Refractory COVID-19 Vaccine Myocarditis: Rapamycin Case Study
Resolution of Refractory COVID-19 Vaccine-Induced Myopericarditis with Adjunctive Rapamycin
Nicolas Hulscher1, Alexander Vickery2, Peter A. McCullough2
OPEN ACCESS
PUBLISHED: 30 November 2024
CITATION: HULSCHER, Nicolas; VICKERY, Alexander; MCCULLOUGH, Peter A.. Resolution of Refractory COVID-19 Vaccine-Induced Myopericarditis with Adjunctive Rapamycin. Medical Research Archives, [S.l.], v. 12, n. 11, nov. 2024. Available at: <https://esmed.org/MRA/mra/article/view/6099>.
COPYRIGHT: © 2025 European Society of Medicine. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
DOI: https://doi.org/10.18103/mra.v12i11.6099.
ISSN 2375-1924
ABSTRACT
COVID-19 vaccine-induced myopericarditis is a rare but serious complication of mRNA vaccines. The mainstay of clinical management involves spike protein deactivation and colchicine for 12 months or longer. Herein, we present a case of a previously healthy 23-year-old male with myopericarditis and chest pain following mRNA-1273 COVID-19 vaccine injection.
Keywords
COVID-19, vaccine, myopericarditis, rapamycin, spike protein
Introduction
The emergence of the COVID-19 pandemic in late 2019 and early 2020 constituted an unprecedented global public health emergency¹. In response, vaccine development progressed at an unparalleled pace, with the mRNA-1273 candidate entering clinical trials after only approximately two months of development². The safety profile of SARS-CoV-2 mRNA vaccines remains a topic of substantial scientific debate. There is a growing body of rigorous, methodologically sound, and credible research that points to the concerning severity and frequency of adverse events associated with these vaccines³. Particularly, cardiovascular adverse effects constitute a preeminent majority of COVID-19 side effects⁴, with 28,641 reports of myopericarditis and 37,966 reports of death in the Vaccine Adverse Event Reporting System (VAERS) at the time of writing⁵. Rose et al. found that COVID-19 vaccination is strongly associated with a serious adverse safety signal of myocarditis, especially in children and young adults resulting in hospitalization and death⁶. Autopsy and population-level studies demonstrate that both clinical and subclinical COVID-19 vaccine-induced myopericarditis can be fatal⁷˒⁸. Accordingly, the development of therapeutic approaches to COVID-19 vaccine-induced myopericarditis is a critical public health objective. Here, we present a case of a previously healthy 23-year-old male with suspected COVID-19 vaccine-induced myopericarditis and heart failure (NYHA II), complicated by pre-existing anxiety and autism. We propose rapamycin as a potential treatment for reducing inflammation and addressing ST elevation.
Case Presentation
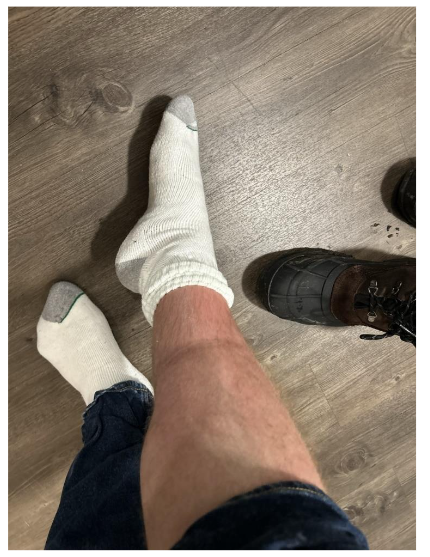
A 21-year-old male diagnosed with autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD) had received one dose of the Moderna mRNA-1273 COVID-19 vaccine (100 mcg – batch: 061E21A) on November 18th, 2021. Approximately two weeks later, the patient began ingesting a combination of atomoxetine (ATX, 200 mg) and dexmethylphenidate (d-MPH, 150 mg, Focalin®). The patient had previously ingested similar doses prior to vaccination, which resulted in expected sympathomimetic effects, including xerostomia, melanoglossia, and anxiety, without any cardiac symptoms. The patient subsequently developed hyperhidrosis, orthostatic intolerance, subjective palpitations, and pitting lower extremity edema (Figure 1). These symptoms spontaneously resolved within 24 hours, and the patient returned home without seeking medical attention. However, the patient experienced recurring cardiovascular symptoms, particularly after the ingestion of d-MPH/ATX, prompting discontinuation of psychostimulants and atomoxetine. Throughout the following months, the patient’s condition deteriorated. He reported frequent episodes of angina pectoris, dyspnea, palpitations, edema, and fatigue without clear inciting factors. He sought emergency department care multiple times, but his symptoms were consistently attributed to anxiety, likely due to his history of anxiety diagnoses. Nine months after mRNA injection, the patient suffered vaccine failure and contracted COVID-19, managed with nirmatrelvir/ritonavir. The infection was moderate, peaking with a temperature of 104°F, and was followed by several non-SARS-CoV-2 viral upper respiratory infections. Seventeen months after mRNA injection, he contracted COVID-19 again, and while the infection was mild, he soon developed symptoms of heart failure (NYHA Class II), including effort intolerance, dyspnea, orthostatic intolerance, and lower extremity edema (Figure 2).
Figure 1: Pitting lower extremity edema observed on Nov. 30th, 2021, approximately 13 days after mRNA-1273 COVID-19 vaccine injection.

Cardiac evaluation at that time included a 48-hour ambulatory ECG that revealed a heart rate of 39-205 bpm, with consistent arrhythmia. Blood pressure was measured at 118/70 mmHg. There were no heart murmurs. SARS-CoV-2 spike protein IgG quantification was 7058 AU/mL. Based on the presentation, a diagnosis of COVID-19 vaccine-induced myopericarditis was made. A “Spike detoxification protocol” was initiated, consisting of bromelain (500 mg once daily), natokinase (2,000 FU twice daily), and curcumin (500 mg twice daily). Given the suspected inflammatory etiology, colchicine (0.6 mg twice daily) was also prescribed. The patient’s response to these treatments was variable, with periods of improvement noted. Followed by relapse, the patient was transitioned to rapamycin (1 mg daily) for 2024. In addition to pharmacological interventions, dietary modifications were made to enhance metabolic capacity and reduce inflammation.
Figure 2: Significant lower extremity edema and erythema observed on April 2023, approximately 519 days after mRNA-1273 COVID-19 vaccine injection. Other symptoms included effort intolerance, dyspnea, orthostatic intolerance, and palpitations.
6-Month Follow-Up and Successful Treatment with Rapamycin
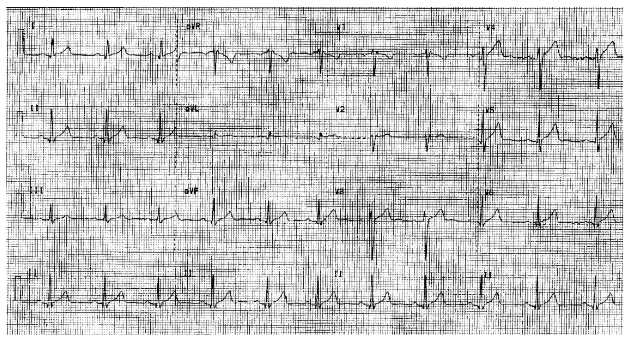
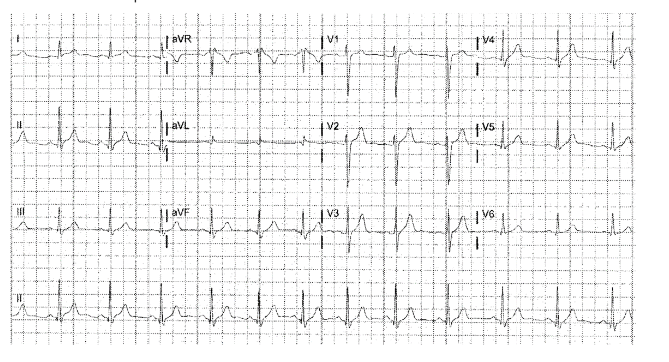
After six months of treatment for myopericarditis, the patient presented with a blood pressure of 122/80 mmHg, a pulse rate of 75 bpm, and an SpO₂ level of 100%. The ECG revealed persistent ST elevation characteristic of active, ongoing myopericarditis (Figure 4). Thoracic ultrasound did not reveal any pericardial effusion, and auscultation was unremarkable for abnormal heart sounds. The patient reported atypical chest pain localized to the seventh left intercostal space. A cardiac MRI with contrast was declined by the patient due to concerns about gadolinium-based contrast agents. SARS-CoV-2 Spike protein IgG quantification had decreased to 2650 AU/ml. Prednisone (20 mg/day) was initiated. The patient experienced improvement in symptoms and resolution of the ST elevation after completing the corticosteroid course. However, due to unfavorable cognitive side effects, the patient chose not to continue corticosteroid therapy, and alternative treatments were explored. Given the patient’s reluctance to continue corticosteroids and the persistence of myopericarditis symptoms, rapamycin (1 mg/day per os) was introduced as an alternative therapeutic intervention. The patient’s HR, SpO₂, and ECG were monitored regularly during treatment. After approximately four weeks of rapamycin administration, ST elevation resolved, and QRS voltage returned to normal (previously showing low voltage). Subjectively, the patient reported durable resolution of symptoms, including chest pain, dyspnea, and effort intolerance. Follow-up labs, including a complete blood count (CBC) and comprehensive metabolic panel (CMP), were performed three months after rapamycin initiation and were unremarkable. A repeat ECG at seven months into rapamycin treatment showed persistent resolution of the ST
elevation (Figure 5). The patient’s condition had stabilized, and no significant side effects from rapamycin, such as depression of white blood cell counts or inhibition of wound healing, were observed.
Figure 4: Electrocardiogram at 6-month follow-up (February 5th, 2024) revealed extensive ST elevation characteristic of pericarditis.
Figure 5: Electrocardiogram at approximately 7 months following initiation of rapamycin treatment (October 16th, 2024) demonstrated complete ST elevation resolution.
Discussion
In this case report, the patient experienced a near-complete resolution of COVID-19 vaccine-induced myopericarditis and heart failure following standard of care Spike protein detoxification plus colchicine, a course of corticosteroids, and approximately 7 months of treatment with rapamycin. This regimen with Spike protein detoxification interventions⁹, including bromelain, curcumin, and nattokinase, and colchicine was designed with the hope of dissolving Spike protein, provision of anti-inflammatory effects, and therapeutic efficacy in resolving COVID-19 vaccine myopericarditis.¹⁰˒¹¹ A structured risk stratification approach for future cardiac events post-COVID-19 vaccination was employed to guide the treatment plan (Figure 6).¹² The patient’s symptomatic improvement was remarkable, with the eventual normalization of electrocardiographic abnormalities and significant relief from effort intolerance, dyspnea, and chest pain. This therapeutic approach appeared to ameliorate the inflammatory processes, reverse the cardiovascular damage, and restore the patient’s quality of life.
Figure 6: Risk Stratification Approach for Future Cardiac Arrest after COVID-19 Vaccination.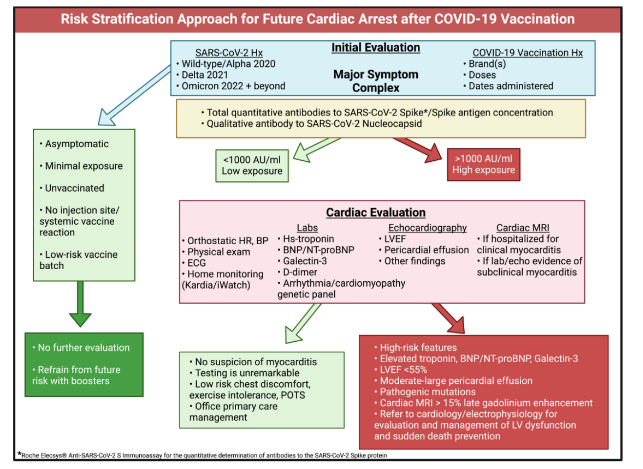
Green boxes indicate clinical features, test results, and patients at lower risk. Red and pink boxes show tests and results indicating higher risk.
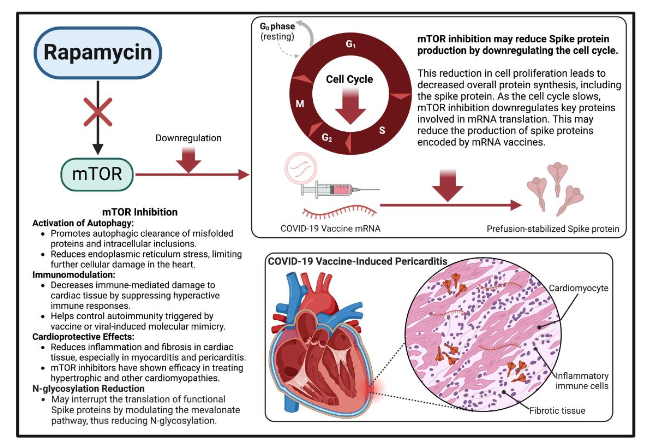
Figure and legend reprinted from McCullough et al. Permission to use this figure has been granted in accordance with the open access Creative Commons CC BY 4.0 license. Created with BioRender.com.This case aligns with a growing body of literature indicating that myopericarditis, particularly in younger individuals, has become a significant post-vaccine complication as a result of Spike protein accumulation in cardiac tissues³˒⁶˒⁹˒¹³–¹⁵. Recurrent COVID-19 infections and booster vaccines can reintroduce Spike protein into circulation, possibly enhancing the risk of myopericarditis¹⁴˒¹⁵. Current vaccines appear to have limited efficacy in preventing these reinfections¹⁶, which exacerbates the risk of further Spike-related inflammation. After COVID-19 vaccination, mRNA has been detected in the bloodstream for up to 28 days post-injection¹⁷, and the vaccine-derived, prefusion-stabilized Spike protein persists in circulation for at least six months¹⁸. This extended presence suggests a considerable window during which myocarditis may develop as a vaccine-related injury. Moreover, cardiac abnormalities have been detected for over a year following the initial diagnosis of COVID-19 vaccine-induced myopericarditis¹⁹, raising concerns about potential long-term effects.The adverse event profile and safety of COVID-19 vaccines have been widely debated, particularly in vaccine batch²⁰–²². The ‘How Bad is My Batch’ tool²³, which queries VAERS data, has proven useful in investigating potential vaccine-related deaths and adverse events²⁴. In this case, we conducted a batch analysis for the Moderna vaccine received by the patient (batch: 061E21A). The analysis revealed 5 reported deaths, 23 hospitalizations, 19 cases of dyspnea, 17 cases of chest pain or discomfort, 8 cases of tachycardia, 5 cases of palpitations, and 1 case of myocarditis. These reported events from other healthcare providers align closely with the symptoms observed in our case. This batch analysis indicates that cardiovascular adverse events are indeed plausible with the vaccine batch administered in this case, 061E21A.Rapamycin likely contributed to the resolution of this case through multiple mechanisms, as illustrated in Figure 7. We initially explored therapeutic options targeting glycosylation processes. Notably, N-glycosylation inhibition has been shown to impede SARS-CoV-2 replication²⁵. Further, alpha-glucosidase inhibition via castanospermine has demonstrated antiviral activity by disrupting the inflammatory processes, reverse the cardiovascular damage, and restore the patient’s quality of life.risk of inducing ER stress due to protein misfolding, which, combined with the narrow therapeutic window and toxicity of such agents, limits their practical use. Consequently, our therapeutic focus turned to mTOR inhibitors, given that SARS-CoV-2 exploitatively stimulates mTOR signaling to facilitate viral replication²⁷. mTOR inhibition has been demonstrated to interrupt SARS-CoV-2 infection²⁸, and as such, mTOR inhibitors have been proposed for drug repurposing in the treatment of SARS-CoV-2²⁹. Additionally, mTOR inhibitors could confer extensive therapeutic effects. mTOR signaling is dysregulated in cardiomyopathy, and mTOR inhibitors have shown promise in managing conditions like hypertrophic cardiomyopathy³⁰. Rapamycin, in particular, has demonstrated therapeutic potential in experimental autoimmune myocarditis (EAM), an induced-autoimmunity model that more closely approximates vaccine-induced myocarditis when vaccinizing mice with myosin peptides in combination with an adjuvant³¹. The pathophysiology in EAM mirrors that of vaccine-related myopericarditis, raising concerns that COVID-19 vaccination may trigger myocarditis presenting after a post-vaccination latency period. Given that EAM and vaccine-induced myopericarditis share immune self-reactivity mechanisms, rapamycin’s immunomodulatory effects likely helped suppress hyperactive immune responses, reducing inflammation and fibrosis in cardiac tissue³¹˒³². Of particular relevance is the potential for autoimmunity triggered by molecular mimicry, as prior research has shown significant overlap between SARS-CoV-2 proteins and human cardiac epitopes³³˒³⁴. This molecular mimicry could provoke a self-reactive immune response, particularly targeting cardiac tissue, resulting in myocardial injury. Coordinate pathophysiological mechanisms likely involve RAAS disruptions (the “Spike effect”)³⁵ and aberrant protein accumulation, which are known to contribute to cardiomyopathies³⁶. Rapamycin’s combined autophagic and immunomodulatory effects make it an ideal candidate for addressing such pathomechanisms³¹˒³⁷. As we advanced our consideration of rapamycin’s therapeutic potential, we also explored its indirect effects on N-glycosylation. mTOR inhibition downregulates sterol regulatory element-binding protein (SREBP) signaling, which controls the expression of HMG-CoA reductase and regulates the mevalonate pathway³⁸˒³⁹. This pathway is responsible for producing isoprenoids, including dolichol, which facilitate N-glycosylation. By inhibiting this pathway, rapamycin could help prevent the functional production of Spike proteins from persistent mRNA or genomic inserts, while concurrently reducing pathological RAAS disruption. mTOR inhibition may also reduce Spike protein production by downregulating the cell cycle. mTOR is a key regulator of cell growth and proliferation, and its inhibition leads to cell cycle arrest, particularly in the G1 phase, thereby reducing overall protein synthesis⁴⁰. This reduction in cell proliferation leads to a decrease in global protein production, including Spike proteins. As the body’s cell cycle slows, mTOR inhibition downregulates key proteins involved in mRNA translation such as 4E-BP1 and S6K1, which are involved in suppression of translational machinery, limiting the production of general proteins and possibly the Spike proteins encoded by mRNA vaccines. Importantly, the patient in this case demonstrated a dramatic reduction in Spike protein antibodies during treatment, suggesting that rapamycin, along with adjunctive treatments such as bromelain, curcumin, nattokinase, and colchicine, may have facilitated the clearance of residual Spike protein from the body. This outcome further highlights the potential of mTOR inhibitors, proteolytic enzymes, and anti-inflammatories in the treatment of COVID-19 vaccine-induced myopericarditis. Moreover, the combination of rapamycin’s autophagic induction, its capacity to reduce N-glycosylation, and cell cycle downregulation may prevent the production of pathological Spike proteins, offering a multifaceted approach to managing vaccine-induced cardiac injury.
Figure 7: Proposed mechanisms supporting the use of rapamycin in treating COVID-19 vaccine-induced pericarditis.
This case report is inherently limited by its focus on a single patient N of 1 trial. While the patient’s positive response to the addition of rapamycin offers encouraging preliminary evidence, it is anecdotal and cannot alone establish causal efficacy. Larger-scale studies and clinical trials are critically needed to confirm these observations and to develop optimized therapeutic protocols for patients facing these emerging and complex conditions. This case presents the challenge of differentiating between the adverse cardiovascular effects of psychostimulants and those induced by COVID-19 vaccination. The safety profile of long-term ADHD treatment with psychostimulants remains unclear, with existing data pointing to the possibility of exacerbating cardiovascular disease⁴². Neuropsychiatric medications may result in more symptomatic syndromes among those with vaccine myopericarditis. Although it is plausible that a stimulant overdose exacerbated the vaccine-induced pathology, the patient had prior tolerance to stimulant use without reported cardiac symptoms.
Conclusion
Base Spike detoxification with nattokinase, bromelain, and curcumin appears to represent a reasonable standard of care for COVID-19 vaccine myopericarditis. If symptoms do not resolve, then rapamycin could be considered as empiric adjunctive treatment. Given its mechanism of action and tolerability at low doses, rapamycin should be investigated for the treatment of cardiac or circulatory injuries resulting from COVID-19 vaccination or infection. Although this is a limited case report involving a single patient, it nevertheless provides a hypothetical foundation for possibility that rapamycin may display efficacy in the treatment of COVID-19 vaccine-induced myopericarditis. Therefore, further studies for the treatment of COVID-19 vaccine-induced myopericarditis with rapamycin are warranted, including in vitro models, in silico models, animal studies, and eventual clinical trials.
Conflict of Interest:The authors have no conflicts of interest to declare.
Funding Statement:
No funding was received for this study.
Acknowledgements:
None
Abbreviations
Hx = history, AU = antibody units per milliliter, HR = heart rate, BP = blood pressure, ECG = electrocardiogram, Hs-troponin = high-sensitivity troponin, BNP/NT-proBNP = brain natriuretic peptide and N-terminal proBNP, LVEF = left ventricular ejection fraction, POTS = Postural orthostatic tachycardia syndrome.
References
1. Van Damme W, Dahake R, Delamou A, et al. The COVID-19 pandemic: diverse contexts; different epidemics-how and why?. BMJ Glob Health. 2020; 5(7):e003098. doi:10.1136/bmjgh-2020-003098
2. Jackson LA, Anderson EJ, Rouphael NG, et al. An mRNA Vaccine against SARS-CoV-2 – Preliminary Report. N Engl J Med. 2020;383(20): 1920-1931. doi:10.1056/NEJMoa2022483
3. Mead M, Seneff S, Rose J, et al. COVID-19 Modified mRNA “Vaccines”: Lessons Learned from Clinical Trials, Mass Vaccination, and the Bio-Pharmaceutical Complex, Part 2. International Journal of Vaccine Theory, Practice, and Research. 2024; 3(2), 1275-1344. doi: 10.56098/w66wjg87
4. Yasmin F, Najeeb H, Naeem U, et al. Adverse events following COVID-19 mRNA vaccines: a systematic review of cardiovascular complication, thrombosis, and thrombocytopenia. Immun Inflamm Dis. 2023; 11:e807. doi:10.1002/iid3.807
5. United States Department of Health and Human Services (DHHS), Public Health Service (PHS), Centers for Disease Control (CDC) / Food and Drug Administration (FDA), Vaccine Adverse Event Reporting System (VAERS) 1990 – 08/30/2024, CDC WONDER On-line Database. Accessed at http://wonder.cdc.gov/vaers.html on Sep 23, 2024
6. Rose J, Hulscher N, McCullough PA. Determinants of COVID-19 vaccine-induced myocarditis. Ther Adv Drug Saf. 2024;15:20420986 241226566. Published 2024 Jan 27. doi:10.1177/2 0420986241226566
7. Hulscher N, Hodkinson R, Makis W, McCullough PA. Autopsy findings in cases of fatal COVID-19 vaccine-induced myocarditis. ESC Heart Fail. Published online January 14, 2024. doi:10.1002 /ehf2.14680
8. Hulscher, N., Cook, M. J., Stricker, R. B., McCullough, P. A. Excess Cardiopulmonary Arrest and Mortality after COVID-19 Vaccination in King County, Washington. J Emerg Med OA. 2024; 2(1), 01-11.
9. Hulscher N, Procter BC, Wynn C, McCullough PA. Clinical Approach to Post-acute Sequelae After COVID-19 Infection and Vaccination. Cureus. 2023;15(11):e49204. Published 2023 Nov 21. doi:10.7759/cureus.49204
10. Behbahani-Nejad O, Mikolich B, Morgenstern D, Mikolich JR. Myocarditis response to colchicine therapy based on cardiac MRI diagnostic criteria. J Am Coll Cardiol. (2021) 77(18_Supplement_1): 1432. doi: 10.1016/S0735-1097(21)02790-X
11. Valore L, Junker T, Heilmann E, et al. Case report: mRNA-1273 COVID-19 vaccine-associated myopericarditis: Successful treatment and re-exposure with colchicine. Front Cardiovasc Med. 2023;10:1135848. Published 2023 Apr 17. doi:10.3389/fcvm.2023.1135848
12. McCullough, P.; Hulscher, N. Risk Stratification for Future Cardiac Arrest after COVID-19 Vaccination. Preprints 2024, 2024080821. doi:10. 20944/preprints202408.0821.v1
13. Faksova K, Walsh D, Jiang Y, et al. COVID-19 vaccines and adverse events of special interest: A multinational Global Vaccine Data Network (GVDN) cohort study of 99 million vaccinated individuals. Vaccine. 2024;42(9):2200-2211. doi:10. 1016/j.vaccine.2024.01.100
14. Parry PI, Lefringhausen A, Turni C, et al. ‘Spikeopathy’: COVID-19 Spike Protein Is Pathogenic, from Both Virus and Vaccine mRNA. Biomedicines. 2023;11(8):2287. Published 2023 Aug 17. doi:10.3390/biomedicines11082287
15. Baumeier C, Aleshcheva G, Harms D, et al. Intramyocardial Inflammation after COVID-19 Vaccination: An Endomyocardial Biopsy-Proven Case Series. Int J Mol Sci. 2022;23(13):6940. Published 2022 Jun 22. doi:10.3390/ijms23136940
16. Wu N, Joyal-Desmarais K, Ribeiro PAB, et al. Long-term effectiveness of COVID-19 vaccines against infections, hospitalisations, and mortality in adults: findings from a rapid living systematic evidence synthesis and meta-analysis up to December, 2022. Lancet Respir Med. 2023;11(5): 439-452. doi:10.1016/S2213-2600(23)00015-2
17. Castruita JAS, Schneider UV, Mollerup S, et al. SARS-CoV-2 spike mRNA vaccine sequences circulate in blood up to 28 days after COVID-19 vaccination. APMIS. 2023;131(3):128-132. doi:10. 1111/apm.13294
18. Brogna C, Cristoni S, Marino G, et al. Detection of recombinant Spike protein in the blood of individuals vaccinated against SARS-CoV-2: Possible molecular mechanisms. Proteomics Clin Appl. 2023;17(6):e2300048. doi:10.1002/prca.202 300048
19. Yu CK, Tsao S, Ng CW, et al. Cardiovascular Assessment up to One Year After COVID-19 Vaccine-Associated Myocarditis. Circulation. 2023; 148(5):436-439. doi:10.1161/CIRCULATIONAHA.1 23.064772
20. Fürst T, Šourek P, Krátká Z, Janošek J. Batch-dependent safety of COVID-19 vaccines in the Czech Republic and comparison with data from Denmark. Eur J Clin Invest. 2024;54(10):e14271. doi:10.1111/eci.14271
21. Schmeling M, Manniche V, Hansen PR. Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine. Eur J Clin Invest. 2023;53(8):e13998. doi:10.1111/eci.13998
22. Jablonowski K, Hooker B. Batch-dependent safety of the BNT162b2 mRNA COVID-19 vaccine in the United States. Science, Public Health Policy and the Law. 2024 Sep 26; v5.2019-2024
23. Knoll F. How Bad is My Batch? [Online]. GitHub; 2024 [cited 2024 Oct 25]. Available at: https://knollfrank.github.io/HowBadIsMyBatch/HowBadIsMyBatch.html
24. Hulscher, N.; McCullough, P. A. Delayed Fatal Pulmonary Hemorrhage Following COVID-19 Vaccination: Case Report, Batch Analysis, And Proposed Autopsy Checklist. Preprints 2024, 2024 021096. doi:10.20944/preprints202402.1096.v1
25. Huang HC, Lai YJ, Liao CC, et al. Targeting conserved N-glycosylation blocks SARS-CoV-2 variant infection in vitro. EBioMedicine. 2021;74:10 3712. doi:10.1016/j.ebiom.2021.103712
26. Whitby K, Pierson TC, Geiss B, Lane K, Engle M, Zhou Y, Doms RW, Diamond MS. Castanospermine, a potent inhibitor of dengue virus infection in vitro and in vivo. J Virol. 2005 Jul;79(14):8698-706. doi: 10.1128/JVI.79.14.8698-8706.2005.
27. Garcia G Jr, Sharma A, Ramaiah A, et al. Antiviral drug screen identifies DNA-damage response inhibitor as potent blocker of SARS-CoV-2 replication. Cell Rep. 2021;35(1):108940. doi:10. 1016/j.celrep.2021.108940
28. Casas-Sanchez, Aitor et al. “Inhibition of Protein N-Glycosylation Blocks SARS-CoV-2 Infection.” mBio vol. 13,1 (2021): e0371821. doi:10.1128/mbio.03718-21
29. Khan N. mTOR: A possible therapeutic target against SARS-CoV-2 infection. Arch Stem Cell Ther. 2021;2(1):5-7.
30. Gu J, Hu W, Song Z-P, Chen Y-G, Zhang D-D and Wang C-Q (2016) Rapamycin Inhibits Cardiac Hypertrophy by Promoting Autophagy via the MEK/ERK/Beclin-1 Pathway. Front. Physiol. 7:104. doi: 10.3389/fphys.2016.00104
31. Maeda K, Shioi T, Kosugi R, et al. Rapamycin ameliorates experimental autoimmune myocarditis. Int Heart J. 2005;46(3):513-530. doi:10.1536/ihj.46.513
32. Maisch B. SARS-CoV-2, vaccination or autoimmunity as causes of cardiac inflammation. Which form prevails?. SARS-CoV-2, Impfung oder Autoimmunität als Ursachen für Herzentzündungen. Welche Form überwiegt?. Herz. 2023;48(3):195-205. doi:10.1007/s00059-023-05182-6
33. Kanduc, Darja. “From SARS-CoV-2 to Myocarditis and Sudden Death via Molecular Mimicry and Immunologic Memory,” Current Practice in Medical Science Vol. 4 (2022): 129-138. doi:10.9734/bpi/cpms/v4/3200B
34. Nunez-Castilla J, Stebliankin V, Baral P, Balbin CA, Sobhan M, Cickovski T, Mondal AM, Narasimhan G, Chapagain P, Mathee K, Siltberg-Liberles J. Potential Autoimmunity Resulting from Molecular Mimicry between SARS-CoV-2 Spike and Human Proteins. Viruses. 2022 Jun 28;14(7): 1415. doi: 10.3390/v14071415
35. Angeli F, Reboldi G, Trapasso M, Zappa M, Spanevello A, Verdecchia P. COVID-19, vaccines and deficiency of ACE2 and other angiotensinases. Closing the loop on the “Spike effect”. Eur J Intern Med. 2022 Sep;103:23-28. doi: 10.1016/j.ejim. 2022.06.015
36. Krämer LM, Brettschneider J, Lennerz JK, et al. Amyloid precursor protein-fragments-containing inclusions in cardiomyocytes with basophilic degeneration and its association with cerebral amyloid angiopathy and myocardial fibrosis. Sci Rep. 2018;8(1):16594. Published 2018 Nov 9. doi:10.1038/s41598-018-34808-7
37. Gao G, Chen W, Yan M, Liu J, Luo H, Wang C, Yang P. Rapamycin regulates the balance between cardiomyocyte apoptosis and autophagy in chronic heart failure by inhibiting mTOR signaling. Int J Mol Med. 2020 Jan;45(1):195-209. doi: 10.3892/ijmm. 2019.4407
38. Porstmann T, Santos CR, Griffiths B, Cully M, Wu M, Leevers S, Griffiths JR, Chung YL, Schulze A. SREBP activity is regulated by mTORC1 and contributes to Akt-dependent cell growth. Cell Metab. 2008 Sep;8(3):224-36. doi: 10.1016/j.cmet. 2008.07.007
39. Madison BB. Srebp2: A master regulator of sterol and fatty acid synthesis. J Lipid Res. 2016 Mar;57(3):333-5. doi: 10.1194/jlr.C066712
40. Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149(2):274-293. doi:10.1016/j.cell.2012.03.017
41. Zoncu R, Efeyan A, Sabatini DM. mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol. 2011;12(1):21-35. doi:10.1038/nrm3025
42. Groenman AP, Schweren LJ, Dietrich A, Hoekstra PJ. An update on the safety of psychostimulants for the treatment of attention-deficit/hyperactivity disorder. Expert Opin Drug Saf. 2017;16(4):455-464. doi:10.1080/14740338. 2017.1301928


 http://orcid.org/0009-0008-0677-7386
http://orcid.org/0009-0008-0677-7386