Real-World Evidence in Wound Care: RCTs vs. RWE
Real-World Evidence Complements Randomized Controlled Trial Data in Complex Wound Care: Early Experience with Three-Dimensional Acellular Wound Matrix (3D-ACM) in a Clinical Registry
Robert J. Snyder, DPM, MSc, MBA1, Joseph Rolley, MSIA2
- Cardiff University School of Medicine, Cardiff, UK
- Principal, JTR Business Consulting, LLC, Doylestown, PA, USA
ABSTRACT
Introduction: Randomized controlled trials (RCT) and real-world evidence represent complementary approaches in clinical research, each with distinct strengths and limitations. Randomized controlled trials (RCT) are regarded as the gold standard for establishing efficacy because of their rigorous design and control over variables, but they are often criticized for limited generalizability to broader patient populations. In contrast, real-world evidence (RWE), derived from routine clinical practice, captures outcomes across diverse, heterogeneous populations and settings, enhancing applicability to clinical decision-making.
Discussion: This manuscript reviews the advantages and limitations of both methodologies, emphasizing their complementary roles in generating robust evidence. RCTs offer strong internal validity through randomization, blinding, and strict eligibility criteria, while RWE provides external validity by reflecting patient diversity, long-term outcomes, and care patterns in everyday practice.
Conclusion: Early clinical experience with a three-dimensional acellular wound matrix illustrates how registry-derived RWE can complement ongoing RCTs in wound care. This dual evidence strategy underscores the importance of aligning trial rigor with real-world complexity to generate clinically relevant, patient-centered data that further supports innovation, regulatory pathways, and reimbursement frameworks.
Keywords: Evidence-Based Practice, Randomized Controlled Trials, Diabetic Foot, Tissue Scaffolds, Wound Healing, Skin Ulcer.
Introduction
High-quality clinical evidence is essential for guiding therapeutic decisions, informing policy, and improving patient outcomes. Yet in many fields including wound care there remains a persistent gap between the data generated in controlled research settings and the complexity of patients encountered in everyday practice. As healthcare delivery becomes increasingly interdisciplinary and outcomes-focused, there is a growing need for evidence that not only demonstrates scientific rigor but also reflects real-world feasibility, applicability, and clinical utility. These dynamics have brought renewed attention to how different forms of evidence contribute to the evaluation of medical interventions.
Randomized controlled trials (RCTs) have long served as the gold standard for assessing safety and efficacy for medical devices, biologics, and pharmaceuticals. The 5th edition of Fundamentals of Clinical Trials by Friedman et al. outlines the core principles that underpin this status including randomization, blinding, and appropriate sample size estimation, each of which is designed to reduce bias and strengthen internal validity. As the authors conclude, well-designed clinical trials remain indispensable for generating reliable medical evidence and advancing patient care. By reducing confounding variables and standardizing study conditions, RCTs can isolate treatment effects with a high degree of confidence.
However, practical limitations surrounding RCT design are increasingly recognized in clinical medicine. Many trials employ highly selective eligibility criteria that exclude older adults, patients with multimorbidity, and individuals with complex medical or social needs. Trial environments often differ substantially from routine practice, and protocolized care may not reflect the variability seen in actual clinical workflows. One review reported that common comorbid conditions triggered exclusion in 81.3% of trials, underscoring the substantial divide between controlled research settings and real-world patient populations. As healthcare systems place greater emphasis on pragmatic, patient-centered decision-making, these limitations highlight the need for evidence frameworks that balance internal validity with external relevance.
Real-world evidence (RWE), which encompasses data generated during routine clinical practice, offers a complementary lens that can broaden understanding of treatment effectiveness across diverse and clinically complex populations. When collected and analyzed using rigorous methodological standards, RWE can provide insights into care patterns, treatment performance, and patient outcomes that may not be captured by RCTs alone, supporting both clinical and policy decision-making.
This clinical review critically examines the strengths and limitations of RCTs and RWE, synthesizing statistical findings and published literature to clarify their respective contributions to evidence development. It further explores how integrating both approaches can support a more comprehensive and clinically meaningful evaluation of interventions including advanced wound care technologies by capturing both controlled efficacy and real-world performance.
Discussion
THE PROS AND CONS OF RANDOMIZED CONTROLLED TRIALS
Randomized controlled trials (RCTs) remain the gold standard of clinical research because they minimize bias through randomization, blinding, and strict eligibility criteria. These features provide high internal validity and enable causal inference that observational studies cannot reliably achieve. RCTs have been pivotal in shaping modern medicine. For example, the Diabetes Control and Complications Trial (DCCT) demonstrated that intensive insulin therapy significantly reduced long-term complications in type 1 diabetes, transforming diabetes management. Similarly, in wound care, randomized trials of negative pressure wound therapy (NPWT), initially cleared via the 510(k) pathway, provided evidence of improved healing outcomes compared with standard therapy. This data strengthened the clinical profile of NPWT and supported payer coverage, illustrating how RCTs can complement regulatory clearance with robust evidence.
Regulators continue to rely heavily on RCTs. In 2020, 92% of newly approved drugs were supported by randomized trial data, underscoring their central role in demonstrating efficacy and safety. Beyond regulatory approval, RCTs remain highly influential in payer and formulary decisions, providing the evidence base for comparative effectiveness and value assessments.
Despite these strengths, RCTs face important limitations. One major concern is generalizability. RCTs often exclude patients with advanced age, multimorbidity, or other complexities common in clinical practice. A systematic review found that only about 5% of patients with chronic conditions would meet eligibility criteria for typical RCTs, highlighting how trial populations may not reflect the real-world patient population. This lack of external validity raises questions about whether trial findings can be applied broadly across healthcare systems.
RCTs are also resource intensive. Large trials often require thousands of patients, cost millions of dollars, and take years to complete. The Tufts Center for the Study of Drug Development has estimated that bringing a new drug to market costs on average $2.6 billion, with clinical trials representing a substantial share of that expense. These costs can delay access to therapies and limit the number of interventions that can feasibly be evaluated in randomized designs.
Finally, ethical challenges can complicate trial design. Randomization is only appropriate when there is genuine uncertainty regarding which treatment is superior. Once preliminary evidence suggests one therapy is better, randomization may be considered unethical. This creates barriers to conducting trials in settings where effective standard therapies already exist, such as oncology or life-threatening infections.
In summary, RCTs remain essential for establishing efficacy and internal validity, but their limited generalizability, high cost, and ethical constraints restrict their ability to answer all questions relevant to patient care.
THE PROS AND CONS OF REAL-WORLD EVIDENCE
Real-world evidence (RWE) offers a complementary perspective by capturing outcomes in diverse populations under routine clinical conditions. Unlike RCTs, which rely on carefully selected cohorts, RWE reflects the heterogeneity of everyday practice, including patients with comorbidities, polypharmacy, and variable adherence. Data sources include registries, electronic health records (EHRs), insurance claims, and patient-reported outcomes, allowing researchers to assess performance in large, heterogeneous cohorts over extended timeframes. For example, large registry-based studies have shown that treatment effectiveness can vary significantly across ethnic groups, an insight often missed in narrowly defined trials.
Recently, RWE has also gained traction with regulators. In a retrospective review, FDA identified 90 device submissions between 2012 and 2019 in which real-world data were used to support regulatory decisions, including several cases where RWE was the primary source of clinical evidence for approval or indication expansion. CMS has applied RWE in its National Coverage Determinations (NCDs), most notably through the Coverage with Evidence Development (CED) pathway. Under CED, Medicare grants provisional coverage for innovative technologies while additional evidence is collected in clinical registries. High-profile examples include transcatheter aortic valve replacement (TAVR) and low-dose CT for lung cancer screening, where registry-based RWE has been integral to ongoing coverage. Local Coverage Determinations (LCDs) are also beginning to reference RWE. For example, Palmetto GBA’s MolDX LCD for the pigmented lesion assay acknowledged that future coverage updates would consider longer-term, practice-based outcome data, illustrating that Medicare Administrative Contractors (MACs) are starting to integrate RWE into evidence frameworks.
Another strength of RWE is efficiency. Studies using existing data sources can often be conducted more quickly and at lower cost than traditional randomized controlled trials. As Sherman and colleagues note, RWE can leverage data already collected during routine care, reducing both the time and expense required to generate evidence compared with initiating new trials. This efficiency allows for more rapid evidence generation, particularly in therapeutic areas where RCTs are impractical, such as rare diseases or rapidly evolving technologies.
Utilizing RWE also enables longitudinal evaluation, providing insights into adherence, durability of response, and long-term outcomes that RCTs often cannot capture due to time and cost constraints. In wound care, registry and claims-based studies have demonstrated how real-world data can illuminate treatment effectiveness and resource use over extended periods. Analyses from the U.S. Wound Registry have shown wide variation in healing rates across outpatient centers, underscoring the complexity of wound care in everyday practice, while Medicare claims analyses have documented the long-term clinical and economic burden of chronic wounds at a population level.
However, RWE has important limitations. Its observational design makes it more vulnerable to bias and confounding, which can obscure causal relationships. Data quality is another challenge: EHRs and claims data may contain missing, inaccurate, or inconsistent information, with some studies estimating error rates of up to 30% in clinical records. Moreover, the lack of standardized definitions and protocols across RWE studies complicates comparisons and can yield conflicting results.
Despite these issues, regulatory bodies increasingly recognize RWE as a valuable complement to RCTs. The FDA’s Real-World Evidence Framework (2018) and subsequent guidance documents explicitly outline how RWE may inform regulatory decisions across drugs, biologics, and devices. When designed with methodological rigor, RWE provides critical external validity, contextualizing trial findings and helping bridge the gap between efficacy and effectiveness. A recent example of leveraging RWE in an FDA decision is its 2023 approval expanding RECELL® labeling for soft tissue reconstruction. The decision was supported not only by randomized trials but also by real-world evidence and post-market safety data which demonstrated consistent effectiveness and safety beyond burns.
Taken together, RCTs and RWE provide complementary strengths. RCTs deliver high internal validity by isolating treatment effects under controlled conditions, while RWE offers external validity by capturing outcomes across diverse patient populations and real-world practice. As of now, neither approach alone is sufficient to fully inform clinical, regulatory, or payer decision-making. Increasingly, stakeholders recognize that a coordinated strategy, combining the rigor of RCTs with the inclusivity and efficiency of RWE, can generate more comprehensive and clinically relevant evidence.
P&T SYNERGISM BETWEEN RANDOMIZED CONTROLLED TRIALS AND REAL-WORLD DATA
The application of RWE in wound care has lagged despite the availability of large observational datasets and registries. Currently regulatory and payer bodies, including FDA and CMS, continue to prioritize RCTs in this space, even though such trials are often difficult to conduct in heterogeneous patient populations and may not reflect real-world practice.
Formulary decision-makers have likewise been cautious in adopting RWE. Commercial Pharmacy & Therapeutics (P&T) committees continue to prioritize randomized controlled trials (RCTs), citing concerns about data quality and methodological rigor. At the health-system level, Value Analysis Committees (VACs) play a similar role for medical devices, emphasizing cost-effectiveness and value, and generally treating RWE as supplementary to trial-based evidence.
According to the VA Formulary Management Process, the VA P&T Committee evaluates available scientific evidence to determine the safety, efficacy, and value of medications considered for the VA National Formulary. Randomized controlled clinical trials (RCTs) are regarded as the preferred evidence base, while other sources of real-world evidence (observational studies, registries, etc.) may be considered when RCT data are insufficient.
This makes clear that while RWE is not excluded from VA formulary decision-making, it is subordinate to RCT evidence and treated as supplementary. This aligns with the broader pattern across U.S. P&T and federal committees where RWE adoption has lagged regulatory acceptance.
These bodies, which are responsible for evaluating clinical value and cost-effectiveness in formulary decisions, traditionally prioritize high-quality RCTs but are beginning to consider RWE in areas where trial evidence is lacking or insufficient. For these stakeholders, economic models are particularly influential. Budget Impact Models (BIMs) and Cost Effectiveness (CE) analyses, often grounded in real-world data, are essential for formulary placement and pricing negotiations but are rarely evaluated by FDA reviewers. Based on our experience, Table 1 illustrates how different types of evidence are weighed by payers, regulators, and purchasers.
| Evidence Type | Payers | Regulators | Purchasers |
|---|---|---|---|
| Randomized Controlled Trials (RCTs) | Helpful, but not sufficient | Gold standard | Baseline requirement |
| Real-world Evidence (RWE) | Highly valued | Supplementary | Useful for implementation decisions |
| Economic Models | Required (Budget Impact Models, Comparative Effectiveness) | Rarely reviewed | Key for budget justification |
While RCTs remain the gold standard for FDA approval and a baseline requirement for purchasers, payers find them helpful but not sufficient. RWE is increasingly valued by payers and is useful for implementing decisions by institutional purchasers. Economic models are required by payers and purchasers for budget justification, even though they are rarely reviewed by regulatory agencies.
A growing body of formal guidance underpins these developments. The FDA has released a series of documents since 2017 detailing how RWE may be used in submissions for medical devices, drugs, and biologics, including its 2018 Framework for Real-World Evidence and more recent guidance on data standards and the evaluation of electronic health record and claims data. CMS has advanced similar efforts, issuing guidance on Coverage with Evidence Development (CED) and convening a dedicated MEDCAC meeting on real-world evidence to inform Medicare coverage policy.
| Agency | Document/Initiative | Date | Relevance | Summary |
|---|---|---|---|---|
| FDA | Framework for FDA’s RWE Program | Dec 2018 | Drugs, Biologics, Devices | Outlines FDA’s strategic framework for using RWE in regulatory decisions; safety and effectiveness assessments. |
| FDA | Use of RWE to Support Regulatory Decision-Making for Medical Devices | Aug 2017 | Devices | Details FDA’s approach to using RWE to support medical device approvals and post-market modifications. |
| FDA | Considerations for the Use of RWD and RWE to Support Regulatory Decision-Making for Drug and Biological Products | Aug 2023 | Drugs, Biologics | Clarifies how FDA evaluates RWE and the underlying RWD for supporting regulatory decisions. |
| FDA | Assessing Electronic Health Records and Medical Claims Data | July 2024 | All | Guidance assessing fitness of EHR and claims data for regulatory purposes. |
| FDA | Data Standards for Drug and Biological Product Submissions with RWD | Dec 2023 | Drugs, Biologics | Provides clarity on acceptable data standards for RWD in drug submissions. |
| CMS | CMS Evidence Development Guidance Document | 2024 | Coverage (All) | Update Coverage (All) CMS Coverage with Evidence Development (CED) website. |
| CMS | MEDCAC Meetings | Varies | Coverage (All) | Public CMS committee meetings evaluating use of RWE in coverage decisions. |
| Joint (FDA/CMS/PCORI) | Sentinel Initiative | Ongoing | Post-Market Safety, Drugs | National data system for post-market safety surveillance using RWE. |
| Joint (FDA/CMS/PCORI) | PCORnet | Ongoing | RWE Infrastructure | Distributed research network to support pragmatic trials and RWE. |
| FDA/CMS | Parallel Review Program | Ongoing | Drugs, Devices, Coverage | Allows simultaneous FDA review and CMS coverage consideration, often including RWE. |
In parallel, federal initiatives such as the FDA’s Sentinel Initiative, the national PCORnet research network, and the joint FDA–CMS Parallel Review Program illustrate a broader government commitment to modernizing evidence frameworks for real-world decision-making. To maximize the strengths of both randomized controlled trials (RCTs) and real-world evidence (RWE), a coordinated approach can be employed. By integrating findings from RCTs with RWE, researchers can enhance the robustness of clinical evidence. For instance, RCTs can establish the initial efficacy required for regulatory approval, while RWE can validate these findings in everyday clinical practice.
One application of this synergistic approach is in post-marketing surveillance. After a product receives regulatory approval based on RCT data, RWE can be used to monitor long-term safety and effectiveness in the general population. The FDA actively utilizes RWE in post-marketing surveillance – for example, through the Sentinel System – to detect rare adverse events that may not have been apparent in RCTs and has issued guidance on how RWE can support regulatory decision-making for both drugs and medical devices. Another area of application is patient-centered outcomes research. Coordinating RCTs and RWE can facilitate research focused on patient-centered outcomes. For example, RWE can inform the design of RCTs by identifying relevant endpoints that matter to patients, ensuring that clinical trials align with real-world patient experiences and preferences.
THREE-DIMENSIONAL ACELLULAR WOUND MATRIX DEVELOPMENT PROGRAM: USING RCTS AND RWD IN TANDEM: A MOVE TOWARDS GENERALIZABILITY
Product Description and Unique Characteristics
The 3D-ACM (Miro3D® Wound Matrix, Reprise Biomedical, Plymouth, MN) represents a three-dimensional acellular collagen matrix (3D-ACM) derived from porcine liver extracellular matrix and processed using perfusion decellularization to retain the organ’s native microvasculature and structural integrity. The resulting scaffold is a 2 cm-thick, porous construct, engineered specifically to conform to irregular, undermined, or volumetric wound spaces, including those with tunneling or deep cavity formation.
Unlike traditional skin substitutes and biologic matrices which are typically flat and thin (1-5 mm) and designed to overlay superficial wound beds, 3D-ACM offers true volumetric structure, enabling clinicians to pack, layer, or tailor the implant to fill complex wound geometries. Its compressibility and tensile strength allow for delivery into narrow sinuses or cavities without collapse, while maintaining contact with the wound base. In early preclinical and clinical experience, 3D-ACM appeared to permit cellular infiltration, neovascularization, and granulation tissue formation across wound volume; ongoing randomized and registry studies are designed to validate these findings.
Collectively, the 3D architecture and handling properties support use where the clinical goal is to provide volume by maintaining apposition to complex wound geometry and encourage granulation through the wound volume rather than simply covering a planar surface.
Recent consensus in the wound care literature has recognized the emergence of CAMPs (cellular, acellular, and matrix-like products) as a new category of materials for soft-tissue reconstruction. The 2025 Journal of Wound Care consensus on CAMPs noted that volumizing and three-dimensional matrices – particularly those designed for deep and tunneling wounds – are increasingly being used as early interventions, rather than salvage options, in multidisciplinary care plans. The expert panel emphasized that acellular scaffolds with bulk structure are uniquely suited for difficult wound geometries that challenge flat sheet matrices and may help obviate the need for more invasive reconstructive procedures such as flaps.
This broader clinical recognition aligns with the intended use of 3D-ACM. In wounds where conventional biologics may rapidly dissolve, fail to reach the full depth of injury, or require multiple stacked applications, a structurally intact volumetric scaffold may offer enhanced advantages in both handling and performance.
Clinical Development Strategy
Given the unique architecture and intended clinical applications of 3D-ACM, a dual-pronged evidence development strategy was adopted to evaluate both its clinical efficacy and real-world performance. The approach comprises:
- Real-World Evidence Generation
The initial body of clinical evidence for 3D-ACM has been established through:- Retrospective case reports and in practice usage data, including wounds with complex features such as deep tunneling, undermining, exposed structures, or history of radiation therapy.
- Scientific poster presentations and podium abstracts at prominent wound care meetings, including recurring sessions at the Symposium for the Advancement of Wound Care (SAWC) and other specialty congresses have helped disseminate clinical outcomes data to expert audiences.
- A peer-reviewed case series publication in the Journal of Wound Care demonstrated rapid closure and granulation in deep diabetic foot wounds, even in the presence of tendon exposure and patient nonadherence.
- An ongoing, prospective clinical registry capturing outcomes in patients with full-thickness wounds and ulcerations, with a preference for undermining/tunneling or cavity defects that are commonly excluded from randomized trials; the registry reflects real-world wound complexity and comorbidities.
- Randomized Controlled Trials
To rigorously assess the safety and efficacy of 3D-ACM under controlled conditions, two prospective RCTs are actively enrolling:- Study 1 (Outpatient DFU RCT): A multicenter, randomized controlled trial comparing 3D-ACM plus standard of care (SOC) vs SOC alone in Wagner Grade 1 diabetic foot ulcers. This study adheres to current CMS endpoint guidance, using complete wound reepithelialization with no drainage and no dressing as the primary endpoint at 12 weeks.
- Study 2 (Inpatient PU/STI RCT): A separate trial evaluating 3D-ACM in pressure ulcers, post-surgical dehiscence, and other soft tissue wounds in an inpatient setting. This study includes a crossover design, allowing patients who do not heal under SOC to receive 3D-ACM after a predefined interval.
These two trials are designed to complement real-world evidence by evaluating product performance and clinical outcomes in more controlled, homogeneous patient populations. Importantly, clinical registry and retrospective experience continue to enroll and accumulate data in parallel with the trials, providing insights into use patterns, healing trajectories, and product performance in wounds that lie outside of conventional RCT inclusion criteria.
This integrated evidence strategy is intended to confirm clinical practice and inform payer evaluation frameworks. It reflects a growing consensus in wound care that both RWE and RCT data are key to fully evaluating product performance, especially for CAMPS intended to address the most complex wound types.
Representative Clinical Experience
To illustrate both the real-world breadth of wounds treated with 3D-ACM and its relevance to the trial population, we present two deliberately contrasted cases. The first, a chronic diabetic foot ulcer with exposed tendon, represents a complex DFU presentation commonly encountered in practice but ineligible for the DFU RCT because the protocol limits enrollment to Wagner Grade 1 wounds (i.e., no exposed tendon, capsule, muscle, or bone) and excludes deeper anatomy. The second, a chronic diabetic foot ulcer managed under standard outpatient conditions, falls within the RCTs eligibility framework and demonstrates outcomes achievable in the controlled trial population. Together, these paired clinical cases “bookend” the spectrum of use, linking controlled efficacy questions to everyday clinical complexity and providing context for how RCT findings may (or may not) generalize to broader care.
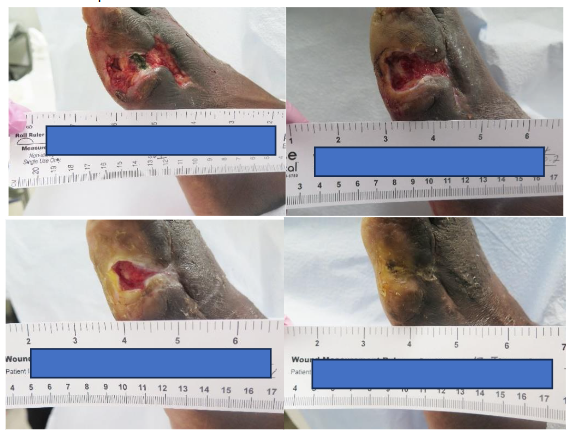
REAL WORLD EVIDENCE: CHRONIC DIABETIC FOOT ULCERS WITH EXPOSED TENDON
A 64-year-old male presented with a chronic diabetic foot ulcer with exposed tendon measuring 5.1 x 3.1 x 0.3 cm (15.81 cm²). The ulcer had been present for 55 days prior to enrollment. Following sharp debridement and standardized offloading, 3D-ACM was applied weekly for four weeks, then bi-weekly through week 13. By week 4, the wound achieved 54.5% percent area reduction (PAR); by week 8, 90.3% PAR (residual area ~1.5 cm²), with robust granulation over the previously exposed tendon. The wound achieved complete closure by week 13 with consistent offloading and dressing adherence.
RANDOMIZED CONTROLLED TRIALS-ELIGIBLE CASE: CHRONIC WAGNER GRADE 1 DIABETIC FOOT ULCER IN A HIGH-RISK, NONADHERENT PATIENT
A 33-year-old male presented with a chronic 24-week-old diabetic foot ulcer (2.2 cm²) located on the left heel. The case was complicated by a history of tobacco use and poor adherence to prescribed offloading, creating a high-risk environment for impaired wound healing. 3D-ACM was applied weekly for the first four weeks in combination with standard of care (SOC) dressing protocols and clinical monitoring.
At week 4, the wound achieved an 80.4% area reduction (PAR) despite suboptimal adherence and environmental challenges such as moisture imbalance. By week 8, the ulcer demonstrated 96.4% PAR, reducing to 0.08 cm² with notable granulation and improvement in peri-wound conditions, including resolution of maceration. Substantial closure was observed by week 9.
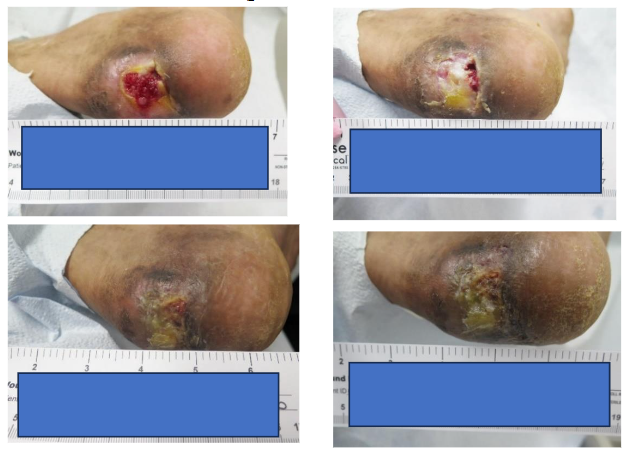
Even with suboptimal adherence, 3D-ACM supported cellular ingrowth and granulation tissue formation, with organization consistent with early remodeling, suggesting a pro-healing microenvironment that promotes epithelialization and mitigates chronic inflammation despite real-world risk factors.
These cases illustrate the value of early response metrics, particularly 4-week percent area reduction (PAR), as potential predictors of subsequent closure. Across DFU and chronic wound studies, achieving ≥50% PAR at 4 weeks is associated with markedly higher 12-week healing rates, whereas wounds below this threshold rarely close by week 12. However, in 120 patients who achieved 50% PAR by week 4, 62 (52%) failed to heal by 12 weeks. In a follow-up retrospective analysis including the intent-to-treat control patient data from two published randomized, diabetic foot ulcer trials, Warriner and colleagues found that wounds experiencing a deviation from the predicted healing course weeks during weeks 5 and 6 (a two-week delay) and those that did not reach 90% PAR by week 8 were not likely to heal by week 12.
These benchmarks provide practical decision points to reassess care and consider escalation to advanced modalities. In the chronic DFU with exposed tendon, PAR reached 54.5% by week 4 and 90.3% by week 8, with complete healing by week 13. In the chronic Wagner Grade 1 DFU, PAR was 80.4% at week 4 and >96% by week 8, aligning with predictive thresholds reported in multicenter cohorts. Taken together, these observations suggest that a volumetric scaffold can provide anatomical conformity in complex beds and help patients reach early response thresholds that guide evidence-informed care.
COMPARING CLINICAL REALITY TO TRIAL ELIGIBILITY
Randomized controlled trials in wound care prioritize internal validity by enforcing narrow eligibility, whereas prospective registries and real-world experience capture broader clinical complexity. To show where evidence originates (and where it does not), we present a single comparative table summarizing inclusion and exclusion constructs for the ongoing outpatient DFU RCT, the prospective post-market registry, and pragmatic RWE use of the 3D-ACM. This allows readers to see how trial populations differ from the patients seen in everyday practice.
| Inclusion Criteria | RCT (DFU/SWD) | Prospective Registry | Real-World Experience |
|---|---|---|---|
| Population – Age | Adults (≥18 years) | Adults (≥18 years) | Typically adults; pragmatic use (not protocolized) |
| Wound type / location | Wagner 1 DFU/SWD on the foot; no exposed deep structures. | Wounds/ulcerations; Wagner 1–2; any site; tunneling/undermining preferred (not required) | Broad spectrum, including complex locations |
| Depth / anatomy | Superficial only (no exposed tendon/muscle/bone) | Any depth; full thickness permitted | Deep/undermined/tunneled; exposed structures as appropriate |
| Tunneling / undermining | Not permitted (Wagner 1) | Allowed; preferred (not required) | Common in practice |
| Wound size | 1-20 cm² (post-debridement) | Recruitment target 1–40 cm² | Variable; often larger/irregular |
| Duration | ≥4 weeks prior to screening; 2-week SOC run-in precedes randomization | Remain open after ≥4 weeks of SOC; no run-in | Typically chronic; ≥4 weeks SOC common |
| Perfusion / vascular status | Adequate perfusion per ABI 0.7-1.2, TBI ≥0.7, TcPO₂ ≥40 mmHg, or biphasic/triphasic Doppler | Adequate perfusion documented as any of: ABI ≥0.7, TBI ≥0.7, TcPO₂ ≥40 mmHg, or biphasic/triphasic Doppler. | Per practice; revascularize/optimize as needed |
| Infection history | Prior infection allowed if controlled (IDSA); active/untreated osteomyelitis excluded | Prior infection allowed if controlled (IDSA); active/untreated osteomyelitis excluded | Managed by IDSA/clinical judgment; treat first, then proceed |
| Treatment objective / wound readiness | Clean, debrided base; offloading (Foot Defender) and compression if edema; one index wound | Clean, debrided base; offloading and/or compression; able to follow-up | Use after wound-bed preparation and limb optimization consistent with TIME and IDSA/IWGDF guidance. |
| Exclusion Criteria | RCT (DFU/SWD) | Prospective Registry | Real-World Experience |
|---|---|---|---|
| Glycemic control | HbA1c ≥12% | Recruitment expects A1c ≤12% (screening target) | Varies; may constrain use |
| Renal status | Dialysis | Dialysis | Case-by-case; often constrained by payer policy |
| Infection / ischemia | Active/uncontrolled osteomyelitis; worsening ischemia/gangrene. | Active/untreated osteomyelitis; worsening ischemia/gangrene. | Managed per clinical judgment. |
| Oncologic / therapy | Malignancy/vasculitis at index site; ongoing chemotherapy | Malignancy/vasculitis at site; ongoing chemotherapy | Typically managed in oncology pathways |
| Prior investigational therapy | Investigational drug/device/biologic ≤30 days | Investigational therapy ≤30 days | – |
| Radiation / hardware (index area) | Prior radiation to index area; exposed internal fixation/implants/hardware | Prior radiation to index area; exposed hardware/implants | Seen in salvage; individualized approach |
| Adherence / follow-up | Documented nonadherence or conditions limiting follow-up | Nonadherence/conditions limiting completion | – |
| Material sensitivity | Porcine sensitivity | Porcine sensitivity | Porcine sensitivity |
| Goals of care | Palliative / comfort-care status | Palliative / comfort-care status | – |
Most patients treated with CAMPs in practice would not qualify for enrollment in typical RCTs. One study showed that over 50% of patients seen in wound care clinics would have been excluded from 15 of 17 reviewed trials, and even after relaxing criteria, 25–50% would still have been ineligible. The cases featured in this manuscript – a chronic DFU with exposed tendon (ineligible for the DFU RCT) and a chronic Wagner Grade 1 DFU that meets RCT criteria – illustrate both ends of the clinical spectrum. The first exemplifies real-world complexity that conventional trials exclude, while the second aligns with the trial population and demonstrates outcomes achievable under those constraints. Together, they underscore the notion that randomized trials and real-world evidence are complementary: RCTs establish efficacy in controlled cohorts, whereas RWE captures performance and applicability in patients who fall outside trial eligibility, enabling a more complete appraisal of safety, effectiveness, and generalizability with a three-dimensional acellular collagen matrix.
Conclusion
Randomized controlled trials (RCTs) provide the regulatory gold standard for establishing efficacy, yet their strict eligibility criteria often limit generalizability to real-world patient populations. Real-world evidence (RWE), derived from registries, case series, and clinical practice data, complements RCTs by capturing outcomes in diverse and complex patients, including those commonly excluded from trials. This distinction is particularly relevant in wound care, where chronic wounds frequently involve comorbidities, tunneling, and variable adherence that challenge traditional trial design. Regulatory and payer bodies increasingly acknowledge the value of RWE, though its integration into decision-making remains cautious. The development of a three-dimensional acellular collagen matrix (3D-ACM) highlights the importance of combining methodologies: ongoing RCTs provide rigorous efficacy data, while parallel real-world registries document performance in complex wounds. Together, RCTs and RWE create a synergistic evidence base that enhances clinical relevance, supports regulatory and reimbursement pathways, and informs patient-centered wound care.
Conflict of Interest Statement
RS and JR report consultancy relationships with Reprise Biomedical. The authors declare no other conflicts of interest related to this work.
Funding Statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Acknowledgements
The authors thank the patients for their willingness to allow their cases to be used for this publication and the clinical staff involved in their care for their support.
Ethics Statement
This work was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent for publication of clinical details and accompanying images was obtained from the patient(s). Institutional review board approval was not required, as this article describes case-based illustrative examples rather than a research study.
References
- Bothwell LE, Greene JA, Podolsky SH, Jones DS. Assessing the gold standard—lessons from the history of randomized controlled trials. N Engl J Med. 2016;374(22):2175-2181. doi:10.1056/NEJMms1604593
- Friedman LM, Furberg CD, DeMets DL, Reboussin DM, Granger CB. Fundamentals of Clinical Trials. 5th ed. New York: Springer; 2015.
- Van Spall HGC, Toren A, Kiss A, Fowler RA. Eligibility criteria of randomized controlled trials published in high-impact general medical journals: a systematic sampling review. JAMA. 2007;297(11):1233-40. doi:10.1001/jama.297.11.1233
- U.S. Food and Drug Administration. Framework for FDA’s Real-World Evidence Program. Silver Spring, MD: FDA; 2018. Available from: https://www.fda.gov/media/120060/download
- Sherman RE, Anderson SA, Dal Pan GJ, Gray GW, Gross T, Hunter NL, et al. Real-world evidence — what is it and what can it tell us? N Engl J Med. 2016;375(23):2293-7. doi:10.1056/NEJMsb1609216
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977-86. doi:10.1056/NEJM199309303291401
- Willy C, Agarwal A, Andersen CA, et al. Closed incision negative pressure therapy: international multidisciplinary consensus recommendations. Int Wound J. 2017;14(2):385-98. doi:10.1111/iwj.12612
- U.S. Food and Drug Administration. Advancing Health Through Innovation: New Drug Therapy Approvals 2020. Silver Spring, MD: FDA; 2021. Available from: https://www.fda.gov/media/145616/download
- Tufts Center for the Study of Drug Development. The cost of developing a new drug. Available at: https://csdd.tufts.edu. Accessed October 2023.
- Wang SV, Schneeweiss S, Berger ML, et al. Reporting to improve reproducibility and facilitate validity assessment for healthcare database studies: a joint ISPOR-ISPE special task force report. Value in Health. 2017;20(8):1009-1022. doi:10.1016/j.jval.2017.08.3018.
- U.S. Food and Drug Administration (FDA). Examples of Real-World Evidence (RWE) Used in Medical Device Regulatory Decisions. Silver Spring, MD: CDRH; 2021.
- Centers for Medicare & Medicaid Services (CMS). Decision Memo for Transcatheter Aortic Valve Replacement (TAVR) (CAG-00430R). Baltimore, MD: CMS; March 26, 2019. Available at: https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&NCAId=293 . Accessed August 11, 2025.
- Centers for Medicare & Medicaid Services (CMS). Decision Memo for Screening for Lung Cancer with Low Dose Computed Tomography (LDCT) (CAG-00439R). February 10, 2022. Available at: https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&NCAId=304. Accessed August 11, 2025.
- Palmetto GBA. MolDX: Pigmented Lesion Assay. Local Coverage Determination (LCD): L38151. Centers for Medicare & Medicaid Services; 2020. Available from: https://www.cms.gov/medicare-coverage-database.
- Sherman RE, Anderson SA, Dal Pan GJ, Gray GW, Gross T, Hunter NL, LaVange L, Marinac-Dabic D, Marks PW, Robb MA, Shuren J, Temple R, Woodcock J, Yue LQ, Califf RM. Real-world evidence —what is it and what can it tell us? N Engl J Med. 2016;375(23):2293–2297. doi:10.1056/NEJMsb1609216.
- Fife CE, Eckert KA, Carter MJ. Publicly reported wound healing rates: the fantasy and the reality. Adv Wound Care (New Rochelle). 2018;7(3):77–94. doi:10.1089/wound.2017.0767.
- Nussbaum SR, Carter MJ, Fife CE, DaVanzo J, Haught R, Nusgart M, Cartwright D. An economic evaluation of the impact, cost, and Medicare policy implications of chronic nonhealing wounds. Value Health. 2018;21(1):27–32. doi:10.1016/j.jval.2017.07.007.
- Weiskopf NG, Weng C. Methods and dimensions of electronic health record data quality assessment: enabling reuse for clinical research. J Am Med Inform Assoc. 2013;20(1):144–151. doi:10.1136/amiajnl-2011-000681.
- Concato J, Corrigan-Curay J. Real-world evidence — where are we now? N Engl J Med. 2022;386(18):1680–1682. doi:10.1056/NEJMp2200089.
- FDA. Summary of Safety and Effectiveness Data (SSED): RECELL® Autologous Cell Harvesting Device (P170031/S016). U.S. Food and Drug Administration. Approved June 6, 2023. Available at: December 22, 2023 Summary of the Safety and Effectiveness Data – RECELL Autologous Cell Harvesting Device
- Franklin JM, Schneeweiss S. When and how can real-world data analyses substitute for randomized controlled trials? Clin Pharmacol Ther. 2017;102(6):924–933. doi:10.1002/cpt.857.
- Perl JR, Sheth KR, Shea KG, Wall J. Hospital value committees: the role of the surgeon in new technology adoption. Surg Innov. 2021;28(4):401–402. doi:10.1177/15533506211031094.
- Radomski TR, Good CB, Thorpe CT, Zhao X, Marcum ZA, Glassman PA, Lowe J, Mor MK, Fine MJ, Gellad WF. Variation in formulary management practices within the Department of Veterans Affairs health care system. J Manag Care Spec Pharm. 2016;22(2):114–120. doi:10.18553/jmcp.2016.14251.
- U.S. Food and Drug Administration (FDA). Use of Real-World Evidence to Support Regulatory Decision-Making for Medical Devices: Guidance for Industry and Food and Drug Administration Staff. Silver Spring, MD: FDA; August 2017. Available at: https://www.fda.gov/media/99447/download.
- U.S. Food and Drug Administration (FDA). Framework for FDA’s Real-World Evidence Program. Silver Spring, MD: FDA; December 2018. Available at: https://www.fda.gov/media/120060/download.
- U.S. Food and Drug Administration (FDA). Real-World Data: Assessing Electronic Health Records and Medical Claims Data To Support Regulatory Decision-Making for Drug and Biological Products. Guidance for Industry. Silver Spring, MD: FDA; September 2021. Available at: https://www.fda.gov/media/152503/download.
- Centers for Medicare & Medicaid Services (CMS). Guidance for the Public, Industry, and CMS Staff: Coverage with Evidence Development (CED). Baltimore, MD: CMS; November 2014. Available at: https://www.cms.gov/medicare-coverage-database/view/medicare-coverage-document.aspx?MCDId=27. Accessed August 2025.
- Centers for Medicare & Medicaid Services (CMS). Medicare Evidence Development & Coverage Advisory Committee (MEDCAC) Meeting: Consideration of Real-World Evidence (RWE). Baltimore, MD: CMS; December 7, 2016. Available at: https://www.cms.gov/medicare-coverage-database/view/medcac-meeting.aspx?medcacId=72. Accessed August 2025.
- U.S. Food and Drug Administration. Considerations for the Use of Real-World Data and Real-World Evidence to Support Regulatory Decision-Making for Drug and Biological Products. Guidance for Industry. Silver Spring, MD: FDA; August 2023. Available at: https://www.fda.gov/media/171667/download. Accessed September 2025.
- U.S. Food and Drug Administration, Center for Devices and Radiological Health (CDRH). Use of Real-World Evidence to Support Regulatory Decision-Making for Medical Devices: Guidance for Industry and Food and Drug Administration Staff. Silver Spring, MD: FDA; August 2017. Available at: https://www.fda.gov/media/99447/download. Accessed September 2025.
- Reprise data on file.
- Chaffin AE, et al. Cellular, acellular and matrix-like products (CAMPs) for soft-tissue reconstruction. J Wound Care. 2025;34(Suppl B):S2-S20.
- Abdo RJ, Couch AL. Use of three-dimensional acellular collagen matrix in deep or tunneling diabetic foot ulcers: a retrospective case series. J Wound Care (North Am Suppl). 2024;33(9):1-6.
- Snyder RJ, Cardinal M, Dauphinee D, Stavosky J. A post-hoc analysis of reduction in diabetic foot ulcer size at 4 weeks as a predictor of healing by 12 weeks. Wound Manag Prev. 2010;56(3):44-48.
- Warriner RA, Snyder RJ, Cardinal MH. Differentiating diabetic foot ulcers that are unlikely to heal by 12 weeks following achieving 50% percent area reduction at 4 weeks. Int Wound J. 2011;8:632-637.
- Sheehan P, Jones P, Caselli A, Giurini JM, Veves A. Percent change in wound area of diabetic foot ulcers over a 4-week period is a robust predictor of complete healing in a 12-week prospective trial. Diabetes Care. 2003;26(6):1879-1882.
- Margolis DJ, Berlin JA, Strom BL. Which randomized controlled trials in wound care are applicable to your patient? Estimating the applicability of wound care randomized controlled trials to general wound-care populations. Wound Repair Regen. 2003;11(6):387-392.




