Pulmonary Rehabilitation for COPD: History and Future Trends
History, Current Initiatives, and Future Directions of Pulmonary Rehabilitation for the Patient with Chronic Obstructive Pulmonary Disease
Antarpreet Kaur, MD1,2, FCCP; Hnin Oo2, MD; Richard L ZuWallack, MD, FCCP1,2
- Division of Pulmonary Critical Care and Sleep medicine, Trinity Health of New England, Hartford CT, USA
- University of Connecticut Medical center, Farmington, CT, USA
OPEN ACCESS
PUBLISHED 31 October 2024
CITATION Kaur, A., Oo, H., et al., 2024. History, Current Initiatives, and Future Directions of Pulmonary Rehabilitation for the Patient with Chronic Obstructive Pulmonary Disease. Medical Research Archives, [online] 12(10). https://doi.org/10.18103/mra.v12i10.5993
COPYRIGHT © 2024 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI https://doi.org/10.18103/mra.v12i10.5993
ISSN 2375-1924
ABSTRACT
Pulmonary rehabilitation (PR) is a comprehensive intervention designed to improve the physical and psychological condition of patients with chronic respiratory disease. Despite proven benefits shown in multiple studies, uptake of PR remains dismal. This comprehensive review highlights the history of PR from the 1960s through the 2020s. This is followed by review of evidence and discussion on approaches to increasing referral and uptake of PR, the use of technology to provide virtual PR as an alternative or adjunct to traditional PR, PR in the management of the COPD patient particularly in the post exacerbation period and a brief review of PR approaches in developing countries.
Keywords
- Pulmonary Rehabilitation
- Chronic Obstructive Pulmonary Disease
- Virtual Rehabilitation
- Healthcare Utilization
- Patient Outcomes
1. Introduction
In order to fully appreciate recent developments in the science and implementation of comprehensive pulmonary rehabilitation (PR), it is first necessary to know what it is and have an idea of its relatively short history. The following is an accepted definition of PR dating back to a 2013 American Thoracic Society and European Respiratory Society Statement, subsequently endorsed in a 2021 American Thoracic Society workshop: “[PR is] a comprehensive intervention based on a thorough patient assessment followed by patient-tailored therapies that include, but are not limited to, exercise training, education, and behavior change, designed to improve the physical and psychological condition of people with chronic respiratory disease and to promote the long-term adherence to health-enhancing behaviors.”
What follows is a brief review of the history of PR from the 1960’s through 2020, emphasizing its transition from an intervention that made sense to astute clinicians but had no evidence base backing it up to its current recognition as a “gold standard” in the treatment of chronic respiratory disease and incorporation in practically all clinical guidelines on COPD. The increase in peer-reviewed papers on PR – including its implementation, scientific underpinnings, outcomes, and worldwide recognition as a therapeutic modality – since the 1960’s has been dramatic, as evidenced in Figure 1.
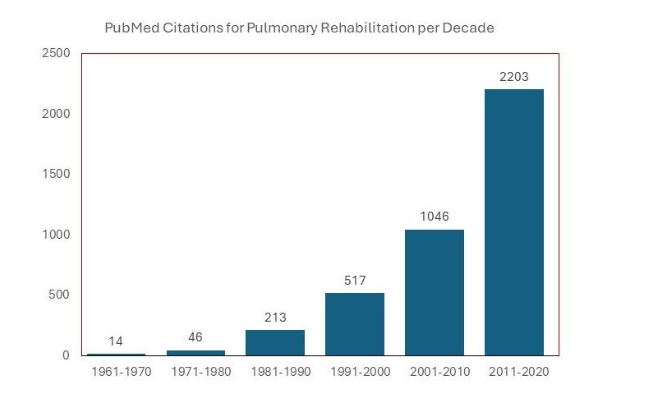
Following this review of the history of PR, we will highlight some of its recent advances, challenges and initiatives for COPD. These include: 1) Approaches to increase referral and uptake of this intervention by providing it in home or community settings; 2) The trend towards promoting less-intense and regular physical activity in addition to standard exercise training; 3) Using technology to provide virtual PR either as a stand-alone or hybrid model; 4) Use of smart phone technology to provide some components of PR; 5) Incorporation of PR in the integrated model of COPD management; 6) PR in the post exacerbation period and discussion on whether this represents the holy grail in PR outcomes and 7) A brief review of PR approaches in developing countries.
2. Key Dates in the Development, Science, and Implementation of Pulmonary Rehabilitation as a Distinct Therapeutic Entity
1960’s:
Astute clinicians in this decade recognized that the commonly utilized reductionist approach to treating people with advanced COPD, such as treating them only with bronchodilators, was not as effective as holistic and comprehensive approaches to management. To meet this challenge, Thomas L Petty (1932 -2009), a respirologist at the University of Colorado who could be considered the father of PR, organized a multidisciplinary pulmonary rehabilitation program to treat individuals with COPD. At the time, this treatment approach did not have evidence-based science backing; rather it reflected the perception by this physician and other clinicians of the systemic effects and comorbid conditions that often drive COPD morbidity and mortality. Some of the therapies in this program included patient education, breathing techniques, structured exercise, supplemental oxygen therapy, bronchial hygiene, and individualized pharmacologic therapy. He subsequently published on this in 1969, reporting (without the backing of a randomized, controlled trial) that it led to marked subjective involvement in symptoms and objective improvement in exercise tolerance and subsequent hospitalizations.
1970’s:
Pulmonary rehabilitation gained traction among some clinicians, and in 1974 a committee of the American College of Chest Physicians came up with the following definition: “art of medical practice, wherein an individually tailored, multidisciplinary program is formulated through accurate diagnosis, therapy, emotional support and education to stabilize or reverse both physiopathological and psychopathological manifestations of pulmonary diseases. Such a program must attempt to return the patient to the highest possible functional capacity allowed by the handicap and overall life situation.” Note that this first definition of PR included the word, “art”. While at that time its supporting evidence base was minimal, this descriptor plagued PR clinicians for decades until the science caught up with the practice.
1980’s:
A clearer understanding of the constituents of a comprehensive PR program were clarified. These included, “proper patient selection, initial assessment to determine needs, development of appropriate goals, development of components of care, assessment of patient progress, and arrangement for long-term follow-up.” Adding to this was the idea that the treatment should be tailored to the needs of the individual respiratory patient. These components are still in effect today, although – beginning in this decade – the importance of exercise training was emphasized.
1990’s:
This decade saw a flurry of activity, both in scientific studies supporting its effectiveness and in official statements recommending its use. Leading the charge of new science, was a paper by Casaburi and colleagues, Reductions in Exercise Lactic Acidosis and Ventilation as a Result of Exercise Training in Patients with Obstructive Lung Disease, a physiologic study which – despite its small number of subjects – demonstrated that individuals with COPD can have physiological improvement from exercise training. Prior to this, a prevailing thought that patients were ventilatory “pump-limited” and could not demonstrate such a benefit. Other studies showed that PR led to increased functional exercise capacity and functional status, exercise endurance training in moderate to severe COPD can reduce exercise-induced lactic acidosis and improve skeletal muscle oxidative capacity, and PR can reduce dyspnea. Supporting these smaller studies was the first randomized trial of PR by Andrew Ries and colleagues, which demonstrated that a comprehensive, 8-week outpatient PR program (compared to education alone) led to improvements in exercise capacity, dyspnea, muscle fatigue, and self-efficacy for walking. These studies and others led to the 1999 publication, Pulmonary rehabilitation-1999: The Official Statement of the American Thoracic Society. Other official statements in this decade included Pulmonary rehabilitation in chronic obstructive pulmonary disease (COPD) with recommendations for its use. Report of the European Respiratory Society Rehabilitation and Chronic Care Scientific Group (1992), and Selection criteria and programmes for pulmonary rehabilitation in COPD patients (1997).
2000 – 2010:
In this decade, PR became a standard of care for COPD, and additional statements and guidelines solidified the rationale and organizational aspects of this intervention. Among these were the joint American Thoracic Society / European Respiratory Society Statement on Pulmonary Rehabilitation (2006), Clinical Competency Guidelines for Pulmonary Rehabilitation Professionals, and Pulmonary Rehabilitation Joint ACCP/AACVPR Evidence-Based Guidelines (2007). Also published in this decade was the largest randomized trial of PR to date, Results at 1 Year of Outpatient Multidisciplinary Pulmonary Rehabilitation: a Randomised Controlled Trial. In 2001, of considerable importance, the very influential Global Initiative for COPD (GOLD) document included PR as an established treatment for COPD in a 2001 publication, and their 2003 update placed it in their algorithm for the recommended management for this disease. Other advances included the importance of self-efficacy training as a stand-alone intervention or component of PR, an appreciation of the importance of increased physical activity as an outcome in PR, the concept of bringing PR into the home and community settings, cost effectiveness of PR, the first recognition of the benefits of PR in the post exacerbation period, and the idea that PR is a major component in the integrated care of the complex COPD patient.
2010-2020:
A review of the effectiveness PR by McCarthy and colleagues, while not the first systematic review on this intervention, was the first to state that based on what they considered unequivocal positive evidence put forth in their article: “It is our opinion that additional RCTs comparing pulmonary rehabilitation and conventional care in COPD are not warranted.” Authors of an extensive survey that outlined similarities and (considerable) differences among PR programs worldwide stressed the need for implementing processes and performance metrics, establishing international benchmarking, and creating international standards based on evidence and best practice. A systematic review by Puhan and colleagues reported that PR – in addition to already established improvements in exercise tolerance and health status – also may reduce health care utilization and even mortality risk, although the quality of evidence was not particularly high.
3. Developments in Pulmonary Rehabilitation since 2020
A. INCREASING REFERRAL AND UPTAKE OF PULMONARY REHABILITATION
Despite its proven benefits, PR utilization remain suboptimal on a global scale with some heterogeneity in its implementation. A scoping review of international studies of 10 developed countries report a referral rate of 35% in over 90% of studies. A recent nationwide study in France showed an 8.6% of PR uptake 90 days after an admission for acute COPD exacerbation. In the United States, studies among Medicare beneficiaries with COPD showed an uptake ranging from 1.9% to 4%. The percentage of Medicare beneficiaries with COPD receiving PR increased from 2.6% in 2003 to 3.7% in 2012 while in a subsequent study showed an increase from 2.5% in 2013 to 4.0% in 2019. Therefore, while there has been some increase in uptake of PR over the years, the numbers remain quite dismal. To explore potential solutions, we must first dissect the potential barriers. The barriers can be broadly classified into factors related to the patient and the surrounding environment, the healthcare professional (HCP) and systemic factors.
Patient and healthcare professional related factors
Uptake of PR must start with initial referral of eligible patients by the HCP and acceptance by the patient. Lack of awareness of PR from both the provider and the patient plays a big role in suboptimal referral to PR. An official ATS/ERS statement by Rochester et al published in 2015 highlighted the lack of knowledge of PR in HCPs and the need for education starting from the trainee stage. Influence of the provider plays an importance role in patient uptake of PR. HCPs therefore must not only know about PR but also be able to inform the patients about the benefits of PR. A study by Bhatt et al showed that patients who had a pulmonologist as a sole provider or co-managing provider was more likely to be referred to PR. However, primary care providers and other allied health professionals play a major role in management of patients with chronic respiratory conditions such as COPD. Therefore, optimizing knowledge of PR and perceived benefits of PR among all such HCPs is the first step in increasing PR utilization. Patient buy-in is the next essential step. An international online survey administered by Rochester et al published in 2018 showed that out of 1685 respondents, nearly 25% have never heard of PR. Limited knowledge of PR, lack of perceived benefits and worries about exercise tolerance may all contribute to poor uptake of PR from a patient’s perspective. These factors could potentially be addressed with patient education. Other factors associated with decreased PR uptake may prove more challenging to remedy. Active cigarette smokers and patients with co-morbidities such as depression have been shown to have lower PR uptake. These patient factors may be harder to address as this requires getting to the root of underlying general non-adherence and addressing underlying mental health issues which affect many of our patients with chronic respiratory disease. Even more challenging to address are disparities among PR availability and uptake which have been demonstrated in multiple studies. Lower socioeconomic status, older age and black race have been shown to be associated with decreased PR uptake. We start this discussion under the heading of “patient factors” as it involves patient characteristics but in fact, there are complex interaction involving many systemic inadequacies in our healthcare system which often fails to cater to our vulnerable patient population. Potential solutions will require a multidisciplinary approach involving patients, HCPs, payers, advocacy groups and policy makers.
Systemic factors
Suboptimal reimbursement for PR is a major limiting factor in the referral and expansion of PR programs. In 2010, Medicare changed its reimbursement policy for PR with the introduction of a bundle payment code (G0424) and also established a payment rate of $50 per unit of G0424 which is approximately half of the reimbursement for cardiac rehabilitation. This certainly does not reflect the complexity of PR catering to a variety of patients with chronic respiratory disease. Efforts must be made to increase awareness in our payers and policy makers of the benefits of PR in reducing cost in the long run so that PR programs can be reimbursed appropriately to ensure sustainability and expansion.
On the topic of expansion, poor access to PR centers is certainly a major factor in suboptimal PR uptake. A study of Medicare beneficiaries with COPD in 2018 by Malla et al revealed that there is one PR center for every 6030 individuals with COPD. Overall, the proportion of patients who had PR centers available within 10 miles was 61.5%. However, access in rural areas is much more limited with PR center availability within 10 miles was 11.6% in this population. Compared to metropolitan areas, rural areas are 95% less likely to have access to PR within 10 miles. This is important to note as a previous study by Spitzer et al showed that patients living more than 10 miles or 30 minutes away from a PR center were half as likely to initiate PR. This is unsurprising as transportation issues are often cited as a major limitation in PR utilization. This again highlights a major disparity in access to PR. Novel solutions such as home PR and telemedicine have been proposed which we will discuss in another section of this paper. However, for this to be successful, we will also need to ensure some standardization in the PR process.
We can attempt to establish more PR centers or attempt novel therapies, however, one potential barrier to effective PR would be lack of standardization of PR and lack of trained providers who can provide PR. A survey completed by 430 centers in over 40 countries showed that there was heterogeneity among different centers including the settings, case mix and composition of the team among other differences. ATS published an official society workshop report defining modern PR which highlighted, among other important issues, the essential components of PR and existing certification processes. There should be some standardization and quality control of PR delivered in varying forms to ensure patients receive the intended benefits.
Potential solutions
To increase uptake of PR, above barriers must be addressed. An official ATS/ERS policy statement was published in 2015 highlighting specific actionable plans to increase utilization of PR. This provides a good framework for increasing referral and uptake of PR. We will highlight some items from this along with suggestions for the future. Pulmonary rehabilitation currently is taught sporadically at both the undergraduate and post-graduate levels. While some post-graduate courses are available, they are not mandatory or standardized. Educational authorities from varying disciplines including primary care providers and allied health professionals, including respiratory therapists and physiotherapists, should develop standardized curricula to ensure PR is a mandatory part of training. For those in clinical practice, societies such as ATS/ERS should offer training sessions, CMEs and other educational material to HCPs. Awareness campaigns should go beyond the pulmonologists as other providers such as primary care providers remain on the frontline in caring for patients who would benefit from PR.
Patient awareness and buy-in could be achieved via dissemination of educational material by societies such as ATS/ERS and advocacy groups in various settings such as community clinics, hospitals, online, media campaigns and partnership with pharmaceutical companies to promote the use of PR. A systematic review of interventions to promote referral, adherence and uptake of PR by Watson et al found that patients held score cards (used to empower patients on their knowledge of COPD), staff education and quality improvement initiatives increased referral to PR. Of note, in patients with underlying anxiety or depression, one RCT found that cognitive behavioral therapy along with PR improved adherence. This highlights the need for additional support for patients with mental health issues. Increasing awareness and addressing co-morbidities are important steps. However, if we do not address systemic problems and disparities which exist, access will remain an issue. Adequate funding is needed to sustain and expand PR programs. This should start with payer awareness of the true cost of PR and re-framing PR as a long-term cost-saving measure. For example, the guideline highlights that in the UK, PR stands at a favorable position in terms of cost saving as compared to other treatments for COPD. In the United States, a study by Mosher et al published in 2022 showed that patients participating in PR after hospitalization for COPD had a potential for net savings of $5721/patient over the course of a lifetime and $1-1.25 billons of savings for Medicare annually. Payers should be made aware of studies such as this and at the local level detailed costs of PR should be communicated to the payers. Collaboration among patients, PR providers, local government, professional societies and advocacy groups is needed to ensure adequate funding for PR centers and ensuring access where this is needed. Adequate funding will ensure sustainability of current programs, training and reimbursement of members of the PR team and increasing programs to improve access.
Data on disparities should be made available to local government and policy makers so that these issues can be addressed. For example, audits of local numbers could be performed with a goal to open a PR center where there is a need. There should also be standardization of the quality of PR programs as previously highlighted. Professional societies should publish expected standards which all PR programs should meet. In addition, patient outcomes at each center should be monitored and reported. A central body should certify all PR programs and members of the team delivering PR should require standardized certification as well. This becomes more challenging as more novel modalities of PR become available. This is one way to address the issue of poor access. Evidence for these modalities is emerging with some favorable outcomes as we will highlight shortly. Especially in rural areas where PR centers are currently not readily available, telemedicine and home-PR are acceptable alternatives while we work to improve access to formal PR centers. We must, however, strike a good balance of improving access by embracing these modalities while maintaining quality standards.
We have highlighted some barriers and potential solutions to increasing referral and uptake of PR. Work has been ongoing and much more need to be done. In order to coordinate these efforts, we agree with the proposal by Carolyn Rochester in the review of barriers to PR that a National PR Action Plan similar to the National COPD Action Plan in 2016 should be implemented. This can be carried out in each country with a goal of having standardized, collaborative efforts among all stakeholders.
B. PROMOTING PHYSICAL ACTIVITY IN ADDITION TO STANDARD EXERCISE TRAINING IN PR
For the purposes of this discussion, we use an accepted definition of physical activity and exercise, with the former referring to bodily movement produced by skeletal muscles that results in energy expenditure, and the latter referring to a generally structured subset of physical activity, undertaken with the goal of achieving physical fitness. Compared with age matched controls, patients with COPD have diminished activity that begins early in the disease and worsens as it progresses. Since its beginnings, exercise training with the goal of increasing functional exercise performance has been a prominent goal of PR. In fact, the authors of a prominent systematic review defined PR as “… exercise training for at least four weeks with or without education and/or psychological support.” This reductionist approach is a bit short-sighted, since a 2021 American Thoracic Society Workshop report based on Delphi process feedback by experts in the discipline listed other program components, such as structured education and self-management strategies. In a PR context, exercise training improves not only functional and maximal exercise performance, but also respiratory specific health status, including domains of symptoms, activity, impacts (SGRQ, and dyspnea, fatigue, mastery, and emotion (CRQ).
Although the importance of regular physical activity in well-being and survival for all people has been known for some time, it received more attention for COPD when studies appeared that demonstrated the relationship between self-reported physical activity and subsequent health care use and mortality. For example, Garcia-Aymerich and colleagues reported in 2006 that individuals from Copenhagen with lung function abnormalities consistent with COPD followed for 20 years who had very low self-reported activity had significantly higher mortality and more hospitalizations over this period than those who reported low, moderate, and high activity. This relationship was present even after controlling for potential confounders. Although causality cannot be determined, this relationship was supported by a host of other studies.
The beneficial effects of higher levels of physical activity in COPD has naturally led to considerable interest in increasing its levels using PR as the modality. However, trials of PR that incorporated physical activity as an outcome have had mixed results, with some showing a beneficial effect and some showing no significant effect. Interpretation of these results is hampered by the heterogeneity of PR approaches. In particular, it appears that the intervention to raise levels of physical activity takes longer than that needed to increase exercise and requires a greater emphasis on behavioral intervention. In this sense, increases in exercise capacity resulting from PR are only permissive of increased activity.
The importance of motivation was emphasized in a non-PR randomized, controlled trial aimed at increasing activity in individuals with COPD. The intervention group received motivational interviews, advice on walking in outdoor public spaces, and some feedback opportunities including pedometers, calendars, brochures, a website, text messages, and walking groups. The control group received usual care. Improvements in activity were noted in only those in the treatment group who exhibited increased motivation, but not in the intent to treat analysis. This underscores the difficulty in changing longstanding maladaptive behaviors.
An interesting research question published in a recent task force include its aspects, involves separate characterizations of its amount and intensity. Amount can be estimated as the number of steps per day, while intensity can be assessed as the amount of time above a certain metabolic equivalent (MET) threshold (either expressed as total time or number of bouts). Although further research is indicated, the consensus among experts was that both factors are important.
C. USING TECHNOLOGY TO PROVIDE VIRTUAL PULMONARY REHABILITATION EITHER AS A STAND-ALONE OR HYBRID MODEL
Pulmonary rehabilitation traditionally is performed in a PR center which is hospital-based or at times, at a stand-alone center. However, as we have previously highlighted, access to one of these centers is limited particularly in rural areas. One solution to this is to leverage information and communication technologies to deliver PR remotely to patients’ homes or close to home (telerehabilitation). This can be done via various modalities including telephone or web-based platforms delivered asynchronously or synchronously with real-time videoconferencing to the home or an intermediary hub such as a local gym. Hybrid models which involve assessment and care at both a traditional center and PR at home have also been tried. Do these models work just as well as traditional PR? Could this then help solve the issue of limited access? Let us first explore the evidence.
Many studies and subsequent systematic reviews have been published on this subject. A Cochrane review by Cox et al looked at 15 studies with 5 different modes of telerehabilitation and concluded that telerehabilitation achieved similar outcomes as center-based PR. Two of the studies looked at a “hub-and-spoke” model where PR was delivered via videoconferencing from a traditional PR center to a local health center. A more recent systematic review and metanalysis by Uzzaman et al looked strictly at home-PR (defined as PR delivered at home and not including those done at a satellite centers) vs usual care or traditional center-based PR. Analysis of six trials showed that functional exercise capacity (6MWT being the most used standardized testing method) was significantly improved in the home-PR group as compared to the usual care group. Health related quality of life using standardized scales was also significantly improved in the home PR group as compared to usual care group. There was no statistically significant difference between home PR and center-based PR in either outcome. There seems to be clear evidence that telerehabilitation is better than usual care (without PR) and at least equivalent to traditional PR. The ATS clinical practice guideline recommends that for adults with stable chronic respiratory disease, patients should be offered a choice of center-based PR or telerehabilitation (strong recommendation, moderate-quality evidence). Note should be made that although more evidence is emerging, the studies are quite heterogenous. Furthermore, all studies involve an in-center assessment prior to the start remote intervention. The studies therefore do not full reflect a fully remote program and perhaps the in-person assessment to determine eligibility for telerehabilitation may need to an initial part of all remote programs. The ATS guideline also states that the current body of evidence for telerehabilitation is not as extensive as center-based PR which should still be offered as first line with telerehabilitation as an alternative. British Thoracic Society (BTS) clinical statement on PR also caution that further research is needed on telerehabilitation, and it should only be offered to those who declined or failed center-based PR. It was also noted that most of the studies are on patients with COPD and further research is needed in patients with other chronic respiratory disease.
What can we take away from the available evidence and guidelines? One could clearly argue that telerehabilitation is better than no rehabilitation at all. However, we must ensure standardization of these programs. With wide availability, we must be cautious of third party telerehabilitation services that may not include all necessary components of PR. One solution is to develop an official certification with a published list of certified telerehabilitation programs. Patient selection will also be key as there are potential safety issues in certain patient populations, e.g. an elderly patient on home O2, performing high intensity exercise at home alone. Caution should also be taken in comparing telerehabilitation with traditional center-based rehabilitation as promotion of telerehabilitation could result in funding taken away from center-based PR which remains the gold-standard. Rather than being a direct comparison, telemedicine should complement center-based PR especially where access is limited.
D. USING SMART PHONE TECHNOLOGY TO PROVIDE SOME COMPONENTS OF PULMONARY REHABILITATION
A discussion on telerehabilitation would not be complete without a brief discussion on smart phone technology. Smart phones have very much become a part of everyday life, and we would be remiss if we did not leverage this in helping our patients access some components of PR on this platform. A recent systemic review and metanalysis by Chung et al looked at clinical efficacy of mobile app-based self-directed PR vs traditional PR. Ten studies were included in the systemic review and nine included in the metanalysis. Studies were small with the largest study population being 343, and some discrepancies in baseline characteristics and outcomes. Different apps were included in the studies with many using new applications and studies from the UK using “myCOPD”- an app approved by the National Health Services. There was heterogeneity in the content of the apps; some apps provided education and symptoms management programs while others provided exercise programs only. Although not statistically significant (apart from CAT score), results were trended towards improved exercise capacity, symptoms scores, quality of life outcomes and rehospitalization in the app-based group. Apps can certainly be used to deliver some components of PR such as education and potential for individualized exercise plans based on built in pedometers in smartphones. We will reiterate that apps, as with any other mode of telerehabilitation, need a process for certification and standardization. This is an emerging platform for telerehabilitation for which more evidence is needed.
E. UTILITY OF PULMONARY REHABILITATION IN THE INTEGRATED CARE OF THE COPD PATIENT
Recognizing the need to remove “silos’ in delivery of medical services, the World Health Organization (WHO) has promoted integrated care, which it defines as: “… a concept bringing together inputs, delivery, management and organization of services related to diagnosis, treatment, care, rehabilitation and health promotion. Integration is a means to improve the services in relation to access, quality, user satisfaction and efficiency.” Integration in this context is especially important during transitions of care, such as when the patient moves from the hospital to the home or extended care facility. To meet this challenge, the Joint Commission, an organization with a mission “… to continuously improve health care for the public, in collaboration with other stakeholders, by evaluating health care organizations and inspiring them to excel in providing safe and effective care of the highest quality and value.” Seamlessly transitioning care is a goal of the commission and, in a publication, it lists insufficiencies in cross-site communication and collaboration, inadequate knowledge and subsequent transfer of specific patient factors (such as lack of knowledge of patient wishes, abilities and goals of care) and medication reconciliation failures.
Poot and colleagues published a systematic review of integrated disease management for COPD in 2021. For their review, the investigators required the interventions to be multi‐disciplinary (two or more healthcare providers) and multi‐treatment (two or more components). This definition overlapped somewhat with the WHO definition provided above but under-emphasized the element of transition of care. With this caveat in mind, the systematic review demonstrated significant improvements in respiratory-specific quality of life, exercise capacity, and respiratory-related and all-cause hospitalizations. Thus, even without the emphasis on transition of care, health care utilization was reduced.
The complex nature of COPD, including its substantial variability in symptoms and treatment requirements, often prominent systemic effects, frequent and debilitating exacerbations, and common comorbidity, requires integration of care for optimal outcome. The need for integration of services is arguably most acute following discharge for COPD exacerbations, when adequate communication among health care providers and systems is often lacking. Participation in PR permits weeks of ongoing interaction between the patient and health care provider, and along with its holistic approach to patient care and strong promotion of collaborative self-management, this comprehensive intervention fosters integrated care. While PR is associated with decreased health care utilization post-hospitalization, it is not known which components of the intervention are responsible for this beneficial outcome. Furthermore, since PR uptake post-hospitalization for COPD is only about 2%, marked increases in this utilization would be necessary for it to have a substantial effect in the societal costs of the exacerbation.
F. PULMONARY REHABILITATION IN THE POST-EXACERBATION PERIOD (DOES IT PROVIDE THE HOLY GRAIL IN PULMONARY REHABILITATION OUTCOMES?)
It has been known for decades that participation comprehensive PR leads to a significant and clinically meaningful increases in exercise tolerance, decreases in distressing symptoms of dyspnea, fatigue, anxiety and depression, and improvements in health status (health-related quality of life) and self-efficacy. In addition, uncontrolled and systematic reviews of controlled studies suggest that, when PR is administered following respiratory exacerbations (when risk for subsequent hospitalization is high) health care utilization is decreased. These outcomes are, of course, of considerable importance to the individual patient.
However, data indicating that PR has a survival benefit would be of obvious significance. To date, systematic reviews of controlled studies of the effect of PR on survival in COPD have not demonstrated consistent benefit in this important outcome area. A systematic review by Puhan and colleagues, published in 2005, reported a relative risk for mortality of 0.45 (95% confidence interval (CI), 0.22 to 0.91). However, the review included only three trials, with follow-up among the trials ranging from 3 to 48 months, and the overall findings strongly influenced by one considerably larger study. A subsequent review published in 2016 with the same first author, now including six trials, showed high heterogeneity in trials and failed to demonstrate a significant effect of PR on mortality: pooled odds ratio = 0.68 (95% CI 0.28 to 1.67). This result was strongly influenced by one large study published in 2014, involving 196 and 193 COPD patients in intervention and usual care groups, respectively. The intervention was exercise rehabilitation begun within 48 hours of hospitalization. Subsequent hospitalizations (primary outcome) were not significantly different between the groups, and mortality (secondary outcome) was actually higher in the intervention group.
The reason(s) for these apparently anomalous results in this single study remain unclear. Survival analyses in PR trials are consistently fraught with difficulty because of their relatively small size making them under-powered for this outcome, heterogeneity in their treatments, and differences in follow up durations.
A study by Lindenauer and colleagues, published in JAMA in 2020, was designed to evaluate the effect of PR initiated post-discharge among U.S. Medicare patients who were hospitalized in 2014 and 2015 with codes indicating exacerbations for COPD. All-cause mortality in those patients who initiated PR within 90 days of discharge (as determined by outpatient Medicare billing) was compared to that of a matched sample of those who did not. Of 197,376 hospitalized patients meeting their criteria, 2721 (1.5%) initiated PR within 90 days of the index hospitalization. Propensity matching included sociodemographic factors, Medicare-Medicaid dual eligibility, tobacco use, comorbidities, frailty, and markers of disease severity. Propensity-matched cohorts (2710 in each of the PR and no-PR groups) were then compared with respect to subsequent all-cause mortality over one year.
Pulmonary rehabilitation initiated within 90 days of hospital discharge was associated with an impressively lower all-cause mortality over the ensuing year: hazard ratio = 0.63 (95% CI, 0.57 to 0.69). The mortality reduction was present across a range of post-hospitalization PR start times. Furthermore, there was a significant, positive association between the number of PR sessions attended and reduction in mortality. In summary, this was a very large, real-world study involving US COPD Medicare patients, generally aged 65 or older that demonstrated a substantial survival benefit in those who participated in PR following hospitalizations for exacerbations. While not a randomized, controlled trial and thus potentially subject to biases and unmeasured confounders, it nevertheless provides important data on this important outcome and some evidence supporting a potential benefit in mortality. The study also points out a glaring statistic: despite a likely benefit in reducing mortality, its uptake following hospital discharge was only 1.5% – a disparity not missed by the editorial accompanying the article.
G. A BRIEF REVIEW OF PULMONARY REHABILITATION IN DEVELOPING COUNTRIES
Access and utilization of pulmonary rehabilitation continues to be suboptimal globally. Barriers to referral, uptake and completion seems to be different in well-developed countries as compared to low- and middle-income countries. In a recent systematic review of 112 publications out of 78 developing and underdeveloped countries, PR was found to be established only in 17 of the included countries. Another recent qualitative study that focuses on factors that affect access to pulmonary rehabilitation in Iran included 11 people with COPD and 9 family members who were their primary caregivers. Inaccessibility of service and inadequate insurance availability to cover the program were highlighted as major factors impairing access to pulmonary rehabilitation in the study. Another review that focused on implementation of rehabilitation services in low-income countries found that limited funding and minimal resources hampers successful delivery of PR. More recently, a qualitative study by Bickton at all explored barriers and enable her to pulmonary rehabilitation and low- and middle-income countries by doing anonymous interviews of healthcare professionals with PR experience. Major barriers included limited resources, low awareness, coronavirus disease 2019 and patient access related cost. Possible enablers for PR expansion include local adaptation, motivated patients, better awareness and recognition, provision for PR training and resource support. Ongoing collaborations between the National Institute of health research in UK along with global partners including India, Uganda, Kyrgyzstan, and Sri Lanka aims at improving access to pulmonary rehab in developing countries while focusing on cultural adaptations. This date is not known as the global RECHARGE core data set (NIH Global health research group on respiratory rehabilitation) aims to collect data for different countries including demographics, lung health measures, comorbidities, health assessment at baseline, physical measures, treatment and outcome measures including functional status as well as rehabilitation completion, duration of the program, attendance as well as adverse events during pulmonary rehabilitation to help guide implementation globally. Use of tele-rehabilitation and home-based models might be extremely beneficial in limited resource countries as well.
Conclusion:
The science of pulmonary rehabilitation, although young in its inception, has been proven to be an effective therapy for improving a multitude of health outcomes in both stable and post exacerbation COPD. For cost effectiveness and COPD care, pulmonary rehabilitation is second to smoking cessation only. However, access and uptake of pulmonary rehabilitation continues to be dismal. Successful implementation of newer technology including smart phones, tele-rehabilitation and home-based pulmonary rehabilitation might drive the future in improving delivery of PR services. Consideration of policy changes and reimbursement drivers will help overcome economic challenges and implementing successful pulmonary rehabilitation programs.
References
- Spruit MA, Singh SJ, Garvey C, et al. An official american thoracic society/european respiratory society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. Oct 15 2013;188(8):e13-64. doi:10.1164/rccm.201309-1634ST
- Holland AE, Cox NS, Houchen-Wolloff L, et al. Defining modern pulmonary rehabilitation. An official American Thoracic Society workshop report. Annals of the American Thoracic Society. 2021;18(5):e12-e29.
- Petty TL, NETT LM, FINIGAN MM, BRINK GA, CORSELLO PR. A comprehensive care program for chronic airway obstruction: methods and preliminary evaluation of symptomatic and functional improvement. Annals of internal medicine. 1969;70(6):1109-1120.
- Hodgkin J. Pulmonary rehabilitation: official ATS Statement. Am Rev Respir Dis. 1981;124:663-666.
- Hodgkin JE, Celli BR, Connors GA. Pulmonary rehabilitation: guidelines to success. Elsevier Health Sciences; 2023.
- Hodgkin JE. Pulmonary rehabilitation: Structure, components, and benefits. Journal of cardiopulmonary rehabilitation and prevention. 1988;8(11):423-434.
- Casaburi R, Wasserman K. Exercise training in pulmonary rehabilitation. Mass Medical Soc; 1986. p. 1509-1511.
- Casaburi R, Patessio A, Ioli F, Zanaboni S, Donner CF, Wasserman K. Reductions in exercise lactic acidosis and ventilation as a result of exercise training in patients with obstructive lung disease. American Review of Respiratory Disease. 1991;143(1):9-18.
- ZuWallack RL, Patel K, Reardon JZ, Clark III BA, Normandin EA. Predictors of improvement in the 12-minute walking distance following a six-week outpatient pulmonary rehabilitation program. Chest. 1991;99(4):805-808.
- Haggerty MC, Stockdale-Woolley R, ZuWallack R. Functional status in pulmonary rehabilitation participants. Journal of cardiopulmonary rehabilitation and prevention. 1999;19(1):35-42.
- Maltais F, LeBlanc P, Simard C, et al. Skeletal muscle adaptation to endurance training in patients with chronic obstructive pulmonary disease. American journal of respiratory and critical care medicine. 1996;154(2):442-447.
- Reardon J, Awad E, Normandin E, Vale F, Clark B, ZuWallack RL. The effect of comprehensive outpatient pulmonary rehabilitation on dyspnea. Chest. 1994;105(4):1046-1052.
- Ries AL, Kaplan RM, Limberg TM, Prewitt LM. Effects of pulmonary rehabilitation on physiologic and psychosocial outcomes in patients with chronic obstructive pulmonary disease. Annals of internal medicine. 1995;122(11):823-832.
- Lareau S, ZuWallack R, SASSI-DAMBRON D, STUBBING D, CARLIN B, CELLI B. Pulmonary rehabilitation-1999: This official statement of the American Thoracic Society was adopted by the ATS board of directors, November 1998. American journal of respiratory and critical care medicine. 1999;159(5):1666-1682.
- Donner CF, Howard P. Pulmonary rehabilitation in chronic obstructive pulmonary disease (COPD) with recommendations for its use. Report of the European Respiratory Society Rehabilitation and Chronic Care Scientific Group (SEPCR Rehabilitation Working Group). European Respiratory Journal. 1992;5(2):266-275.
- Donner C, Muir J. Selection criteria and programmes for pulmonary rehabilitation in COPD patients. Rehabilitation and Chronic Care Scientific Group of the European Respiratory Society. European Respiratory Journal. 1997;10(3):744-757.
- Nici L, Donner C, Wouters E, et al. American thoracic society/European respiratory society statement on pulmonary rehabilitation. American journal of respiratory and critical care medicine. 2006;173(12):1390-1413.
- Nici L, Limberg T, Hilling L, et al. Clinical competency guidelines for pulmonary rehabilitation professionals: American Association of Cardiovascular and Pulmonary Rehabilitation position statement. Journal of cardiopulmonary rehabilitation and prevention. 2007;27(6):355-358.
- Ries AL, Bauldoff GS, Carlin BW, et al. Pulmonary Rehabilitation: Joint ACCP/AACVPR Evidence-Based Clinical Practice Guidelines. Chest. 2007/05/01/ 2007;131(5, Supplement):4S-42S. doi:https://doi.org/10.1378/chest.06-2418
- Griffiths TL, Burr ML, Campbell IA, et al. Results at 1 year of outpatient multidisciplinary pulmonary rehabilitation: a randomised controlled trial. The Lancet. 2000;355(9201):362-368.
- ZuWallack R. A history of pulmonary rehabilitation: back to the future. Advances in Respiratory Medicine. 2009;77(3):298-301.
- Bourbeau J, Julien M, Maltais F, et al. Reduction of hospital utilization in patients with chronic obstructive pulmonary disease: a disease-specific self-management intervention. Archives of internal medicine. 2003;163(5):585-591.
- Bourbeau J, Van Der Palen J. Promoting effective self-management programmes to improve COPD. Eur Respiratory Soc; 2009. p. 461-463.
- Pitta F, Troosters T, Probst VS, Langer D, Decramer M, Gosselink R. Are patients with COPD more active after pulmonary rehabilitation? Chest. 2008;134(2):273-280.
- de Blok BM, de Greef MH, ten Hacken NH, Sprenger SR, Postema K, Wempe JB. The effects of a lifestyle physical activity counseling program with feedback of a pedometer during pulmonary rehabilitation in patients with COPD: a pilot study. Patient education and counseling. 2006;61(1):48-55.
- Maltais F, Bourbeau J, Shapiro S, et al. Effects of home-based pulmonary rehabilitation in patients with chronic obstructive pulmonary disease: a randomized trial. Annals of internal medicine. 2008;149(12):869-878.
- Man WD, Polkey MI, Donaldson N, Gray BJ, Moxham J. Community pulmonary rehabilitation after hospitalisation for acute exacerbations of chronic obstructive pulmonary disease: randomised controlled study. Bmj. 2004;329(7476):1209.
- Griffiths TL, Phillips C, Davies S, Burr ML, Campbell I. Cost effectiveness of an outpatient multidisciplinary pulmonary rehabilitation programme. Thorax. 2001;56(10):779-784.
- Golmohammadi K, Jacobs P, Sin DD. Economic evaluation of a community-based pulmonary rehabilitation program for chronic obstructive pulmonary disease. Lung. 2004;182:187-196.
- Seymour JM, Moore L, Jolley CJ, et al. Outpatient pulmonary rehabilitation following acute exacerbations of COPD. Thorax. 2010;65(5):423-428.
- Wouters EF. Pulmonary rehabilitation and integrated care. Sage Publications Sage CA: Thousand Oaks, CA; 2005. p. 177-179.
- Spruit MA, Vanderhoven-Augustin I, Janssen PP, Wouters EF. Integration of pulmonary rehabilitation in COPD. The Lancet. 2008;371(9606):12-13.
- Nici L, Raskin J, Rochester CL, et al. Pulmonary rehabilitation: what we know and what we need to know. Journal of cardiopulmonary rehabilitation and prevention. 2009;29(3):141-151.
- McCarthy B, Casey D, Devane D, Murphy K, Murphy E, Lacasse Y. Pulmonary rehabilitation for chronic obstructive pulmonary disease. Cochrane database of systematic reviews. 2015;(2)
- Spruit MA, Pitta F, Garvey C, et al. Differences in content and organisational aspects of pulmonary rehabilitation programmes. European Respiratory Journal. 2014;43(5):1326-1337.
- Puhan MA, Gimeno‐Santos E, Cates CJ, Troosters T. Pulmonary rehabilitation following exacerbations of chronic obstructive pulmonary disease. Cochrane database of systematic reviews. 2016;(12)
- Milner SC, Boruff JT, Beaurepaire C, Ahmed S, Janaudis-Ferreira T. Rate of, and barriers and enablers to, pulmonary rehabilitation referral in COPD: A systematic scoping review. Respir Med. Apr 2018;137:103-114. doi:10.1016/j.rmed.2018.02.021
- Guecamburu M, Coquelin A, Rapin A, et al. Pulmonary rehabilitation after severe exacerbation of COPD: a nationwide population study. Respir Res. Apr 7 2023;24(1):102. doi:10.1186/s12931-023-02393-7
- Spitzer KA, Stefan MS, Priya A, et al. Participation in Pulmonary Rehabilitation after Hospitalization for Chronic Obstructive Pulmonary Disease among Medicare Beneficiaries. Ann Am Thorac Soc. Jan 2019;16(1):99-106. doi:10.1513/AnnalsATS.201805-332OC
- Nishi SP, Zhang W, Kuo YF, Sharma G. Pulmonary Rehabilitation Utilization in Older Adults With Chronic Obstructive Pulmonary Disease, 2003 to 2012. J Cardiopulm Rehabil Prev. Sep-Oct 2016;36(5):375-82. doi:10.1097/hcr.0000000000000194
- Bhatt SP, Westra J, Kuo YF, Sharma G. Pulmonary Rehabilitation Utilization in Older Adults with Chronic Obstructive Pulmonary Disease, 2013-2019. Ann Am Thorac Soc. May 2024;21(5):740-747. doi:10.1513/AnnalsATS.202307-601OC
- Rochester CL, Vogiatzis I, Holland AE, et al. An Official American Thoracic Society/European Respiratory Society Policy Statement: Enhancing Implementation, Use, and Delivery of Pulmonary Rehabilitation. Am J Resp Crit Care Med. Dec 1 2015;192(11):1373-86. doi:10.1164/rccm.201510-1966ST
- Keating A, Lee A, Holland AE. What prevents people with chronic obstructive pulmonary disease from attending pulmonary rehabilitation? A systematic review. Chron Respir Dis. 2011;8(2):89-99. doi:10.1177/1479972310393756
- Rochester CL, Vogiatzis I, Powell P, Masefield S, Spruit MA. Patients’ perspective on pulmonary rehabilitation: experiences of European and American individuals with chronic respiratory diseases. ERJ Open Res. Oct 2018;4(4)doi:10.1183/23120541.00085-2018
- Cox NS, Oliveira CC, Lahham A, Holland AE. Pulmonary rehabilitation referral and participation are commonly influenced by environment, knowledge, and beliefs about consequences: a systematic review using the Theoretical Domains Framework. J Physiother. Apr 2017;63(2):84-93. doi:10.1016/j.jphys.2017.02.002
- Rochester CL. Barriers to Pulmonary Rehabilitation. Respir Care. May 28 2024;69(6):713-723. doi:10.4187/respcare.11656
- Spitzer KA, Stefan MS, Priya A, et al. A Geographic Analysis of Racial Disparities in Use of Pulmonary Rehabilitation After Hospitalization for COPD Exacerbation. Chest. May 2020;157(5):1130-1137. doi:10.1016/j.chest.2019.11.044
- Malla G, Bodduluri S, Sthanam V, Sharma G, Bhatt SP. Access to Pulmonary Rehabilitation among Medicare Beneficiaries with Chronic Obstructive Pulmonary Disease. Ann Am Thorac Soc. Apr 2023;20(4):516-522. doi:10.1513/AnnalsATS.202204-318OC
- Holland AE, Cox NS, Houchen-Wolloff L, et al. Defining Modern Pulmonary Rehabilitation. An Official American Thoracic Society Workshop Report. Ann Am Thorac Soc. May 2021;18(5):e12-e29. doi:10.1513/AnnalsATS.202102-146ST
- Watson JS, Jordan RE, Gardiner L, Adab P, Jolly K. A Systematic Review of the Effectiveness of Interventions to Promote Referral; Adherence; and Uptake of Pulmonary Rehabilitation for Patients with Chronic Obstructive Pulmonary Disease. Int J Chron Obstruct Pulmon Dis. 2023;18:1637-1654. doi:10.2147/copd.S396317
- Mosher CL, Nanna MG, Jawitz OK, et al. Cost-effectiveness of Pulmonary Rehabilitation Among US Adults With Chronic Obstructive Pulmonary Disease. JAMA Netw Open. Jun 1 2022;5(6):e2218189. doi:10.1001/jamanetworkopen.2022.18189
- Caspersen CJ, Powell KE, Christenson GM. Physical activity, exercise, and physical fitness: definitions and distinctions for health-related research. Public Health Rep. Mar-Apr 1985;100(2):126-31.
- Goldstein R, Jardim JR, Nici L, Raskin J, Spruit MA, ZuWallack R. The importance of addressing physical activity and exercise intolerance in our patients with COPD. Eur Respiratory Soc; 2024.
- Xiang X, Huang L, Fang Y, Cai S, Zhang M. Physical activity and chronic obstructive pulmonary disease: a scoping review. BMC pulmonary medicine. 2022;22(1):301.
- Belman MJ. Exercise in Chronic Obstructive Pulmonary Disease. Clinics in chest medicine. 1986/12/01/ 1986;7(4):585-597. doi:https://doi.org/10.1016/S0272-5231(21)00453-6
- Fishman A. Pulmonary rehabilitation research. American journal of respiratory and critical care medicine. 1994;149(3):825-833.
- Clini EM, Crisafulli E. Exercise capacity as a pulmonary rehabilitation outcome. Respiration. 2009;77(2):121-128.
- Celli BR. Pulmonary rehabilitation in patients with COPD. American journal of respiratory and critical care medicine. 1995;152(3):861-864.
- Morris J, Heady J. Mortality in relation to the physical activity of work: a preliminary note on experience in middle age. British journal of industrial medicine. 1953;10(4):245.
- Warburton DE, Nicol CW, Bredin SS. Health benefits of physical activity: the evidence. Cmaj. 2006;174(6):801-809.
- Garcia-Aymerich J, Lange P, Benet M, Schnohr P, Antó JM. Regular physical activity reduces hospital admission and mortality in chronic obstructive pulmonary disease: a population based cohort study. Thorax. 2006;61(9):772-778.
- Waschki B, Kirsten A, Holz O, et al. Physical activity is the strongest predictor of all-cause mortality in patients with COPD: a prospective cohort study. Chest. 2011;140(2):331-342.
- Vaes AW, Garcia-Aymerich J, Marott JL, et al. Changes in physical activity and all-cause mortality in COPD. European Respiratory Journal. 2014;44(5):1199-1209.
- Garcia-Rio F, Rojo B, Casitas R, et al. Prognostic value of the objective measurement of daily physical activity in patients with COPD. Chest. 2012;142(2):338-346.
- Donaire-Gonzalez D, Gimeno-Santos E, Balcells E, et al. Benefits of physical activity on COPD hospitalisation depend on intensity. European Respiratory Journal. 2015;46(5):1281-1289.
- Moy ML, Gould MK, Liu I-LA, Lee JS, Nguyen HQ. Physical activity assessed in routine care predicts mortality after a COPD hospitalisation. ERJ open research. 2016;2(1).
- Spruit MA, Pitta F, McAuley E, ZuWallack RL, Nici L. Pulmonary rehabilitation and physical activity in patients with chronic obstructive pulmonary disease. American journal of respiratory and critical care medicine. 2015;192(8):924-933.
- Arbillaga-Etxarri A, Gimeno-Santos E, Barberan-Garcia A, et al. Long-term efficacy and effectiveness of a behavioural and community-based exercise intervention (Urban Training) to increase physical activity in patients with COPD: a randomised controlled trial. European Respiratory Journal. 2018;52(4):1800063. doi:10.1183/13993003.00063-2018
- Demeyer H, Mohan D, Burtin C, et al. Objectively measured physical activity in patients with COPD: recommendations from an international task force on physical activity. Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation. 2021;8(4):528.
- Bhatt SP. Counterpoint: In-Home Pulmonary Rehabilitation Is an Attractive Alternative. Respir Care. May 28 2024;69(6):763-771. doi:10.4187/respcare.11957
- Cox NS, Dal Corso S, Hansen H, et al. Telerehabilitation for chronic respiratory disease. Cochrane Database Syst Rev. Jan 29 2021;1(1):Cd013040. doi:10.1002/14651858.CD013040.pub2
- Uzzaman MN, Agarwal D, Chan SC, et al. Effectiveness of home-based pulmonary rehabilitation: systematic review and meta-analysis. Eur Respir Rev. Sep 30 2022;31(165)doi:10.1183/16000617.0076-2022
- Rochester CL, Alison JA, Carlin B, et al. Pulmonary Rehabilitation for Adults with Chronic Respiratory Disease: An Official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. Aug 15 2023;208(4):e7-e26. doi:10.1164/rccm.202306-1066ST
- Man W, Chaplin E, Daynes E, et al. British Thoracic Society Clinical Statement on pulmonary rehabilitation. Thorax. Oct 2023;78(Suppl 4):s2-s15. doi:10.1136/thorax-2023-220439
- Bhatt SP, Casaburi R, Mosher CL, Rochester CL, Garvey C. Telehealth Pulmonary Rehabilitation: A Call for Minimum Standards. Am J Respir Crit Care Med. Jul 15 2024;210(2):145-146. doi:10.1164/rccm.202402-0392VP
- Chung C, Lee JW, Lee SW, Jo MW. Clinical Efficacy of Mobile App-Based, Self-Directed Pulmonary Rehabilitation for Patients With Chronic Obstructive Pulmonary Disease: Systematic Review and Meta-Analysis. JMIR Mhealth Uhealth. Jan 4 2024;12:e41753. doi:10.2196/41753
- Grone O, Garcia-Barbero M. WHO EOFIHC Services (2001),“Integrated care: a position paper of the WHO European office for integrated health care services”. International Journal of Integrated Care. 1:e21.
- Anonymous. Who We Are. Accessed 23 July 2024, 2024. https://www.jointcommission.org/who-we-are/
- Sheikh F, Gathecha E, Bellantoni M, Christmas C, Lafreniere JP, Arbaje AI. A call to bridge across silos during care transitions. The Joint Commission Journal on Quality and Patient Safety. 2018;44(5):270-278.
- Poot CC, Meijer E, Kruis AL, Smidt N, Chavannes NH, Honkoop PJ. Integrated disease management interventions for patients with chronic obstructive pulmonary disease. Cochrane Database of Systematic Reviews. 2021;(9)
- Kripalani S, LeFevre F, Phillips CO, Williams MV, Basaviah P, Baker DW. Deficits in Communication and Information Transfer Between Hospital-Based and Primary Care Physicians: Implications for Patient Safety and Continuity of Care. Jama. 2007;297(8):831-841. doi:10.1001/jama.297.8.831
- ZuWallack R, Nici L. Integrated care of the COPD patient: a pulmonary rehabilitation perspective. Breathe. 2010;6(4):313-319.
- Stefan MS, Pekow PS, Priya A, et al. Association between Initiation of Pulmonary Rehabilitation and Rehospitalizations in Patients Hospitalized with Chronic Obstructive Pulmonary Disease. Observational Study Research Support, N.I.H., Extramural. Am J Respir Crit Care Med. Nov 1 2021;204(9):1015-1023.
- Lindenauer PK, Stefan MS, Pekow PS, et al. Association between initiation of pulmonary rehabilitation after hospitalization for COPD and 1-year survival among Medicare beneficiaries. Jama. 2020;323(18):1813-1823.
- Nici L, Lareau S, ZuWALLACK R. Pulmonary rehabilitation in the treatment of chronic obstructive pulmonary disease. American Family Physician. 2010;82(6):655-660.
- Raskin J, Spiegler P, McCusker C, et al. The effect of pulmonary rehabilitation on healthcare utilization in chronic obstructive pulmonary disease: the Northeast Pulmonary Rehabilitation Consortium. Journal of cardiopulmonary rehabilitation and prevention. 2006;26(4):231-236.
- Group CPRC. Effects of pulmonary rehabilitation on dyspnea, quality of life, and healthcare costs in California. Journal of cardiopulmonary rehabilitation and prevention. 2004;24(1):52-62.
- Puhan MA, Scharplatz M, Troosters T, Steurer J. Respiratory rehabilitation after acute exacerbation of COPD may reduce risk for readmission and mortality–a systematic review. Respiratory research. 2005;6:1-12.
- Troosters T, Gosselink R, De Paepe K, Decramer M. Pulmonary rehabilitation improves survival in COPD patients with a recent severe acute exacerbation. Am J Respir Crit Care Med. 2002;165(8):A16.
- Greening NJ, Williams JE, Hussain SF, et al. An early rehabilitation intervention to enhance recovery during hospital admission for an exacerbation of chronic respiratory disease: randomised controlled trial. Bmj. 2014;349
- Rochester CL, Holland AE. Pulmonary rehabilitation and improved survival for patients with COPD. Jama. 2020;323(18):1783-1785.
- Farah, R.; Groot, W.; Pavlova, M. Barriers to access to pulmonary rehabilitation in developing countries: A systematic review. Virtual Congress 2020—Optimising the benefits of pulmonary rehabilitation. Eur. Respir. J. 2020, 56, 884.
- Sami, R.; Salehi, K.; Hashemi, M.; Atashi, V. Exploring the barriers to pulmonary rehabilitation for patients with chronic obstructive pulmonary disease: A qualitative study. BMC Health Serv. Res. 2021, 21, 828.
- Habib, G.M.M.; Rabinovich, R..; Pinnock, H. Systematic review of clinical effectiveness, components, and delivery of pulmonary rehabilitation in low-resource settings. NPJ Prim. Care Respir. Med. 2020.
- Bickton F; Shannon H. Barriers and Enablers to Pulmonary Rehabilitation in Low- and Middle-Income Countries: A Qualitative Study of Healthcare Professionals. Int J Chron Obstruct Pulmon Dis. 2022; 17: 141–153.
- Orme, M.W.; Free, R.C.; Manise, A. Global RECHARGE: Establishing a standard international data set for pulmonary rehabilitation in low- and middle-income countries. J. Glob. Health 2020, 10, 020316.




