Prognostic Factors in CNS Metastasis from Colorectal Cancer
Study of Prognostic Factors in Patients with Colorectal Cancer and Central Nervous System Metastasis: A Retrospective Analysis
Daniella DS Arruda1, Mauro DS Donadio2, Maria Leticia G Silva3, Celso AL Mello (in memoriam)1
- Instituto Brasileiro de Controle do Câncer, São Paulo, Brazil
- Centro Paulista de Oncologia – Oncoclinicas, São Paulo, Brazil
- AC Camargo Cancer Center, São Paulo, Brazil
OPEN ACCESS
PUBLISHED: 31 December 2024
CITATION: Arruda, DDS., Donadio, MDS., et al., 2024. Study of Prognostic Factors in Patients with Colorectal Cancer and Central Nervous System Metastasis: A Retrospective Analysis. Medical Research Archives, [online] 12(12). https://doi.org/10.18103/mra.v12i12.6102
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i11.6102
ISSN 2375-1924
ABSTRACT
Background: Metastasis of colorectal cancer to the central nervous system (CNS) is relatively rare but leads to shorter survival for these patients, even with improvements in palliative treatments.
Objectives: The primary objective of this study was to assess the overall survival of patients with brain metastasis from colorectal cancer treated at the A.C. Camargo Cancer Center. The secondary objectives were to identify clinical and epidemiological factors, correlate clinicopathological factors with overall survival, and describe prognostic factors associated with CNS metastasis.
Methods: We retrospectively evaluated patients with metastatic colorectal cancer and CNS involvement treated from January 2002 to August 2020 at a single cancer center.
Results: We identified 46 patients with a median age of 63.9 years; 50% were male, and 80% had a good performance status. The predominant primary site was the left colon (71.7%). Sixty percent were stage IV, 71.7% had previously presented with lung metastasis, and 76.7% had a high volume of lung disease. RAS mutations were detected in 56.5% of the pts. Progression to the CNS occurred after a median of three treatment lines, with a median time of 49.5 months between diagnosis and progression to the CNS. Sixty percent had supratentorial lesions, and 55.6% had only one lesion. After a median follow-up time of 27.2 months, the overall survival was 6.4 months. Surgery and radiation were associated with improved overall survival (HR 0.24, 95% CI 0.09-0.67; p = 0.006; and HR 0.20, 95% CI 0.08-0.5; p = 0.001, respectively).
Conclusion: Our study found that most patients with CNS metastasis experienced progression after three lines of palliative treatment, had prior high-volume lung metastasis, and presented with left-sided tumors. Surgery and radiation may improve overall survival.
Keywords
colorectal cancer, CNS metastasis, palliative care
Introduction
In 2020, colorectal cancer (CRC) accounted for over 1.9 million new cases and 900,000 deaths, making it the third most common cancer and the second leading cause of cancer-related mortality globally. Notable geographical disparities exist in incidence and mortality rates, as well as in trends over time and projected future burdens of CRC across various countries and regions. In Brazil, CRC is the third most common neoplasm. It is estimated that for each year in the 2020-2022 triennium, there will be 20,520 cases of CRC in men and 20,470 in women, corresponding to an estimated risk of 19.63 new cases per 100,000 men and 19.03 per 100,000 women.
In the progression of this tumor, the most common sites of metastasis are the liver, lungs, lymph nodes, and less frequently the central nervous system (CNS). However, the incidence of brain metastasis has increased thanks to advances in the systemic treatment of advanced CRC, particularly following the emergence of anti-EGFR (epidermal growth factor receptor) therapies and anti-vascular endothelial growth factor (anti-VEGF) therapy, which have improved life expectancy and prognosis. Among gastrointestinal tumors, CRC has the highest incidence of brain metastasis, though it remains uncommon, at around 2-3%.
Certain genetic mutations associated with a favorable microenvironment promote the emergence of metastasis in the CNS. It is known that mutations and amplifications of the EGFR are associated with the tumorigenesis of brain metastasis; likewise, KRAS mutations, which are most common in CRC and sometimes have a higher prevalence than in primary tumors. The latter is associated with a worse prognosis. In a case series from the Memorial Sloan Kettering Cancer Center, it was observed that 75% of patients with brain metastasis had mutated RAS, thus presenting an independent risk factor for this complication.
Most metastatic lesions in the CNS from CRC occur in the cerebral hemispheres (48-58%), followed by the cerebellum (18-43%), and less frequently, multiple lesions (23-33%). The most common symptoms include headaches, seizures, motor or visual disturbances, and aphasia, depending on the affected area. Typically, the patient profile includes those whose primary tumor is located in the rectum, with other extracranial lesions, primarily in the lungs, and who develop symptomatic brain lesions approximately 25 months after diagnosis.
The prognosis for patients with metastatic CRC becomes guarded following the emergence of metastasis in the CNS, typically around six months. However, certain factors indicate a poorer prognosis, including: being under 65 years of age, a short interval between the diagnosis of CRC and the onset of metastasis, elevated carcinoembryonic antigen (CEA) levels, the presence of more than three intracranial lesions, and whether the lesions are resectable. Additionally, the number of treatment lines administered before diagnosis serves as an independent risk factor for survival.
Prognostic tools such as recursive partitioning analysis (RPA), diagnosis-specific graded prognostic assessment (DS-GPA), the Radiation Therapy Oncology Group staging system, and the Score Index for Brain metastasis (SIR) are useful in determining prognosis. The RPA categorizes patients into three classes, with progressively worse prognosis. Class 1 includes patients with a Karnofsky Performance Status greater than 70, under 65 years old, with controlled primary tumors and no evidence of extracranial metastasis. Class 3 encompasses those with a KPS less than 70, while Class 2 includes all others. Studies by Fountzilas et al. demonstrated that patients in RPA Class 1 have a median overall survival (OS) of 28.7 months, whereas those in Class 2 average 3.2 months, and Class 3 patients have the shortest survival, around 1.4 months.
Managing these metastasis should involve a multidisciplinary discussion. Literature consistently supports improved survival outcomes when patients receive local treatment; those who undergo combined local therapy have better survival rates compared to those receiving single-modality treatments like surgery or radiotherapy. Despite advancements in systemic and local therapies and imaging methods for early diagnosis, there remains a need for better identification of predictive factors for the development of this complication, as well as other clinical-pathological prognostic factors.
Objectives
The primary objective of this study was to evaluate the OS of patients diagnosed with brain metastasis from CRC treated at the A.C. Camargo Cancer Center. The secondary objectives were to identify clinical, epidemiological, and pathological factors; correlate clinical-pathological factors with OS; and describe prognostic factors associated with CNS metastasis.
Methods
This was a descriptive, analytical, retrospective, single-center study of patients treated at the A.C. Camargo Cancer Center from January 2002 to August 2020. Inclusion criteria were: patients with colorectal adenocarcinoma and brain metastasis treated at the A.C. Camargo Cancer Center during the specified period; age over 18; and sufficient medical record data for analysis. The exclusion criterion was patients with a second primary cancer, except for those classified as in situ. Collected clinical data included: sex, performance status according Eastern Cooperative Oncology Group (ECOG), laterality of the primary tumor, staging at diagnosis, pathological and molecular characteristics (mutations in RAS, BRAF, MSI), use of anti-EGFR therapy, number of lines of systemic treatment, presence and volume of pulmonary metastasis, and characteristics of CNS metastasis (number of lesions, location, treatment administered, and recurrence).
Descriptive statistics were used to summarize absolute and relative frequencies and medians. The chi-square test or Fisher’s exact test was employed to compare binary variables, when necessary. Survival curves were generated using the Kaplan-Meier method, with the log-rank test employed for group comparisons. Multivariate analysis was conducted using Cox regression. SPSS software version 24 was utilized, and statistical tests were considered significant at p < 0.05 with a 95% confidence interval (CI).
Results
From 2002 to 2020, a total of 46 patients with metastatic CRC involving the CNS were included in the study. (Table 1) Of these, 50% were male; 36 patients (78.2%) had good performance status (ECOG 0 or 1); and 33 patients (71.7%) had their primary tumor in the left colon.
| Characteristic | Value |
|---|---|
| Sex | Male: 23 (50%), Female: 23 (50%) |
| T stage | T1 – T2: 4 (9.0%), T3 – T4: 26 (59%) |
| ECOG | 0-1: 36 (78.2%), ≥ 2: 9 (19.6%) |
| Lymph Node stage | N0: 5 (11%), N1: 18 (39.1%), N2: 12 (26.1%) |
| Sidedness | Left: 33 (71.7%), Right: 13 (8.3%) |
| RAS status | Wild: 20 (43.5%), Mutated: 26 (56.5%) |
| Initial staging | I – III: 17 (39.5%), IV: 26 (60.5%) |
| BRAF status | Wild: 10 (21.7%), Mutated: 2 (4.3%), Unknown: 34 (73.9%) |
| Lung metastasis | No: 13 (28.3%), Yes: 33 (71.7%) |
| MSI status | Yes: 1 (2.2%), No: 29 (64%), Unknown: 16 (33%) |
| Lung disease burden | Low (≤2 lesions): 7 (23.3%), High (> 2 lesions): 23 (76.7%) |
| anti-EGFR therapy | No: 16 (34.8%), Yes: 30 (65.2%) |
| Previous lines | 1: 7 (15.6%), 2: 14 (31.1%), ≥3: 23 (51%) |
| CNS location | Supratentorial: 28 (60.9%), Infratentorial: 6 (13%), Both: 12 (26.1%) |
| CNS lesions number | 1: 25 (55.6%), ≥2: 20 (44.4%) |
| CNS recurrence | No: 30 (65.2%), Yes: 16 (34.8%) |
At diagnosis, 26 patients (60.5%) were already metastatic, with 26 (59%) exhibiting tumors that invaded the muscularis propria up to pericolic tissue (T3) or perforated the visceral peritoneum (T4), while 65.2% had lymph node involvement. RAS mutations were present in 26 patients (56.5%), and 30 patients (65.2%) had been treated with some form of anti-EGFR therapy. Only 2.2% of patients showed microsatellite instability. The majority (73.9%) had unknown BRAF status, and among those tested, only 4.3% were mutated.
Most patients (71.7%) had pulmonary metastasis, with 76.7% of these cases categorized as high-volume disease (defined as having two or more lesions, whether bilateral or not). Additionally, 51% developed CNS metastasis after three or more lines of systemic treatment. The most common site for metastasis was the supratentorial region, with 25 patients (55.6%) having a single brain lesion. Notably, 65.2% of patients did not experience recurrence in the CNS.
Exclusive palliative care was provided to 19.6% of patients who were not fit for other procedures. Isolated surgery was performed on 15.2% of patients, while 17.3% underwent surgery followed by radiotherapy. The majority received radiotherapy alone, with 40.9% proposed for whole brain radiation, 27.2% for stereotactic radiation, and 31.8% for radiosurgery.
The median time from CRC diagnosis to progression to CNS involvement was 49.5 months, while the median time from first-line systemic treatment to CNS progression was 33 months. The median time from pulmonary metastasis diagnosis to CNS metastasis diagnosis was 19.3 months, and 15.6% of patients were asymptomatic.
After a median follow-up of 27.2 months, the OS following the diagnosis of CNS metastasis was 6.4 months (1.61 – 11.13 months).
Lymph node involvement (hazard ratio [HR] 1.74, 95% CI 0.79-3.83; p: 0.17), bilateral pulmonary involvement (HR 1.97, CI 0.67-5.8; p: 0.21), ECOG (HR 2.7, 95% CI 0.89-8.19; p: 0.11), and exposure to multiple lines of systemic treatment (HR 1.61, 95% CI 0.76-3.39; p: 0.71) showed no significant correlation with OS.
Surgery was associated with improved OS of 20.1 months, reducing the risk of death by 76% (HR 0.24, 95% CI 0.09-0.67; p: 0.006)
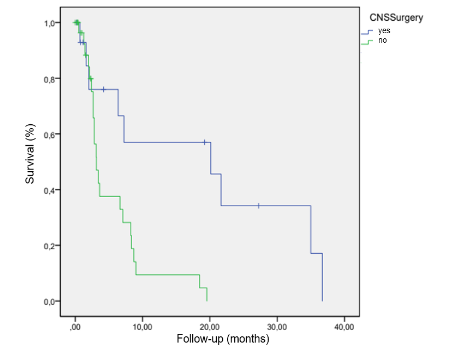
.
Radiotherapy also correlated with increased OS (8.2 versus 2.6 months) with a 80% reduction in the risk of death (HR 0.20; 95% CI 0.08-0.5; p: 0.001)
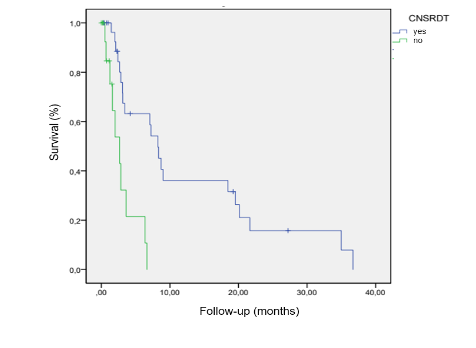
.
In multivariate analysis, both surgery and radiotherapy emerged as independent factors associated with increased OS (HR 0.29, 95% CI 0.10-0.86; p: 0.002; and HR 0.17, 95% CI 0.06-0.51; p: 0.025, respectively).
Discussion
Our study described the profile of patients associated with the development of CNS metastasis at the A.C. Camargo Cancer Center between 2002 and 2020. Most patients had a primary tumor on the left side, presented with high-volume pulmonary metastasis, and had undergone more than two lines of prior treatment, with CNS lesions primarily located in the supratentorial region. The median survival of these patients was slightly over six months, and both surgery and radiotherapy were linked to improved overall survival.
The majority of our patients had a good performance status (ECOG 0 or 1) at the time of cancer diagnosis, consistent with findings from the study by Nozawa. In contrast, the Brazilian study by Bonadio et al reported a higher percentage of patients with poorer prognosis, likely due to the nature of public healthcare services, where patients often present with more advanced disease.
Most patients in our study had primary tumors located in the left colon, similar to findings in other studies. This may be related to the fact that CNS metastasis from left-sided tumors can develop at an earlier stage due to the vascular drainage of the rectum, which directly connects to the internal iliac vein, providing direct access to the lungs and CNS. In contrast, drainage from the right colon occurs via the mesenteric vein. Furthermore, the poorer prognosis associated with right-sided colorectal cancer may result in patients succumbing to the disease before CNS metastasis appears, which typically occurs at a later stage.
Indeed, the majority of our patients were diagnosed with more advanced disease, whether metastatic, as seen in the studies by Bonadio et al and Chahine et al, or presenting with T3 and T4 tumors. Additionally, nearly 90% of our population had lymph node involvement, which was more pronounced than what is reported in the literature.
Regarding mutation status, our study aligns with a research conducted in Melbourne involving 148 patients, which primarily aimed to evaluate pulmonary metastasis. A sub-analysis of that study found a higher incidence of RAS mutations in patients with brain metastasis. Similarly, another study at the Memorial Sloan Kettering Cancer Center, which evaluated 441 patients with RAS mutations, revealed that these patients had a greater incidence of pulmonary, bone, and cerebral metastasis. By the end of the follow-up period, 29 cases of brain metastasis were identified in mutated patients compared to 9 cases in wild-type patients.
The association between CNS metastasis and pulmonary metastasis observed in our study is well-supported in the literature, with multiple studies, including two meta-analyses with larger populations, showing similar findings. Generally, a progression of metastasis has been noted, beginning with liver and pulmonary involvement, followed by bone and CNS metastasis, likely due to the vascular dissemination route where tumor cells initially enter the portal and pulmonary circulation before reaching the brain.
After the diagnosis of metastatic disease to the central nervous system, the OS of our patients was 6.4 months, which is consistent with data from Imaizumi and colleagues. In contrast, the study by Bonadio et al. involving 247 patients reported a poorer survival rate, likely due to the worse performance status of their population, with nearly half having an ECOG score of 3 or 4 and over half already classified as stage IV at diagnosis.
Despite the generally poor prognosis, locoregional treatment of CNS metastasis from colorectal cancer can provide benefits for symptom control and survival. The advantages of surgery and radiotherapy that we observed are also documented in other series. A meta-analysis conducted by Mege et al., which included over 1,800 patients with similar characteristics, indicated an overall survival of approximately 16 months for those who underwent surgery. However, patients with brain metastasis often present worse performance status, more lines of prior treatments, and less likelihood of being candidates for neurosurgery, which was the case for nearly 20% of our series.
The roles of radiotherapy and surgery for brain metastasis in colorectal cancer prognosis are well-established in the literature. Damiens et al. evaluated 48 patients with similar demographic characteristics and demonstrated a global survival benefit of 10 months for those exposed to radiotherapy, regardless of the modality.
Our study has several limitations that must be acknowledged. It is a retrospective, single-center study with a small sample size and no control group. Additionally, there was no central and independent review of the histopathological and imaging reports. Nonetheless, it holds significance for addressing a still underexplored area with growth tendency.
There is still much to learn regarding patients at risk for developing such metastasis, which could improve screening and lead to earlier treatment and enhanced quality of life for these patients.
Conclusion
In our study, the majority of patients with CNS metastasis experienced progression in the CNS after three lines of palliative treatment, had high-volume prior pulmonary metastasis, and had their primary tumor located in the left colon. The median time between diagnosis and the onset of CNS lesions was over four years. Our data suggest that patients with CRC exhibiting this profile should be monitored more closely for potential CNS metastasis, as timely treatment can reduce mortality. However, our findings need to be validated in a larger patient cohort.
Declarations
Authors’ Contribution: All authors have made a significant contribution to this manuscript, have seen and approved the final manuscript, and agree to its submission to the Journal.
Conflict of interest: none.
Financial support: none to declare.
Funding: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Warnings: The opinions expressed in the report presented are those of the authors and do not necessarily represent the official position of the institution to which they belong.
Acknowledgement: This study was approved by AC Camargo Cancer Center IRB: registry 092541/2020.
References:
- Ervik M, Lam F, Laversanne M, et al. Global Cancer Observatory: Cancer Over Time. Available online: https://gco.iarc.fr/overtime (accessed on 23 October 2024).
- Ferlay J, Ervik M, Lam F, et al. Global Cancer Observatory: Cancer Today. Available online: https://gco.iarc.fr/today (accessed on 23 October 2024).
- Ferlay J, Laversanne M, Ervik M, et al. Global Cancer Observatory: Cancer Tomorrow. Available online: https://gco.iarc.fr/tomorrow (accessed on 23 October 2024).
- Estimativa 2020: incidência de câncer no Brasil / Instituto Nacional de Câncer José Alencar Gomes da Silva – Rio de Janeiro: INCA, 2019.
- Christensen T, Spindler K, Palshof J, et al. Systematic review: brain metastases from colorectal cancer—Incidence and patient characteristics. BMC Cancer, 2016;1:16:260.
- Grothey A, Sargent D, Goldberg R, Schmoll H-J. Survival of patients with advanced colorectal cancer improves with the availability of fluorouracil-leucovorin, irinotecan, and oxaliplatin in the course of treatment. J. Clin. Oncol. 2004;22:1209–14.
- Zang Y, Gu X, Xiang J, et al. Brain Metastases from Colorectal Cancer: Microenvironment and Molecular Mechanisms. Int J Mol Sci. 2012;13(12):15784-800.
- Ogino S, Shima K, Meyerhardt J, et al. Predictive and prognostic roles of BRAF mutation in stage III colon cancer: results from intergroup trial CALGB 89803. Clin Cancer Res. 2012;18:890-900.
- Yaeger R, Cowell E, Chou J, et al. RAS mutations affect pattern of metastatic spread and increase propensity for brain metastasis in colorectal cancer. Cancer 2015;121:1195-203.
- Qiu M, Hu J, Yang D, et al. Pattern of distant metastases in colorectal cancer: a SEER based study. Oncotarget 2015;6:38658-66.
- Mongan J, Fadul C, Cole B, et al. Brain metastases from colorectal cancer: Risk factors, incidence, and the possible role of chemokines. Clin. Colorectal Cancer. 2009;8,100–5.
- Gaspar L, Scott C, Rotman M, et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys. 1997;37(4):745-51.
- Fountzilas C, Chang K, Hernandez B, et al. Clinical characteristics and treatment outcomes of patients with colorectal cancer who develop brain metastasis: a single institution experience. J Gastrointest Oncol. 2017;8(1):55-63.
- Cagney D, Martin A, Catalano P, et al. Incidence and prognosis of patients with brain metastases at diagnosis of systemic malignancy: a population-based study. Neuro Oncol. 2017;19(11):1511-21.
- Nozawa H, Ishihara S, Kawai K, et al. Brain Metastasis from Colorectal Cancer: Predictors and Treatment Outcomes. Oncology. 2017;93(5):309-14.
- Bonadio R, Freitas G, Batista D, et al. Epidemiology and Outcomes of Patients With Brain Metastases From Colorectal Cancer—Who Are These Patients? Clin Colorectal Cancer. 2021;20(3):e195-e200.
- Teixeira B, Rider A, Holbrook T, et al. Overall cancer survival inequalities in the state of São Paulo: A comparison between public and private systems. JCO 40, 6525-6525(2022). doi: 10.1200/JCO.2022.40.16_suppl.6525
- L. Bittencourt Hinrichsen, M.T. Tsukazan, A. Vigo, et al. P12.03 Public Versus Private Care Patients Treated in the Same Hospital. Are the Patients the Same? Journal of Thoracic Oncology, Volume 16, Issue 3, S325. doi: 10.1016/j.jtho.2021.01.499
- Tin Tin S, Elwood JM, Lawrenson R, et al. Differences in Breast Cancer Survival between Public and Private Care in New Zealand: Which Factors Contribute? PLoS One. 2016 Apr 7;11(4):e0153206. doi: 10.1371/journal.pone.0153206.
- Huerta L, Manrique A, Szafranska J, et al. Brain metastases in colorectal cancer: prognostic factors and survival analysis. Int J Colorectal Dis. 2018;33(11):1517-23.
- Merrie A, Phillips L, Yun K, et al. Skip metastases in colon cancer: assessment by lymph node mapping using molecular detection. Surgery. 2001 Jun;129(6):684-91. doi: 10.1067/msy.2001.113887.
- Read TE, Mutch MG, Chang BW, et al. Locoregional recurrence and survival after curative resection of adenocarcinoma of the colon. J Am Coll Surg. 2002 Jul;195(1):33-40. doi: 10.1016/s1072-7515(02)01224-3.
- Warwick R and Page R. Resection of pulmonary metastases from colorectal carcinoma. Eur J Surg Oncol. 2007 Dec;33 Suppl 2:S59-63. doi: 10.1016/j.ejso.2007.09.018.
- Chahine G, Ibrahim T, Felefly T, et al. Colorectal cancer and brain metastases: An aggressive disease with a different response to treatment. Tumori. 2019;105(5):427-33.
- Meguid R, Slidell M, Wolfgang C, et al. Is there a difference in survival between right- versus left-sided colon cancers? Ann Surg Oncol. 2008 Sep;15(9):2388-94. doi: 10.1245/s10434-008-0015-y.
- Asghari-Jafarabadi M, Wilkins S, Plazzer JP, et al. Prognostic factors and survival disparities in right-sided versus left-sided colon cancer. Sci Rep. 2024 May 29;14(1):12306. doi: 10.1038/s41598-024-63143-3.
- Bourakkadi Idrissi M, El Bouhaddouti H, Mouaqit O, et al. Left-Sided Colon Cancer and Right-Sided Colon Cancer: Are They the Same Cancer or Two Different Entities? Cureus. 2023 Apr 14;15(4):e37563. doi: 10.7759/cureus.37563.
- Mangone L, Pinto C, Mancuso P, et al. Colon cancer survival differs from right side to left side and lymph node harvest number matter. BMC Public Health. 2021 May 12;21(1):906. doi: 10.1186/s12889-021-10746-4.
- Janssens K, Fransen E, Van Camp G, et al. A Belgian Population-Based Study Reveals Subgroups of Right-sided Colorectal Cancer with a Better Prognosis Compared to Left-sided Cancer. Oncologist. 2023 Jun 2;28(6):e331-e340. doi: 10.1093/oncolo/oyad074.
- Tie J, Lipton L, Desai J, et al. KRAS Mutation Is Associated with Lung Metastasis in Patients with Curatively Resected Colorectal Cancer. Clin Cancer Res. 2011;17(5):1122-30.
- Müller S, Köhler F, Hendricks A, et al. Brain Metastases from Colorectal Cancer: A Systematic Review of the Literature and Meta-Analysis to Establish a Guideline for Daily Treatment. Cancers (Basel). 2021 Feb 21;13(4):900. doi: 10.3390/cancers13040900.
- Imaizumi J, Shida D, Narita Y, et al. Prognostic factors of brain metastases from colorectal cancer. BMC Cancer. 2019;19(1):755.
- Mege D, Sans A, Ouaissi M, et al. Brain metastases from colorectal cancer: characteristics and management. ANZ J Surg. 2018;88(3):140-5.
- Damiens K, Ayoub J, Lemieux B, et al. Clinical features and course of brain metastases in colorectal cancer: an experience from a single institution. Curr Oncol. 2012;19(5):254-8.

