema, leaky gut, cholesterol, triglycerides, and systemic inflammation.
- Lactobacillus sporogenes (Bacillus coagulans): Effective against rotaviral, travelers, and antibiotic-associated diarrhea, IBS, IBD, Helicobacter pylori infections, and respiratory tract infections. Enhances immune response and serves as an adjuvant to increase vaccine efficacy.
- Lactobacillus rhamnosus: Reduces gastrointestinal and viral infections by modulating the Th1/Th2 immune response. Offers broad-spectrum antimicrobial action and enhances vaccine-related immunity.
- Lactobacillus plantarum: Mitigates seasonal allergies, IBS, hypertension, anxiety, influenza (including H1N1 and coronaviruses), cancer risk, diabetes, and obesity. Strongly binds to intestinal epithelial cells, boosting immunity and resisting pathogenic colonization.
- Lactobacillus casei: Reduces viral infections, respiratory tract infections (including pneumonia), and rotavirus. Enhances immune response, particularly in the elderly, by countering immunosenescence.
- Lactobacillus paracasei: Exhibits potent immunomodulatory effects beneficial in IBD. Adheres well to intestinal epithelium and acts as an effective para-probiotic.
- Lactobacillus helveticus: Reduces blood pressure, arthritis, anxiety, depression, and allergies. Supports bone health and resists gut dysbiosis due to its acid and bile tolerance.
- Lactobacillus reuteri: Reduces inflammatory diseases through suppressing production of pro-inflammatory cytokines, strengthens the intestinal barrier, also promoting T-Reg cell development and activity.
Bifidobacterium Species
- Bifidobacterium bifidum: Treats IBS, constipation, H. pylori infections, ulcerative colitis, necrotizing enterocolitis, and lung infections.
- Bifidobacterium longum: Reduces inflammation, infection, and oxidative stress through immune system stimulation.
Streptococcus and Enterococcus Species
- Streptococcus thermophilus: Alleviates lactose intolerance, mucositis, gastritis, ulcerative colitis, and antibiotic-associated diarrhea. Enhances skin hydration and stimulates innate immunity (macrophages and natural killer cells).
- Streptococcus faecium (Enterococcus faecium): Prevents infections by Clostridium difficile, Listeria, Salmonella, and H. pylori via broad-spectrum bacteriocin production. Promotes gut integrity and immune modulation through butyrate production and T-reg cell activation.
Lactococcus Species
- Lactococcus lactis subsp. lactis: Reduces allergies, bronchitis, alveolar inflammation, hypertension, LDL cholesterol, hearing loss, and viral infections. Stimulates dendritic and NK cells to enhance both innate and adaptive immunity.
- Lactococcus lactis subsp. cremoris: Mitigates depression, anxiety, and oxidative stress. Inhibits Listeria monocytogenes and boosts antioxidant production (e.g., folate, glutathione).
- Lactococcus lactis subsp. lactis var. diacetylactis: Strengthens immune response via cytokine and macrophage activation. Possesses antimicrobial and antifungal effects on gram negative pathogenic bacteria, as well as pathogenic yeast and molds.
Other Beneficial Bacteria
- Leuconostoc mesenteroides subsp. cremoris: Strong antimicrobial properties and excellent adhesion to intestinal epithelial cells. It significantly reduces gastrointestinal inflammation.
- Pediococcus acidilactici: Treats diarrhea, constipation, and autoimmune conditions such as encephalomyelitis by inducing IL-10–producing regulatory T-cells.
- Propionibacterium shermanii: Balances intestinal microbiota, scavenges mycotoxins, inhibits pathogens, and fosters Bifidobacterium growth. Produces propionic acid with antifungal and antimutagenic effects.
- Brevibacterium linens: Stimulates immune response, enhances protein and fat digestion, lowers cholesterol, inactivates RNA viruses (e.g., coronaviruses), and reduces GI tumors.
Yeasts and Molds
- Saccharomyces boulardii: A probiotic yeast that improves mineral bioavailability, detoxifies mycotoxins, reduces inflammation and oxidative stress, and lowers risk for cardiovascular diseases, Alzheimer’s disease, and cancer.
- Penicillium camembertii: A food-grade mold producing potent lipolytic and proteolytic enzymes. Supports digestion, gastrointestinal regeneration, and inflammation control.
- Penicillium roquefortii: A food grade mold, which produces Andrastatins A–D, with Andrastatin A showing strong anti-tumor effects and others acting as natural statins to inhibit cholesterol biosynthesis.
Although it is basic, I would like to present microscopic images of the probiotics Streptococcus thermophilus and Lactobacillus bulgaricus to illustrate their morphological features, both in growth media and within the gastrointestinal (GI) tract. These probiotics, when in single-cell form, are more metabolically active than when in chains, due to their larger surface area, which enhances nutrient uptake and the discharge of metabolic end products. The details of which are presented in Figure 1.

It is important to note that the plasmids are synthesized by the probiotic bacteria themselves to encode specific enzymes necessary for their survival. These plasmid-encoded enzymes not only offer survival advantages to the bacteria but are also responsible for the therapeutic effects of probiotics. In pathogenic bacteria, similar plasmids may be responsible for multiple antibiotic resistance.
Figure 3 illustrates the human intestinal tract, showing the approximate distribution of microbiota from the stomach to the distal end of the large intestine. This allows one to appreciate the abundance and localization of microorganisms within the GI tract, which perform a range of functions including digestion and immune support.
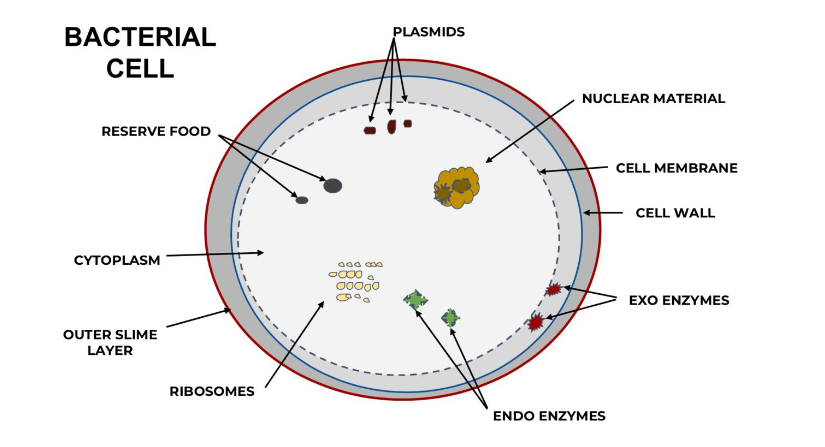
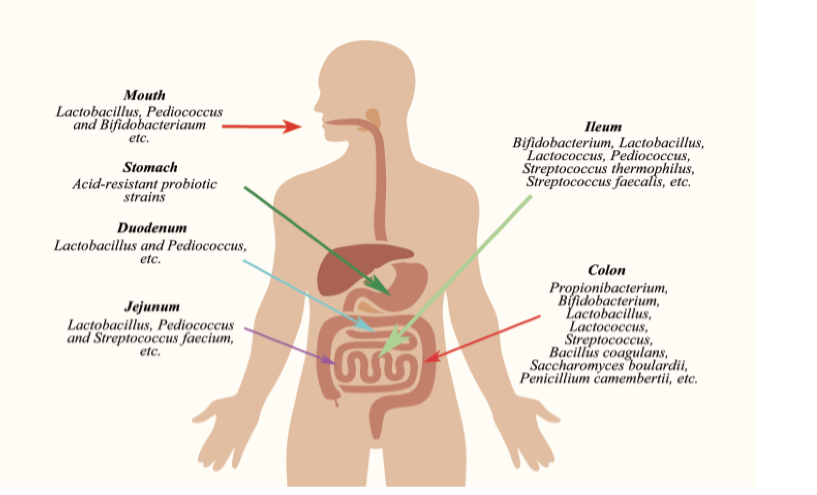
Figure 4 presents the approximate distribution, location, and adhesion sites of specific probiotics in the human GI tract. The total number of microorganisms in the GI tract is estimated to be around 100 trillion, of which approximately 20%—or 20 trillion—are probiotics.

It is distinctly clear from Figure 4 that most of the major functional probiotic bacteria are located in the ileum and colon. Overall, species belonging to the genus Lactobacillus and Pediococcus are located throughout the GI tract, starting from the duodenum to the distal end of the colon. The species belonging to genus Lactobacillus, Bifidobacterium and Streptococcus are mostly located in the jejunum and ileum, where they exhibit their maximum therapeutic effects. It is worthwhile to note that species belonging to the genus Propionibacterium and beneficial yeast and molds are also located in the colon. The total number of probiotics in the GI tract are roughly 20 trillion out of 100 trillion microorganisms constituting the Microbiota and Microbiome.
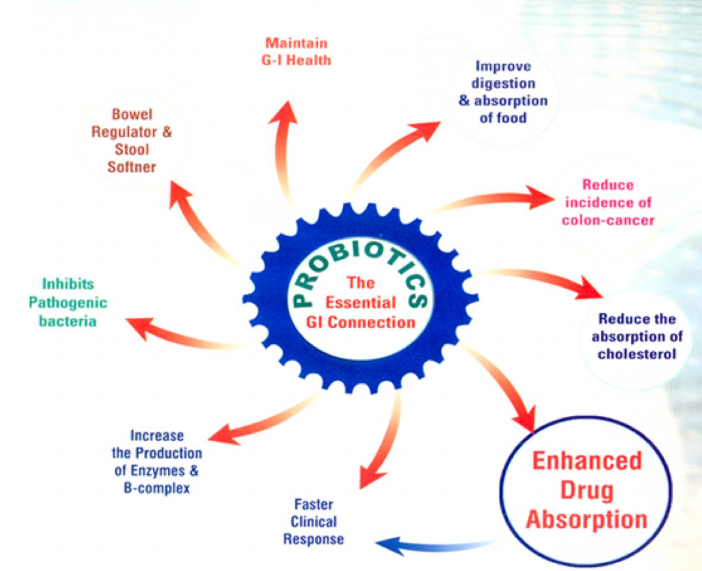
Figure 5 is a simplified illustration summarizing the major functions of probiotics in the human body. Since the primary focus of this article is to highlight the role of probiotics in preventive and clinical health, it is worthwhile to include these fundamental aspects of their morphology and distribution in the GI tract—particularly for readers with minimal background in microbiology.
The significant general functions of probiotics in the GI tract are digestion, reduction of cholesterol absorption, production of vitamins and beneficial enzymes. In addition, they facilitate the absorption of therapeutic drugs (specifically herbal and nutraceuticals) by eliminating destruction by other saprophytic organisms in the distal end of the GI tract. General functions of probiotics also include facilitating the absorption of minerals by maintaining a proper pH.
Finally, Table 1 in Part II of this article lists various therapeutic functions of probiotics.
The pathophysiology of a specific disease(s) and physiological molecular mechanism of probiotic therapeutic action are described under several specific headings in a chronological order.
Part II: Specific Proven Therapeutic Functions of Probiotics to Prevent/Treatment of Various Diseases
The specific proven therapeutic functions of probiotics are summarized in Table 1 below, after which we will expand upon each property in greater detail.
| Therapeutic Functions | Probiotic Strains |
|---|---|
| Immunomodulation | Lactobacillus rhamnosus, Lactobacillus plantarum |
| Reduction of Intestinal Infections | Lactobacillus acidophilus, Bifidobacterium bifidum |
| Reduction of Lactose Intolerance | Lactobacillus acidophilus |
| Suppression of Helicobacter pylori | Lactobacillus casei |
| Control of Cytokine Storms and COVID-19 | Lactobacillus rhamnosus, Lactobacillus plantarum |
| Control of Diabetes | Lactobacillus acidophilus, Bifidobacterium longum |
| Control of Hypertension | Lactobacillus helveticus |
| Control of Obesity | Lactobacillus rhamnosus, Bifidobacterium lactis |
| Prevention and Control of Allergies | Bifidobacterium bifidum, Lactobacillus casei |
| Improving Anti-Aging | Lactobacillus acidophilus, Lactobacillus helveticus |
| Prevention/Treatment of Hospital-Acquired Infections | Lactobacillus rhamnosus, Lactobacillus plantarum |
| Prevention or Suppression of Cancer | Lactobacillus rhamnosus, Bifidobacterium longum |
1) IMMUNOMODULATION (BY PROBIOTICS)
This is a critical and essential function induced by probiotics. The immune-stimulating activity is primarily attributed to their bacterial cell envelope constituents, such as peptidoglycan. Various studies indicate that probiotics stimulate the production of antibodies, enhance systemic macrophage activity, increase interferon levels, and elevate the number of natural killer cells. To influence the immune system effectively, probiotics must activate the lymphoid cells of the gut-associated lymphoid tissue (GALT), which are diffusely distributed among the epithelial cells and populate the lamina propria and submucosa.
The immunogenic properties of probiotic bacteria, such as Propionibacterium, do not reside in extracellular slime. Rather, their cell walls possess antigenic properties, further supporting the idea that the immunomodulatory effect of probiotics is a function of their cell wall composition—which can vary between strains. It is also noteworthy that even dead probiotic cells can exert immunomodulatory effects to some extent due to their retained cell wall components. Thus, probiotics exert superior immunomodulation through the production of their immunomodulins. Immunomodulation by probiotics is an essential requisite for human survival.
2) REDUCTION OF INTESTINAL INFECTIONS
These infections are commonly caused by pathogenic bacteria. Under normal circumstances, such pathogens are suppressed by the beneficial microflora of the gastrointestinal (GI) tract. A key component of this beneficial flora is probiotics, which have the innate ability to outcompete harmful bacteria.
Probiotic therapy has been shown to control antibiotic-induced diarrhea effectively. Lactobacillus and Bifidobacterium species have been used successfully in both children and adults to treat intestinal infections. Probiotics act via multiple mechanisms: competition for nutrients, secretion of antimicrobial substances, lowering of intestinal pH via short-chain fatty acid production, blocking of pathogen adhesion and toxin receptor sites, immune stimulation, and attenuation of bacterial and viral virulence.
In this connection, it is worthwhile to mention regarding the probiotic-produced bacteriocins and their role in inhibiting pathogenic bacteria involved in several infections. The bacteriocins are ribosomal synthesized antimicrobial peptides produced and excreted by probiotics. These bacteriocins exhibit a wide range of inhibitory effects on several pathogens. However, unlike antibiotics, they are non-toxic and non-allergenic and can be ultimately inactivated in the gastrointestinal (GI) tract after they exert their functions. Unlike antibiotics, pathogenic bacteria cannot develop resistance to bacteriocins. The specific bacteriocins produced by several strains of probiotics are listed in Table 6 towards the end of this publication. Selecting proper probiotic strains which are intended to be used as therapeutic agents to cure a specific disease must be based on the bacteriocins they produce.
3) REDUCTION OF LACTOSE INTOLERANCE
Lactose is a disaccharide composed of glucose and galactose. It is hydrolyzed by the enzyme lactase (or β-galactosidase), which is produced by the epithelial cells of the ileum. In some individuals, lactase is absent or produced in insufficient quantities. Consequently, undigested lactose causes gastrointestinal symptoms such as bloating, gas, and diarrhea—collectively known as lactose intolerance. It affects nearly half of the global population.
Probiotic organisms, especially lactic acid-producing bacteria, can help by producing lactase. This lactase is often encoded by extrachromosomal plasmids in the bacteria. When these probiotics colonize the ileum, they can compensate for the host’s lactase deficiency, thereby alleviating symptoms. Lactose intolerance is particularly prevalent in the elderly due to reduced endogenous lactase production. Therefore, multiple mixed-strain probiotics should be administered routinely to older adults. Additionally, it has been observed that probiotic levels naturally decline with age due to physiological factors. Deficiency of lactase enzyme is one of the prime reasons for the impaired absorption of calcium in the ilium due to the impairment of maintaining a proper pH. Derangement of calcium absorption leads to osteopenia and osteoporosis, in addition to hypertension. Thus, it is vital to maintain proper probiotics in the GI tract.
4) SUPPRESSION OF HELICOBACTER PYLORI
H. pylori is a gram-negative, spiral-shaped bacterium that colonizes the stomach lining by creating microcolonies generating high ammonia production from urea (via urease enzyme). It is a well-established cause of chronic gastritis, peptic ulcers, gastric atrophy, and gastric cancer.
In 1994, the International Agency for Research on Cancer classified H. pylori as a Group 1 carcinogen, the definitive cause of gastric cancer. Some probiotic strains can suppress or inactivate H. pylori. The likely mechanisms include the production of bacteriocins, antimicrobial peptides, and other inhibitory substances. It is an excellent and desirable practice to administer probiotics as a part of a preventative therapy to reduce the incidences of H.pylori infection.
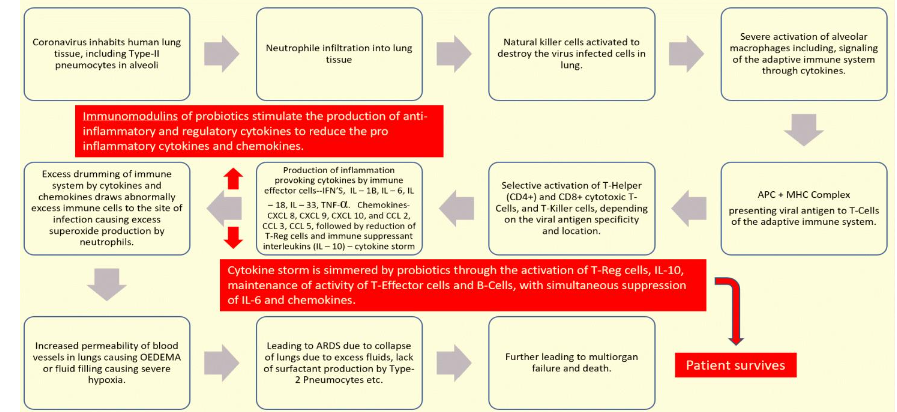
5) CONTROL OF CYTOKINE STORMS AND COVID-19 (BY PROBIOTICS)
A cytokine storm is a dangerous, systemic inflammatory response characterized by the over-activation of immune cells and excessively high levels of circulating cytokines. This uncontrolled immune reaction can damage healthy tissues and contribute to various severe conditions, including autoimmune diseases and complications from infections like COVID-19.
Cytokines (“cyto” means cells and “kinos” means moving) are crucial cell-signaling molecules, acting as messengers within the immune system. They are categorized into:
- Pro-inflammatory cytokines (e.g., IL-1, IL-6, IL-8, IL-12, IL-18, TNF-alpha, GM-CSF, IFN-gamma): These are produced mainly by T-Helper cells (TH) and Macrophages. They promote, upregulate and amplify inflammatory responses. IL-6 is a particularly significant pro-inflammatory cytokine.
- Anti-inflammatory cytokines (e.g., IL-4, IL-10, IL-11, IL-13, TGF-beta): These work to suppress inflammation and restore immune balance. IL-10 is a key anti-inflammatory cytokine.
- Regulatory cytokines (e.g., IL-21, IL-35, and dual roles by TGF-beta and IL-10): These orchestrate a balanced and optimal immune system response.
In a nutshell, when the body encounters a pathogen, the innate immune system (e.g., neutrophils, macrophages) triggers the production of cytokines, which then activate the adaptive immune system (T-cells, B-cells) to produce more cytokines. While typically functioning at very low concentrations (Pico molar), during severe infections or inflammation, cytokine levels can surge dramatically (nano and micro molar), leading to a cytokine storm leading to death and decay. The only T-cells capable of dampening this storm are T-regulatory (T-reg) cells, which suppress pro-inflammatory cytokine production and enhance anti-inflammatory and regulatory cytokines.
5.1: Related Immunology Terminologies used in Cytokine Storm
Interferon vs. Interleukin
An Interferon is denoted as IFN. These Interferons are mainly involved in inhibiting viruses by making the neighboring cells non-permeable to such infections. The examples of Interferons are IFN-alpha and IFN-gamma.
An Interleukin is denoted by IL. The meaning of Interleukin is “inter” means between and “Leukin” means Leucocytes or White Blood Cells. The examples of Interleukins are IL-6, IL-10 etc. They serve as Messenger molecules between several immune cells.
Chemokines
The term “Chemo” means “chemical “and “Kinos” means moving. Chemokines also come under Cytokines. The cytokines are the general category of Messenger molecules, whereas Chemokines are special Cytokines involved in the migration of White Blood Cells to the infected or damaged tissues. The inflammatory Chemokines include: CCL2, CCL3, CXCL1, CXCL2, CXCL8, and CXCL10, and they are produced in Lymphoid tissue and Thymus. Thus, the general term Cytokines include Chemokines, Interferons, and Interleukins etc.
Most of the aforementioned diseases are as a result of the immune system mistakenly attacking healthy tissues triggering the cytokine storm, which will ultimately induce severe symptoms and cause multi organ failures.
Out of all the diseases outlined above, I would like to focus on the Pathophysiology of the recent Pandemic created Covid-19 infection, which induced ARDS due to severe Cytokine storm, and also attempt to explain the physiology behind the preventative and curative aspect of these syndromes with the use of probiotics and their immunomodulins.
5.3: Specific Details of Cytokine Storm during Covid-19 infection
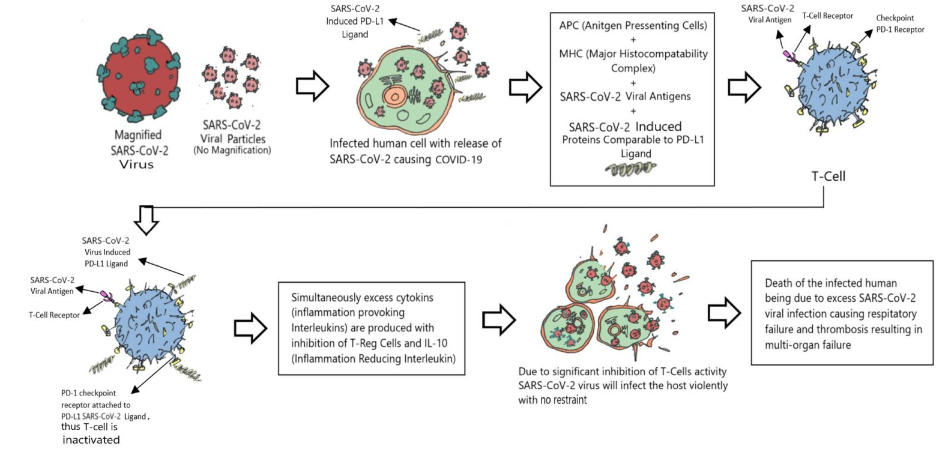
Induction of cytokine storm, due to Covid-19 disease causing SARS-CoV-2 corona virus starts initially with the reaction of innate immune cells to inactivate the virus. At the same time, the innate immune cells produce cytokines to activate the adaptive immune system. The corona viral antigens attached to MHC-2 (major histocompatibility complex-2) on the antigen presenting cells, activate CD 4 +T cells and they in turn activate TH-17 cells. The activated TH-17 in turn recruits and sends more Neutrophiles and Macrophages (innate immune cells) to the infection site with the aid of Interleukins and chemokines. In addition, CD-4 + T cells also produce abnormal number of T-killer cells to attack the SARS-CoV-2 virus present in the lung tissue, leading to more damage to the lung. Thus, the combination of excess Neutrophile infiltration and leakage of plasma from blood vessels results in the preliminary stages of ARDS (acute respiratory distress syndrome). Meanwhile CD-8+ T cells, after recognizing SARS-CoV-2 viral peptides in the infected lung tissue, releases excess number of cytotoxic molecules to stop further spread of the virus. Adding insult to the wound, the corona virus also selectively induces Macrophages to produce inflammation provoking IL-6, which in turn regulates the PD-1 and CTL-4 immune check point receptors on T-cells with the aid of the virus infected cell induced ligands to make the T-cells infective. This will cause severe decrease in lymphocyte count from 3000 per microliter to roughly 1000, resulting in lymphocytopenia. Thus, cytokine storm results in causing T- lymphocyte exhaustion and severe lung tissue damage, ultimately end up with severity of disease -Covid-19 with resulting ARDS and death. The exact mechanisms of how the corona virus propagates in the infected Eukaryotic cells, and how the corona virus induced host cell ligands inactivate T-cells to induce lymphocytopenia during Covid-19 corona viral infection is presented in Figure 6 and Figure 7.

5.4: Covid-19 Disease Progression
The infective pattern and multiplication of SARS-CoV-2 virus in Eukaryotic cell is presented in Fig-1. The corona virus spike attaches to the ACE-2 receptor site on the eukaryotic cell and thus releases the single stranded positive sense RNA molecule. The viral RNA starts multiplying, making several progenies. The fully formed virions are released through exocytosis out of the infected cell and continue the infection at a rapid pace. Since SARS-CoV-2 virus has roughly 90 spikes, the chances of the viral spike attachment to host cell ACE-2 receptor is significantly greater and after attachment viral RNA is integrated into the host cell. It takes roughly 15 minutes from the time it attaches to the cell and inject viral RNA into the host eukaryotic cell. From the time the viral RNA is injected into the eukaryotic cell to the time the release and appearance of the newly made virion is called the eclipse period. Generally, the eclipse period in corona virus is roughly 12-36 hours. The number of viral particles released from each viral infected cell is called burst size. It has been reported that the burst size of the corona virus is roughly 700 virions from each infected cell. The molecular details of the infection pattern are outlined in depth in the publication of Reddy.
5.5: Role of probiotics along with their immunomodulins for preventing or treating Covid-19 infection due to SARS-CoV-2 virus through immunomodulation, and the prevention of Lymphocytopenia
Probiotics, along with their immunomodulins (substances produced by probiotics that influence the immune system), are believed to mitigate cytokine storms through a sophisticated process of immunomodulation. Essentially, they aim to restore the delicate balance of the immune system by:
- Up-regulating beneficial immune responses: Probiotics can increase the production of anti-inflammatory cytokines (like IL-10) and regulatory cytokines. They also promote the activity and proliferation of T-regulatory cells, which are essential for reining in excessive inflammation. Furthermore, they can enhance the function of T-effector cells and B-cells, ensuring an effective, but controlled, immune response.
- Down-regulating harmful immune responses: Concurrently, probiotics work to reduce the production of pro-inflammatory cytokines (such as IL-6) and chemokines (specialized cytokines that direct immune cells to sites of infection), thereby suppressing the uncontrolled inflammatory cascade characteristic of a cytokine storm.
This entire mechanism of cytokine storm control by probiotics is presented in Figure 8.
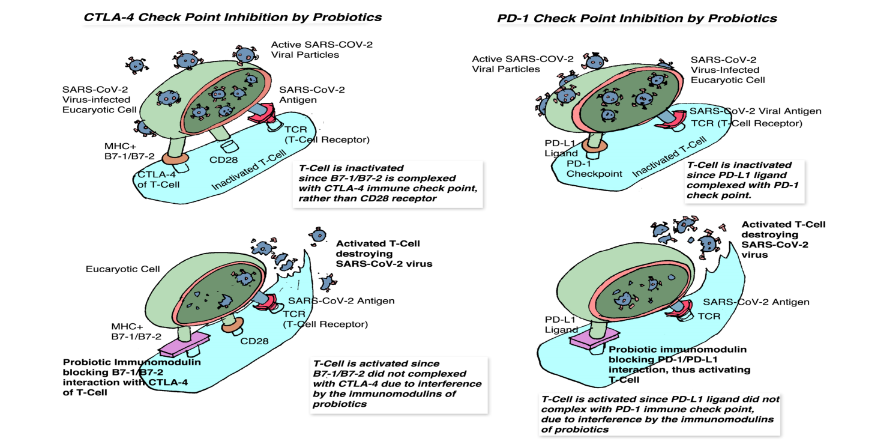
5.6: The Role of Probiotics to reduce Lymphocytopenia in COVID-19
In conditions like severe COVID-19, the SARS-CoV-2 virus can cause a significant decrease in lymphocyte count (lymphocytopenia), leading to T-cell exhaustion and compromised immune function. This occurs partly because the virus can inactivate T-cells by interacting with their immune checkpoints (like PD-1 and CTLA-4 receptors). These checkpoints normally act as “brakes” on T-cell activation, preventing the immune system from attacking the body’s own tissues. However, in viral infections, ligands produced by infected cells (PDL-1 and B7-1/B7-2) can bind to these check points (PD-1 and CTLA-4) on T-cells, effectively deactivating them and allowing the virus to multiply unchecked.
Multiple mixed-strain probiotics and their immunomodulins may act as checkpoint inhibitors themselves, much like therapeutic antibodies. By occupying or modulating these immune checkpoints on T-cells, probiotics could prevent the virus-induced ligands from attaching and inactivating the T-cells. This would help preserve T-cell function, prevent their exhaustion, and support the host’s ability to mount an effective immune response against the virus, thereby mitigating lymphocytopenia and the severity of the disease. The details on how probiotics can act as check point inhibitors is presented in Figure 9.
5.7: Control of Diabetes (BY PROBIOTICS)
There are two types of Diabetes Mellitus: Type 1 Diabetes (T1DM) and Type 2 Diabetes (T2DM). By definition, Diabetes Mellitus is a chronic metabolic disorder resulting from either the body’s inability to produce enough insulin or its inability to effectively use the insulin it produces, leading to elevated blood sugar levels. As of mid-2025, approximately 537 million adults globally are living with diabetes, a number projected to rise to 783 million by 2045. This disorder is characterized by blood glucose dysregulation.
The beta cells of the Islets of Langerhans in the pancreas produce hormone insulin, which controls the entry of blood glucose into eukaryotic cells to generate ATP through the glycolytic cycle. In T1DM, the pancreas produces little or no insulin, leading to high blood sugar levels. In contrast, in T2DM, the pancreas may produce insufficient insulin, or even a normal level of insulin, which may not effectively facilitate glucose uptake into cells due to insulin resistance, resulting in excess glucose in the blood (hyperglycemia).
According to the literature, 90% of diabetic cases are T2DM, with roughly 10% being T1DM. T2DM is currently a global pandemic. The WHO (World Health Organization) defines it as a metabolic disorder of multiple etiologies characterized by chronic hyperglycemia with disturbances of carbohydrate, fat, and protein metabolism, resulting from defects in insulin secretion, insulin action, or both. It has been identified as a leading cause of blindness, end-stage renal disease, lower limb amputation, and cardiovascular disease. Although diabetes was first reported in Egypt over 3,000 years ago, the distinction between T1DM and T2DM was established only in 1936.
6.1: Etiology and Pathophysiology of Type 2 Diabetes
T2DM is largely caused by impaired insulin production and secretion by pancreatic beta cells, as well as peripheral tissue insulin resistance. Before I go into how probiotics can reduce hyperglycemia, a brief description of the genesis of hyperglycemia is presented.
The following etiological factors have been attributed to the onset of T2DM:
- Obesity: Generally considered a primary factor, as obese individuals have a 90% chance of developing T2DM, due to insulin resistance.
- Stress and Depression: These increase the risk of T2DM.
- Environmental Factors: Pollution and excessive use of food preservatives have also been implicated.
- Pathogenic Viruses: Viruses such as Herpes Simplex and Hepatitis C have been attributed as causative factors. Specifically, a liver infected with Hepatitis C virus promotes severe insulin resistance.
Out of all several etiological factors, I would like to discuss the role of Obesity and insulin resistance in the onset of T2DM.
Diabetic onset is partly due to obesity, which induces peripheral insulin resistance. Obesity is characterized by elevated levels of cytokines and fatty acids. Initially, the onset of insulin resistance increases the demand for insulin production by the beta cells of the Islets of Langerhans in the pancreas, achieved through increasing the volume of cells. Under chronic insulin resistance, beta cells eventually cannot cope with the demand for insulin production, thus significantly slowing down production and undergoing apoptosis. This results in hyperglycemia.
In addition, low insulin levels also alter normolipidemic states (normal triglycerides, HDL, LDL, and cholesterol in the blood). Higher levels of these lipids due to decreased insulin levels can lead to cardiovascular complications. Another indication of T2DM is an increase in proinsulin levels in the blood. Proinsulin is the inactive precursor to insulin produced in the pancreas. It is a single-chain protein that converts into insulin and C-peptide (connecting peptide) through the removal of C-peptide by prohormone convertase enzymes. Normally, small amounts of proinsulin enter the blood along with insulin, typically higher after meals and in individuals with insulin resistance or early stages of T2DM. In healthy individuals, proinsulin in the blood can be roughly 20% of total insulin; however, in T2DM, it can reach up to 50%, indicating beta cell dysfunction. The lack of conversion of proinsulin into insulin deficiency leading to T2DM.
6.2: Insulin Resistance (IR)
The term insulin resistance (IR) refers to why glucose levels remain elevated even when there is no insulin deficiency. Normally, blood glucose is reduced by cellular uptake using GLUT transporters. Some cells have transporters that are responsive to insulin (e.g., GLUT4 in muscle and adipose cells), while others have transporters that do not require insulin for activation (e.g., glucose-dependent cells including red blood cells, white blood cells, renal papilla cells, nervous system cells, cardiac muscle cells, etc.). Some muscle cells possess both non-insulin responsive and insulin-responsive (GLUT4) transporters. As ironic as it sounds, it has been attributed that Hyperinsulinemia, due to excess production and more concentration in the blood, is one of the causes of insulin resistance in T2DM.
According to available literature, although the etiology of diabetes is multifactorial, certain facts have been established. T1DM is primarily due to the total destruction of pancreatic beta cells, largely due to autoimmune reactions and genetic factors. In contrast, T2DM is attributed to reduced insulin production due to partial cessation of pancreatic beta cell function, often linked to obesity-related insulin resistance, resulting in hyperglycemia and hyperinsulinemia. Additionally, inflammation, pathogenic viral infections, and depression have been listed as etiological factors for the genesis of T2DM.
6.3: Mechanism by which Probiotics Reduce Blood Sugar Levels to Prevent or Treat T2DM
Probiotics and their immunomodulins have a significant effect, both directly and indirectly, on the prevention, control, and/or treatment of diabetes. This can be explained logically using the following available experimental evidence in relation to Inflammation reduction, reduction of viral infections, controlling depression and stress, and regulation of fat metabolism by probiotics.
6.4: Inflammation Reduction
As discussed in relation to reducing COVID-19 infection, a similar physiological mechanism applies to reducing inflammation in T2DM. The primary culprit in inflammation is predominantly the cytokine storm, due to an overactive immune system, characterized by elevated pro-inflammatory cytokines such as IL-6 and TNF-alpha. This cytokine storm, often related to obesity, ultimately leads to the slowing of beta cell functions, which are already overburdened due to increased demand for insulin production. Such cytokine storm and related chain reactions increases the accumulation of misfolded proteins in the endoplasmic reticulum (ER), leading to ER stress. Oxidative stress and ER stress can cause apoptotic cell death, leading to the progressive failure of even residual beta cells in the pancreas. This reduced insulin level leads to hyperglycemia, which further damages beta cells due to glucose toxicity. Additionally, hyperglycemia demands more insulin production, resulting in hyperinsulinemia, which can also contribute to beta cell damage.
Probiotics and their immunomodulins, through the activation of anti-inflammatory cytokines such as IL-10 and TGF-beta, can simmer inflammation. Simultaneously, probiotics also reduce the production and activity of pro-inflammatory cytokines to protect beta cells from deterioration.
It has also been hypothesized that our own T-cells can exhibit autoimmunity, destroying beta cells by misreading self-antigens as foreign proteins. Probiotics possess the innate ability to maintain and control immune checkpoints, which serve as negative regulators of T-cells, preventing them from attacking beta cells in the Islets of Langerhans. Furthermore, probiotics have the intrinsic ability to activate T-regulatory cells, which can significantly mitigate abnormal T-cell activity encountered in a cytokine storm. Thus, peripheral insulin resistance can be brought under control by restoring the function and repair of beta cells with the aid of probiotics and their immunomodulins.
6.5: Reduction of Pathogenic Viral Infections
Viral infections, such as herpes and hepatitis C, have been attributed as causative factors that increase insulin resistance, leading to T2DM. Probiotics have a significant effect on reducing viral infections by activating the immune system through immunomodulation. A detailed description of how probiotics can inhibit viral multiplication through immunomodulation was presented earlier.
6.6: Reduction of Depression and Stress
Depression and stress have also been identified as causative factors for T2DM. These can be brought under control by probiotics. This is accomplished by maintaining the cell wall integrity of the intestinal epithelial cells by probiotics and their immunomodulins, allowing tryptophan to be absorbed to generate serotonin, which acts as a nerve stimulant to prevent or suppress depression.
It has been reported that stress can alter the composition of intestinal microbiota, resulting in dysbiosis, which ultimately negatively influences the immune system and the health of human cells and tissues, including pancreatic beta cells. Probiotics and their immunomodulins can override the stress effect on the immune system by restoring the composition of microbiota and microbiome.
6.7: Regulating Fat Metabolism
Finally, the disturbance in fat metabolism due to T2DM, characterized by an increase in blood triglycerides, LDL, and total cholesterol, can be brought under control through the proven benefits of probiotics in reducing triglycerides and hypercholesterolemia. Probiotics can correct hypercholesterolemia by adsorbing cholesterol, through hydrolyzing the bile salts and thus, eliminating the cholesterol in feces, and also by assimilating the dietary cholesterol in the gastrointestinal tract.
6.8: Practical Illustration
As a practical illustration, a controlled experiment during the COVID-19 pandemic demonstrated that probiotics, along with their immunomodulins, reduced blood sugar levels in a diabetic patient using a significantly reduced dosage of medication. Readers are referred to US Patent # 11,077,052 B1 for trial details. This experiment definitively proved that probiotic therapy could function as an adjuvant alongside medicaments to lower or cure T2DM. Similar results have been observed by several investigators, although the exact mechanism was not always determined.
The number of diabetic cases is significantly increasing worldwide, and it can be controlled through the infusion of probiotics as a preventative measure. Although it can also be used both therapeutically or as an adjuvant in conjunction with the allopathic diabetic therapy.
7) CONTROL OF HYPERTENSION
The simple definition of blood pressure is the amount of force the blood uses to get through the arteries. It has been established that the normal blood pressure in adults should be below 120/80 mm Hg. Most people with high blood pressure will have no symptoms. Some of the causative factors for high blood pressure are stress, dysbiosis, obesity and diabetes etc. The results of hypertension are heart attacks, stroke, thrombosis, kidney problems, eye problems, dementia etc. According to the American Heart Association the stage one hypertension is 130-139 mm Hg Systolic/ Diastolic below 80 mm Hg, and stage two hypertension is if Systolic number is 140mm Hg or higher, and Diastolic number is 90 mm Hg or higher.
Lack of exercise and overeating leads to obesity. The inflammation goes up significantly due to obesity with the onset of excess cytokines in the body leading to hypertension. The current discussion is concentrated mainly on pathophysiology of hypertension and the role of probiotics to prevent or control or treat hypertension.
Although the pathophysiology of hypertension is multifactorial, in my opinion it is linked to dysbiosis, which is an imbalance in the gut microbiome leading to hypertension through several interconnected physiological mechanisms. These include oxidative stress, increased inflammation, and disruptions to the gut-brain axis, altered metabolite production, change in the gut permeability, and changes in the Renin Angiotensin-Aldosterone system. I would like to delve into the role of the Renin Angiotensin-Aldosterone system and Dysbiosis as the causative factors for inducing hypertension.
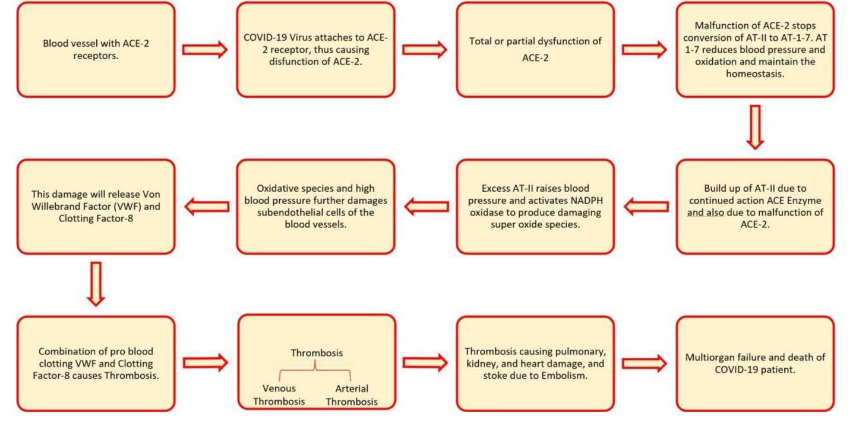
7.1: Renin-Angiotensin-Aldosterone System
Angiotensinogen gets converted to Angiotensin-1 by the action of renin enzyme in the liver. Angiotensin Converting Enzyme (ACE) acts on Angiotensin-1 to produce Angiotensin-2. This Angiotensin-2 serves as a substrate to be acted by ACE-2 enzyme present in the blood vessels resulting in Angiotensin 1-7. The cumulation of Angiotensin-2 constricts the blood vessels and thus raise the blood pressure. If not corrected, the excess activity of Angiotensin-2 will damage the vascular endothelial cells through NADPH Oxidase and thus induce severe oxidation of the blood vessels resulting in blood clots. Such blood clots ultimately block the blood flow resulting in heart attacks and strokes. Angiotensin 1-7 has completely opposite effect of Angiotensin-2 in that it causes vasodilation of blood vessels to lower or maintain the blood pressure.
7.2: Role of Dysbiosis to induce Hypertension
If the composition of Gastrointestinal micro-flora is altered due to toxins or excess pathogenic micro-flora in relation to saprophytic and probiotic bacterial microflora, the GI tract endothelial cells gets damaged. Such damage results in the weakening of the gut, which can cause hypertension perhaps by disrupting the function of ACE-2 enzyme in the blood vessels. In addition, if the number of probiotic bacteria are significantly reduced in the GI tract, the intestinal peristalsis gets altered to result in constipation. Constipation encourages the growth of non-beneficial microflora, which can convert the amino acids histidine and Tyrosine to histamine and Tyramine. These amines tend to induce hypertension through severe vasoconstriction. Dysbiosis can also weaken the intestinal barrier by increasing its permeability allowing pathogenic bacteria and their products like lipopolysaccharides (LPS) into the blood stream triggering inflammation and endothelial dysfunction resulting in hypertension.
Dysbiosis increases the pro-inflammatory bacteria and a concomitant decrease in the anti-inflammatory bacteria. This imbalance will trigger a chronic inflammatory response in the blood vessels resulting in vasoconstriction and hypertension.
Dysbiosis, by disrupting the microbial composition in the gut, can negatively alter the production of short chain fatty acids such as acetate and butyrate, which have anti-inflammatory and blood pressure lowering effects. It can also lead to the production of harmful metabolites due to the action of certain microbes in the GI tract, like Trimethylamine N-Oxide (TMAO) and hydrogen sulfide, which can promote vascular dysfunction and inflammation.
Dysbiosis can also disrupt the gut-brain axis communication (gut microbiome interaction with nervous system), which can affect the hormones (cortisone) to elevate blood pressure, thus inducing hypertension. For easy understanding the pathophysiology of hypertension due to disruption of hormonal-enzyme dependent vascular homeostasis due to SARS-CoV-2 corona viral infection is illustrated in Figure 10.
7.3: How do Probiotics reduce Hypertension?
Out of 100 trillion bacteria in the GI tract, roughly 20 trillion can be categorized as probiotics. I have listed several of these probiotic bacteria and their therapeutic effects earlier in this article. Probiotics and their immunomodulins can recognize pathogenic bacteria with the aid of receptors on their cell walls called “Pattern Recognition Receptors” (PRR5) to recognize pathogen associated molecular patterns (PAMPS) on the surface of the pathogenic bacteria. After recognizing the pathogens, probiotics trigger their own immune mechanism to inhibit or destroy the pathogens, through production of antimicrobial agents. In addition, they also compete for nutrients and the attachment sites in the GI tract thus outcompeting pathogenic bacteria. Thus, probiotics can correct the dysbiosis to reduce or eliminate inflammation to control hypertension.
In addition, probiotics also stimulate the immune system to eliminate inflammation by enhancing the anti-inflammatory cytokines such as IL-10 and TGF-beta. At the same time, probiotics significantly reduce the pro-inflammatory cytokines such as IL-6 and TNF-alpha. In addition, probiotics play a significant role in orchestrating immune modulation with the aid of T-regulatory cells. The immunomodulins or the growth end products of probiotics can up regulate the production of ACE-2 enzymes to convert angiotensin-2 into angiotensin 1-7, thus, to increase vasodilation and retard the oxidation by the NADPH oxidase enzyme to protect the blood vessels from hypertension.
It has also been cited in the literature that probiotics significantly reduce the production of angiotensin-2 from angiotensin-1 by down regulating the production of angiotensin converting enzyme (ACE), to reduce the excess buildup of angiotensin-2, which is a vasoconstrictor.
Probiotics through production of short chain fatty acids protect the intestinal epithelial cell barrier from letting the entry of the vasoconstriction inducing toxins into the blood to protect the host from hypertension. In addition, probiotics reduce constipation through improved peristalsis and also prevent conversion of amino acids (such as tyrosine) into hypertension creating amine (tyramine).
Probiotics also improve the absorption of calcium through maintaining the gut pH in the acid range to reduce the hypertension since bio-available calcium controls and maintains the blood pressure in the normal range. The mechanism of how probiotics can prevent or control hypertension due to corona viral infection is presented next in Figure 11. Similar mechanisms can be exhibited by probiotics in various different viral infections besides Covid-19.
As a practical illustration, a controlled experiment conducted during Covid-19 pandemic demonstrated that probiotics, along with their immunomodulins, reduced hypertension using a significantly reduced dosage of blood pressure medication. The details and data are presented in US Patent # 11,077,052 B1. It goes to prove that probiotics have a significant effect on treating hypertension when used as adjuvant therapy. In addition, probiotics can also be used as preventive therapeutic aids to eliminate or control blood pressure in normal healthy range.
8) CONTROL OF OBESITY (BY PROBIOTICS)
Obesity is now recognized as a pathological condition and is associated with several diseases, including Type 2 Diabetes, Cancer, and Cardiac Anomalies. It has been both postulated and proven that gut microbiota significantly influences the onset of obesity. By 2030, it is estimated that 38% of the adult population will be overweight and 20% will be obese worldwide.
The human gastrointestinal tract contains over 100 trillion microorganisms. Approximately 90% of the gut microbiota belongs to the phyla Firmicutes and Bacteroidetes. Firmicutes are typically Gram-positive, indicating they have thick layers of peptidoglycan in their cell walls, whereas Bacteroidetes are Gram-negative, with thin peptidoglycan layers sandwiched between two phospholipid bilayers. Thus, Bacteroidetes are difficult to inhibit compared to Firmicutes either with antibiotics or other inhibitory agents.
Research using the 16S rRNA amplification technique has tentatively shown that the ratio of Firmicutes to Bacteroidetes (F/B) plays a crucial role in determining the onset of either obesity or intestinal disorders, particularly Inflammatory Bowel Disease (IBD), which includes Ulcerative Colitis (UC) and Crohn’s Disease.
As a general rule:
- If the F/B ratio is significantly greater than that of one, the individual is more likely to develop obesity.
- If the F/B ratio is significantly less than one, the individual is more susceptible to intestinal disorders due to inflammation and cytokine storms.
Rule of Thumb: Maintaining a roughly equal number of Firmicutes and Bacteroidetes (F/B ≈ 1) is optimal for gut health.
High-fat diets tend to promote the growth of Firmicutes while decreasing the growth of Bacteroidetes. This microbial imbalance increases plasma lipopolysaccharides (LPS), which contributes to obesity by activating TLR-4 and upregulating pro-inflammatory cytokines. On the other hand, increasing Bacteroidetes enhances endogenous production of Glucagon-Like Peptide-2 (GLP-2) and strengthens intestinal tight junctions, thereby reducing LPS absorption and preventing obesity. However, excessive Bacteroidetes can also cause intestinal irritation due to a significant reduction in Firmicutes.
The optimal F/B ratio, therefore, lies close to 1:1, balancing the benefits of both phyla.
8.1: Determining the F/B Ratio:
The F/B ratio can be assessed using Gram staining of fecal samples under a microscope:
- Gram-positive (Firmicutes): purple
- Gram-negative (Bacteroidetes): red
Examples to determine the F/B ratio under microscope in a given field:
- 10 purple and 10 red bacteria = 1 (ideal)
- 15 purple and 5 red → F/B = 3 (indicative of obesity)
- 5 purple and 15 red → F/B = 0.33 (indicative of dysbiosis)
F/B ratios can be easily estimated through serial dilutions of fecal samples, which can be viewed accurately upon gram staining under microscope.
8.2: Probiotics and Their Role in Restoring the F/B Ratio
Controlling dysbiosis caused by excess Firmicutes through several mechanisms:
- Direct inhibition of Firmicutes overgrowth.
- Improving gut barrier integrity to reduce LPS absorption.
- Inducing anti-inflammatory cytokines such as IL-10 and TGF-beta while suppressing pro-inflammatory cytokines such as IL-6 and TNF-alpha.
- Nutritional competition to maintain a balanced F/B ratio.
- Production of bacteriocins to selectively inhibit excess Firmicutes.
- Generation of short-chain fatty acids (SCFAs) to strengthen the intestinal lining.
- Immunomodulation to suppress inflammation.
- Encouraging Bacteroidetes growth when F/B ratio is significantly higher than 1 by inhibiting Firmicutes selectively.
- Probiotic strains like Lactobacillus rhamnosus have shown effectiveness in reducing the F/B ratio to correct obesity. Similarly, Saccharomyces boulardii helps reverse obesity by modulating the F/B ratio.
8.3: Controlling excessive Bacteroidetes to Optimize the F/B Ratio
Increased Bacteroidetes in comparison to Firmicutes, can also lead to immunological disorders. These bacteria may adhere to mucosal epithelial cells, triggering inflammatory responses and inducing TNF-alpha production by monocytes and macrophages. Suppressing TNF-alpha by proper probiotics can lead to remission in IBD patients.
SCFAs produced by probiotics promote the secretion of IL-10, an immunosuppressive cytokine, by T-effector cells. Most studies show:
- Bacteroidetes exhibit pro-inflammatory properties via endotoxins.
- Firmicutes display anti-inflammatory effects and help slow IBD progression.
- Selective beneficial probiotics can offset the ill effects created by such dysbiosis.
8.4: Examples of beneficial probiotics:
- L. rhamnosus, L. paracasei, L. salivarius, and S. boulardii are effective when the F/B ratio is high (obesity-related) to bring to the optimal level to correct obesity.
- L. reuteri, L. plantarum, L. casei, L. acidophilus, Bifidobacterium bifidum, and B. lactis help when the F/B ratio is low (IBD-related), to correct the intestinal inflammation.
In conclusion, understanding the F/B ratio is key to managing obesity and intestinal disorders. Multiple mixed strain probiotics along with their immunomodulins can dynamically adjust the microbiota composition through immune modulation, helping either increase or decrease the F/B ratio as needed. Dysbiosis and altered F/B ratios are now also implicated in long COVID, reinforcing the importance of microbiome balance.
Thus, multiple mixed strain probiotic therapy, in combination with proper nutrition and exercise, is a powerful tool in managing obesity, and other intestinal disorders, as an essential therapeutic aid in both preventive and clinical medicine settings.
9) PREVENTION AND CONTROL OF ALLERGIES
Food allergies have become a common public health problem. It can be classified into IgE-mediated, non-IgE-mediated, and mixed IgE-mediated types. In general, clinical practice, IgE-mediated allergies are more common. Approximately 4% of Americans are affected by IgE-mediated food allergies, and the prevalence is as high as 8% in children. Globally, between 10% (800 million) and 40% (3.2 billion) of the population are affected by some form of allergy. Allergies are more prevalent in Western countries than in Asian countries. The “Hygiene Hypothesis” suggests that reduced exposure to microbes in early childhood may contribute to higher allergy rates in Western populations.
An allergy is an adverse immune reaction to normally harmless substances known as allergens. When an allergic individual is exposed to an allergen, their immune system overreacts, causing various symptoms such as sneezing, itchy eyes, wheezing, runny nose, hives, and even life-threatening anaphylaxis. It is important not to confuse food allergies with food intolerances. For instance, lactose intolerance is not a food allergy, while a reaction to casein (a milk protein) is considered an allergy. Allergic reactions typically appear quickly, whereas symptoms of intolerances take longer to manifest.
When someone (with allergies) is exposed to an allergen, their immune system produces specific IgE antibodies against that allergen. This IgE production results from the overactivity of TH2 immune cells. These IgE antibodies bind to mast cells and basophils, priming them for a reaction. Upon re-exposure to the same allergen, the allergen cross-links with

